
Sperm Whale
Physeter macrocephalus

2025 Red list status
Vulnerable
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Physeter macrocephalus – Linnaeus, 1758
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – PHYSETERIDAE – Physeter – macrocephalus
Common Names: Sperm Whale, Spermacet Whale, Pot Whale, Cachelot (English), Potvis (Afrikaans), Ballena Esperma, Cachalote (Spanish; Castilian), Cachalot (French), Capodoglio (Italian), Gabdoll (Maltese), Kashalot (Albanian), Kaşalot (Turkish), Ulješura (Croatian), İspermeçet balinası (Turkish), φυσητήρας (fysitíras) (Greek, Modern (1453-)), ראשתן (roshtan) (Hebrew), عنبر (anbar) (Arabic)
Synonyms: Physeter catodon Linnaeus, 1758
Species name changed to macrocephalus, based on priority rule
Taxonomic Note:
Although Physeter catodon is still occasionally used in the literature, P. macrocephalus is recommended (Rice 1989). Both names are listed on the same page of the original description by Linnaeus (1758), and priority is unclear. However, P. macrocephalus is preferable because it is used much more frequently, and this will support nomenclatural stability.
Red List Status: VU – Vulnerable, A1d (IUCN version 3.1)
Assessment Information
Assessors: Shabangu, F.1,2, Purdon, J.2,3 & da Silva, J.4
Reviewer: Smith, C.5
Institutions: 1Department of Forestries, Fisheries and the Environment, 2The Whale Unit, University of Pretoria, 3TUT Nature Conservation, 4South African National Biodiversity Institute, 5Endangered Wildlife Trust
Previous Assessors: Elwen, S., Findlay, K., Meyer, M., Oosthuizen, H. & Plön, S.
Previous Reviewer: Child, M.F.
Previous Contributor: Relton, C.
Assessment Rationale
Although the population is recovering, commercial whaling in the Antarctic within the last three generations (90 years) reduced the global abundance of species significantly. As commercial whaling has ceased, the species is evaluated under the A1 criterion. Model results revealed a 6% probability for Endangered, a 54% probability of Vulnerable, and a 40% probability of Near Threatened. Thus, the species is listed as Vulnerable A1d based on historical decline in line with the global assessment. Circumpolar surveys estimate around 8,300 mature males, which is extrapolated to around 40,000 individuals in total. Within the assessment region, the historical depletion may have created a skewed sex ratio, which may make this species more vulnerable to minor threats. For example, systematic surveys from the Antarctic showed no significant population increase between 1978 and 1992. Modelling results corroborate the Sperm Whale’s slow recovery rate, where a small decrease in adult female survivorship could lead to a declining population. Ongoing loss of mature individuals from entanglement in fishing nets and plastic ingestion could be hindering population recovery in certain areas. Furthermore, marine noise pollution may be an emerging threat that could suppress population recovery, as this reduces the foraging and communication behaviour of the species. The effects should continue to be monitored. Overall, the overexploitation of Sperm Whales has ceased, and they usually remain fairly far from anthropogenic effects due to their deep-sea distribution, and the large-scale commercial fishing industry does not target major Sperm Whale food sources. However, given their historical depletion, their slow growth rate (possibly only 1% per year) and their modelled sensitivity to disturbance, current abundance and population trend estimates are urgently needed, and this species should be reassessed once such data are available.
Regional population effects: Sperm Whales are highly migratory and wide-ranging. There are no barriers to dispersal, thus rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Shabangu F, Purdon J & da Silva JM. 2025. A conservation assessment of Physeter macrocephalus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Sperm whales have a broad geographic range (Rice 1989) and may be present in nearly every marine region from the tropics to high latitudes. Typically, they are located in deeper waters or along the continental slope, avoiding waters shallower than 300 m. Their geographic range also includes several enclosed or partially enclosed areas, including both the Mediterranean and Baltic Seas, the Sea of Okhotsk and the Gulf of Mexico. Their distributions vary according to sex and size, where males have been observed closer to inshore areas than females (Best 1999). Additionally, females and their young are often restricted in their range to regions between 40°N and 40°S, while males may migrate as far as 70°N and 70°S in summer, and larger males appear to extend further north or south than smaller individuals. Sperm Whales are known to travel substantial distances, with one individual reported to have covered a straight-line distance of 7,400 km (Ivashin 1967). Although some overlap in geographic distribution is known to occur between Northern and Southern Hemisphere stocks, populations are thought to be genetically isolated because seasonal breeding periods occur six months apart.
