Antarctic Fur Seal
Arctocephalus gazella

2025 Red list status
Least Concern
Regional Population Trend
Change compared
to 2016
No Change
Overview
Arctocephalus gazella – (Peters, 1875)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – OTARIIDAE – Arctocephalus – gazella
Common Names: Antarctic Fur Seal, Kerguelen Fur Seal (English), Antarktiese Pelsob (Afrikaans), Otarie de Kerguelen, Arctocéphale de Kerguelen (French)
Synonyms: Arctophoca gazella Peters, 1875; Arctocephalus tropicalis ssp. gazella Peters, 1875
Taxonomic Note:
Antarctic Fur Seals were formerly considered a subspecies of Arctocephalus tropicalis and were known as A. t. gazella (Repenning et al. 1971). Rice (1998) listed the species as A. gazella. In 2011 the genus of this, and most other species of Fur Seals was changed to Arctophoca, Peters 1866 (Committee on Taxonomy 2011) based on evidence presented in Berta and Churchill (2012). However, in 2013, based on genetic evidence presented in Nyakatura and Bininda-Emonds (2012), this change was considered to be premature, and these species were returned to the genus Arctocephalus pending further research (Committee on Taxonomy 2013).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: de Bruyn, P.J.N.1 & Hofmeyr, G.J.G.2
Reviewer: Kerley, G.I.H.3
Contributors: da Silva, J.M.4 & Patel, T.5
Institutions:1University of Pretoria, 2Port Elizabeth Museum at Bayworld, 3Nelson Mandela University, 4South African National Biodiversity Institute, 5Endangered Wildlife Trust
Previous Assessors and Reviewers: Hofmeyr, G.J.G., de Bruyn, N., Bester, M.N. & Wege, M.
Previous Contributors: Kirkman, S., Oosthuizen, H., Meyer, M., Seakamela, M., Lowry, L., Page-Nicholson, S. & Child, M.F.
Assessment Rationale
Nationally, due to the increasing size of the population at the Prince Edward Islands (PEI), their links with other populations, and a lack of apparent threats, the Antarctic Fur Seal is listed Least Concern. While this species was listed as Near Threatened in 2004, exponential growth has increased the population (Wege et al. 2016). The current total PEI population is estimated at 15,360. The latest values for pups production are 810 in 2008/2009 for PEI itself, and approximately 2,500 for Marion Island in 2019/2020 (MIMMP unpublished data). The main colony on Marion Island is responsible for some two thirds of pup production.
The Prince Edward Islands group was proclaimed a Special Nature Reserve in 1995, under the South African Environmental Conservation Act (No. 73 of 1989), and a Marine Protected Area in 2013. Antarctic Fur Seals are further protected by various items of legislation.
Although this species does not currently face any major threats within the assessment region, it may be vulnerable to climate change due to the possible impact on populations of prey species. In addition, recent Southern Hemisphere avian influenza (H5N1) outbreak events are cause for concern which should be monitored. With recent slowing of their rate of increase at the assessment site, they should be monitored both in light of the continued decline in abundance of this species at Bird Island, South Georgia, and the recent decline in the sympatric population of Subantarctic Fur Seals (A. tropicalis) at Marion Island. Given the scarcity of pup production data from other islands and the Antarctic Fur Seals’ ability to travel extreme distances, the effects of migration on population estimates at the above-mentioned sites cannot be discounted.
Regional population effects: Antarctic Fur Seals are thought to have a continuous range and therefore the potential exists for immigrants from other subpopulations in the Southern Ocean to augment the Prince Edward Island population. This especially since Antarctic Fur Seals have been recorded at locations distant from their breeding colonies (Drehmeer and De Oliveira 2000, Stewardson 2007, Bester et al. 2014). Genetic evidence indicates that the large population at South Georgia may have been a partial source of immigrants that established the Prince Edward Islands population (Wynen et al. 2000), although newer genetic evidence suggests that perhaps geographically closer (but unknown) relic populations may have been more important (Paijmans et al. 2020). The arrival and birth of leucistic individuals (de Bruyn et al. 2007; Wege et al. 2015), a colour morph common at South Georgia (Bonner 1968), together with an exponential growth rate of this population at the end of the 20th century (Hofmeyr et al. 2006), indicates that such immigration was likely
