Tree Rat
Thallomys paedulcus

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Thallomys paedulcus – (Sundevall, 1846)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Thallomys – paedulcus
Common Names: Tree Rat, Acacia Rat (English), Boomrot (Afrikaans)
Synonyms: No Synonyms
Taxonomic Note: Thallomys paedulcus possibly represents a complex of several similar species. This species has been controversial and there have been many changes in its definition (Perrin 2013). Further studies are needed to clarify the taxonomic status of populations currently allocated to this species, and the relationships between this species and others of its genus (Monadjem et al. 2015). As T. nigricauda was included in T. paedulcus until recently, some information for the latter may actually refer to the former (Perrin 2013).
Red List Status: LC – Least Concern
Assessment Information
Assessor: Taylor, P.1 & da Silva, J.M.2
Reviewer: Smith, C.3
Institutions:1University of the Free State,2South African National Biodiversity Institute,3Endangered Wildlife Trust
Previous Assessors and Reviewers: Perrin, M., Child, M.F. & Mondajem, A.
Previous Contributors: Relton, C., Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
Listed as Least Concern in view of its wide distribution within the assessment region, and its occurrence in numerous protected areas, including Kruger National Park. However, this species may become threatened in the near future as encroaching human settlements and industries degrade the woodlands needed by this species. Selective harvesting of tall trees for firewood and charcoal production, as well as wholesale destruction of woodlands by mining and agricultural expansion, threaten to cause population decline. For example, it is estimated that, between 1990 and 2006, 20% of woodland cover was lost to pine and Eucalyptus plantations and residential expansion in the Soutpansberg, Limpopo Province. Assuming the rate of loss is linear, 1.25% of woodland is lost per year in the region, which, if extrapolated across the province, could lead to a 12.5% decline in woodland cover over the next ten years and a suspected population decline. Similarly, models of fuelwood extraction from the Bushbuckridge region in Mpumalanga Province indicate that biomass in the area will be exhausted within 13 years at current rates of extraction. However, the relationship between woodland loss and population density is unknown, and more research is needed to estimate potential population decline. Remote sensing techniques (for example, Landsat imagery or LiDAR) should be used to assess tall tree loss at finer spatial scales and field surveys should be used to ground-truth the suspected impact on this species’ population. These data can then be used to estimate both the area of occupancy, and the rate of population decline for this species. A reassessment will be needed when such data are available, as we suspect that this species may qualify for a threatened category. This species is a dietary specialist with a complex gastric anatomy containing microbial symbionts, which may negatively affect the colonisation of new woodland sites, particularly those containing different species composition. Thus, significant rescue effects are doubtful. Key interventions for this species include the conservation of tall tree structure through protected area expansion, biodiversity stewardship and community engagement, and active rehabilitation of degraded sites.