The International Whaling Commission (IWC) recognises nine Sperm Whale divisions for the Southern Hemisphere, which have been based more specifically on data available from commercial whaling, rather than actual biological factors (Donovan 1991). Although Sperm Whales migrate long distances, and exhibit low genetic differentiation between ocean basins (Lyrholm et al. 1999; Mesnick et al. 1999; Drouot et al. 2004a), some studies infer a high degree of geographic structure among populations across many regions (Bannister & Mitchell 1980; Kasuya & Miyashita 1988; Rendell & Whitehead 2003; Whitehead 2003). This is corroborated by recent molecular analyses that suggest females show site fidelity to coastal basins while males disperse widely for breeding (Engelhaupt et al. 2009). Within South African waters, Sperm Whales are present across the majority of the Exclusive Economic Zone (EEZ), frequently in deep waters off the west coast, and excluding shallow regions along the continental shelf. Records from Durban (30°S) and Donkergat (33°S) found that males and females reveal varied seasonality where large males were often caught in this region in spring, while juvenile males and females were more frequently caught earlier in winter. Passive acoustic monitoring data also show year-round presence of Sperm Whale clicks off the west coast of South Africa with presence peak in spring through summer (Shabangu and Andrew 2020). A recent distribution modelling study (Purdon et al. 2020a) indicated that sperm whales come closer to shore throughout the assessment area during winter, but still remaining off the shelf. In summer their distribution covers a wider area within the assessment area.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): 0
Depth Upper Limit (in metres below sea level): ≥3000
Depth Zone: Bathypelagic
Map
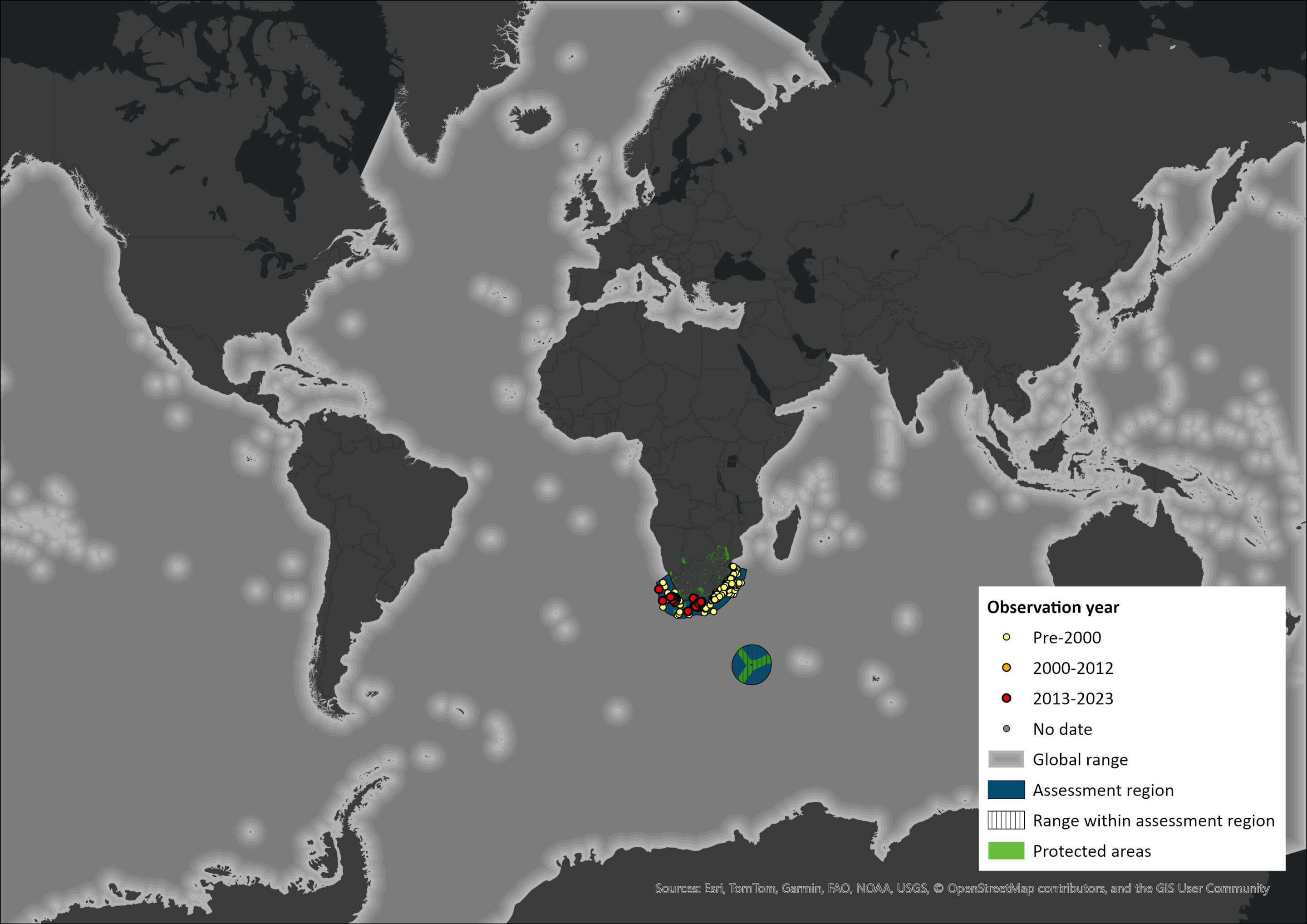 Figure 1. Distribution records for Sperm Whale (Physeter macrocephalus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Sperm Whale (Physeter macrocephalus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Antarctic, Australasian, Indomalayan, Nearctic, Neotropical, Oceanian, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Albania | Extant | Native | – | – |
| Algeria | Extant | Native | – | – |
| American Samoa | Presence Uncertain | Native | – | – |
| Angola | Extant | Native | – | – |
| Anguilla | Presence Uncertain | Native | – | – |
| Antarctica | Extant | Native | – | – |
| Antigua and Barbuda | Extant | Native | – | – |
| Argentina | Extant | Native | – | – |
| Aruba | Presence Uncertain | Native | – | – |
| Australia | Extant | Native | – | – |
| Bahamas | Extant | Native | – | – |
| Bangladesh | Extant | Native | – | – |
| Barbados | Extant | Native | – | – |
| Belgium | Extant | Native | – | – |
| Belize | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Bermuda | Presence Uncertain | Native | – | – |
| Bonaire, Sint Eustatius and Saba | Extant | Native | – | – |
| Bosnia and Herzegovina | Presence Uncertain | Native | – | – |
| Brazil | Extant | Native | – | – |
| British Indian Ocean Territory | Presence Uncertain | Native | – | – |
| Brunei Darussalam | Extant | Native | – | – |
| Cabo Verde | Extant | Native | – | – |
| Cambodia | Presence Uncertain | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Canada | Extant | Native | – | – |
| Cayman Islands | Presence Uncertain | Native | – | – |
| Chile | Extant | Native | – | – |
| China | Extant | Native | – | – |
| Christmas Island | Presence Uncertain | Native | – | – |
| Cocos (Keeling) Islands | Presence Uncertain | Native | – | – |
| Colombia | Extant | Native | – | – |
| Comoros | Extant | Native | – | – |
| Congo | Presence Uncertain | Native | – | – |
| Congo, The Democratic Republic of the | Presence Uncertain | Native | – | – |
| Cook Islands | Presence Uncertain | Native | – | – |
| Costa Rica | Extant | Native | – | – |
| Croatia | Extant | Native | – | – |
| Curaçao | Extant | Native | – | – |
| Cyprus | Extant | Native | – | – |
| Côte d’Ivoire | Presence Uncertain | Native | – | – |
| Denmark | Extant | Native | – | – |
| Djibouti | Extant | Native | – | – |
| Dominica | Extant | Native | – | – |
| Dominican Republic | Extant | Native | – | – |
| Ecuador | Extant | Native | – | – |
| Egypt | Extant | Native | – | – |
| El Salvador | Extant | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Eritrea | Presence Uncertain | Native | – | – |
| Falkland Islands (Malvinas) | Extant | Native | – | – |
| Faroe Islands | Extant | Native | – | – |
| Fiji | Extant | Native | – | – |
| France | Extant | Native | – | – |
| French Guiana | Presence Uncertain | Native | – | – |
| French Polynesia | Presence Uncertain | Native | – | – |
| French Southern Territories | Presence Uncertain | Native | – | – |
| Gabon | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Germany | Presence Uncertain | Native | – | – |
| Ghana | Extant | Native | – | – |
| Gibraltar | Extant | Native | – | – |
| Greece | Extant | Native | – | – |
| Greenland | Extant | Native | – | – |
| Grenada | Extant | Native | – | – |
| Guadeloupe | Presence Uncertain | Native | – | – |
| Guam | Presence Uncertain | Native | – | – |
| Guatemala | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Guyana | Extant | Native | – | – |
| Haiti | Extant | Native | – | – |
| Honduras | Extant | Native | – | – |
| Hong Kong | Presence Uncertain | Native | – | – |
| Iceland | Extant | Native | – | – |
| India | Extant | Native | – | – |
| Indonesia | Extant | Native | – | – |
| Iran, Islamic Republic of | Extant | Native | – | – |
| Ireland | Extant | Native | – | – |
| Israel | Extant | Native | – | – |
| Italy | Extant | Native | – | – |
| Jamaica | Extant | Native | – | – |
| Japan | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Kiribati | Extant | Native | – | – |
| Korea, Democratic People’s Republic of | Extant | Native | – | – |
| Korea, Republic of | Extant | Native | – | – |
| Lebanon | Extant | Native | – | – |
| Liberia | Extant | Native | – | – |
| Libya | Extant | Native | – | – |
| Madagascar | Extant | Native | – | – |
| Malaysia | Extant | Native | – | – |
| Maldives | Extant | Native | – | – |
| Malta | Extant | Native | – | – |
| Marshall Islands | Extant | Native | – | – |
| Martinique | Presence Uncertain | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Mauritius | Extant | Native | – | – |
| Mayotte | Presence Uncertain | Native | – | – |
| Mexico | Extant | Native | – | – |
| Micronesia, Federated States of | Extant | Native | – | – |
| Monaco | Extant | Native | – | – |
