
Zulu Pipistrelle Bat
Neoromicia zuluensis

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Neoromicia zuluensis – (Roberts, 1924)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – VESPERTILIONIDAE – Neoromicia – zuluensis
Common Names: Zulu Serotine, Zulu Pipistrelle Bat, Aloe Bat, Aloe Serotine, Aloe Serotine Bat (English), Kaapse dakvlermuis (Afrikaans), Pipistrelle zouloue, Sérotine des aloes (French)
Synonyms: Eptesicus zuluensis Roberts, 1924; Pipistrellus zuluensis (Roberts, 1924)
Taxonomic Note:
This species is closely related to Neoromicia somalica (Thomas 1901), and although previous authors have considered these species conspecific (Meester et al. 1986), interspecific chromosomal data recommended that zuluensis is specifically distinct from somalica (Rautenbach et al. 1993). More recent molecular DNA studies using mitochondrial genes have confirmed this distinction (Monadjem et al. 2020). Furthermore, Taylor et al. (2022) found deep mitochondrial sequence divergence between N. zuluensis from southern Africa and N. cf. zuluensis from East Africa; however further integrative taxonomic studies are required within these groups across Africa before formally describing cryptic lineages within these species.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Balona, J.1, Richards L.R.2 & da Silva, J.M.3
Reviewer: Richardson, E.4
Institutions: 1Gauteng and Northern Regions Bat Interest Group, 2Durban Natural Science Museum, 3South African National Biodiversity Institute, 4Independent Consultant at Richardson & Peplow Environmental
Previous Assessors and Reviewers: Monadjem, A., Jacobs, D., Cohen, L., MacEwan, K., Richards, L.R., Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Relton, C., Raimondo, D. & Child, M.F.
Assessment Rationale
Listed as Least Concern in view of its distribution outside of the assessment region (estimated extent of occurrence within the assessment region is 253,648 km²) and because there are no major identified threats that could cause widespread population decline. It occurs in many protected areas across its range and appears to have a degree of tolerance for human modified habitats. More research is needed into the roosting behaviour of this species to identify key roost sites and monitor population trends.
Regional population effects: Its range is believed to be continuous with Zimbabwe and Mozambique through transfrontier parks, and thus dispersal is assumed to be occurring. However, it has relatively low wing loading (Norberg & Rayner 1987; Schoeman & Jacobs 2008), so significant rescue effects are uncertain.
Red List Index
Red List Index: No change
Recommended citation: Balona J, Richards LR & da Silva JM. 2025. A conservation assessment of Neoromicia zuluensis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species is widespread in East and southern Africa but limited to the north-eastern part of the assessment region. However, there is some evidence to suggest that individuals from southern Africa (Angola, Eswatini and presumably South Africa) are distinct from East African populations (Taylor et al. 2022). The eastern distribution ranges from Ethiopia and South Sudan to Uganda and Kenya (Happold et al. 2013). The southern range extends from Zambia and the southern parts of the Democratic Republic of the Congo to eastern South Africa, and from eastern Angola to central Zambia, Zimbabwe, northern Botswana and northeastern Namibia (Monadjem et al. 2020). In southern Africa, it is recorded from scattered, and sometimes seemingly isolated, localities in Namibia, Botswana, Zimbabwe and South Africa (Happold et al. 2013). In the assessment region, the species is recorded from Limpopo, Mpumalanga, North West and KwaZulu-Natal provinces of South Africa, as well as in Eswatini. The type specimen is from Umfolozi Game Reserve, KwaZulu-Natal (Monadjem et al. 2020). Estimated extent of occurrence within the assessment region is 253,648 km².
