Smith’s Red Rock Hare
Pronolagus rupestris

2025 Red list status
Least Concern
Regional Population Trend
Declining
Change compared
to 2016
No Change
Overview
Pronolagus rupestris – (A. Smith, 1834)
ANIMALIA – CHORDATA – MAMMALIA – LAGOMORPHA – LEPORIDAE – Pronolagus – rupestris
Common Names: Smith’s Red Rock Hare, Smith’s Red Rock Rabbit, Smith’s Red Rockhare (English), Smith Se Rooiklipkonyn (Afrikaans)
Synonyms: Pronolagus curryi (Thomas, 1902); Pronolagus fitzsimonsi (Roberts, 1938); Pronolagus melanurus (Rüppell, 1842); Pronolagus mülleri (Roberts, 1938); Pronolagus nyikae (Thomas, 1902); Pronolagus vallicola Kershaw, 1924
Taxonomic Note:
There are no subspecies. Hoffmann and Smith (2005) recognised four subspecies: Pronolagus rupestris curryi, P. r. nyikae, P. r. saundersiae, and P. r. vallicola. P. saundersiae has recently been distinguished as a good species. There has been some contention as to the number of possible subspecies of P. rupestris; however, until the biological and exact taxonomic status of these clades is clearer, the treatment of Meester et al. (1986) is provisionally upheld. Although the species occurs in both southern Africa and east Africa, it is unlikely that there is any gene flow between the two populations.
| Red List Status |
| LC – Least Concern, (IUCN version 3.1) |
Assessment Information
Assessors: Wilson-Hartmann, B.1, Matthee, C.M.2 & da Silva, J.3
Reviewer: Moodley, S.4
Institutions: 1McGregor Museum, Kimberley, 2Stellenbosch University, 3South African National Biodiversity Institute, 4Private
Previous Assessors and Reviewers: Matthee, C.M., Wilson, B., Robinson, T.J. & Child, M.F.
Previous Contributors: Roxburgh, L. & Smith, A.
Assessment Rationale
Red rock hares are widespread but patchily distributed within the assessment region due to their restriction to areas of rocky habitat. While no estimates of population size or trend are available, we infer a large population, given their extent of occurrence, and retain the Least Concern status. However, while their rocky habitats are largely inaccessible and unlikely to be transformed, increasing hunting pressure (inferred from expanding human settlements and anecdotal reports of decline), loss of foraging areas from agricultural expansion, and habitat degradation from overgrazing may be causing local declines and possible extinctions. These species are easily hunted and are thought to be substantially utilised for subsistence or sport hunting. For example, anecdotal reports from the Northern Cape suggest Smith’s Red Rock Hare have declined over the past 30 years. Similarly, the Natal Red Rock Hare occurs in areas of high human population density and may be increasingly locally threatened by hunting. Emerging threats to Smith’s Red Rock Hare may be increased by grazing pressure from goats and sheep, and increased predation from mesocarnivores due to lack of holistic management.
Although local declines may be occurring due to overhunting for bushmeat in some areas, there was no evidence to suggest a significant widespread population decline during previous assessments. However, with the outbreak of Rabbit Haemorrhagic Disease Virus strain 2 (RHDV-2) in November 2022 in the Northern Cape Province, South Africa that subsequently spread to all the provinces (DALRRD 2024), urgent investigations are needed to ascertain the effect on populations and subpopulations.
Concerted research into population size, densities and trends, as well as quantifying the identified threats, should be conducted before specific interventions are implemented if needed. Specifically, the area of occupancy and population trends of Smith’s Red Rock Hares should be determined and reassessed once such data are available. Long-term monitoring sites should be established to detect subpopulation trends in different regions.