| Montenegro | Presence Uncertain | Native | – | – |
| Montserrat | Presence Uncertain | Native | – | – |
| Morocco | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Myanmar | Presence Uncertain | Native | – | – |
| Namibia | Extant | Native | – | – |
| Nauru | Extant | Native | – | – |
| Netherlands | Extant | Native | – | – |
| New Caledonia | Presence Uncertain | Native | – | – |
| New Zealand | Extant | Native | – | – |
| Nicaragua | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Niue | Extant | Native | – | – |
| Norfolk Island | Presence Uncertain | Native | – | – |
| Northern Mariana Islands | Presence Uncertain | Native | – | – |
| Norway | Extant | Native | – | – |
| Oman | Extant | Native | – | – |
| Pakistan | Extant | Native | – | – |
| Palau | Extant | Native | – | – |
| Palestine, State of | Presence Uncertain | Native | – | – |
| Panama | Extant | Native | – | – |
| Papua New Guinea | Extant | Native | – | – |
| Peru | Extant | Native | – | – |
| Philippines | Extant | Native | – | – |
| Pitcairn | Presence Uncertain | Native | – | – |
| Portugal | Extant | Native | – | – |
| Puerto Rico | Presence Uncertain | Native | – | – |
| Qatar | Presence Uncertain | Native | – | – |
| Russian Federation | Extant | Native | – | – |
| Réunion | Presence Uncertain | Native | – | – |
| Saint Barthélemy | Extant | Native | – | – |
| Saint Helena, Ascension and Tristan da Cunha | Extant | Native | – | – |
| Saint Kitts and Nevis | Extant | Native | – | – |
| Saint Lucia | Extant | Native | – | – |
| Saint Martin (French part) | Extant | Native | – | – |
| Saint Pierre and Miquelon | Presence Uncertain | Native | – | – |
| Saint Vincent and the Grenadines | Extant | Native | – | – |
| Samoa | Extant | Native | – | – |
| Sao Tome and Principe | Extant | Native | – | – |
| Saudi Arabia | Presence Uncertain | Native | – | – |
| Senegal | Extant | Native | – | – |
| Seychelles | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Singapore | Extant | Native | – | – |
| Sint Maarten (Dutch part) | Extant | Native | – | – |
| Slovenia | Extant | Native | – | – |
| Solomon Islands | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| South Georgia and the South Sandwich Islands | Presence Uncertain | Native | – | – |
| Spain | Extant | Native | – | – |
| Sri Lanka | Extant | Native | – | – |
| Sudan | Presence Uncertain | Native | – | – |
| Suriname | Extant | Native | – | – |
| Sweden | Presence Uncertain | Native | – | – |
| Syrian Arab Republic | Extant | Native | – | – |
| Taiwan, Province of China | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Thailand | Extant | Native | – | – |
| Timor-Leste | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Tokelau | Presence Uncertain | Native | – | – |
| Tonga | Extant | Native | – | – |
| Trinidad and Tobago | Extant | Native | – | – |
| Tunisia | Extant | Native | – | – |
| Turks and Caicos Islands | Presence Uncertain | Native | – | – |
| Tuvalu | Extant | Native | – | – |
| Türkiye | Extant | Native | – | – |
| United Arab Emirates | Presence Uncertain | Native | – | – |
| United Kingdom of Great Britain and Northern Ireland | Extant | Native | – | – |
| United States Minor Outlying Islands | Presence Uncertain | Native | – | – |
| United States of America | Extant | Native | – | – |
| Uruguay | Extant | Native | – | – |
| Vanuatu | Extant | Native | – | – |
| Venezuela, Bolivarian Republic of | Extant | Native | – | – |
| Viet Nam | Extant | Native | – | – |
| Virgin Islands, British | Presence Uncertain | Native | – | – |
| Virgin Islands, U.S. | Presence Uncertain | Native | – | – |
| Wallis and Futuna | Presence Uncertain | Native | – | – |
| Western Sahara | Presence Uncertain | Native | – | – |
| Yemen | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
| Presence | Origin | Formerly Bred | Seasonality | |
| 21. Atlantic – northwest | Extant | Native | – | – |
| 27. Atlantic – northeast | Extant | Native | – | – |
| 31. Atlantic – western central | Extant | Native | – | – |
| 34. Atlantic – eastern central | Extant | Native | – | – |
| 37. Mediterranean and Black Sea | Extant | Native | – | – |
| 41. Atlantic – southwest | Extant | Native | – | – |
| 47. Atlantic – southeast | Extant | Native | – | – |
| 48. Atlantic – Antarctic | Extant | Native | – | – |
| 51. Indian Ocean – western | Extant | Native | – | – |
| 57. Indian Ocean – eastern | Extant | Native | – | – |
| 58. Indian Ocean – Antarctic | Extant | Native | – | – |
| 61. Pacific – northwest | Extant | Native | – | – |
| 67. Pacific – northeast | Extant | Native | – | – |
| 71. Pacific – western central | Extant | Native | – | – |
| 77. Pacific – eastern central | Extant | Native | – | – |
| 81. Pacific – southwest | Extant | Native | – | – |
| 87. Pacific – southeast | Extant | Native | – | – |
| 88. Pacific – Antarctic | Extant | Native | – | – |
Climate change
Sperm whales are ecosystem sentinels (Hazen et al. 2019) and can therefore indicate ecosystem change. Close monitoring of this species to determine distribution changes could highlight areas that are more susceptible to climate change. For example, a study by Posdaljian et al. (2022) showed that between the years of 2015 and 2019 in Baffin Bay of the Canadian Arctic, acoustic presence of sperm whales increased steadily. They related this increase to the minimum sea ice concentration each year. In this area their distribution is likely expanding, however in other areas this may not be the case. In New Zealand a study showed that under the worst-case scenario of climate change RCP 8.5 (where nothing is done to curb greenhouse gas emissions by 2100) 61% of sperm whale habitat would be lost. Climate change poses a threat to sperm whales due to their longevity and sensitivity to their prey species’ distribution and abundance (Peters et al. 2022) and could therefore impact their recovery from whaling.
