Suricate
Suricata suricatta

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Suricata suricatta – (Schreber, 1776)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – HERPESTIDAE – Suricata – suricatta
Common Names: Suricate, Meerkat, Slender-tailed Meerkat (English), Stokstertmeerkat, Graatjie-meerkat (Afrikaans), Xarab, !Naixarab (Damara, Nama), Todi, Letoto (Sesotho), Kôtôkwê, Kôtôkô, Lekôtôkô (Tswana), Scharrtier (German), Xarab, !Naixarab (Damara, Nama)
Synonyms: Viverra suricatta Schreber, 1776
Taxonomic Note:
Meester et al. (1986) recognised two subspecies: S. s. suricatta and S. s. marjoriae Bradfield 1936. The nominate subspecies occupies the whole distributional range, excluding the Namib Desert and pro-Namib north of Swakopmund, where S. s. marjoriae occurs; the latter might be replaced by S. s. iona Cabral 1971 in Angola (Skinner & Chimimba 2005).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Adams, E.C.1 & da Silva, J.M.2
Reviewer: Do Linh San, E.3
Institutions: 1Endangered Wildlife Trust, 2South African National Biodiversity Institute, 3Sol Plaatje University
Previous Assessors a& Reviewers: Jordan, N.R., O’Riain, J., Martins, V., Page-Nicholson, S. & Do Linh San, E.
Previous Contributor: Madikiza, Z.
Assessment Rationale
The Suricate is listed as Least Concern as it is relatively widespread in the assessment region, is present in several protected areas (notably in the Kgalagadi Transfrontier Park), and there are currently no major threats to the species. It occurs in habitats and regions that are largely intact and unlikely to be extensively transformed. Climate change may adversely affect this species if rainfall in semi-arid areas becomes more unpredictable and the annual average is reduced. The predicted extension of the current semi-arid region of southern Africa in an easterly direction may result in an easterly shift of its current distribution.
Regional population effects: The species’ range is assumed to be continuous within southern Africa and hence it is likely that there is dispersal across regions. However, there is a lack of research on the actual range and whether there are breaks in the distribution that may result from unsuitable edaphic factors, mountains or large perennial rivers (e.g. the Orange River) and/or the absence of other species that may be important for creating suitable microhabitats – for example Cape Ground Squirrels (Xerus inauris), whose burrows Suricates use as sleeping and denning sites.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Adams EC & da Silva JM. 2025. A conservation assessment of Suricata suricatta. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
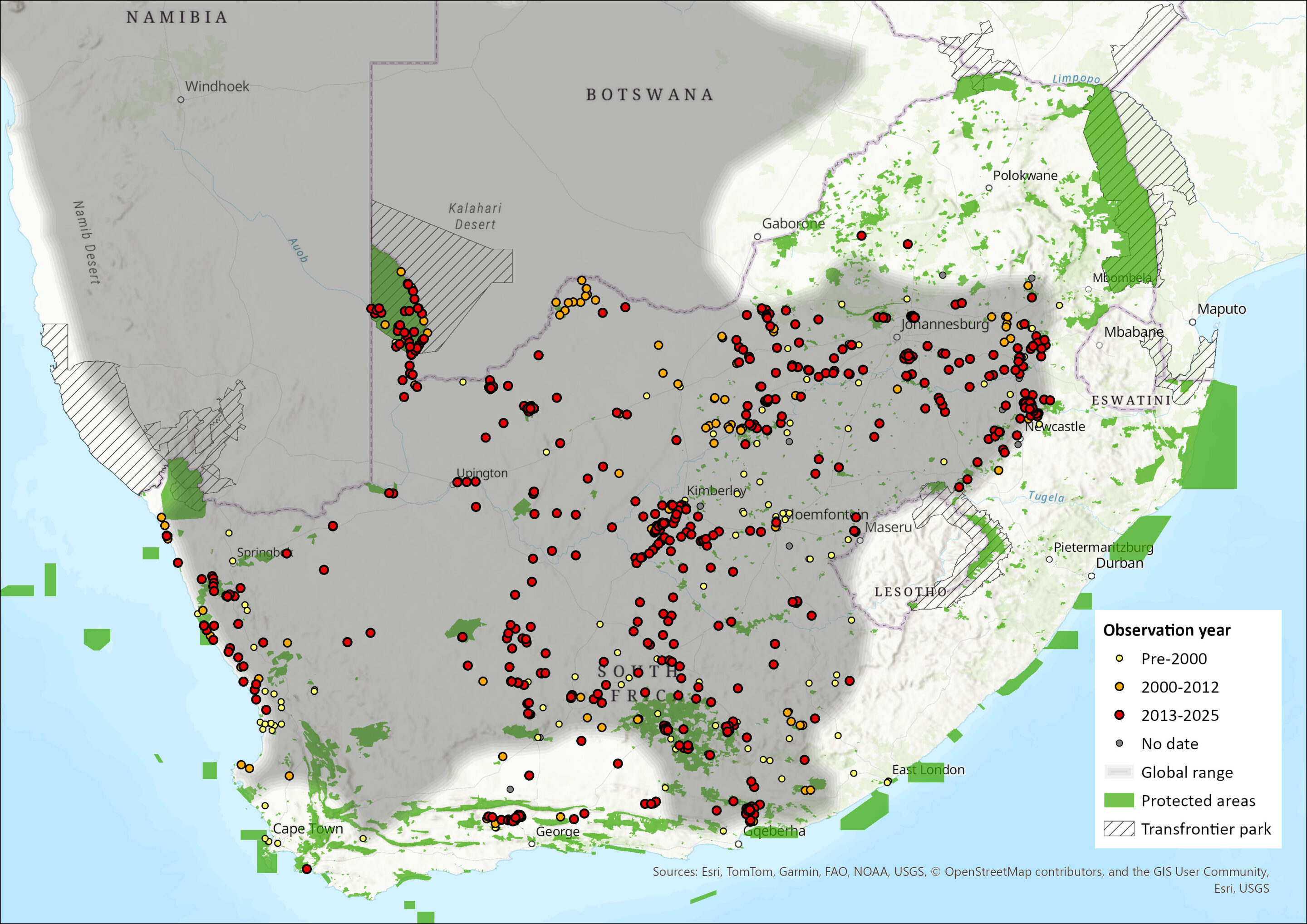
This species is widespread in the western parts of southern Africa, including western and southern Namibia, southwestern Botswana, and north and west South Africa, with a very marginal intrusion into extreme southwestern Angola (Macdonald 2013). Lynch (1994) suggested that this species may occur in the lowlands of Lesotho, but there have been no records so far (N.L. Avenant pers. comm. 2013).