Regional population effects: For Smith’s Red Rock Hare, given the approximately 1,750 km separation between the East African and South Africa populations, it is possible that these are two different species and no rescue effect can occur. If this is the case, then the South African population would be near endemic.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation:Wilson-Hartmann B, Matthee CM & da Silva JM. 2025. A conservation assessment of Pronolagus rupestris. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The close confinement of all Pronolagus species to rocky areas means their distribution is discontinuous and naturally fragmented with vast areas of land within their extent of occurrence unsuitable for them (Skinner & Chimimba 2005). As these species exist in habitat patches, area of occupancy should be calculated using land-cover data and remote sensing.
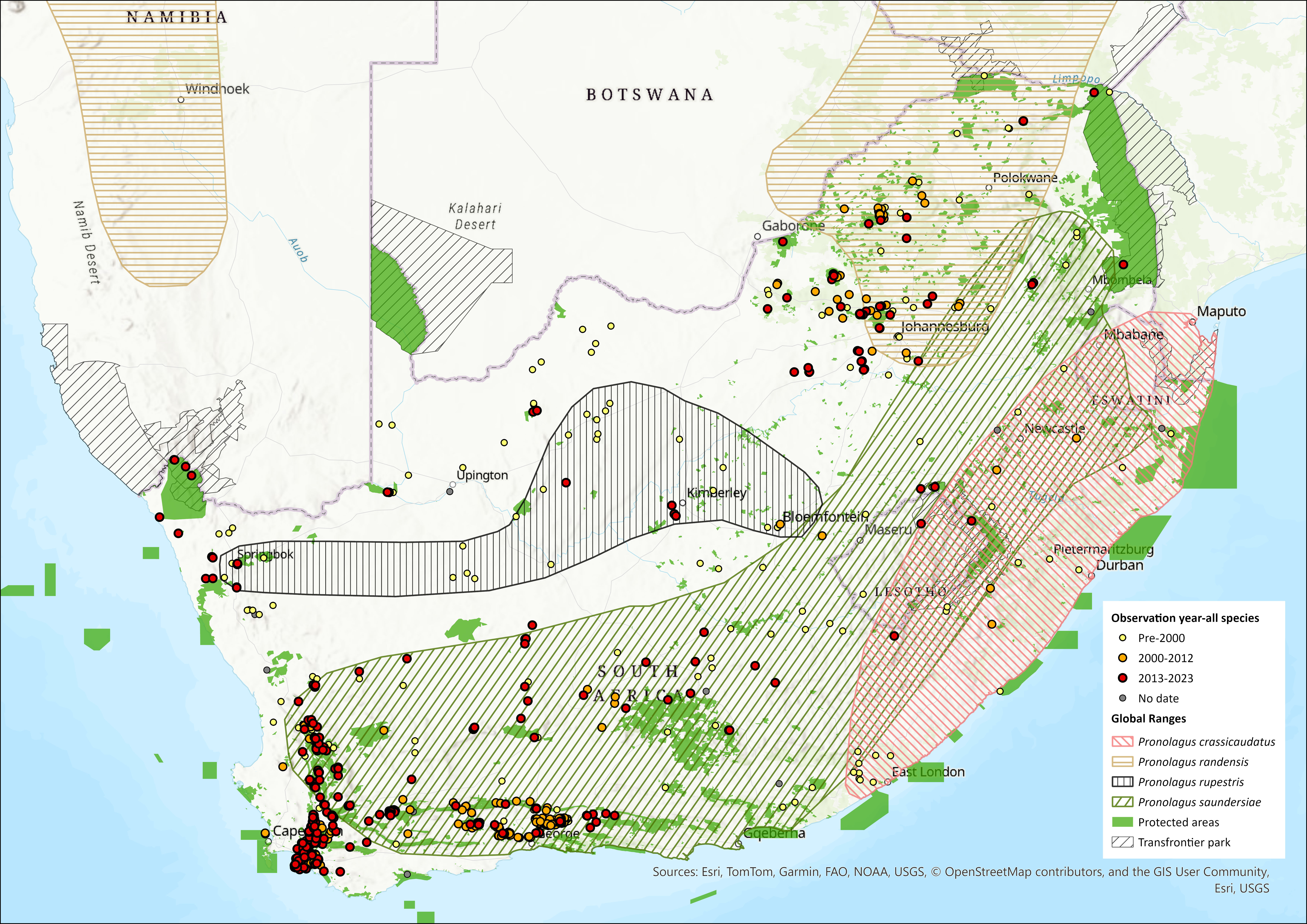
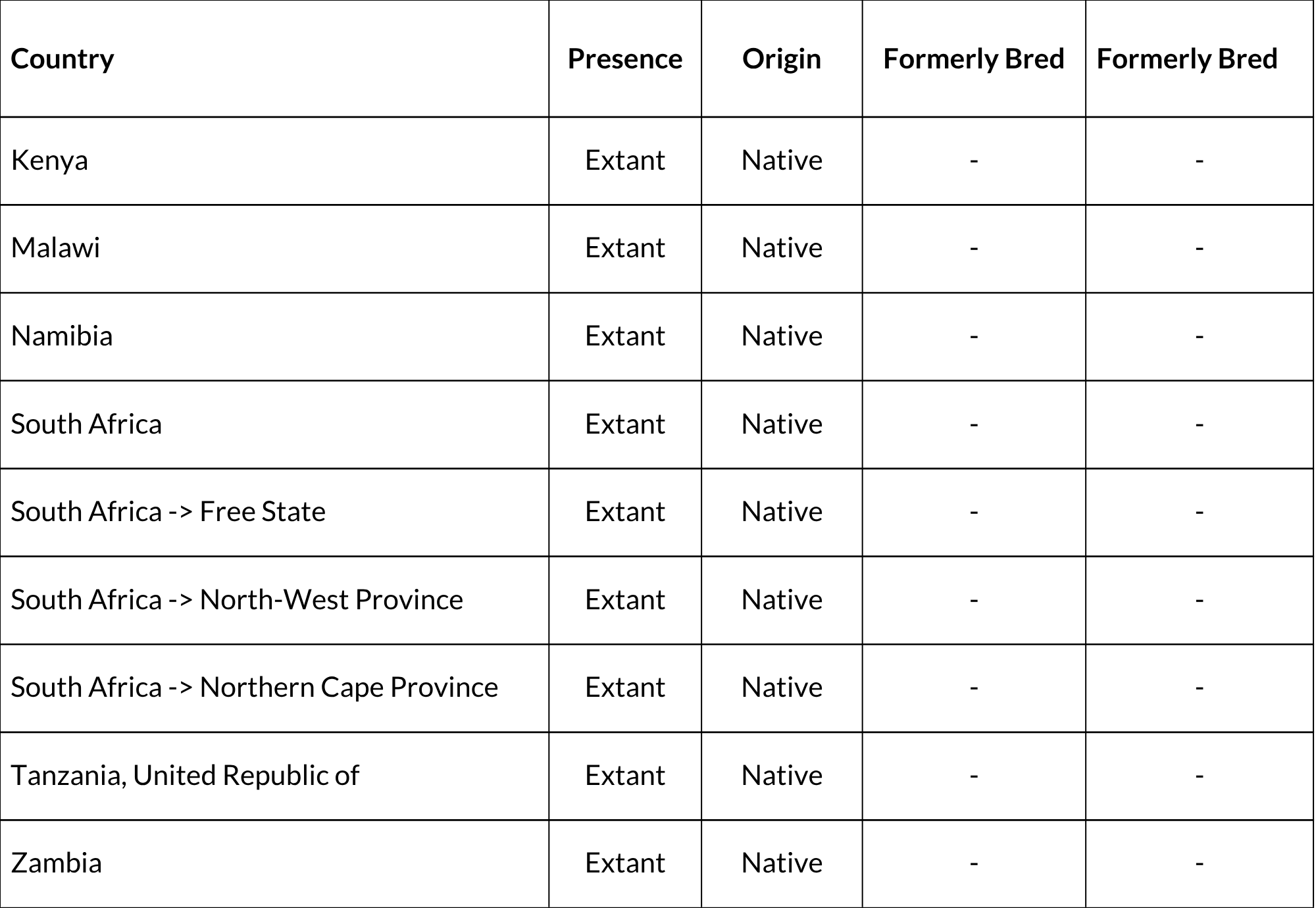
P. rupestris occurs in South Africa (Free State, North West, and Northern Cape provinces), and in a separate, isolated population in southwestern Kenya, central Tanzania, eastern Zambia, and Malawi (Duthie & Robinson 1990; Robinson & Matthee 2005). The two populations are separated by 1,200 km and thus require taxonomic resolution (Happold 2013). Although previously thought to no longer occur in Namibia (Bronner et al. 2003; Happold 2013), there is one confirmed record from Keetmanshoop (C. Matthee unpubl. data). Field surveys are needed to determine the continued existence, extent and viability of the Namibian population. The species is restricted to rocky outcrops, so the exact area of occupancy is probably lower than indicated from its wide extent of occurrence. Much of the habitat in the region of the distribution map is characterised by isolated rocky outcrops.