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: de Bruyn PJN & Hofmeyr GJG. 2025. A conservation assessment of Arctocephalus gazella. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Antarctic Fur Seals are widely-distributed in waters south, and in some areas, slightly north, of the Antarctic Convergence (Bonner 1968). While most Antarctic Fur Seals breed at South Georgia (Boyd 1993) colonies are also found on the South Shetland Islands (Goebel et al. 2008), the South Orkney Islands (Boyd 1993), the South Sandwich Islands (Holdgate 1963), the Prince Edward Islands (Bester et al. 2003, Hofmeyr et al. 2006), Îles Crozet (Guinet et al. 1994), Îles Kerguelen (Guinet et al. 2000), Heard Island (Page et al. 2003), McDonald Island (Johnstone 1982), Macquarie Island (Goldsworthy et al. 1999) and Bouvetøya (Hofmeyr et al. 2005). Vagrants have been recorded at Gough Island (Wilson et al. 2006, Bester and Reisinger 2010), Tristan da Cunha (Bester et al. 2014) and on the coasts of Antarctica (Shaughnessy and Burton 1986), southern South America (Drehmer and De Oliviera 2000) and Australia (Shaughnessy et al. 2014). Antarctic Fur Seals disperse widely when at sea (Boyd et al. 1998, Staniland et al. 2012, Arthur et al. 2018).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0 m
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): ~250m below sea level foraging
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
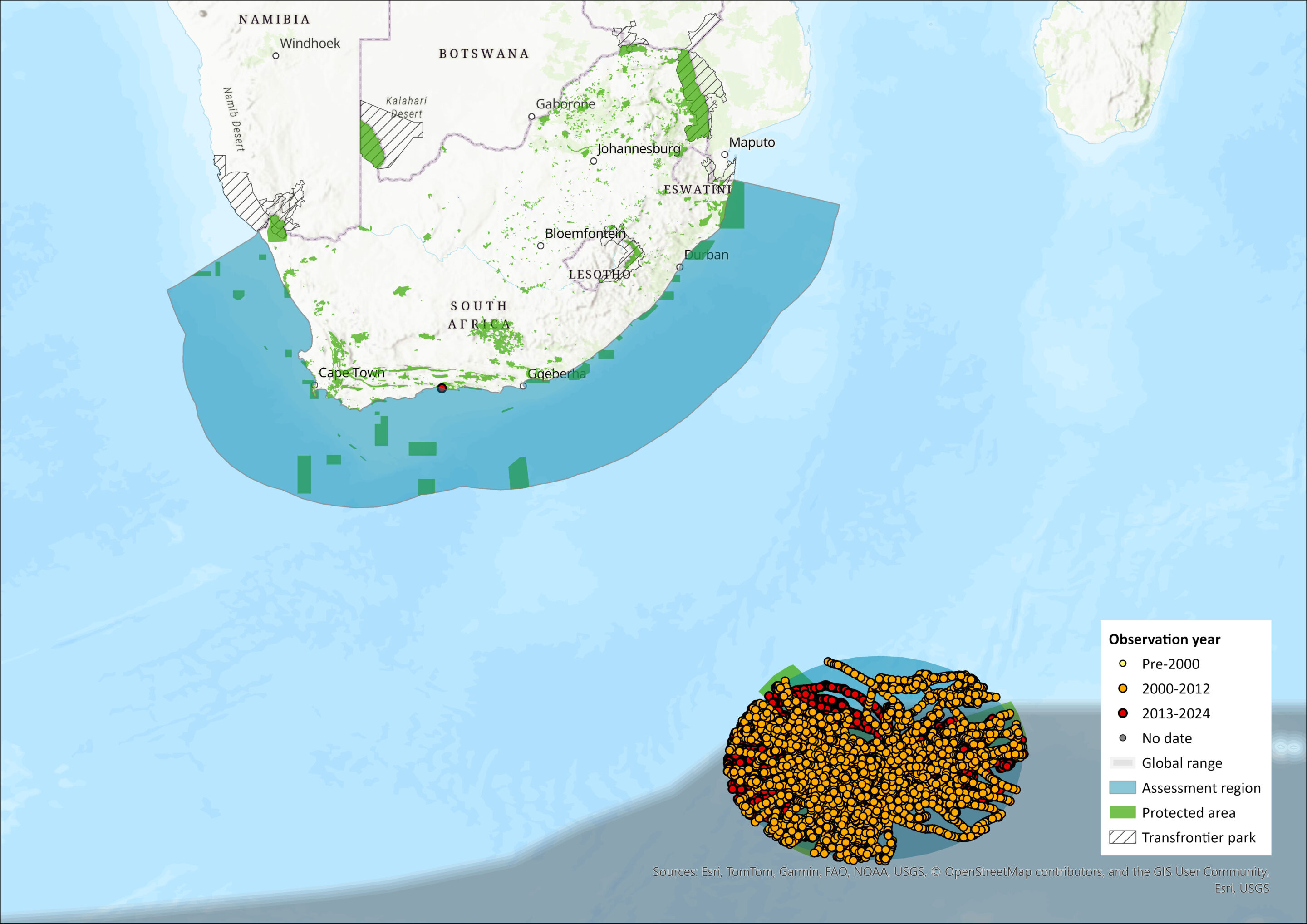
Map
Figure 1. Distribution records for Antarctic Fur Sea (Arctocephalus gazella) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified
Biogeographic Realms
Biogeographic Realm: Antarctic, Sub-Antarctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Antarctica | Extant | Vagrant | – | – |
| Argentina | Extant | Vagrant | – | – |
| Australia | Extant | Vagrant | – | – |
| Australia -> Macquarie Is. | Extant | Native | – | – |
| Bouvet Island | Extant | Native | – | – |
| Brazil | Extant | Vagrant | – | – |
| Chile | Extant | Vagrant | – | – |
| Chile -> Juan Fernández Is. | Extant | Native | – | – |
| French Southern Territories | Extant | Native | – | – |
| French Southern Territories -> Crozet Is. | Extant | Native | – | – |
| French Southern Territories -> Kerguelen | Extant | Native | – | – |
| Heard Island and McDonald Islands | Extant | Native | – | – |
| Saint Helena, Ascension and Tristan da Cunha | Extant | Vagrant | – | – |
| Saint Helena, Ascension and Tristan da Cunha -> Tristan da Cunha | Extant | Vagrant | – | – |
| South Africa | Extant | Vagrant | – | – |
| South Africa -> Marion-Prince Edward Is. | Extant | Native | – | – |
| South Georgia and the South Sandwich Islands | Extant | Native | – | – |
| South Georgia and the South Sandwich Islands -> South Georgia | Extant | Native | – | – |
| South Georgia and the South Sandwich Islands -> South Sandwich Is. | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: None
FAO Area Occurrence
| Presence | Origin | Formerly Bred | Seasonality | |
| 41. Atlantic – southwest | Extant | Native | – | – |
| 48. Atlantic – Antarctic | Extant | Native | – | – |
| 58. Indian Ocean – Antarctic | Extant | Native | – | – |
| 61. Pacific – northwest | Extant | Native | – | – |
.
Climate change
As the sole otariid indicator species contributing to the Ecosystem Monitoring Program (CEMP) of the Commission for the Conservation of Antarctic Marine Living Resources (CCAMLR), Antarctic Fur Seal studies are essential for informing the potential effects of both short-term oceanographic events (for example, El Niño Southern Oscillation and Southern Annular Mode events) and long-term changes (for example, climate change, fisheries exploitation) in the Southern Ocean. The demographic drivers of changes in Antarctic Fur Seal abundance and behaviour require continued monitoring. This for assessing, both ecosystem changes and the survival of this species in light of the decline in abundance at South Georgia (Forcada et al. 2023). Forcada and Hoffman (2014) attributed this decline to climate change. However, Boyd (2014) challenged this interpretation and suggested that the decline was more likely due to density-dependent factors acting on a population that had been growing exponentially for decades. The role of climate change is unknown but there is evidence of a possibly related shift in the prey base (de Lima et al. 2022). However, the plasticity in the Antarctic Fur Seal diet between populations throughout its range (e.g. Friscourt et al. 2024), and even within populations (e.g. Reisinger et al. 2018), complicates assessment of the strength of climate change as a driver. In examining the potential redistribution of a community of marine predators by comparing current and future habitat distribution projections, Reisinger et al. (2022) showed how Antarctic Fur Seal foraging habitat use is predicted to shift substantially due to changing climate. At present, the local increasing population of this species at the Prince Edward Islands and their known dietary plasticity suggest current changing conditions remain within their tolerance range, although the extent of that tolerance range is unknown.