In South Africa little has been done to determine the effects of climate change on sperm whales. Purdon et al. (2020b) conducted a study on the anthropogenic effects on cetacean species richness. They found that in the assessment area, climate change stressors which include increasing sea surface temperature, ocean acidification and rising sea levels pose high risks to species richness. It was also noted that ocean acidification and sea level rise increased throughout the assessment area with the highest trends along the west and south coast, areas where sperm whale presence is high. Ocean acidification may affect phytoplankton and zooplankton affecting top marine predators (Simmonds and Isaac, 2007) like sperm whales. Future research should include modelling the effects of climate change on sperm whale distribution as well as their prey’s distribution.
Population information
Using historical trajectories, an abundance model for global Sperm Whale populations was developed to estimate the population decline between 1700 and 2022 (Whitehead and Shin 2022). This model includes only the threat of legal commercial whaling and does not consider any other anthropogenic threats to this species, such as ship strikes, illegal whaling in the North Pacific and Antarctica, climate change, pollution, entanglement in fishing gear, or the persistent effects of social disruption and sexually skewed population structure. These factors may limit population recovery in many areas. Estimates of trends in the post-whaling era (from 1982) suggest that sperm whale populations not facing much human impact are recovering slowly, but populations may be declining in areas with substantial anthropogenic footprint (Whitehead and Shin 2022). Although there is uncertainty, the model estimates a global pre-exploitation (1710) population of 1.9 million Sperm Whales declining to 840,000 in 2022 (57% decline) with most of this decline occurring during the period of heavy industrialized whaling between 1950-1975. The three generation (1940-2022) decline is estimated as 48%. Thus, the species remains close to Vulnerable status under the A1 criterion.
Two major global Sperm whaling operations were driven by the high commercial value attached to this species: the primitive “open-boat” hunt from 1712–1920 (Best 1983), and the modern whaling expeditions from 1910–1988 (Tønnessen & Johnsen 1982). Modern whaling operations did not impact all Sperm Whale populations. For example, populations in the western North Atlantic remain at reasonably high densities and show evidence of successful reproduction (Gordon et al. 1998). After the decline of other large rorqual species and the invention of new uses for Sperm Whale oil, commercial whaling of this species was intensified until 1964, when an annual peak of 29,255 individuals were caught. Limits imposed by the IWC after 1968 coincided with the development of synthetic alternatives for Sperm Whale oil, resulting in a decline in Sperm whaling efforts. Commercial Sperm whaling is currently prohibited by the IWC. Under special permit, only one Sperm Whale was recorded as caught in the 2009/10 season by a Japanese whaling vessel in the North Pacific (IWC unpubl. data). Although, the effect on the Sperm Whale stocks by small-scale recent operations is negligible, the value of these activities is severely questioned.
There is some concern that a few locations are still in decline, and there is no clear quantitative evidence suggesting that the global population has shown a recovery since large-scale whaling ceased in 1980 (Taylor et al. 2008). There is also no evidence to the contrary. Future population assessments are required to address the doubt surrounding the recovery of this species. Within the assessment region, we assume that the population is at the depleted level suggested by the global assessment, although evidence from the circumpolar surveys indicates that the population is recovering (IWC unpubl. data). The historical depletion may have created a skewed sex ratio, which may make this species more vulnerable to minor threats (for example, plastic pollution, ship strikes, entanglements). While the Antarctic population should have repopulated from less heavily exploited breeding populations at lower latitudes following the end of large-scale commercial whaling (Taylor et al. 2008), systematic surveys of Sperm Whales in the Antarctic showed no substantial or statistically significant increase between 1978 and 1992 (Branch & Butterworth 2001). We infer the population trend to be stable but current surveys are required to assess population trends. Corroborating the empirically estimated slow recovery rate, a recent population model revealed that Sperm Whale populations grow slowly and are potentially sensitive to survivorship rates of adult females, where a slight decline in survivorship could lead to a declining population (Chiquet et al. 2013).
| Continuing decline in mature individuals? | Qualifier | Justification |
| No | – | – |
Current population trend: Stable
Number of mature individuals in population: c. 8,300 from circumpolar surveys.
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
A population genetic study examining the structure and diversity of sperm whales across its range, and specifically within South Africa, revealed a single population throughout the assessment region, and this population differs genetically to those in the Atlantic and Indo-Pacific (Neveceralova et al. 2022). The African population (i.e., including Namibia) was also found to possess the most genetic variation; however, there was evidence of increased inbreeding over the past decade.
Based on this information, the proportion of populations maintained indicator can be calculated as 1.0 (1 of 1 populations maintained); however, because there are no known population size estimates for the region and contemporary effective population size (Ne) has not been calculated, the Ne 500 indicator cannot be estimated. Should updated count data be acquired, caution should be taken in estimating Ne given the observed levels of inbreeding. It is rather advisable to quantify Ne based on available genetic data.
Habitats and ecology
With the exception of the Black Sea and probably the Red Sea, sperm whales occur within all major marine water bodies deeper than 1,000 m including areas covered by ice sheets (Rice 1989; Whitehead 2003, Giorli and Pinkerton 2023). They are predominantly located in deeper waters of the open sea. However, they (especially males) may occasionally frequent shallower waters of the western North Atlantic (Scott & Sadove 1997). Females and young are most commonly limited to waters exhibiting sea surface temperatures greater than 15 °C (Rice 1989); and at latitudes lower than 40°N and 40°S. Their abundance and habitat selection usually increases with primary productivity (Jaquet et al. 1996; Rendell et al. 2004).
Sperm Whales are sexually dimorphic. Males may reach lengths of up to 18.3 m, while mature females may have a mass three times less than that of mature males, reaching lengths of 12.5 m. A 13.3 m male Sperm Whale weighed on a railway loading truck in Durban was 31,450 kg (Gambell 1970), but the heaviest recorded Sperm Whales included a male of 18.1 m at 57,000 kg and a female of 11.0 m at 24,000 kg. Sperm Whales have a substantial ecological footprint, and may consume roughly the same amount of biomass of marine resources as humans (Whitehead 2003).