All museum records need vetting following taxonomic resolution of these species to accurately delineate the distribution maps.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realm: Afrotropical
Map
Figure 1. Distribution records for Smith’s Red Rock Hare (Pronolagus rupestris) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
The effects of climate change on Pronolagus sp. are currently not well researched, however it can be assumed that climate change per se was unlikely to have a dramatic effect on the Natal Rock Hare during this assessment period. Climate change does however affect the spread of infectious diseases beyond their typical geographic reach. Whilst this may not be the cause of the outbreak of Rabbit Haemorrhagic Disease Virus strain 2 (RHDV-2) in South Africa at the end of 2022, it can assist in the spread of the virus and increase the susceptibility of hares and rabbits to the effects of droughts and periods of flooding. Often, the cross-border spread of infectious diseases is further exacerbated by the lack of global governance policies or a consensus to mitigate climate change. Future focus should be placed on prioritising infectious disease research and making mitigation recommendations to address possible exacerbation of virus spread that can result from the effects of climate change.
Population information
Smith’s Red Rock Hare is thought to have large subpopulations in the Kuruman and Springbok areas, but the majority of anecdotal reports collected from farmers across the Northern Cape report a decline in numbers over the past 30 years (B. Wilson unpubl. data.). Happold (2013) reports the total population size as being above 10,000 mature individuals in South Africa, but this needs investigation. There is no evidence of gene flow between the East African and South African populations. It may be experiencing local declines due to encroachment of human settlements and subsequent hunting pressure. In addition, the recent outbreak of RHDV-2 may lead to significant declines throughout the range.
Population Information
|
Continuing decline in mature individuals? |
Qualifier |
Justification |
|
Yes |
Inferred |
Hunting pressure and competition for food resources. Mortalities likely related to RHDV-2 |
Current population trend: Declining
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: Suspected
All individuals in one subpopulation: No
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: 2
Severely fragmented: Yes. Ranges are naturally fragmented across mountainous and rocky areas.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
The species has been looked at in a phylogenetic context (Robinson & Matthee 2005), and also using mtDNA phylogeoraphy (Matthee & Robinson 1996). The animals within the assessment region belong to the southern population of the species, with no evidence of gene flow with the northern population (Duthie & Robinson 1990; Matthee & Robinson 1996). The population within the assessment region exists as a metapopulation. Due to the absence of population size estimates, effective population size (Ne) cannot be quantified using proxy methods. It is highly recommended that a population genetic study be undertaken to better inform the genetic health and status of this species.