Regional population effects: Possible through contiguous habitat or patches linked by corridors and dispersal across Botswana, Zimbabwe and Mozambique.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Taylor P & da Silva JM. 2025. A conservation assessment of Thallomys paedulcus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
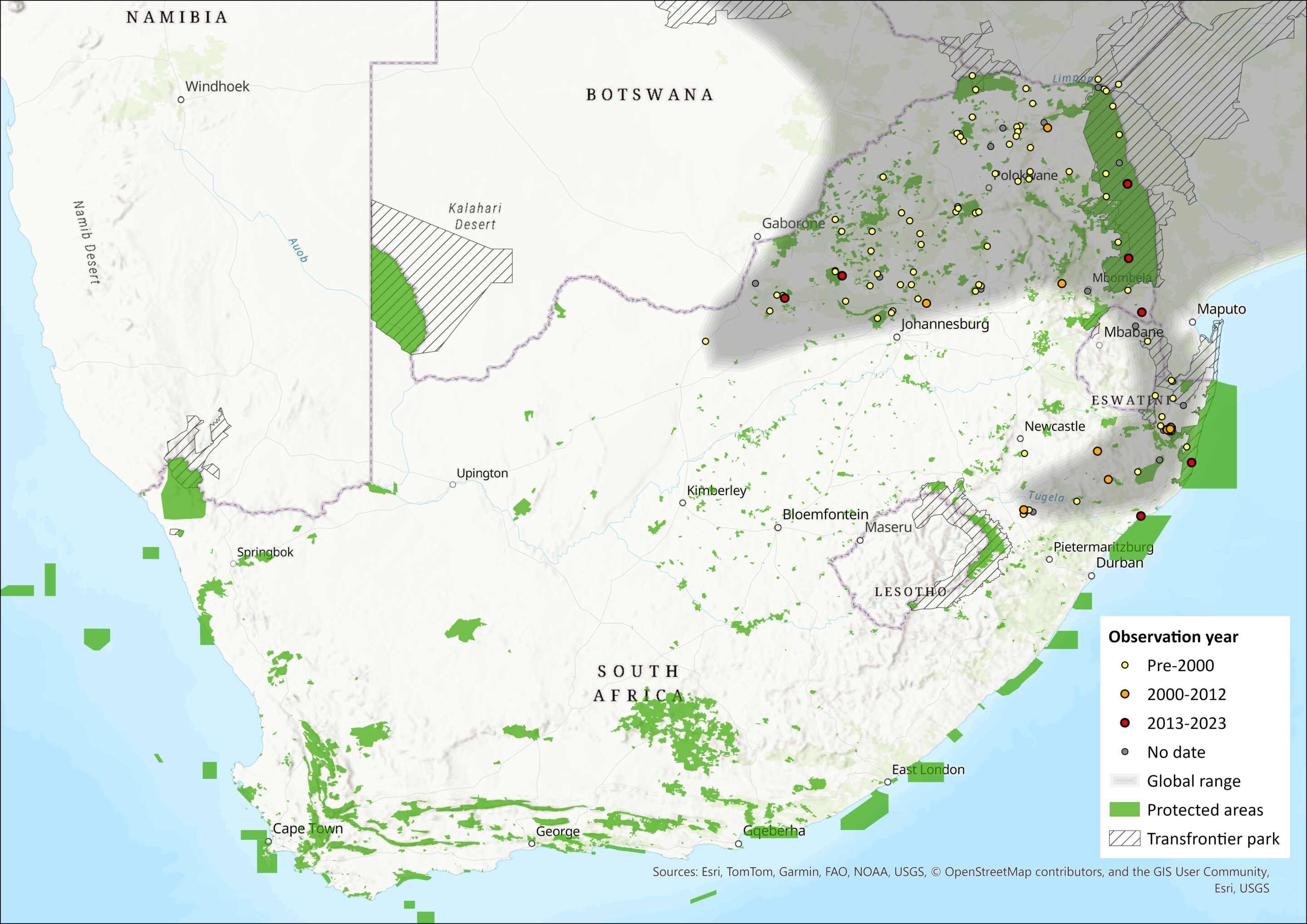
This species ranges widely from southern Ethiopia and southern Somalia in the north, through East Africa, to southern parts of the Democratic Republic of the Congo, Angola and much of Zambia. From here, it ranges south to northeastern South Africa (northern KwaZulu-Natal, Mpumalanga, Limpopo, Gauteng and North West provinces) and Eswatini. The western limit of its distribution within the assessment region is unclear and may be more or less extensive than shown in Figure 1 (see Perrin 2013; Monadjem et al. 2015). For example, Power (2014) captured this species in the northeastern bushveld of the North West Province, where it often associates with human habitation, but there were no captures in the western district in the Kalahari, where one could expect them to be and all grass nests examined belonged to the Namaqua Rock Mouse Micaelamys namaquensis. Thallomys paedulcus and T. nigricauda are believed to be parapatric, possibly overlapping partially in a narrow contact zone (Taylor et al. 1995). Further vetting of museum records is required to delimit the respective distributions of T. nigricauda and T. paedulcus.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Tree Rat (Thallomys paedulcus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
As with T. nigricauda, the impact of climate change on Acacia savanna woodland is unknown but denudation of woodlands and especially large trees with cavities for fuelwood extraction would have a much larger impact on populations. In situ measurements of body temperature in T. paedulcus tree rats from Weenen Nature Reserve in KwaZulu-Natal showed physiological responses to a highly variable thermal environment, e.g. the amplitude of their diurnal cycle was 259.7% of that expected (Coleman & Downs, 2010). Individuals forage nocturnally and are able to lower their body temperature whilst resting in cavities during the day. Whilst cavities provide moderate thermal buffering effect to allow behavioural thermoregulation, more extreme high temperatures in the future may minimise this energy saving and could potentially place animals under high physiological stress. At Weene Nature Reserve individuals were observed living in cavities in the wall space of a building.