Within the assessment region, the species occurs in the arid western and southern areas, highland grasslands, and Eastern Cape (Figure 1). Suricates are restricted to the southern parts of the North West Province, Gauteng and Mpumalanga (Skinner & Chimimba 2005). However, they do not extend eastwards as far as the Eswatini border. Suricates are widespread in both the Free State and Northern Cape provinces. As the species favours arid, open country, it occurs widely through the Nama-Karoo and Succulent Karoo biomes. Within the Western Cape, it occurs in the Fynbos Biome and extends eastwards into the Savanna Biome in areas where annual rainfall exceeds 600 mm (Skinner & Chimimba 2005). While Suricates occur in the Western and Eastern Cape provinces, they are absent along the coastline, from Cape Town to Port Elizabeth; this absence does extend for some distance inland.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Suricate (Suricata suricatta) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Lesotho | Presence Uncertain | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
A study by Thorley et al. (2025) examined the effects of climate change on Kalahari Suricates over a period of 25 years. During the study period they recorded a monthly temperature increases of 1.5 to 3.2 °C as well as fluctuating rainfall without a discernible trend. The main determinant of female fecundity and the recruitment of pups was rainfall, as this was seen to affect the group size and population density. This effect was most evident during droughts, which saw a profound decline in the population density and a local extinction of some groups (Thorley et al. 2025). Paniw et al. (2022) also noted the correlation between increasing air temperatures and the prevalence of tuberculosis outbreaks within wild Kalahari Suricate groups, as a result this is expected to affect survival and reproduction, and in turn more than double overall group extinction risk. Vital rates (such as survival, growth, reproduction, stage transitions and emigration) were also found to be positively correlated with high rainfall in the Kalahari (Paniw et al. 2019; Thorley et al. 2025). As Suricate body mass is also linked to climate fluctuations and could have an effect on vital rates in each season, this could either serve as a buffer against extinction or facilitate it (Paniw et al. 2019). Van de Ven et al. (2019) also noted that pup body mass gains were lower under hotter temperatures due to the increased cost of thermoregulation. It is also noted that if droughts become more prevalent (as is suggested by the predictive models), the Suricates in the Kalahari could be at risk of extinction (Thorley et al. 2025). The effects of climate change may be lessened by co-operative breeding and alloparental care as displayed by Suricates (Groenewoud & Clutton-Brock 2020).