Population information
Globally, Antarctic Fur Seals breed at numerous sites on 11 islands or island groups. While some 95% of Antarctic Fur Seal pup production is on the island of South Georgia, eight other subpopulations are estimated to support breeding populations (Forcada & Hoffman 2014; SCAR_EGS 2014; Forcada et al. 2023). Although Antarctic Fur Seals are still the most abundant species of fur seals with an estimated population in excess of 3.5 million animals in 2009 (Forcada et al. 2023), the numbers of adult females at Bird Island, South Georgia are estimated to have declined by 24% between 1984 and 2012 (Forcada & Hoffman 2014). Since 2009, this population at Bird Island (off South Georgia) has continued to decline at 7.2% per annum (Forcada et al. 2023). Should this be representative of the South Georgia population as a whole, this would effectively represent a decline in the global population. Forcada and Hoffman (2014) attributed this decline and an accompanying increase in population genetic diversity to climate change. However, Boyd (2014) challenged these conclusions and suggested the decline is most likely in response to density-dependent factors acting upon a population that had been growing exponentially for several decades.
Within the assessment region, both islands (Marion & Prince Edward) within the Prince Edward Islands Group support increasing breeding rookeries and the total population (2022/23) is estimated at over 15,360 animals using a conversion factor of 4.8 (pups to total population) (Kerley 1983). Pup production in the 2008/09 summer at Prince Edward Island itself was estimated at 810 having grown by 11.4% since 2000/01 (the most recent estimate). While pup production on Marion Island increased by 4.0% between 2009/10 and 2012/13 to 1,553, it had slowed by 17% between 2003/04 and 2012/13 (Wege et al. 2016). This was attributed to saturation at the main rookery. However, the current assessment shows that total pup production has since increased by 35% to ~2400 in 2022/23. This is a result of growth both at the main rookery, which is currently responsible for some two thirds of island pup production and at other rookeries. The latter have increased by some 50% from 355 pups (2012/13) to 716 (2022/23). It is noteworthy that the main rookery now constitutes a reduced proportion of the total island population than at the last assessment (67% compared to 77–81%). Two other breeding rookeries produce in excess of 300 and 100 pups annually, while the remainder of the island hosts tiny but increasingly numerous rookeries. Although the breeding rookeries are fragmented along the island coastlines, gene flow occurs as individuals occasionally move between rookeries (Mammal Research Institute unpubl. data).
Population Information
Current population trend: Increasing
Continuing decline in mature individuals? No
Extreme fluctuations in the number of subpopulations: No. Subpopulations are relatively stable
Continuing decline in number of subpopulations: No.
All individuals in one subpopulation: No, although a major proportion (~67%) exist in one large rookery on Marion Island
Number of mature individuals in largest subpopulation: 9120
Number of Subpopulations: Antarctic Fur Seals likely have a continuous global range with no distinct subpopulations.
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Very low
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Very low
Probability of extinction in the wild within 100 years: No projections
Population genetics
By the late 19th century, the Antarctic Fur Seal was rendered almost extinct due to more than a century of commercial sealing. This resulted in concerns over the consequences of a genetic bottleneck effect as the global population recovered (Kovacs et al. 2012). Populations survived this period in small numbers at, at least, three sites: South Georgia, Bouvet Island and Îles Kerguelen (Wynen et al. 2000; Hofmeyr et al. 2005). It is also possible that the species survived at a fourth site: the South Shetland Islands (Bonin et al. 2013). Coalescent simulations suggest that all these four known relic populations experienced severe bottlenecks down to effective population sizes of around 150–200 (Paijmans et al. 2020). Despite this global population bottleneck (Wynen et al. 2000), the persistence, even in small numbers, of these few relic subpopulations resulted in unexpectedly high levels of genetic diversity remaining (Bonin et al. 2013; Paijmans et al. 2020). This is also true for the population at the Prince Edwad Islands (Maboko 2009). Humble et al. (2018) found evidence for population structuring across the species range, with additional structure detected between South Georgia, the South Shetlands and Bøuvetoy islands using ddRAD compared to microsatellites. The study did not incorporate animals from the assessment region, but animals from Kerguelen, Heard and Macquarie Islands formed part of the same subpopulation. It is likely animals from the assessment region fall within a single subpopulation, possibly connected with the Kerguelen, Heard and Macquarie cluster.
By interrogating a genetic polymorphism (S291F) responsible for cream-coloured phenotypes that are especially prevalent in the South Georgia population, Hoffman et al. (2018) suggested that emigration from this relic population to recolonise former breeding sites was likely fairly localised. Their results suggest that the South Georgia relic populations were unlikely to have played a major role in population recovery of geographically more distant former breeding sites (including the Prince Edward Islands). This raises the tantalising possibility of other unidentified relic populations surviving the sealing efforts, corroborated by Humble et al.’s (2018) findings. Locally relevant for this assessment, genetic differentiation within Marion Island rookeries is near zero showing a high level of connectivity between rookeries (Marion Island Marine Mammal Programme, unpubl. data).
Based on the available information, the Convention on Biological Diversity’s Global Biodiversity Framework’s genetic diversity indicators can be calculated. Since no subpopulations have gone extinct within the assessment region, the complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (1/1 subpopulations remain). Based on the total population within the assessment region of 15,360, and applying an effective population size (Ne) to census size (Nc) ratio between 0.1-0.3, the Ne for this species is estimated at 1 536 –4 608. This exceeds the Ne 500 threshold for a healthy, stable genetic population. The GBF’s headline indicator – proportion of species with an effective population size (Ne) greater than 500 – is therefore 1.0 (1/1 population with Ne> 500). It is highly recommended a population genetic study be undertaken to more accurately quantify the Ne for the population given that bottlenecks can considerably impact upon Ne due to high levels of inbreeding.