Mesopelagic squid form the principal food source for Sperm Whales; however, in certain regions bottom dwelling fish are also commonly taken (Roe 1969). In South African waters, squid with an average weight of 0.5– 0.6 kg are usually consumed, while in the Antarctic, much larger squid (about 7.0 kg) are commonly preyed upon. The largest recorded squid found in the belly of a Sperm Whale was a Giant Squid (Architeuthis spp.) weighing 184 kg and 4.94 m long. Additional Sperm Whale food sources documented off the west coast of South Africa include crabs and tunicates (most commonly eaten by males), mysids and oilfish (more frequently eaten by females), and rays, angler fish and lancet fish (only eaten by males) (Best 1999). Adult males and females are thought to consume approximately 2% and 3% of their body mass per day, respectively. Bottom-dwelling sharks found in the stomach of a Sperm Whale collected in Durban suggest that the whale would have dived over 3,000 m deep.
The breeding season of Sperm Whales in the Southern Hemisphere peaks between October and December.
Females usually produce calves every three or five years; however, this duration increases with age (Rice 1989). Following a gestation period of 15–16 months, one calf (about 4 m in length) is born. Solid food is consumed before the calf reaches a year old; however, the stomachs of some individuals still contained evidence of milk at ages of 7.5 years and 13 years for females and males, respectively. Sexual maturity is reached at an age of 7–13 years (lengths of 8.5 m) for females, and at around 20 years (lengths of 12.5 m) for males; however, males only reach the status of a “breeding bull” once they are around 25 years old (lengths of 13.7 m).
Ecosystem and cultural services: Marine mammals integrate and reflect ecological variation across large spatial and long temporal scales, and therefore they are prime sentinels of marine ecosystem change; migratory mysticete whales may be used to investigate broad-scale shifts in ecosystems (Moore 2008). Sperm Whales are also important reservoirs of, and vectors for, nutrients (Roman et al. 2014), thus influencing oceanic ecosystem functioning. A Sperm Whale is the main antagonist (or protagonist, depending on your point of view) in the classic novel Moby Dick by Herman Melville.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 9.1. Marine Neritic -> Marine Neritic – Pelagic | – | Suitable | Yes |
| 10.1. Marine Oceanic -> Marine Oceanic – Epipelagic (0-200m) | – | Suitable | Yes |
| 10.2. Marine Oceanic -> Marine Oceanic – Mesopelagic (200-1000m) | – | Suitable | Yes |
Life History
| Generation Length | Justification | Data Quality |
| 27.3 | – | – |
Age at Maturity: Female or unspecified: 7–13 years
Age at Maturity: Male: 20 years
Size at Maturity (in cms): Female: 850
Size at Maturity (in cms): Male: 1250
Longevity: 60-70 years
Average Reproductive Age: 9-10 years
Maximum Size (in cms): 2,000
Size at Birth (in cms): 400
Gestation Time: 15–16 months
Reproductive Periodicity: 5-7 years
Average Annual Fecundity or Litter Size: One calf
Natural Mortality: 0.05 to 0.09 per year
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? Yes
Movement Patterns
Movement Patterns: Daily movement
Congregatory: Yes, up to 50 individuals
Systems
System: Marine
General Use and Trade Information
Extensive commercial Sperm whaling has ceased, however, small-scale hunting for Sperm Whales continues in Japan and Indonesia. Under the IWC’s Special Permit, 10 individuals are taken per annum by Japanese whaling fleets (Clapham et al. 2003). During the whaling era, sperm whale blubber was used for products such as lamp oil, margarine, cooking oil, candles, soaps, and cosmetics, while whale meat was sold for animal feed and fertilisers.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| No | – | – | – |
National Commercial Value: No
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | – | – |
| 5. Manufacturing chemicals | – | true | – | – |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: (Not specified)
Threats
Commercial whaling, historically the most substantial threat to the livelihood of this species, has ceased. Nonetheless, a range of additional factors threaten the remaining diminished populations of Sperm Whales. Considering that Sperm Whales are a large-scale migrating species, threats affecting this species in other areas of the world may impact populations that frequent the waters around South Africa, particularly those in the Atlantic, Indian and Southern Oceans. Similarly, population recovery is hindered by slow growth rates, possibly c. 1% per year (Whitehead 2002).
Similar to other cetaceans, Sperm Whales are vulnerable to entanglement in fishing gear, specifically gillnets, across a range of areas. This is particularly problematic in the Mediterranean Sea where Sperm Whales were one of the most commonly recorded non-target species caught in driftnets (Northridge 1991) before they were banned in 1990. Since 1990, illegal driftnet fisheries still occur in the Mediterranean but the frequency of Sperm Whale entanglements has declined considerably (Drouot et al. 2004b). Other cases of Sperm Whale entanglements have been recorded in Ecuador (Haase & Félix 1994) and California (Barlow & Cameron 2003), but are also considered to be a minor threat to this species within the assessment region. Although occasional catches in small scale gillnet fisheries are not currently expected to have a large effect on the global population, it is likely there are many more cases of Sperm Whale entanglements in unregulated driftnet fisheries in deeper waters, which are not recorded.
Occasionally, Sperm Whales engage in an activity known as “depredation”, when they actively remove fish from fishing gear (most commonly from demersal long-lines). This appears to be an increasing and risky phenomenon, which may result in entanglements, injury, death (Hucke-Gaete et al. 2004), and hostility from fishermen (including shooting of Sperm Whales) (Donoghue et al. 2003). It has been documented in a number of marine regions across the globe, including the North Atlantic, Chile, southeast Alaska, South Georgia, as well as other Southern Ocean islands and waters within the assessment region (Meÿer et al. 2011). In the Prince Edwards Islands (south of South Africa), Sperm Whales have been known to remove Patagonian Toothfish (Dissostichus eleginoides) from longline fishing vessels, which occasionally results in the illegal use of dynamite or thunder flashes to deter Sperm Whales (Ashford et al. 1996; Kock et al. 2006).
The sensitivity of Sperm Whales to noise is largely unconfirmed, where studies have shown contrasting evidence of high sensitivities (Watkins et al. 1985; Bowles et al. 1994) versus little to no effects on the species (Madsen & Møhl 2000; Madsen et al. 2002). There is a lack of research investigating the long-term effects of noise, sonar and seismic surveys on Sperm Whales, and, as yet, no mortality has been observed as a result of these disturbances. Off the west coast of South Africa, Sperm Whale clicks are masked by both high and low frequency underwater noise produced by marine traffic (Shabangu et al. 2022). On the other hand, this possible threat is increasing globally and thus the sensitivity of Sperm Whales to noise should be treated with caution.