Habitats and ecology
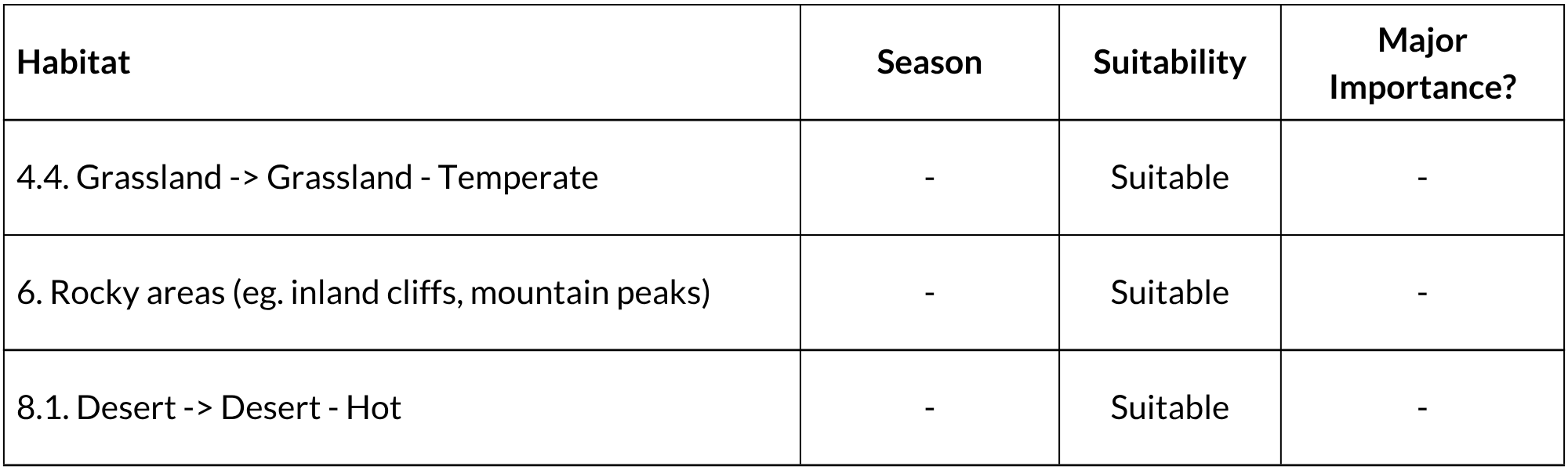
All species of Pronolagus seem to have similar habitat requirements in that they are confined to rocky areas that provide shelter and occur in krantzes, rocky boulder-strewn hillsides, rocky ravines and amongst rocks in dry riverbeds (Skinner & Chimimba 2005). Such areas must provide palatable grasses (they are grazers) and some cover of scrub bushes. They are predominantly nocturnal, emerging at dusk to feed but never forage far from their shelters, although they forage around the base of hills looking for fresh sprouting grasses (Skinner & Chimimba 2005). Shortridge (1934) remarked on their ability to ‘vanish like shadows behind rocks or down rock crevices on the slightest alarm’. Also characteristic of the genus are their flattened round dung pellets deposited on flat spaces amongst rocks (Lynch 1983). They use latrines established away from their resting sites. Rock hares are unique in their ability to run and jump over rocks and can run up steep rock faces to reach crevices when fleeing. Key vegetation types are those typical of mountainous and rocky terrain, including Afromontane and Afroalpine areas.
Smith’s Red Rock Hare is confined to rocky outcrops and hills covered with grass and shrubs (Lynch 1983). Much like the other members of the red rock hare family, this species is a medium-sized rabbit-like hare with thick, dense woolly pelage typically cinnamon-rufous in colour. Generally, the feet and tail are darker red with the soles covered in very dense dark grey hair. They tend to be more vocal than other red rock hare species. Nests are characteristically built in shallow excavations in the open at the base of a shrub and lined with fur from the mother’s belly and flanks over a grass and stick structure. They are partial to young, sprouting grass and can be seen grazing on recently burnt areas. Breeding is restricted to warmer months during which time a female could produce 3–4 litters after a 35 to 40 day gestation. Litter size is 1–2 young (Duthie and Robinson 1990) and the young are born naked (altricial).