Population
Suricate densities can fluctuate greatly across the range and are influenced by rainfall and predation (Doolan & Madonald 1997; Clutton-Brock et al. 1999b). In the South African section of the Kgalagadi Transfrontier Park, densities declined from 1 individual / km2 in April 1994 to 0.32 individual / km2 in May 1995 following a reduction in rainfall (Clutton-Brock et al. 1999b). Generation length is estimated to be six years (Jordan & Do Linh San 2015).
However, Suricates are generally considered to be relatively common across their known range and may have been favoured by the elimination of many of their natural predators (e.g. raptors and mesopredators) by small livestock farmers. Although the population is currently considered to be stable, climate change may cause population decline in the future because Suricates’ reproduction is significantly affected by rainfall (Clutton-Brock et al. 1999a; Russell et al. 2002).
Population Information
Current population trend: Unknown, but probably stable based on wide extent of occurrence and lack of major threats.
Continuing decline in mature individuals: Unknown, but unlikely
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: It is not currently possible to determine the extent or number of subpopulations.
Severely fragmented: No. Suricates have a relatively broad habitat tolerance and can exist in agricultural and rural landscapes; in addition, their favoured habitats are largely connected across their range.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
The complete mitochondrial genome of the Suricate has been sequenced, as well as its phylogeny within the Herpestidae family (Derežanin et al. 2020). While no population genetic study has yet been undertaken, with its wide range and dispersal capabilities, the species is currently thought to exist as a single population (or metapopulation) within the assessment region and connecting with neighbouring countries. Based on this information and no knowledge of subpopulations going extinct, the Convention on Biological Diversity’s GBF’s complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (all populations remaining).
While precise population sizes (e.g. census [Nc]) or effective population size (Ne) data does not exist, the knowledge that this species is relatively common across its range and may have increased in parts due to the elimination of some of their natural predators, suggests that there may be at least 10,000 individuals within the assessment region. Even though the 10,000 value is not an actual census count, this quantity can still be used as a proxy for quantifying the GBF’‘s headline genetic indicator – proportion of populations with an Ne > 500. Based on the assumption that the species is likely to occur as a single metapopulation, the Ne of the species is ~1,000 –3,000 individuals, based on a Ne/Nc conversion ratio of 0.1-0.3. Having the 1 population above Ne 500, translates to an indicator score of 1.0 (1/1 population with Ne > 500).
Further population genetic analyses are needed to understand the genetic structure and health of this species, and to verify the indicator values estimated broadly here.
Habitats and ecology
The Suricate is an inhabitant of arid, open country, characterised by short grasses and sparse woody growth. It is absent from true desert and forested areas, and also from mountainous terrain (Macdonald 2013). Suricates in the Kalahari preferentially select for the areas where clay-sand and sand dunes meet (Bateman et al. 2015). It is carnivorous, feeding mostly on invertebrates. The diet of the species consists predominantly of insects (mainly Coleoptera adults and larvae), supplemented with arachnids, myriapods (e.g. centipedes, Scolopendra morsitans, and millipedes), reptiles (e.g. lizards), birds and some plant material (Skinner & Chimimba 2005). Small mammals may also be consumed locally and seasonally, especially in winter when arthropod abundance and consumption drops. Suricates locate potential prey by means of olfactory cues (Leclaire 2017).