Population Information
It can be locally common in suitable habitats, but populations are generally small and isolated (Perrin 2013). Although southern Africa is considered a stronghold for this species, it is uncommon. It is difficult to catch, and researchers should consider putting traps in trees. For example, P.J. Taylor (unpubl. data) has not captured any individuals in the Soutpansberg Mountains, Limpopo Province, across a gradient of land-use types despite similar overall rodent densities. Worryingly, this corresponds to general woodland loss in the region as estimated by Landsat imagery (Munyati & Kabanda 2009).
Population Information
Current population trend: Unknown
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
No population genetic study has been conducted of this species although such a study has been conducted in the related T. nigricauda (Meyer et al. 2009). Given connectivity with neighbouring countries and within the assessment region is possible due to contiguous habitat or patches linked by corridors enabling dispersal, the species is considered a single metapopulation. However, limited gene flow due to fragmentation of habitat leading to isolation, may be affecting their ability to successfully disperse; hence the genetic structure within the species should be verified. Based on the metapopulation scenario, the Convention on Biological Diversity’s complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (1/1 population remains).
While the species is locally common in some areas, the lack of population size information prevents any inference of the population’s effective population size. Hence, the GBF’s headline indicator – proportion of species with an effective population size (Ne) greater than 500 – cannot be estimated.
Habitats and ecology
This is an arboreal species associated with established woodlands, particularly Acacia bushland. It prefers woodland trees and does not inhabit shrublands or ecotones. Although T. nigricauda may be confined to arid savannahs, aside from some degree of distributional overlap, T. paedulcus generally inhabits broad-leaved and Acacia savannahs with an average annual precipitation of approximately 500 mm (Lovegrove 1997). They nest in cavities in the stems of large trees (Dean et al. 1999). In East Africa, they are mainly associated with Acacia xanthophloa and A. tortilis (Perrin 2013).
This species is a dietary specialist with a complex gastric anatomy containing microbial symbionts, which may negatively affect the colonisation of new woodland sites, particularly those having a different species composition (Perrin 1986). In most respects the behaviour, feeding ecology and reproductive biology of the Tree Rat is similar to that of its sibling species, the Black-tailed Tree Rat. They are both nocturnal and forage mainly in the canopies of Acacia trees (Perrin 2013), but small families of Tree Rats are thought to be less selective in their choice of nesting tree and may nest in any tree offering a suitable fork or hollow (Lovegrove 1997). Additional research is required to reliably validate disparities in the ecology and morphological characteristics of these sister species (Skinner & Chimimba 2005).
Ecosystem and cultural services: No specific ecosystem or cultural services have been identified for this species, however, it is expected that, similar to other arboreal Thallomys species, this species may represent a valuable prey species for opportunistic predators such as tree roosting owls (e.g. the Spotted Eagle Owl, Bubo africanus; Reed 2005), small carnivores and snakes.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
The major threat to this species is woodland loss and degradation (Driver et al. 2012), especially of large mature trees that are potential cavity nesting sites (sensu Dean et al. 1999). Ongoing illegal commercial harvesting of tall trees for firewood or charcoal production is causing a decline in the habitat quality for this species, which is a noted problem in the North West Province (DACE 2008). Fuelwood extraction and plantation forestry has led to a 20% decrease in woodland cover in the Soutpansberg, Limpopo from 1996–2006 (Munyati & Kabanda 2009). In Limpopo Province specifically, urban expansion and agriculture, particularly overgrazing in ranchlands, are key drivers of woodland loss (Munyati & Kabanda 2009). Encroaching human settlements also put pressure on woodlands for supplementary firewood and charcoal production. For example, in the Bushbuckridge region of Mpumalanga, airborne light detection and ranging (LiDAR) modelling indicates that biomass will be exhausted within 13 years given unsustainable fuelwood extraction (Wessels et al. 2013).