Habitats and ecology
Antarctic Fur Seals are an extremely sexually dimorphic species and are highly polygynous. Males arrive at the rookeries in early November to establish territories, with adult females arriving some 2-3 weeks later. Males continue to arrive and challenge one another for territories through much of the season. Territories are acquired and held by use of vocalisations, threat postures and fighting (Bonner 1968). Adult females give birth within days of their arrival, weaning their pups some four months later (Kerley 1987). The median pupping date at the Prince Edwards Islands is ~6th December (Hofmeyr et al. 2006). At the Prince Edward Islands the duration of the attendance cycle by lactating adult females varies between years, with mean onshore periods varying between 1.5 and 2.2 days, and mean foraging trips varying between 6.0 and 9.4 days, becoming progressively longer towards weaning (Bester & Bartlett 1990; Kirkman et al. 2003).
Following weaning, most adult seals disperse widely at sea. However, a few subadults and adult males can be seen ashore at all times of the year (Bonner 1968; Payne 1977; Kerley 1983). The diet of Antarctic Fur Seals varies by season and location. At South Georgia and Bouvet Island, Antarctic Fur Seals are krill specialists (Bonner 1968; North et al. 1983; Kirkman et al. 2000; Hofmeyr et al. 2010). However, at Heard Island (Green et al. 1989, 1991), Macquarie Island (Robinson et al. 2003) and the Prince Edward Islands (Klages & Bester 1998; Makhado et al. 2008; Reisinger et al. 2018), krill is not available, and they follow a generalist diet, preying primarily on cephalopods and fish such as myctophids and notothenids. Antarctic Fur Seals have also been known to eat penguins at a number of sites (Bonner 1968; Green et al. 1989), including at Marion Island (Hofmeyr & Bester 1993). Marion Island is the first location where Antarctic Fur Seals were observed to catch penguins on land (Hofmeyr & Bester 1993; also see Haddad et al. 2015), but this behaviour has subsequently been recorded on Îles Crozet (Charbonnier et al. 2010).
Antarctic Fur Seals are sympatric with Subantarctic Fur Seals at the Prince Edward Islands (Hofmeyr et al. 2006) and Îles Crozet (Guinet et al. 1994) and with both Subantarctic Fur Seals and New Zealand Fur Seals (A. forsteri) at Macquarie Island (Goldsworthy 1999). Hybridisation occurs at all three sites (Hofmeyr et al. 2006; Kingston & Gwilliam 2007; Lancaster et al. 2010).
Ecosystem and cultural services: The Antarctic Fur Seal is an important predator in the Southern Ocean (Boyd & Murray 2001; Goldsworthy et al. 2001; Reid et al. 2004; Lea et al. 2006). It is also a prey species for Leopard Seals (Hydrurga leptonyx) (Boveng et al. 1998) and Killer Whales (Orcinus orca) (Staniland & Robinson 2008; Reisinger et al. 2016). Antarctic Fur Seals are an important component of the Convention for the Conservation of Marine Living Resources (CCAMLR) Ecosystem Monitoring Programme (CEMP) (Agnew 1997).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 9.1. Marine Neritic -> Marine Neritic – Pelagic | – | Suitable | Yes |
| 10.1. Marine Oceanic -> Marine Oceanic – Epipelagic (0-200m) | – | Suitable | Yes |
| 12.1. Marine Intertidal -> Marine Intertidal – Rocky Shoreline | – | Suitable | Yes |
| 13.1. Marine Coastal/Supratidal -> Marine Coastal/Supratidal – Sea Cliffs and Rocky Offshore Islands | – | Suitable | Yes |
Life History
Generation Length: ~10 years
Age at Maturity: Female or unspecified: Sexual maturity 3-4 years
Age at Maturity: Male: Sexual maturity 3-4 years, but social maturity 6-10 years
Size at Maturity (in cms): Female: 120 – 140 cm standard length
Size at Maturity (in cms): Male: 180 cm standard length
Longevity: males ~15 years; females ~23 years
Average Reproductive Age: (Not specified)
Maximum Size (in cms): ~200 cm
Size at Birth (in cms): 60 – 75 cm
Gestation Time: 8-9 months (~12 months including delayed implantation)
Reproductive Periodicity: Annual
Average Annual Fecundity or Litter Size: Single pup, twins are very rare
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial, Marine
General Use and Trade Information
Although this species was previously overexploited in the 19th century, it has not been exploited since 1927, partly being protected by the isolation of their habitat.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Unknown | – | Unknown | – |
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
By the late 19th century, the Antarctic Fur Seal was almost extinct due to commercial sealing (Kovacs et al. 2012). Populations survived in small numbers at at least three sites: South Georgia, Bouvet Island and Îles Kerguelen (Wynen et al. 2000; Hofmeyr et al. 2005; Paijmans et al. 2020). It is also possible that the species survived at a fourth site: the South Shetland Islands (Bonin et al. 2013). Despite a population bottleneck (Wynen et al. 2000), the persistence, even in small numbers, of a few relic populations resulted in unexpectedly high levels of genetic diversity (Bonin et al. 2013; Paijmans et al. 2020), including at the Prince Edward Islands (Maboko 2009).
Few legal fisheries exploit the range of the Antarctic Fur Seal. However, illegal fishing in the area is likely to increase (Hanchet et al. 2003). Antarctic Fur Seals have been recorded entangled in marine debris (including discarded fishing line, nets and other foreign objects) (Jepsen & de Bruyn 2019). As the fisheries expand, it is further likely that this threat too will increase. The most recent estimate of entanglement rates of Antarctic Fur Seals at South Georgia is 0.4% (Arnould & Croxall 1995). At the Prince Edward Islands, the rate of entanglement (together with the sympatric Subantarctic Fur Seal) was estimated to be lower, at approximately 0.24% for the period 1996–2001 (Hofmeyr et al. 2002). Entangled animals are expected to die as a result of their injuries, unless the entanglements are removed early (Bonner & McCann 1982; Croxall et al. 1990; Hofmeyr et al. 2002; Jepsen & de Bruyn 2019).