Suricates are diurnal obligate cooperative breeders that live in social groups of 2–50 individuals (Clutton-Brock et al. 2008; Photo 1). Each group usually has a dominant male and a dominant female, who produce the vast majority (75%) of the group’s surviving young (Clutton-Brock et al. 2001; Manser 2001; Russell et al. 2002). Subordinates assist in raising the offspring of dominants by babysitting (Clutton-Brock et al. 2000) and provisioning pups (Manser & Avey 2000) including by allonursing (MacLeod et al. 2013). Helpers do not seem to specialise in the kind of help they provide (Clutton-Brock et al. 2003). Infanticide occurs, with dominant females killing the young of subordinate females (O’Riain et al. 2000; Clutton-Brock et al. 1998; Young & Clutton-Brock 2006; Young et al. 2008). However, reproductive control tends to be concentrated during the dominant’s pregnancy, when subordinate females are forcibly evicted from their social group (O’Riain et al. 2000; Clutton-Brock et al. 1998). Litters of three to five pups emerge on average, and are born throughout the year except July, with a peak between March and September (Clutton-Brock et al. 1999a) after a 60 day gestation (Clutton-Brock et al. 1999a). Breeding frequency is related to rainfall (Clutton-Brock et al. 1999a). Although there is no sexual dimorphism, individuals adjust their growth to the size of their closest same-sex competitor (Huchard et al. 2016), and this competitive growth appears to drive adaptive size-modification in dominant females (Russell et al. 2004).
Suricates tend to move around within their range, which is approximately 2 km2 (Jordan et al. 2007). Territorial residence may be communicated through a series of latrines and scent marking that are distributed non-randomly throughout the range (Jordan et al. 2007; Bateman et al. 2015). While the Suricate group may utilise as many as 10 burrow systems, only two or three burrows are generally used to sleep and raise young in (Skinner & Chimimba 2005). Suricates also maintain over 1,000 bolt holes throughout their territory, appearing to remember the locations of these sites and using the closest one to escape approaching predators (Manser & Bell 2004). Burrows at the centre of their territory are used more often than those along the periphery of their territory, providing enhanced safety and access to resources (Strandburg-Peshkin et al. 2020).
Ecosystem and cultural services: The Suricate is a flagship species for the Kalahari in South Africa. In addition, Suricates consume many invertebrates which may be considered as pest species. Lastly, at least two tourism operations have opened, offering “walking with wild Suricates” experiences for commercial and educational returns.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 8.1. Desert -> Desert – Hot | – | Suitable | – |
Life History
Generation Length: Estimated to be 6 years
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 60 days
Reproductive Periodicity: Throughout the year with a peak in March to September
Average Annual Fecundity or Litter Size: 3-5 pups per litter
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Suricates are used as research subjects for understanding the costs and benefits of social behaviour in mammals and they are charismatic animals for ecotourism businesses. Suricates are also sold as live animals for the entertainment industry or as pets. They can be habituated and are often used in wildlife documentaries. Trade in live animals is not expected to negatively impact on the population, but there are welfare issues with keeping the species as a pet. It is also important to mention that Suricates do not adapt well to traditional pet care standards of average people. In addition, they generally tend to urinate (scent-mark) in multiple locations in the house, may chew wires, carpets and even furniture, and can be aggressive towards visitors (O.A.E. Rasa pers. comm. 2016) and new pets.
Wildlife ranching may increase the habitat available for the species, but no study has demonstrated this yet.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Suricates are not currently facing any major threats. There is no evidence so far that infection with tuberculosis (probably Mycobacterium bovis), which may be common in this species (Drewe 2010), has led to any direct persecution in farming areas, or that a present small trade in Suricates as pets has affected wild populations.
Climate change and habitat transformation may pose long-term threats to Suricate populations across their distribution. Increasing aridity may adversely affect reproduction (see Clutton-Brock et al. 1999a) and recruitment and may therefore cause local extinctions in already arid areas. No information is currently available to suitably evaluate the possible impacts of land transformation for agriculture on Suricates. Changing the habitat and hence the food resources that are reliant on natural habitat could prove negative for this species. On the other hand, since there are fewer or no large predators on most farmlands (both due to persecution and lack of large, natural prey resulting from habitat change), Suricates can potentially thrive. If mesopredator release occurs, however, this could lead to increased predation by the medium-sized carnivores and/or increased competition for food with other small carnivore species. The conversion of land for crops through irrigation projects in arid areas is likely to have the most direct short-term effects as these activities may eliminate many of the Suricates natural prey in addition to displacing important ecosystem engineer species such as Cape Porcupine (Hystrix africaeaustralis), Common Warthog (Phacochoerus africanus), Aardvark (Orycteropus afer) and especially Cape Ground Squirrels that may benefit Suricates through the provision of burrow systems and bolt holes, respectively.