Sexually-skewed whaling efforts may have long lasting effects on the reproductive rates (Whitehead 2003) and complex social cohesion of certain stocks, including those of the assessment region (Best 1979; Clarke et al. 1980; Whitehead et al. 1997); however, over time this inequality is likely to correct itself automatically. The population recovery and growth of Sperm Whales is fairly low, in fact, the maximum rate of increase is predicted to be around 1% per annum (Whitehead 2002). Population recovery since the end of commercial whaling, although inferred, remains purely theoretical for this species. Additionally, the severely depleted population of large, mature whales in the high latitudes of the Antarctic was assumed to have recovered from lower latitude areas where Sperm Whales were less heavily exploited following the end of commercial whaling. Contrastingly though, systematic surveys conducted for this species in the Antarctic did not reveal any significant increase between 1978 and 1992 (Branch & Butterworth 2001).
Collisions with ships impact Sperm Whales at a more regional scale, and have been specifically documented off the Canary Islands (André & Potter 2000) and in the Mediterranean (Pesante et al. 2002). Sperm Whales were listed as the most affected species by ship strikes near the island of Tenerife (Canary Islands), representing 48.8% of the total collision cases. Sperm whales are susceptible to ship strikes due their large bodies and the prolonged periods that they spend above water. In the assessment area, according to MRI unpublished data there has only been one sperm whale stranded whose death was attributed to a ship strike in 1999. Shipping pressure is high in the assessment area and overlaps with 87.40 % of predicted sperm whale distribution. This high level of overlap indicates that there are possibly many more undocumented deaths of sperm whales caused by ship strikes in the assessment area. This is probably due to these high-risk areas for ship strikes occurring offshore on the shelf edge, especially along the west coast.
The ingestion of marine debris, particularly plastic, is also known to be an increasing threat to this species (Viale et al. 1992; Simmonds 2011; de Stephanis et al. 2013). In 2008, the stomachs of two Sperm Whales stranded on the coast of California were found to contain substantial quantities of plastic debris, fishing net scraps and rope (Jacobsen et al. 2010). Gastric impaction as a result of this ingested debris was the most probable cause of death for both individuals (Jacobsen et al. 2010). Walker and Coe (1990) found that Sperm Whales are primarily affected by problems associated with the ingestion of marine debris, and this may cause a specific threat within the assessment region. Sperm Whale tissues also have high levels of some contaminants (O’Shea 1999; Nielsen et al. 2000), but it is uncertain whether this has an effect on the population level.
Current habitat trend: Declining in quality due to pollution and climate change (MacLeod 2009).
Conservation
The major historic threat to Sperm Whales (commercial whaling) has largely ceased. Thus, this species seems relatively secure from this threat in the short and medium term. Sperm Whales largely avoid anthropogenic effects, as they are mostly located in deeper waters away from the coastline. Additionally, much of this species’ food resources are safe from overexploitation by humans, as they feed predominantly on deep-water squid and fish in mesopelagic and benthic-pelagic habitats (Clarke 1977). This species is also listed on Appendix I of the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) and Appendices I and II of the Convention on the Conservation of Migratory Species of Wild Animals (CMS).
In order to reduce ship strikes, determining the distribution and population estimations of Sperm Whales in areas of high vessel traffic is required. Additionally, high speed vessels may require dedicated on-board observers in order to detect the presence of Sperm Whales and other cetaceans along the trajectory of the vessel. Finally, detailed on-board monitoring reports of cetacean collisions are essential to assess the severity of this threat to cetacean populations within the assessment region.
There have been a number of legislative interventions to limit marine pollution. A key intervention is the 1978 Protocol to the International Convention for the Prevention of Pollution from Ships (MARPOL), which acknowledges that ships add a significant and manageable quantity of pollution into marine environments (Lentz 1987). Annex V of MARPOL aims to “restrict at sea discharge of garbage and bans at sea disposal of plastics and other synthetic materials such as ropes, fishing nets, and plastic garbage bags with limited exceptions”. Nevertheless, this legislation is largely ignored and Clarke (1977) estimated that 6.5 million tons of plastic is discarded into the ocean each year. Enforcement and international cooperation is essential to ensure that all marine vessels comply with the Annex V policy.
Education and community involvement is also a powerful tool with which to approach the threat of marine (particularly plastic) pollution. Considering that land-based pollution usually ends up in the ocean, awareness, education and terrestrial-based action is often more effective in mitigating the problem compared to the development of addition legislative policies (for example, Ross & Swanson 1994).
Recommendations for managers and practitioners:
- Systematic monitoring: design and implement a monitoring programme (acoustic and sightings) that can detect population size and trends. For example, by using the recently developed single nucleotide polymorphism markers (Morin et al. 2007).
- Develop best practice guidelines for seismic surveys and enforce regulations.
Research priorities:
- Population size and trends. Given the long and deep diving behaviour of male Sperm Whales, the global estimate is almost certain to be an underestimate, as it is based on a sighting survey.
- Effects of marine noise pollution on Sperm Whale populations.
- Understanding the effects of minor threats (pollution, ship strikes, entanglements etc.) to this species, as well as the rates and trends associated with population recovery.
- Conduct more acoustic surveys for sperm whale detections in the assessment area. Information from Posdaljin et al. (2022) study indicates that acoustic detections for sperm whales obtain more information than visual sightings.
Encouraged citizen actions:
- Whale-watching operators could contribute to photo ID catalogues and behavioural observations.
- Report strandings to relevant authorities.
- Participate as volunteers in Sperm Whale research projects.
- Avoid using plastic bags, participate in beach and river clean-up initiatives, and raise awareness of the environmental threats associated with marine and terrestrial litter.
Bibliography
André, M. and Potter, J. R. 2000. Fast-ferry acoustic and direct physical impact on cetaceans: evidence, trends and potential mitigation. In: M. E. Zakharia, P. Chevret and P. Dubail (eds), Proceedings of the fifth European conference on underwater acoustics, ECUA 2000, pp. 491-496. Lyon, France.
Ashford JR, Rubilar PS, Martin AR. 1996. Interactions between cetaceans and longline fishery operations around South Georgia. Marine Mammal Science 12: 452-457.
Bannister, J. and Mitchell, E. 1980. North Pacific sperm whale stock identity: distributional evidence from Maury and Townsend charts. Reports of the International Whaling Commission Special Issue 2: 219-230.
Barlow J, Cameron GA. 2003. Field experiments show that acoustic pingers reduce marine mammal bycatch in the California drift gill net fishery. Marine Mammal Science 19: 265-283.