While tourism at Subantarctic islands is minimal (Shirihai 2002; Hofmeyr & Bester 2008), its increase could cause disturbance. However, the South African government prohibits tourism at the Prince Edward Islands (Prince Edward Islands Management Plan 2010).
Although this species does not currently face any major threats within the assessment region, it may be vulnerable to climate change due to the possible impact on populations of prey species (Siniff et al. 2008; Kovacs et al. 2012; McDonald et al. 2012). This has been suggested to be responsible for the decline in the major subpopulation of the species at South Georgia (Forcada & Hoffman 2014; Forcada et al. 2023). Climate change may also negatively impact the abundance of prey species (Kovacs et al. 2012). Growing populations of other krill-feeding predators, such as whales, recovering from previous century’s harvesting might also compete with Antarctic Fur Seals for food.
Antarctic Fur Seals are sympatric with Subantarctic Fur Seals at the Prince Edward Islands and hybridisation occurs. However, levels are very low and thought to be decreasing (Hofmeyr et al. 2006).
Global climate change may result in additional future negative impacts (Learmonth et al. 2006). The risk of disease transmission may increase (Lavigne & Schmitz 1990). Due to their colonial habits, otariids, such as the Antarctic Fur Seal, are at greater risk of future disease outbreaks, though the isolation of their haulout sites may afford some protection. The recent outbreak of High Pathogenicity Avian Influenza (HPAI) in the Southern Hemisphere could be a major threat (Banyard et al. 2024). Detection of the virus in various species of seabirds at Marion Island is cause for concern (Duvenage 2024). This virus has caused mass mortalities of pinnipeds elsewhere (Alava et al. 2024, Campagna et al. 2024). Rabies is also a risk. This disease has recently become endemic in Cape Fur Seals (Van Helden 2024). Southern Ocean pinnipeds are regular vagrants to the South African coast (Bester 1989), and so could potentially come into contact with Cape Fur Seals. The return of infected vagrants to Subantarctic islands may result in the further distribution of the disease. Cape Fur Seals have also been recorded as a vagrant on Marion Island (Kerley 1983).
Conservation
Governments that claim island territories manage breeding sites of Antarctic Fur Seals as protected areas. The species is also secured by the South African Seabirds and Seals Protection Act (Prince Edward Islands Management Plan 2010), the Marine Living Resources Act (Act no. 18 of 1998; Policy on the management of seals, seabirds and shorebirds. (Government Gazette No. 30534, 2007) and the Threatened or Protected Species Regulations (Government Gazzette No. 40876 of 2017) of the National Environmental Management: Biodiversity Act (10/2004). Furthermore, these islands are a special nature reserve and a marine protected area (Chown & Froneman 2008; DEA 2013). The management plan restricts visitor access to specific areas, including all of Prince Edward Island itself and parts of Marion Island (Prince Edward Islands Management Plan 2010). No specific interventions are required at present, but the population should continue to be monitored.
Recommendations for land managers and practitioners:
Continuation of the monitoring and research programme conducted over the past two decades to date by the Marion Island Marine Mammal Programme under the auspices of the South African Polar Research Infrastructure (SAPRI) in association with the Mammal Research Institute, University of Pretoria. Aspects of this work feeds into an international monitoring programme run by the Commission for the Conservation of Antarctic Marine Living Resources (CCAMLR): the CCAMLR Ecosystem Monitoring Programme (CEMP).
Research priorities:
- The effects of global climate change on the foraging and breeding behaviour of this species.
- The effects of global climate change on the prey species of the Antarctic Fur Seal.
- The effects of local fisheries on prey populations.
- Characteristics and causes of population changes.
- Monitor the occurrence and impacts of H5N1 and develop contingency management plans.
Encouraged citizen actions:
Due to the isolation of Antarctic Fur Seal habitat, citizen actions are limited. However, citizens can report potential sightings on virtual museum platforms (for example, iNaturalist and MammalMAP).
Bibliography
Agnew, D.J. 1997. The CCAMLR ecosystem monitoring programme. Antarctic Science, 9(3), pp.235-242.
Alava, J.J., Tirapé, A., Denkinger, J., Calle, P., Rosero R, P., Salazar, S., Fair, P.A. and Raverty, S. 2024. Endangered Galápagos sea lions and fur seals under the siege of lethal avian flu: a cautionary note on emerging infectious viruses in endemic pinnipeds of the Galápagos Islands. Frontiers in Veterinary Science, 11, p.1457035.
Arnould, J.P.Y. and Croxall, J.P. 1995. Trends in entanglement of Antarctic fur seals (Arctocephalus gazella) in man-made debris at South Georgia. Marine Pollution Bulletin 30: 707-712.
Arthur, B., Hindell, M., Bester, M., De Bruyn, P.N., Goebel, M.E., Trathan, P. and Lea, M.A. 2018. Managing for change: using vertebrate at sea habitat use to direct management efforts. Ecological Indicators, 91, pp.338-349.
Banyard, A.C., Bennison, A., Byrne, A.M., Reid, S.M., Lynton-Jenkins, J.G., Mollett, B., De Silva, D., Peers-Dent, J., Finlayson, K., Hall, R. and Blockley, F. 2024. Detection and spread of high pathogenicity avian influenza virus H5N1 in the Antarctic Region. Nature Communications, 15(1), p.7433.
Berta, A. and Churchill, M. 2012. Pinniped taxonomy: review of currently recognized species and subspecies, and evidence used for their description. Mammal Review 42: 207-234.
Bester, M.N. 1989. Movements of southern elephant seals and subantarctic fur seals in relation to Marion Island. Marine Mammal Science, 5(3), pp.257-265.
Bester MN, Bartlett PA. 1990. Attendance behaviour of Antarctic and subantarctic fur seal females at Marion Island. Antarctic Science 2: 309–312.
Bester MN, Reisinger RR. 2010. Vagrant Antarctic fur seals at Gough Island in 2009. Polar Biology 33: 709–711.
Bester, M. N., Ryan, P. G. and Dyer, B. M. 2003. Population numbers of fur seals at Prince Edward Island, Southern Ocean. African Journal of Marine Science 25: 549-554.
Bester, M.N., Ryan, P.G., Bester, W.A. and Glass, T. 2014. Vagrant Antarctic fur seals at the Tristan da Cunha Islands. Polar Biology 37: 1701-1703.