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 17 m asl (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 1,666 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
Map
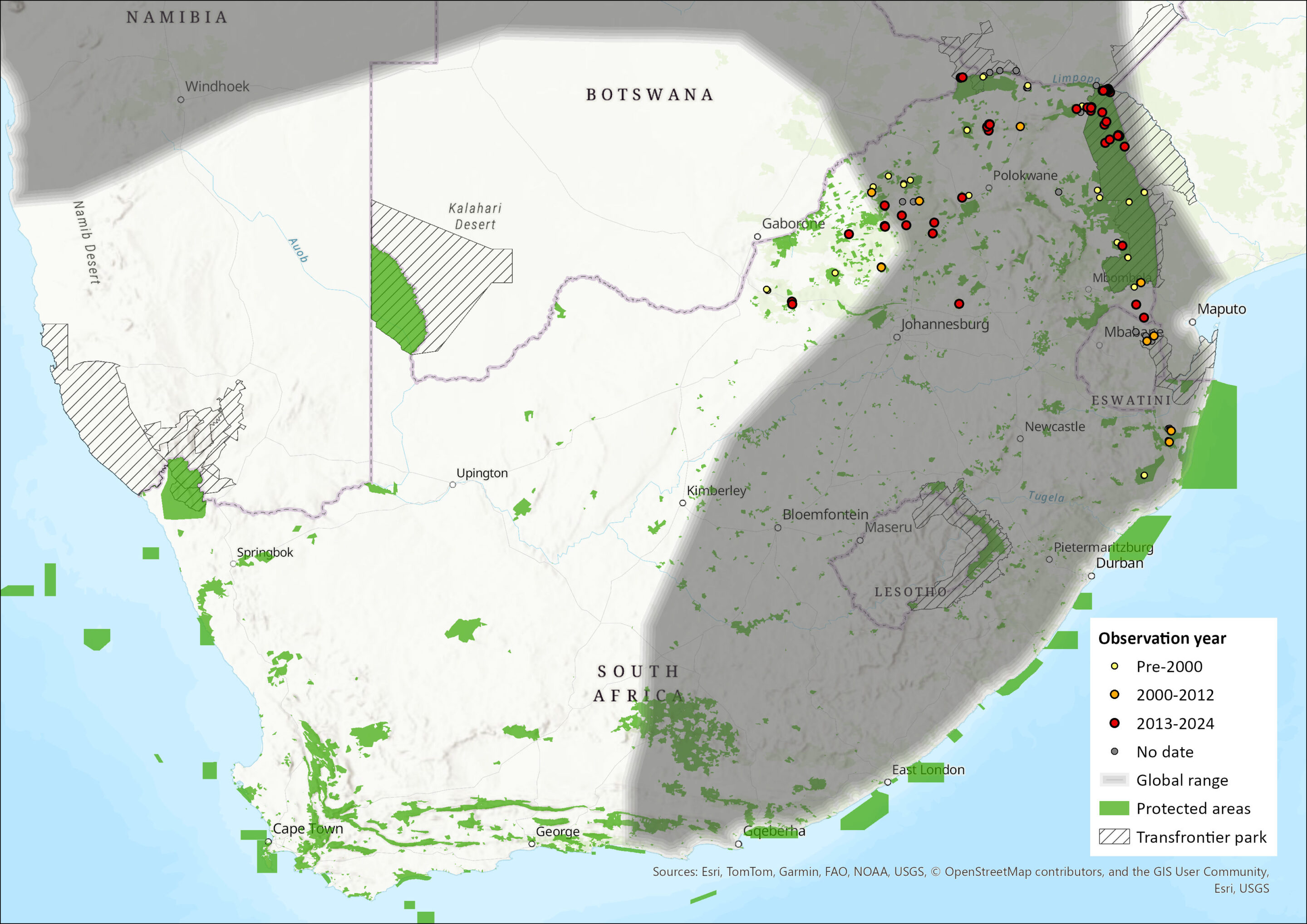 Figure 1. Distribution records for Zulu Pipistrelle Bat (Neoromicia zuluensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Zulu Pipistrelle Bat (Neoromicia zuluensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Eswatini | Extant | Native | – | Resident |
| Ethiopia | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
No formal studies have investigated the potential effects of climate change on the species. Individuals from the Namib Desert have a high urine concentrating ability, reportedly the highest know for any bat species at the time (Happold and Happold 1988). This suggests that the species may be more efficient in conserving water than others, although it is likely to still depend on wild sources of drinking water to counter evaporative water loss (Happold and Happold, 2013). The species has been netted near water sources (T. Kearney pers. obs.; L.R. Richards pers. obs) and it is suggested that individuals forage over water (Fenton and Bogdanowicz 2002).