Best PB. 1999. Food and feeding of sperm whales Physeter macrocephalus off the west coast of South Africa. South African Journal of Marine Science 21: 393-413.
Best, P. B. 1979. Social organization in sperm whales, Physeter macrocephalus. In: H. E. Winn and B. L. Olla (eds), Behavior of marine animals, Volume 3: Cetaceans, pp. 227-289. Plenum Press.
Best, P. B. 1983. Sperm whale stock assessments and the relevance of historical whaling records. Reports of the International Whaling Commission Special Issue 5: 41-56.
Bowles, A. E., Smultea, M., Wursig, B., Demaster, D. P. and Palka, D. 1994. Relative abundance and behavior of marine mammals exposed to transmissions from the Heard Island Feasibility Test. Journal of the Acoustical Society of America 96: 2469-2484.
Branch, T.A. and Butterworth, D.S. 2001. Estimates of abundance south of 60°S for cetacean species sighted frequently on the 1978/79 to 1997/98 IWC/IDCR-SOWER sighting surveys. Journal of Cetacean Research and Management 3(3): 251-270.
Chiquet, R. A., Ma, B., Ackleh, A. S., Pal, N., and Sidorovskaia, N. 2013. Demographic analysis of sperm whales using matrix population models. Ecological Modeling 248: 71-79.
Clapham, P. J., Berggren, P., Childerhouse, S., Friday, N. A., Kasuya, T., Kell, L., Kock, K.-H., Manzanilla-Naim, S., Notarbartolo di Sciara, G., Perrin, W. F., Read, A. J., Reeves, R. R., Rogan, E., Rojas-Bracho, L., Smith, T. D., Stachowitsch, M., Taylor, B. L., Thiele, D., Wade, P. R. and Brownell Jr., R. L. 2003. Whaling as science. Bioscience 53: 210-211.
Clarke, M. R. 1977. Beaks, nets and numbers. Symposia of the Zoological Society of London 38: 89-126.
Clarke, R., Aguayo, A. and Paliza, O. 1980. Pregnancy rates of sperm whales in the southeast Pacific between 1959 and 1962 and a comparison with those from Paita, Peru, between 1975 and 1977. Reports of the International Whaling Commission, Special Issue 2: 151-158.
Donoghue M, Reeves RR, Stone GS. 2003. Report of the workshop on interactions between cetaceans and longline fisheries, Apia, Samoa, November 2002. New England Aquarium Press.
Donovan, G.P. 1991. A review of IWC stock boundaries. Reports of the International Whaling Commission 13: 39-68.
Drouot, V., Bérubé, M., Gannier, A., Goold, J.C., Reid, R.J. and Palsbøll, P.J. 2004. A note on genetic isolation of Mediterranean sperm whales (Physeter macrocephalus) suggested by mitochondrial DNA. Journal of Cetacean Research and Management 6(1): 29-32.
Drouot, V., Gannier, A. and Goold, J.C. 2004. Summer social distribution of sperm whales (Physeter macrocephalus) in the Mediterranean Sea. Journal of the Marine Biological Association of the United Kingdom 84(3): 675-680.
Engelhaupt, D., Hoelzel, A.R., Nicholson, C., Frantzis, A., Mesnick, S., Gero, S., Whitehead, H., Rendell, L., Miller, P., de Stefanis, R., Canadas, A., Airoldi, S. and Mignucci-Giannoni, A. 2009. Female philopatry in coastal basins and male dispersion across the North Atlantic in a highly mobile marine species, the sperm whale (Physeter macrocephalus). Molecular Ecology 18: 4193-4205.
Gambell R. 1970. Weight of a sperm whale, whole and in parts. South African Journal of Science 66: 225-227.
Giorli, G. and Pinkerton, M.H., 2023. Sperm whales forage year-round in the Ross Sea region. Frontiers in Remote Sensing, 4, p.940627.
Gordon, J., Moscrop, A., Caroson, C., Ingram, S., Leaper, R., Matthews, J. and Young, K. 1998. Distribution, movements and residency of sperm whales off the Commonwealth of Dominica, Eastern Caribbean: implications for the development and regulation of the local whalewatching industry. Reports of the International Whaling Commission 48: 551-557.
Haase, B. and Felix, F. 1994. A note on the incidental mortality of sperm whales (Physeter macrocephalus) in Ecuador. Reports of the International Whaling Commission Special Issue 15: 481-484.
Hazen, E.L., Abrahms, B., Brodie, S., Carroll, G., Jacox, M.G., Savoca, M.S., Scales, K.L., Sydeman, W.J. and Bograd, S.J., 2019. Marine top predators as climate and ecosystem sentinels. Frontiers in Ecology and the Environment, 17(10), pp.565-574.
Hucke-Gaete, R., Moreno, C. A. and Arata, J. 2004. Operational interactions of sperm whales and killer whales with the Patagonian toothfish industrial fishery off southern Chile. CCAMLR Science 11: 127-140.
IUCN (International Union for Conservation of Nature). 2012. Physeter macrocephalus. The IUCN Red List of Threatened Species. Version 3.1. Available at: http://www.iucnredlist.org. Downloaded on 21 February 2016
Ivashin MV. 1967. Whale globe-trotter. Priroda (Moscow) 8: 105-107.
Jacobsen, J.K., Massey, L. and Gulland, F. 2010. Fatal ingestion of floating net debris by two sperm whales (Physeter macrocephalus). Marine Pollution Bulletin 60: 765-767.
Jaquet, N., Whitehead, H. and Lewis, M. 1996. Coherence between 19th century sperm whale distributions and satellite-derived pigments in the tropical Pacific. Marine Ecology Progress Series 145: 1-10.
Kasuya, T. and Miyashita, T. 1988. Distribution of sperm whale stocks in the North Pacific. Scientific Reports of the Whales Research Institute 39: 31-75.
Kock K-H, Purves MG, Duhamel G. 2006. Interactions between cetacean and fisheries in the Southern Ocean. Polar Biology 29: 379-388.
Lentz SA. 1987. Plastics in the marine environment: legal approaches for international action. Marine Pollution Bulletin 18: 361-365.
Lyrholm T, Leimar O, Johanneson B, Gyllensten U. 1999. Sex– biased dispersal in sperm whales: contrasting mitochondrial and nuclear genetic structure of global populations. Proceedings of the Royal Society of London B: Biological Sciences 266: 347-354.
MacLeod, C.D. 2009. Global climate change, range changes and potential implications for the conservation of marine cetaceans: a review and synthesis. Endangered Species Research 7: 125-136.
Madsen, P. T. and Møhl, B. 2000. Sperm whales (Physeter catodon L. 1758) do not react to sounds from detonators. Journal of the Acoustic Society of America 107: 668-671.