Conservation
Suricates are present in several large and well-managed protected areas, including the Kgalagadi Transfrontier Park, Tswalu Game Reserve, Mokala National Park, and Addo Elephant National Park which are respectively located at the northern extreme, centre and southern border of their current distribution within the assessment region. There are no specific conservation interventions geared towards their conservation nationally, but local organisations that engage in both ecotourism (e.g. Klein Karoo) and research (e.g. Kuruman River Reserve) are important for educating the public on the ecological role and biological significance of this species. Awareness and education campaigns might help reducing the pet trade, but paradoxically could also – along with media attention – exacerbate this threat.
Recommendations for land managers and practitioners:
- Monitor and regulate their use in the pet trade.
- Educate the public on the pitfalls of keeping Suricates as pets and encourage people to report the trade of meerkats for the pet industry.
- Monitor sites for potential population declines from climate change.
Research priorities:
This is a well-studied African small carnivore (see review in Macdonald 2013) and no particular knowledge is urgently needed in view of managing or conserving the species. However, the majority of behavioural ecology research is derived from studies carried out in the Kalahari Desert. The following research topics will therefore assist in improving our knowledge on this species and gathering information that may be relevant for conservation in the future:
- Study Suricate ecology and behaviour in other biomes (e.g. Karoo, Highveld Grassland or Fynbos).
- Survey this species distribution and status in the Great Karoo and the central and northern parts of the Eastern Cape.
- Assess the potential impacts that climate change will have on the population.
- Assess the drivers and extent of the pet trade.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas. As confusion with Banded Mongoose (Mungos mungo) and other mongoose species is possible, a photograph is required for confirmation of identification, especially when sightings are made in areas where the distribution ranges of these two mongoose species overlap.
Bibliography
Bateman, A.W., Lewis, M.A., Gall, G., Manser, M.B. and Clutton‐Brock, T.H. 2015. Territoriality and home‐range dynamics in meerkats, Suricata suricatta: a mechanistic modelling approach. Journal of Animal Ecology, 84(1), 260-271.
Clutton Brock, T.H., Maccoll, A.D.C., Manser, M., Brotherton, P.N.M., Gaynor, D., Mcilrath, G.M., Kansky, R., Chadwick, P. and Skinner, J.D. 1999. Predation, group size and mortality in a cooperative mongoose, Suricata suricatta. Journal of Animal Ecology 68: 672-683.
Clutton-Brock, T.H., Brotherton, P.N.M., O’Riain, J.M., Griffin, A.S., Gaynor, D., Kansky, R., Sharpe, L., McIllrath, G.M. 2001. Contributions to cooperative rearing in meerkats. Animal Behaviour 61: 705–710.
Clutton-Brock, T.H., Brotherton, P.N.M., O’Riain, J.M., Griffin, A.S., Gaynor, D., Sharpe, L., Kansky, R., Manser, M. and McIlrath, G.M. 2000. Individual contributions to babysitting in a cooperative mongoose, Suricata suricatta. Proceedings of the Royal Society of London 267(Series B): 301–305.
Clutton-Brock TH, Brotherton PNM, Smith R, McIlrath GM, Kansky R., Gaynor D, O’Riain JM, Skinner JD. 1998. Infanticide and expulsion of females in a cooperative mammal. Proceedings of the Royal Society of London 265(Series B): 2291–2295.
Clutton-Brock TH, Hodge SJ, Flower TP. 2008. Group size and subordinate reproduction in Kalahari meerkats. Animal Behaviour 76: 689–700.
Clutton-Brock TH, Maccoll A, Chadwick P, Gaynor D, Kansky R, Skinner JD. 1999a. Reproduction and survival of suricates (Suricata suricatta) in the southern Kalahari. African Journal of Ecology 37: 69–80.
Clutton-Brock TH, Russell AF, Sharpe LL. 2003. Meerkat helpers do not specialise in particular activities. Animal Behaviour 66: 531–540.
Derežanin, L., Fickel, J. and Förster, D. 2020. The complete mitochondrial genome of the meerkat (Suricata suricatta) and its phylogenetic relationship with other feliform species. Mitochondrial DNA Part B, 5(1), 1100-1101.
Doolan SP, Macdonald DW. 1997. Breeding and juvenile survival among slender-tailed meerkats (Suricata suricatta) in the south-western Kalahari: ecological and social influences. Journal of Zoology 242: 309–327.