In general, climate change has been identified as an increasing global threat to bat species as it amplifies water scarcity issues and negatively affects primary plant production and prey availability (Sherwin et al. 2013) and may similarly impact N. zuluensis. More research is needed to determine the likely impacts to the species.
Population Information
Species abundance within the assessment region is uncertain (Happold et al. 2013), however it is generally reported as an uncommon species (ACR 2024). It appears that species is more common elsewhere in its range, having been reported as a frequently recorded species from Angola during recent survey efforts (Taylor et al. Weier et al. 2020). It is not as common as Laephotis capensis in southern Africa (Taylor 2000). It is, however, well represented in museums with over 160 individuals assessed in Monadjem et al. (2020).
Current population trend: Stable
Continuing decline in mature individuals? Cannot be determined
Extreme fluctuations in the number of subpopulations: Uncertain
Continuing decline in number of subpopulations: Uncertain
All individuals in one subpopulation: Recent genetic studies show some divergence between southern African and East African populations (see population genetics).
Number of mature individuals in largest subpopulation: Uncertain
Number of Subpopulations: 1 within the assessment region
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Cannot be determined
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Cannot be determined
Probability of extinction in the wild within 100 years: Cannot be determined
Population Genetics
A phylogenetic study revealed possible cryptic diversity within N. zuluensis with deep mitochondrial sequence divergence separating N. zuluensis from southern Africa from individuals from East Africa; however further integrative taxonomic studies are required within these groups across Africa before formally describing cryptic lineages within these species (Taylor et al. 2022).
No population genetic studies have been conducted on the species, but it is presumed to be capable of dispersing to adjacent colonies and therefore likely exists as a metapopulation within the assessment region. The effective population size cannot be estimated due to limited knowledge of population size; however, it is presumed to be fairly large, likely connected to colonies in adjacent countries, and hence likely exceeds the Ne 500 threshold.
Habitats and ecology
Populations of this bat have been recorded from dry and moist savannah, savannah woodland (including miombo woodland), and into more arid shrublands (Happold et al. 2013), where surface water sources or riparian corridors are available (Monadjem et al. 2020). In the assessment region, it is recorded from the Mopane Bioregion, Lowveld, Highveldand Central Bushveld (Seamark & Kearney 2008; Kearney et al. 2019; Taylor et al. 2013; de Jong et al. 2025). Roosting sites of this species are not well known (Monadjem et al. 2020). They are insectivorous: in the Kruger National Park, their diet consisted mostly of Coleoptera and a lesser proportion of Lepidoptera (Aldridge & Rautenbach 1987). Seamark and Bogdanowicz (2002) reported that their faeces may contain vegetative matter. Morphometric data and field studies reveal that this species forages over water, as well as highly cluttered vegetation, tree canopies, open spaces between lower branches and tree-trunks, as well as in clearings around buildings (Fenton & Bogdanowicz 2002; Happold et al. 2013). Although little information is available for the reproductive ecology of this species, females are known to give birth at the end of November and the beginning of December (Skinner & Chimimba 2005).