Madsen, P. T., Mohl, B., Nielsen, B. K. and Wahlberg, M. 2002. Male sperm whale behaviour during exposures to distant seismic survey pulses. Aquatic Mammals 28(3): 231-240.
Mesnick, S. L., Taylor, B. L., Nachenberg, B., Rosenberg, A., Peterson, S., Hyde, J. and Dizon, A. E. 1999. Genetic relatedness within groups and the definition of sperm whale stock boundaries from the coastal waters off California, Oregon and Washington. Southwest Fisheries Center Administrative Report LJ-99-12: 10 pp.
Meÿer MA, Best PB, Anderson-Reade MD, Cliff G, Dudley SFJ, Kirkman SP. 2011. Trends and interventions in large whale entanglement along the South African coast. African Journal of Marine Science 33: 429-439.
Moore SE. 2008. Marine mammals as ecosystem sentinels. Journal of Mammalogy 89: 534-540.
Morin PA, Aitken NC, Rubio-Cisneros N, Dizon AE, Mesnick S. 2007. Characterization of 18 SNP markers for sperm whale (Physeter macrocephalus). Molecular Ecology Notes 7: 626-630.
Nielsen, J. B., Nielsen, F., Joergensen, P.-J. and Grandjean, P. 2000. Toxic metals and selenium in blood from pilot whales (Globecephala melas) and sperm whales (Physeter catodon). Marine Pollution Bulletin 40: 348-351.
Northridge SP. 1991. Driftnet fisheries and their impacts on nontarget species: a world-wide review. Page 115. Technical Paper 320. FAO Fisheries.
O’Shea, T.J. (ed.). 1999. Environmental contaminants and marine mammals. In: J. E. Reynolds III and S. A. Rommel (eds), Biology of Marine Mammals, pp. 485-564. Smithsonian University Press.
Pesante, G., Collet, A., Dhermain, F., Frantzis, A., Panigada, S., Podestà, M. and Zanardelli, M. 2002. Review of Collisions in the Mediterranean Sea. Proceedings of the Workshop: “Collisions between Cetaceans and Vessels: can we Find Solutions?” 40 (Special issue): 5-12. Rome, Italy.
Rendell L, Whitehead H, Escribano R. 2004. Sperm whale habitat use and foraging success off northern Chile: evidence of ecological links between coastal and pelagic systems. Marine Ecology Progress Series 275: 289-295.
Rendell, L. and Whitehead, H. 2003. Vocal clans in sperm whales (Physeter macrocephalus). Proceedings of the Royal Society of London B Biological Sciences B: 1-7.
Rice, D.W. 1989. Sperm whale Physeter macrocephalus Linneaus, 1758. In: S.H. Ridgway and R. Harrison (eds), Handbook of Marine Mammals, Vol. 4: River dolphins and the larger toothed whales, pp. 177-234. Academic Press.
Roe HSJ. 1969. The food and feeding habits of the sperm whales (Physeter catodon L.) taken off the west coast of Iceland. Journal du Consei 33: 93-102.
Roman J, Estes JA, Morissette L, Smith C, Costa D, McCarthy J, Nation J, Nicol S, Pershing A, Smetacek V. 2014. Whales as marine ecosystem engineers. Frontiers in Ecology and the Environment 12: 377-385.
Ross SS, Swanson RL. 1994. The impact of the Suffolk County, New York, plastics ban on beach and roadside litter. Journal of Environmental Systems 23: 337-351.
Scott, T. M. and Sadove, S. S. 1997. Sperm whale, Physeter macrocephalus, sightings in the shallow shelf waters off Long Island, New York. Marine Mammal Science 13(2): 317-320.
Simmonds MP. 2011. Eating Plastic: a preliminary evaluation of the impact on cetaceans of ingestion of plastic debris. Pages 1– 14. Submission to the IWC Scientific Committee
Shabangu, F.W. and Andrew, R.K., 2020. Clicking throughout the year: sperm whale clicks in relation to environmental conditions off the west coast of South Africa. Endangered Species Research, 43, pp.475-494.
Shabangu, F.W., Yemane, D., Best, G. and Estabrook, B.J., 2022. Acoustic detectability of whales amidst underwater noise off the west coast of South Africa. Marine Pollution Bulletin, 184, p.114122.
Taylor, B. L., Baird, R., Barlow, J., Dawson, S. M., Ford, J., Mead, J. G., Notarbartolo di Sciara, G., Wade, P., and Pitman, R. L. 2008. Physeter macrocephalus. In ‘The IUCN Red List of Threatened Species’. Version 2008. Available at: www.iucnredlist.org. (Accessed: 11 November 2008).
Taylor, B.L., Chivers, S.J., Larese, J. and Perrin, W.F. 2007. Generation length and percent mature estimates for IUCN assessments of cetaceans. NOAA, Southwest Fisheries Science Center Administrative Report LJ-07-01. La Jolla, California.
Tønnessen, J.N. and Johnsen, A.O. 1982. The History of Modern Whaling. University of California Press, Berkeley and Los Angeles, CA, USA.
Viale, D., Verneau, N. and Tison, Y. 1992. Stomach obstruction in a sperm whale beached on the Lavezzi islands: macropollution in the Mediterranean. Journal de Recherche Oceanographique 16: 100-102.
Walker WA, Coe JM. 1990. Survey of marine debris ingestion by odontocete cetaceans. Proceedings of the Second International Conference on Marine Debris, 2–7 April 1989. US Department of Commerce. NOAA Tech. Memo. NMFS, NOAA-TM-NMFS-SWFSC-154, Honolulu, Hawaii.
Watkins, W. A., Moore, K. E. and Tyack, P. 1985. Sperm whale acoustic behaviors in the southeast Caribbean. Cetology 49: 15 pp.
Whitehead, H. 2002. Estimates of the current global population size and historical trajectory for sperm whales. Marine Ecology Progress Series 242: 295-304.
Whitehead, H. 2003. Sperm Whales: Social Evolution in the Ocean. University of Chicago Press, Chicago, IL, USA.
Whitehead, H., Christal, J., and Dufault, S. 1997. Past and distant whaling and the rapid decline of sperm whales off the Galapagos Islands. Conservation Biology 11: 1387-1396.
de Stephanis, R., Gimenez, J., Carpinelli, E., Gutierrez-Exposito, C. and Canadas, A. 2013. As main meal for sperm whales: plastics debris. Marine Pollution Bulletin 69(1-2): 206-214.
Whitehead, H. and Shin, M., 2022. Current global population size, post-whaling trend and historical trajectory of sperm whales. Scientific Reports, 12(1), p.19468.