Bonin, C.A., Goebel, M.E., Forcada, J., Burton, R.S. and Hoffman, J.I. 2013. Unexpected genetic differentiation between recently recolonized populations of a long-lived and highly vagile marine mammal. Ecology and Evolution 3: 3701-3712.
Bonner, W. N. 1968. The fur seal of South Georgia. British Antarctic Survey Report 56: 1-95.
Bonner, W. N. and McCann, T. S. 1982. Neck collars on fur seals, Arctocephalus gazella, at South Georgia. British Antarctic Survey Report 57: 73-77.
Boveng, P. L., Hiruki, L. M., Schwartz, M. K. and Bengston, J. L. 1998. Population growth of Antarctic fur seals: limitation by a top predator, the leopard seal? Ecology 79: 2863-2877.
Boyd, I.L., McCafferty, D.J., Reid, K., Taylor, R. and Walker, T.R. 1998. Dispersal of male and female Antarctic fur seals (Arctocephalus gazella). Canadian Journal of Fisheries and Aquatic Sciences, 55(4), pp.845-852.
Boyd IL, Murray AWA. 2001. Monitoring a marine ecosystem using responses of upper trophic level predators. Journal of Animal Ecology 70: 747–760.
Boyd, I.L. 2014. The climate change bogie – a comment on Forcada and Hoffman. Available at: https://ianlboyd.wordpress.com. (Accessed: 5 November 2014).
Campagna, C., Uhart, M., Falabella, V., Campagna, J., Zavattieri, V., Vanstreels, R.E. and Lewis, M.N. 2024. Catastrophic mortality of southern elephant seals caused by H5N1 avian influenza. Marine Mammal Science, 40(1).
Charbonnier Y, Delord K, Thiebot JP. 2010. King size fast food for Antarctic fur seals. Polar Biology 33: 721–724.
Chown SL, Froneman PW. 2008. The Prince Edward Islands in a global context. In: Chown SL, Froneman PW. (ed.), The Prince Edward Islands. Land-sea Interactions in a Changing Ecosystem., pp. 1–16. Sun Media, Stellenbosch, South Africa.
Clapham, P.J., Young, S.B. and Brownell Jr., R.L. 1999. Baleen whales: conservation issues and the status of the most endangered populations. Mammal Review 29: 35–60.
Croxall, J. P., Rodwell, S. and Boyd, I. L. 1990. Entanglement in man-made debris of Antarctic fur seals at Bird Island, South Georgia. Marine Mammal Science 6: 221-223.
de Bruyn PJN, Pistorius PA, Tosh CA, Bester MN. 2007. Leucistic Antarctic for seal Arctocephalus gazella at Marion Island. Polar Biology 30:1355–1358.
de Lima, R.C., Cebuhar, J.D., Negrete, J., Ferreira, A., Secchi, E.R. and Botta, S. 2022. Ecosystem shifts inferred from long-term stable isotope analysis of male Antarctic fur seal Arctocephalus gazella teeth. Marine Ecology Progress Series, 695, pp.203-216.
Department of Environmental Affairs. 2013. Prince Edward Islands declared a Marine Protected Area.
Drehmer, C. J. and de Oliveira, L. R. 2000. Syncranial osteology of Arctocephalus gazella (Pinnipedia, Otariidae) from Rio Grande do Sul, Brazil. Iheringia. Serie Zoologia 88: 51–59.
Duvenage, E. 2024. Avian flu suspected on South Africa’s remote Marion Island. Nat. Afr, 10.
Forcada, J. and Hoffman, J.I. 2014. Climate change selects for heterozygosity in a declining fur seal population. Nature 511: 462-465.
Forcada, J., Hoffman, J.I., Gimenez, O., Staniland, I.J., Bucktrout, P. and Wood, A.G. 2023. Ninety years of change, from commercial extinction to recovery, range expansion and decline for Antarctic fur seals at South Georgia. Global Change Biology, 29(24), pp.6867-6887.
Friscourt, N., Lea, M.A., Cherel, Y., Wotherspoon, S., Brewer, E.A., Oosthuizen, W.C., de Bruyn, P.N., Wege, M., Goebel, M.E., Trathan, P.N. and Walters, A. 2024. Seasonal and ocean basin-scale assessment of amino acid δ15N trends in a Southern Ocean marine predator. Marine Ecology Progress Series, 747, pp.151-169.
Goebel, M.E., Krause, D., Freeman, S., Burner, R., Bonin, C., Vasquez del Mercado, R., Van Cise, A.M. and Gafney, J. 2008. Pinniped research at Cape Shirreff, Livingston Island, Antarctica, 2008/09.
Goldsworthy, S. D. 1999. Maternal attendance behaviour of sympatrically breeding Antarctic and subantarctic fur seals, Arctocephalus spp., at Macquarie Island. Polar Biology 21: 316-325.
Goldsworthy, S. D., He, X., Lewis, M., Williams, R., and Tuck, G. 2001. Trophic interactions between Patagonian toothfish, its fishery and seals and seabirds around Macquarie Island. Marine Ecology Progress Series 218: 283-302.
Green, K, Burton, H. R. and Williams, R. 1989. The diet of Antarctic fur seals Arctocephalus gazella (Peters) during the breeding season at Heard Island. Antarctic Science 1(3): 17-324.
Green, K., Williams, R. and Burton, H. R. 1991. The diet of Antarctic fur seals during the late autumn and early winter around Heard Island. Antarctic Science 3: 359-362.
Guinet, C., Jouventin, P. and. Georges, J.-Y. 1994. Long term population changes of fur seals Arctocephalus gazella and Arctocephalus tropicalis on subantarctic (Crozet) and subtropical (St. Paul and Amsterdam) Islands and their possible relationship to El Nino Southern Oscillation. Antarctic Science 6(4): 473-478.
Guinet, C., Lea, M.A. and Goldsworthy, S.D. 2000. Mass change in Antarctic fur seal (Arctocephalus gazella) pups in relation to maternal characteristics at the Kerguelen Islands. Canadian Journal of Zoology, 78(3), pp.476-483.