Conservation
It is present within a number of protected areas, including Kruger National Park. Although no interventions are necessary at present, several interventions could be trialled to prevent this species from becoming threatened in the future. For example, protected area expansion to protect and connect woodlands. This can be done formally or informally through conservancy formation and biodiversity stewardship schemes. Wildlife ranches and conservancies may be helping to conserve habitat for this species by protecting tall trees from charcoal harvesting. This should be encouraged. Harvest management for fuelwood extraction should also be considered. For example, Wessels et al. (2013), in their study area in Mpumalanga, suggested that a 15% annual reduction in consumption for eight years is required to reach sustainable fuelwood extraction levels.
Recommendations for land managers and practitioners:
- Subpopulations should be monitored to record any changes in abundance and distribution. However, it should be considered that live-trapping in trees off the ground can be time-consuming, and population densities are likely to fluctuate.
- Land managers should be encouraged to conserve woodlands and old trees.
Research priorities:
- Rate of woodland loss across the species’ range and its effect on population trend. This can be achieved through the combined use of Landsat imagery and field surveys.
- Urgent molecular analysis and taxonomic resolution of the species complex.
- Analysis of museum records to more accurately delimit distribution.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas. However, this species cannot be readily distinguished from T. nigricauda based on morphological characteristics.
- Landowners can preserve Acacia trees, both living and dead.
Bibliography
Anderson MD, Anderson TA. 2001. Too much, too quickly? Doubts about the sustainability of the camelthorn wood harvest. African Wildlife 55: 21–23.
DACE. 2008. North West Province: Environment Outlook. North West Department of Agriculture, Conservation & Environment, Mmabatho, South Africa.
Dean WRJ, Milton SJ, Jeltsch F. 1999. Large trees, fertile islands, and birds in arid savanna. Journal of Arid Environments 41: 61–78.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Lovegrove BG. 1997. Tree Rat Thallomys paedulcus. In: Mills G, Hes L (ed.), The Complete Book of Southern African Mammals, pp. 147. Struik Publishers, Cape Town, South Africa.
Munyati C, Kabanda TA. 2009. Using multitemporal Landsat TM imagery to establish land use pressure induced trends in forest and woodland cover in sections of the Soutpansberg Mountains of Venda region, Limpopo Province, South Africa. Regional Environmental Change 9: 41–56.
Perrin MR. 1986. Gastric anatomy and histology of an arboreal, folivorous, murid rodent; the black-tailed tree rat, Thallomys paedulcus (Sundevall, 1846). Zeitschrift für Säugetierkunde 51: 224–236.
Perrin MR. 2013. Thallomys paedulcus Sundevall’s Acacia Rat (Acacia Thallomys). In: Happold DCD (ed.), Mammals of Africa. Volume III: Rodents, Hares and Rabbits, pp. 561–562. Bloomsbury Publishing, London, UK.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Reed DN. 2005. Taphonomic implications of roosting behavior and trophic habits in two species of African owl. Journal of Archaeological Science 32: 1669–1676.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor, P. J., Rautenbach, I. L., Gordon, D., Sink, K. and Lotter, P. 1995. Diagnostic morphometrics and southern African distribution of two sibling species of tree rat, Thallomys paedulcus and Thallomys nigricauda (Rodentia: Muridae). Durban Museum Novitates 20: 49-62.
von Staden L. and Raimondo D. 2015. Vachellia erioloba (E. Mey.) P.J.H. Hurter. National Assessment: Red List of South African Plants. South African National Biodiversity Institute, South Africa.