Ecosystem and cultural services: As this species is insectivorous, it may contribute to controlling insect populations that damage crops (Boyles et al. 2011; Kunz et al. 2011). Ensuring a healthy population of insectivorous bats can thus decrease the need for pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Marginal | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 8.1. Desert -> Desert – Hot | – | Marginal | – |
Life History
Generation Length: Unknown
Age at Maturity: Female or unspecified: Uncertain
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Female: Mean forearm length = 3.02 ± 0.14 cm; mean total length = 8.00 ± 0.36 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Mean forearm length = 3.00 ± 0.083 cm; mean total length = 7.46 ± 0.39 cm (Monadjem et al. 2020)
Longevity: Unknown
Average Reproductive Age: Unknown
Maximum Size (in cms): Unknown
Size at Birth (in cms): Unknown
Gestation Time: Unknown
Reproductive Periodicity: Reportedly exhibits seasonal monoestry with births coinciding with the rainy summer months (Happold et al. 2013)
Average Annual Fecundity or Litter Size: Two pups per litter (Happold et al. 2013)
Natural Mortality: No available information.
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Unknown
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence to suggest that this species is traded or harvested within the assessment region.
Local Livelihood: None reported
National Commercial Value: Likely to contribute to ecosystem services, in the form of pest insect population control where it occurs in agroecosystems.
International Commercial Value: Likely to assist with insect population control in respect of exported produce.
End Use: None reported
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: N/A
Threats
There appears to be no immediate major threats to this species (ACR 2024). However, disturbance to key roost sites has been recognised as a common threat to other bat species in the assessment region; but further information into the roosting behaviour of N. zuluensis is required in order to understand whether disturbance is a serious threat. Further research should be directed towards land transformation and overexploitation of woodland habitats in the eastern areas of the assessment region, and the possible ill effects on the species.
Conservation
In the assessment region, the species is recorded from many protected areas, including Kruger National Park, Baobab Tree Reserve, Mapungubwe National Park, Blouberg Nature Reserve (Vhembe Biosphere Reserve), Percy Fyfe Nature Reserve, Borakalalo National Park, Pilanesberg National Park and Hluhluwe-iMfolozi Park. It seems probable that this species is present within a number of additional protected areas. No direct conservation measures are currently needed for the species, but research into its habitats and ecology is recommended.
Recommendations for land managers and practitioners:
- Reduce pesticide use in agricultural landscapes.
- Conservation of key habitats (e.g. woodlands and water sources)
Research priorities:
- Enhanced integrative taxonomic studies, inclusive of South African specimen material, to ascertain the taxonomic status of southern and East African genetic lineages.
- Increased sampling throughout the predicted species range in the assessment region (see Monadjem et al. 2010)
- Identifying roost site selection and basic ecology.
- Determining population size, trends and potential threats.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Report any grounded bats to your nearest bat interest group and lodge specimens with a natural science collection institute.
Bibliography
ACR. 2024. African Chiroptera Report. 2024. AfricanBats NPC, Pretoria. i – xv; 1 – 1397.
Adams, R.A. and Hayes, M.A. 2021. The importance of water availability to bats: climate warming and increasing global aridity. In 50 years of bat research: foundations and new frontiers (pp. 105-120). Cham: Springer International Publishing.
Aldridge, H.D.J.N. and Rautenbach, I.L. 1987. Morphology, echolocation and resource partitioning in insectivorous bats. The Journal of Animal Ecology 56: 763–778.
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
De Jong, G., Melville, H., and Richards, L. 2024. The Insectivorous Bat Assemblage in Telperion Nature Reserve, Mpumalanga, South Africa: Species Inventory and Call Library. Acta Chiropterologica (2): 315–327,
Driver, A., Sink, K.J., Nel, J.N., Holness, S., van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An Assessment of South Africa’s Biodiversity and Ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Fenton, M.B. and Bogdanowicz, W. 2002. Relationships between external morphology and foraging behaviour: bats in the genus Myotis. Canadian Journal of Zoology 80: 1004–1013.
Happold, D.C.D. and Happold, M.J.J.Z., 1988. Renal form and function in relation to the ecology of bats (Chiroptera) from Malawi, Central Africa. Journal of Zoology, 215(4), pp.629-655.