Haddad, W.A., Reisinger, R.R., Scott, T., Bester, M.N. and de Bruyn, P.N. 2015. Multiple occurrences of king penguin (Aptenodytes patagonicus) sexual harassment by Antarctic fur seals (Arctocephalus gazella). Polar Biology, 38(5), pp.741-746.
Hanchet, S., Horn, P. and Stevenson, M. 2003. Fishing in the ice: is it sustainable? Water & Atmosphere 11: 24–25.
Hofmeyr, G. J. G., De Maine, M., Bester, M. N., Kirkman, S. P., Pistorius, P. A. and Makhado, A. B. 2002. Entanglement of pinnipeds at Marion Island, Southern Ocean, 1991-2001. Australian Mammalogy 24: 141-146.
Hofmeyr, G.J.G. and Bester M.N. 1993. Predation on king penguins by Antarctic fur seals. South African Journal of Antarctic Research 23(1- 2): 71-74.
Hofmeyr, G.J.G. and Bester M.N. 2008. Subantarctic Islands. In: M. Lück (ed.), Encyclopaedia of Tourism and Recreation in Marine Environments, pp. 456-457. CABI, Wallingford, UK.
Hofmeyr, G.J.G., Bester, M.N., Kirkman, S.P., Lydersen, C. and Kovacs, K.M. 2010. Intraspecific differences in the diet of Antarctic fur seals at Nyrøysa, Bouvetøya. Polar Biology 33: 1171-1178.
Hofmeyr, G.J.G., Bester, M.N., Makhado, A.B. and Pistorius, P.A. 2006. Population changes in Subantarctic and Antarctic fur seals at Marion Island. Polar Biology 17: 150-158.
Hofmeyr, G.J.G., Krafft, B.A., Kirkman, S.P., Bester, M.N., Lydersen, C. and Kovacs, K.M. 2005. Population changes of Antarctic fur seals at Nyrøysa, Bouvetøya. Polar Biology 28: 725-731.
Holdgate, M.W. 1963. Observations in the South Sandwich Islands, 1962. Polar Record, 11(73), pp.394-405.
Humble, E., Dasmahapatra, K.K., Martinez-Barrio, A., Gregório, I., Forcada, J., Polikeit, A.C., Goldsworthy, S.D., Goebel, M.E., Kalinowski, J., Wolf, J.B. and Hoffman, J.I. 2018. RAD sequencing and a hybrid Antarctic fur seal genome assembly reveal rapidly decaying linkage disequilibrium, global population structure and evidence for inbreeding. G3: Genes, Genomes, Genetics, 8(8), pp.2709-2722.
Jepsen, E.M. and de Bruyn, P.N. 2019. Pinniped entanglement in oceanic plastic pollution: a global review. Marine Pollution Bulletin, 145, pp.295-305.
Johnstone, G.W. 1982. Zoology. Pp. 33-39. In: Expedition to the Australian territory of Heard and McDonald Islands 1980. Compiled by C. Veenstra and J. Manning. Division of National Mapping, Technical Report 31.
Kerley, G.I.H. 1983. Comparison of seasonal haulout patterns of fur seals Arctocephalus tropicalis and A. gazella on Subantarctic Marion Island. South African Journal of Wildlife Research 13: 71-77.
Kerley, G.I.H. 1983. Record for the Cape fur seal Arctocephalus pusillus pusillus from subantarctic Marion Island. S. Afr. J. Zool. 18:139-140.
Kerley, G.I.H. 1987. Arctocephalus tropicalis on the Prince Edward Islands. In: J.P. Croxall and R.L. Gentry (eds), Status, Biology and Ecology of Fur Seals; Proceedings of an International Symposium and Workshop, Cambridge, England, 23-27 April 1984. NOAA Technical Report NMFS. 51:61-64.
Kingston, J.J. and Gwilliam, J. 2007. Hybridization between two sympatrically breeding species of fur seal at Iles Crozet revealed by genetic analysis. Conservation Genetics 8: 1133-1145.
Kirkman, S.P., Bester, M.N., Makhado, A.B. and Pistorius, P.A. 2003. Female attendance patterns of Antarctic fur seals at Marion Island. African Zoology 38: 402-405.
Kirkman, S.P., Wilson, W., Klages, N.T.W., Bester, M.N. and Isaksen, K. 2000. Diet and estimated food consumption of Antarctic fur seals at Bouvetøya during summer. Polar Biology 23: 745-752.
Klages, N. T. W. and Bester, M. N. 1998. Fish prey of fur seals Arctocephalus spp. at subantarctic Marion Island. Marine biology 131: 559-566.
Kovacs, K.M., Aguilar, A., Aurioles, D., Burkanov, V., Campagna, C., Gales, N.J., Gelatt, T., Goldsworthy, S.D., Goodman, S.J., Hofmeyr, G.J.G., Härkönen, T., Lowry, L., Lydersen, L., Schipper, J., Sipilä, T., Southwell, C., Thompson, D. and Trillmich, F. 2012. Global threats to pinnipeds. Marine Mammal Science 28: 414-436.
Lancaster, M. L., Goldsworthy, S. D., and Sunnucks, P. 2010. Two behavioural traits promote fine-scale species segregation and moderate hybridisation in a recovering sympatric fur seal population. BMC Evolutionary Biology 2010 10: 143.
Lavigne D.M. and Schmitz, O.J. 1990. Global warming and increasing population densities: a prescription for seal plagues. Marine Pollution Bulletin 21: 280-284.
Lea MA, Guinet C, Cherel Y, Duhamel G, Dubroca L, Pruvost P, Hindell M. 2006. Impacts of climatic anomalies on provisioning strategies of a Southern Ocean predator. Marine Ecology Progress Series 310: 77–94.
Learmonth, J.A., Macleod, C.D., Santos, M.B., Pierce, G.J., Crick, H.Q.P. and Robinson, R.A. 2006. Potential effects of climate change on marine mammals. Oceanography and Marine Biology: An Annual Review 44: 431-464.
Maboko, V.J. 2009. Genetic diversity and hybridisation estimates of Arctocephalus tropicalis and A. gazella from Marion Island. University of Pretoria (South Africa).