Ecosystem and cultural services:
- The endemic near-endemic P. rupestris is a flagship species for its region.
- Hares and rabbits are recognised as important seed dispersal agents, particularly in harsh environments.
IUCN Habitats Classification Scheme
Life History
Generation Length: (Not specified)
Age at Maturity: Female: 6 months
Age at Maturity: Male: 6 months)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): 43-65cm
Size at Birth (in cms): (Not specified)
Gestation Time: 35-40 days
Reproductive Periodicity: Warmer months: Sept-Feb
Average Annual Fecundity or Litter Size: LS=1-2 with up to 4 litters
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
These species are likely to be fairly important in subsistence communities where they are easily obtained. Maliehe (1993) specifically mentioned rock hares as being an important bush meat item in Lesotho and in Zululand, but this is likely to be true throughout their ranges. As a slow-moving species, they are easily hunted with dogs. At night, they tend to freeze when chanced upon in spotlights and can be easily shot or captured in nets (B. Wilson pers. obs.). During the day, it is also possible to hunt them in their refuges, using a coiled piece of barbed wire that hooks and drags the animal out of its shelter (B. Wilson pers. obs.). Whilst having a particularly warm and thick fur, pelts from these species are prone to hairslip and do not cure well, making them unsuitable for hardwearing fur items.
National Commercial Value: No
International Commercial Value: No
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
The main threat to rock hares is hunting, both for bushmeat and for sport. For example, the survival of Hewitt’s Red Rock Hare in Lesotho is threatened due to hunting pressure from herders with packs of dogs (Lynch 1994). While skittish, they can be caught by hand when hiding under rock ledges or boulders (Skinner & Chimimba 2005) and thus may be susceptible to being hunted (see Use and Trade). Hunting pressure is thought to be more severe in areas of high-density human settlements. Additionally, agricultural and rural settlement expansion may be increasing incidental predation on rock hares by domestic dogs. Hunting is suspected to be causing local declines (and possibly local extinctions) in all red rock hare species, but it is uncertain whether there is a net decline in the populations. Long-term monitoring should be established to quantify subpopulation trends across species.
Sheep farming may also impact the habitat quality (reducing foraging areas) for Smith’s Red Rock Hare. In areas where there is intensive sheep farming, the species may face grazing competition (B Wilson pers. obs.). Additionally, because of available food resources, many sheep-farming areas also report higher than normal rates of predators such as Black-backed Jackals (Lupulella mesomelas) and Caracals (Caracal caracal), which is exacerbated by indiscriminate and non-holistic predator control methods (Minnie et al. 2016). These predators pose a threat to the rock hares when at abnormally high numbers. Since rock hare species occur at relatively low densities, it is unlikely that they are considered a significant competitive grazing species and thus are not expected to be persecuted by farmers.