Happold, M., van Cakenberghe, V., Kearney, T. 2013. Pipistrellus zuluensis Zulu Pipistrelle (Aloe Bat). Pages 657–659 in Happold M, Happold DCD, editors. Mammals of Africa. Volume IV: Hedgehogs, Shrews and Bats. Bloomsbury Publishing, London, UK.
Kearney, T.C., Keith, M., Markotter, W., Pretorius, M. and Seamark, E.C.J. 2019. Bat species (Mammalia: Chiro –
ptera) occurring at Telperion Nature Reserve. Annals of the Ditsong National Museum of Natural History, 8: 30–42.
Kearney, T.C., Volleth, M., Contrafatto, G., Taylor, P.J. 2002. Systematic implications of chromosome GTG-band and bacula morphology for southern African Eptesicus and Pipistrellus and several other species of Vespertilioninae (Chiroptera: Vespertilionidae). Acta Chiropterologica 4: 55–76.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T., Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of southern African mammals. Transvaal Museum Monographs 5: 1–359.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2010. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. University of Witwatersrand Press, Johannesburg.Monadjem, A., Demos, T., Dalton, D., Webala, P., Musila, S., Kerbis Peterhans, J. and Patterson, B. 2021: 25. A revision of Pipistrelle-like bats (Mammalia: Chiroptera: Vespertilionidae) in East Africa with the description of new genera and species, Zoological Journal of the Linnean Society.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2010. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis, Second Edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Norberg, U.M. and Rayner, J.M. 1987. Ecological morphology and flight in bats (Mammalia; Chiroptera): wing adaptations, flight performance, foraging strategy and echolocation. Philosophical Transactions of the Royal Society B: Biological Sciences 316: 335–427.
Rautenbach, I.L., Bronner, G.N. and Schlitter, D.A. 1993. Karyotypic data and attendant sympatric implications for the bats of southern Africa. Koedoe 36: 87–104.
Schoeman, M.C. and Jacobs, D.S. 2008. The relative influence of competition and prey defenses on the phenotypic structure of insectivorous bat ensembles in southern Africa. PLoS One 3: e3715.
Seamark, E.C.J. and Bogdanowicz, W. 2002. Feeding ecology of the common slit-faced bat (Nycteris thebaica) in KwaZulu-Natal, South Africa. Acta Chiropterologica 4: 49–54.
Seamark, E.J. and Kearney, T.C. 2008. Bats of Messina Nature Reserve, Limpopo Province, South Africa. African Bat Conservation News 18: 5-7.
Sherwin, H.A., Montgomery, W.I., and Lundy, M.G. 2013. The impact and implications of climate change for bats. Mammal Review 43: 171–182.
Simmons, N.B. 2005. Order Chiroptera . Johns Hopkins University Press, Baltimore, USA.
Skinner, J.D. and Chimimba, C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Taylor, P.J. 2000. Bats of Southern Africa: Guide to Biology, Identification, and Conservation. University of Natal Press, Pietermaritzburg, South Africa.
Taylor, P.J., Schoeman, M.C. and Monadjem, A. 2013. Diversity of bats in the Soutpansberg and Blouberg Mountains of northern South Africa: complementarity of acoustic and non-acoustic survey methods. South African Journal of Wildlife Research, 43(1): 12-26.
Taylor, P.J., Neef, G., Keith, M., Weier, S., Monadjem, A. and Parker, D.M. 2018. Tapping into technology and the biodiversity informatics revolution: updated terrestrial mammal list of Angola, with new records from the Okavango Basin. Zookeys 10, 51–88.
Weier, S.M., Keith, M., Neef, G.G., Parker, D.M. and Taylor, P.J. 2020. Bat species richness and community composition along a mega-transect in the Okavango River Basin. Diversity, 12(5), p.188.