Makhado, A.B., Bester, M.N., Kirkman, S.P., Pistorius, P.A., Ferguson, J.W.H. and Klages, N.T.W. 2008. Prey of the Antarctic fur seal Arctocephalus gazella at Marion Island. Polar Biology 31: 575-581.
McDonald, B.I., Goebel, M.E., Crocker, D.E. and Costa, D.P. 2012. Biological and environmental drivers of energy allocation in a dependent mammal, the Antarctic fur seal pup. Physiological and Biochemical Zoology 85: 134-147.
North, A. W., Croxall, J. P. and Doidge, D. W. 1983. Fish prey of the Antarctic fur seal Arctocephalus gazella at South Georgia. Bulletin of the British Antarctic Survey 61: 27-38.
Nyakatura, K. and Bininda-Emonds, O.R.P. 2012. Updating the evolutionary history of Carnivora (Mammalia): a new species-level supertree complete with divergence time estimates. BMC Biology 10: 12.
Page, B., Welling, A., Chambellant, M., Goldsworthy, S.D., Dorr, T. and van Veen, R. 2003. Population status and breeding season chronology of Heard Island fur seals. Polar Biology, 26(4), pp.219-224.
Paijmans, A.J., Stoffel, M.A., Bester, M.N., Cleary, A.C., De Bruyn, P.N., Forcada, J., Goebel, M.E., Goldsworthy, S.D., Guinet, C., Lydersen, C. and Kovacs, K.M. 2020. The genetic legacy of extreme exploitation in a polar vertebrate. Scientific Reports, 10(1), p.5089.
Payne, M. R. 1977. Growth of a fur seal population. 279: 67-79.
Reid K, Sims M, White RW, Gillon KW. 2004. Spatial distribution of predator/prey interactions in the Scotia Sea: implications for measuring predator/fisheries overlap. Deep Sea Research Part II: Topical Studies in Oceanography 51: 1383–1396.
Reisinger RR, Gröcke DR, Lübcker N, McClymont EL, Hoelzel AR, de Bruyn PN. 2016. Variation in the diet of killer whales Orcinus orca at Marion Island, Southern Ocean. Marine Ecology Progress Series 549: 263–274.
Reisinger, R.R., Landman, M., Mgibantaka, N., Smale, M.J., Bester, M.N., De Bruyn, P.N. and Pistorius, P.A. 2018. Overlap and temporal variation in the diets of sympatric Antarctic and Subantarctic fur seals (Arctocephalus spp.) at Marion Island, Prince Edward Islands. Polar Research, 37(1), p.1451142.
Reisinger, R.R., Corney, S., Raymond, B., Lombard, A.T., Bester, M.N., Crawford, R.J., Davies, D., de Bruyn, P.N., Dilley, B.J., Kirkman, S.P. and Makhado, A.B. 2022. Habitat model forecasts suggest potential redistribution of marine predators in the southern Indian Ocean. Diversity and Distributions, 28(1), pp.142-159.
Repenning, C.A., Peterson, R.S. and Hubbs, C.L. 1971. Contributions to the systematics of the southern fur seals, with particular reference to the Juan Fernández and Guadalupe species. In: W.H. Burt (ed.), Antarctic Pinnipedia, pp. 1-34. Antarctic Research Series 18, American Geophysical Union, New York, USA.
Rice, D.W. 1998. Marine mammals of the world. Systematics and distribution. The Society for Marine Mammalogy Special Publication Number 4. 231 pp.
Robinson SA, Goldsworthy SG, van den Hoff J, Hindell MA. 2003. The foraging ecology of two sympatric fur seal species, Arctocephalus gazella and Arctocephalus tropicalis, at Macquarie Island during the austral summer. Marine and Freshwater Research 53: 1071–1082.
SCAR_EGS. 2014. Scientific Committee for Antarctic Research – Expert Group on Seals Report.
Shaughnessy, P. D. and Burton, H. R. 1986. Fur seals Arctocephalus spp. At Mawson Station, Antarctica, and in the Southern Ocean. Polar Records 23: 79-81.
Shaughnessy, P.D., Kemper, C.M., Stemmer, D. and McKenzie, J. 2014. Records of vagrant fur seals (family Otariidae) in South Australia. Australian Mammalogy, 36(2), pp.154-168.
Shirihai, H. 2002. A complete guide to Antarctic wildlife. Alula Press.
Siniff, D.B., Garrott, R.A., Rotella, J.J., Fraser, W.R. and Ainley, D.G. 2008. Opinion: Projecting the effects of environmental change on Antarctic seals. Antarctic Science 20: 425-435.
Staniland, IJ, Robinson, SL. 2008. Segregation between the sexes: Antarctic fur seals, Arctocephalus gazella, foraging at South Georgia. Animal Behaviour 75: 1581–1590.
Staniland, I.J., Robinson, S.L., Silk, J.R.D., Warren, N. and Trathan, P.N. 2012. Winter distribution and haul-out behaviour of female Antarctic fur seals from South Georgia. Marine Biology, 159(2), pp.291-301.
Van Helden, L. 2024. Rabies in the Western Cape: Seals, Dogs and Jackals. South Africa.
Wege M, Etienne M-P, Chris Oosthuizen W, Reisinger RR, Bester MN, Bruyn PJ. 2016. Trend changes in sympatric Subantarctic and Antarctic fur seal pup populations at Marion Island, Southern Ocean. Marine Mammal Science 32: 960–982.
Wege M, Postma M, Tosh CA, de Bruyn PJN, Bester MN. 2015. First confirmed record of a leucistic Antarctic fur seal pup born outside the Scotia Arc Islands. Polar Biology 38: 569–571.
Wickens, P. and York, A.E. 1997. Comparative population dynamics of fur seals. Marine Mammal Science 13(2): 241-292.
Wilson, J. W., Burle, M.-H. and Bester, M. N. 2006. Vagrant Antarctic pinnipeds at Gough Island. Polar Biology 29: 905–908.
Wynen, L. P., Goldsworthy, S. D., Guinet, C., Bester, M. N., Boyd, I. L., Gjertz, I., Hofmeyr, G. J. G., White, R. W. G. and Slade, R. W. 2000. Post sealing genetic variation and population structure of two species of fur seals (Arctocephalus gazella and A. tropicalis). Molecular Ecology 9: 299-314.