However, a more urgent concern has recently emerged for this restricted population. In October 2022, reports of unusual Lepus deaths were received from the Middelpos, Springbok and Fraserburg areas in the Northern Cape. In November 2022 affected carcasses tested by Onderstepoort Veterinary Research Laboratory, the deaths were confirmed to as the result of the highly contagious and acute fatal hepatitis of Leporids called Rabbit Haemorrhagic Disease Virus 2 strain (RHDV-2). The new variant RHDV-2 affects rabbits, but also causes fatal RHD in various Lepus species, including Sardinian Cape hares (L. capensis mediterraneus), Italian hares (L. corsicanus), and mountain hares (L. timidus) (Rocchi & Dagleish 2018). This disease, which originated in China in 1984, primarily affects all rabbit and hares. Caused by a calicivirus, it was previously absent in South Africa. Infection typically occurs by the oral route. It spreads very easily, normally through direct contact between individuals but is also carried by insects, birds, scavengers feeding on carcasses, contaminated food or urine and faeces, and by humans on clothes, shoes, tools and car tyres. The virus can persist in infected meat (even if frozen) and for long periods in decomposing carcasses (Gleeson & Petritz 2020). Incubation varies from one to three days, and death usually occurs 12-36 hours after the onset of fever. The main clinical manifestations of the acute infection are nervous and respiratory signs, apathy and anorexia. In RHDV-2, clinical signs and mortality are observed even in young animals from 7 to 15 days of age onwards. Indirect control of the disease is easily achieved by vaccination. RHDV has never been reported in humans and other mammals. Outbreaks tend to be seasonal (typically peak breeding seasons) in wild rabbit and hare populations where adults have survived infection and are immune (Kerr & Donnelly 2013). As young kits grow up and stop nursing, they no longer receive the antibodies in their mother’s milk and become susceptible to infection. Mortality rates are high and up to 90% of infected individuals die (WOAH 2025). Not all rabbits exposed to RHDV-2 become overtly ill. A small proportion of infected rabbits clears the virus without developing signs of disease (Kerr & Donnelly 2013). Asymptomatic carriers also occur, and can continue to shed virus for months, thereby infecting other animals. Surviving rabbits develop a strong immunity to the specific viral variant with which they were infected (Gleeson & Petritz 2020).
This outbreak is of concern for indigenous rabbit and hare species due to their historic naivety to the virus and thus susceptibility to the virus. Whilst a vaccine is available for captive situations overseas, there is no specific treatment for RHDV-2, and management is limited to preventing further spread. Often, the cross-border spread of infectious diseases is further exacerbated by the lack of global governance, a factor that led to the introduction of this disease into South Africa and allowed it to spread to neighbouring countries.
Conservation
Rock hares exist in numerous national and provincial protected areas, as well as, presumably, private protected areas and conservancies. However, these should be collated and confirmed to estimate current occupancy. Smith’s Rock Hare is protected seasonally by Provincial Nature Conservation agencies in the Northern Cape and Free State to a certain degree but not in the North West Provinces in its southern range. The main intervention at this stage is research to quantify potential threats, area of occupancy, population size and trends. Until such data have been collected, no specific conservation interventions can be proposed. However, the following general interventions will benefit the rock hare species:
- Set aside land under crop and livestock agriculture to conserve foraging areas around rocky areas or dry riverbeds.
- Enforce legislation restricting residential development in rocky habitat or hilly slopes.
- Employ ecological stocking rates to reduce habitat degradation and grazing pressure.
- Employ holistic management of predators to reduce heightened interspecific predation and competition.
- The continued formation of conservancies should be encouraged to protect rocky habitat and reduce localised grazing pressure.
Recommendations for land managers and practitioners:
- Holistic management of ranch lands through reduction in stocking rates, predator control or areas of set-aside habitat.
Research priorities:
- Taxonomic revision is suggested to determine whether the disjunct populations of P. rupestris are genetically distinct – all inferences about geneflow have not been tested.
- Fine scale distributional studies across the range.
- Studies on changes in density across a spectrum of habitat quality. Quantification of population size and trends.
- Studies into the dispersal abilities and survival of subadult individuals in different habitats.
- Levels of direct persecution by farmers and subsistence hunters, and the efficacy of education and awareness programmes targeted at landowners.
- Long-term monitoring of population and subpopulation trends.
- Vetting of museum records to revise distribution maps.
- Potential of wildlife ranching and the private sector in conserving rock hares.
- Determination of the impact of RHDV-2 on the southern African population, particularly at subpopulation level.
Encouraged citizen actions:
- Refrain from having too many dogs on farms.
- Report sightings, especially outside protected areas, on virtual museum platforms (for example, iNaturalist and MammalMAP). Look out for their disc-shaped pellets as an indication of their presence.
- Balanced farming methods to prevent changes in predation pressures experienced by the hares.
- Report unusual death outbreaks potentially linked to RHDV-2 and prevent the spread of the disease.