Drewe, J.A. 2010. Who infects whom? Social networks and tuberculosis transmission in wild meerkats. Proceedings of the Royal Society B: Biological Sciences 277: 633-642.
Groenewoud, F. and Clutton‐Brock, T. 2021. Meerkat helpers buffer the detrimental effects of adverse environmental conditions on fecundity, growth and survival. Journal of Animal Ecology, 90(3), 641-652.
Huchard E, English S, Bell MBV, Thavarajah N, Clutton-Brock TH. 2016. Competitive growth in a cooperatively breeding mammal. Nature 533: 532–534.
Jordan NR, Cherry MI, Manser MB. 2007. Latrine distribution and patterns of use by wild meerkats: implications for territory and mate defence. Animal Behaviour 73: 613-622.
Jordan NR, Do Linh San E. 2015. Suricata suricatta. The IUCN Red List of Threatened Species 2015.
Leclaire, S. 2017. Recognition of prey odor in wild meerkats. Chemoecology, 27(2), 85-90.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
MacLeod KJ, Nielsen JF, Clutton-Brock TH. 2013. Factors predicting the frequency, likelihood and duration of allonursing in the cooperatively breeding meerkat. Animal Behaviour 86: 1059–1067.
Macdonald, D.W. 2013. Suricata suricatta Meerkat (Suricate). In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. V. Carnivores, Pangolins, Equids and Rhinoceroses, pp. 347-352. Bloomsbury, London, UK.
Manser M. 2001. The acoustic structure of suricates’ alarm call varies with predator type and the level of response urgency. Proceedings of the Royal Society of London 268 (Series B): 2315–2324.
Manser MB, Avey G. 2000. The effect of pup vocalisations on food allocation in a cooperative mammal, the meerkat (Suricata suricatta). Behavioural Ecology and Sociobiology 48: 429-437.
Manser MB, Bell MB. 2004. Spatial representation of shelter locations in meerkats (Suricata suricatta). Animal Behaviour 68: 151–157.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum, Pretoria, South Africa.
O’Riain MJ, Bennett NC, Brotherton PNM, McIlrath G, Clutton-Brock TH. 2000. Reproductive suppression and inbreeding avoidance in wild population of cooperatively breeding meerkats (Suricata suricatta). Behavioural Ecology and Sociobiology 48: 471-477.
Paniw, M, Maag, N, Cozzi, G, Clutton-Brock, T and Ozgul, A. 2019. Life history responses of meerkats to seasonal changes in extreme environments. Science, 363(6427), 631-635.
Paniw, M, Duncan, C, Groenewoud, F, Drewe, JA, Manser, M, Ozgul, A and Clutton-Brock, T. 2022. Higher temperature extremes exacerbate negative disease effects in a social mammal. Nature Climate Change, 12(3), 284-290.
Russell AF, Carlson AA, McIlrath GM, Jordan NR, Clutton-Brock TH. 2004. Adaptive size modification by dominant female meerkats. Evolution 58: 1600–1607.
Russell AF, Clutton-Brock TH, Brotherton M, Sharpe LL, Mcilrath GM, Dalerum FD, Cameron EZ, Barnard JA. 2002. Factors affecting pup growth and survival in co-operatively breeding meerkats Suricata suricatta. Journal of Animal Ecology 71: 700–709.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Strandburg-Peshkin, A., Clutton-Brock, T. and Manser, M.B. 2020. Burrow usage patterns and decision-making in meerkat groups. Behavioral Ecology, 31(2), 292-302.
Thorley, J., Duncan, C., Gaynor, D., Manser, M.B. and Clutton-Brock, T. 2025. Disentangling the effects of temperature and rainfall on the population dynamics of Kalahari meerkats. Oikos, e10988.
Van de Ven, T. M., Fuller, A. and Clutton‐Brock, T.H. 2020. Effects of climate change on pup growth and survival in a cooperative mammal, the meerkat. Functional Ecology, 34(1), 194-202.
Young, A.J., Clutton-Brock, T. 2006. Infanticide by subordinates influences reproductive sharing in cooperatively breeding meerkats. Biology Letters 2: 385–387.
Young, A.J., Monfort S.L., Clutton-Brock T.H. 2008. Physiological suppression among female meerkats: a role for subordinate restraint due to the threat of infanticide? Hormones and Behavior 53: 131–139.