Bibliography
Anderson, M.D. 2000. Raptor conservation in the Northern Cape Province, South Africa. Ostrich 71: 25–32.
Bronner, G.N., Hoffman, M., Taylor, P.J., Chimimba, C.T., Best, P.B., Matthee, C.A. and Robinson, T.J. 2003. A revised systematic checklist of the extant mammals of the southern African subregion. Durban Museum Novitates 28: 56-106.
Department Agriculture, Land Reform and Rural Development. 2024. Rabbit haemorrhagic disease outbreak update report. Directorate Animal Health, 27 February 2024.
Duthie, A.G. and Robinson, T. J. 1990. The African rabbits. In: J.A. Chapman and J.E.C. Flux (eds), Rabbits, Hares and Pikas: Status Survey and Conservation Action Plan, pp. 121-127. IUCN, Gland, Switzerland.
GeoTerraImage. 2015. 1990-2013/14 South African National Land-Cover Change. DEA/CARDNO SCPF002: Implementation of Land-Use Maps for South Africa. Project Specific Data Report.
Gleeson, M. and Petritz, O.A. 2020. Emerging Infectious Diseases of Rabbits. Veterinary Clinics of North America: Exotic Animal Practice. 23 (2): 249–261.
Happold, D.C.D. 2013. Pronoloagus rupestris Smith’s Red Rock Hare. Pages 715–716 in Happold DCD, editor. Mammals of Africa. Volume III: Rodents, Hares and Rabbits. Bloomsbury Publishing, London, UK.
Hoffmann, R.S. and Smith, A.T. 2005. Order Lagomorpha. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World, pp. 185-211. Johns Hopkins University Press, Baltimore, Maryland, USA.
Lynch, C.D. 1983. The mammals of the Orange Free State, South Africa. Navorsinge van die Nasionale Museum Bloemfontein 18: 1-218.
Lynch CD. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum, Bloemfontein 10: 177–241.
Maliehe, T.M. 1993. Non-wood forest products in South Africa. Paper prepared for the Regional Expert Consultation on non-wood forest products for English-speaking African countries. Commonwealth Science Council, Arusha, Tanzania.
Matthee, C.A. and Robinson, T.J. 1996. Mitochondrial DNA differentiation among geographical populations of Pronolagus rupestris, Smith’s red rock rabbit (Mammalia: Lagomorpha). Heredity 76: 514–524.
Matthee, C., Collins, K. and Keith, M. 2004. Pronolagus rupestris. In: Y. Friedman and B. Daly (eds), Red Data Book of the Mammals of South Africa: A Conservation Assessment, pp. 420-421. CBSG Southern Africa, Conservation Breeding Specialist Group (SSC/IUCN), Endangered Wildlife Trust, South Africa.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum, Pretoria, South Africa.
Minnie L, Gaylard A, Kerley GI. 2016. Compensatory life-history responses of a mesopredator may undermine carnivore management efforts. Journal of Applied Ecology 53: 379-387.
Rautenbach, I.L. 1982. Mammals of the Transvaal. No. 1, Ecoplan Monograph, Pretoria, South Africa.
Robinson,. TJ. and Matthee, C.A. 2005. Phylogeny and evolutionary origins of the Leporidae: a review of cytogenetics, molecular analyses and a supermatrix analysis. Mammal Review 35: 231–247.
Robinson, T.J. 1982. Key to the South African Leporidae (Mammalia: Lagomorpha). South African Journal of Zoology 17: 220–222.
Shortridge, G.C. 1934. The Mammals of South West Africa. Heinemann, London, UK.
Skinner, J.D. and Chimimba, C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Taylor, P.J. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.
Whiteford, C.E.M. 1995. Molecular phylogeny of the Genus Pronolagus (Mammalian: Lagomorpha) and the use of morphological and molecular characters in the delineation of P. rupestris. M.Sc. University of Pretoria.
World Organisation for Animal Health. Rabbit Haemorrhagic Disease.