
Rüppel’s Pipistrelle
Vansonia rueppellii

2025 Red list status
Near Threatened
Regional Population Trend
Unknown
Change compared
to 2016
Uplisted
Overview
Vansoniarueppellii – (Fischer, 1829)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – VESPERTILIONIDAE – Vansonia – rueppellii
Common Names: Rüppel’s Pipistrelle, Ruppell’s Pipistrelle, Rüppel’s Pipistrelle Bat, Rüppell’s Bat (English), Rüppell se vlermuis (Afrikaans)
Synonyms: Vespertilio temminckii (Cretzschmar, 1827); Vespertilio rüppellii (J. B. Fischer, 1829); Scotophilus temminkii (H. R. Schinz, 1844); rueppellii (J. A. Wagner, 1855); Vesperugo hypoleuca (von Heuglin & Fitzinger, 1866); Vesperugo sennaariensis (von Heuglin in von Heuglin & Fitzinger, 1866); Vesperugo senarensis (von Heuglin,1877; Vesperugo temminckii (Heuglin, 1877); Pipistrellus temminckii (Thomas 1901); Pipistrellus rueppelli (Anderson 1902); Scotozous rüppelii (Allen 1917); P[ipistrellus] rüppelli (Thomas 1919); Pipistrellus (Scotozous) rueppelli (Ellerman, Morrison-Scott and Hayman, 1953)
Taxonomic Note:
Pipistrellus rueppellii was previously included in the subgenus Vansonia Roberts, 1946 (ACR 2024). However, taxonomic revision is warranted, as molecular genetic evidence supports the reallocation of rueppellii into a distinct genus (Helbig et al. 2010; Koubínová et al. 2013). Benda et al. (2014) further advocated the transfer of rueppellii to the genus This reassignment, however, remained tentative pending the inclusion of specimens from the type locality of P. rueppellii (i.e., Sudan) and P. vernayi—the type species of Vansonia—from Maun, Botswana. Although Monadjem et al. (2020a, 2021) did not include material from Sudan or Botswana in their cytochrome b sequence analyses, their results indicate that rueppellii is genetically distinct from other African Pipistrellus species.
The species is easily identified by its striking white venter and darkened upper parts (Monadjem et al. 2020a). Meester et al. (1986) listed two subspecies, including P. r. vernayi Roberts 1932 from northeastern Botswana, and the nominate P. r. rueppellii from eastern Zimbabwe and the northern regions of Kruger National Park (South Africa). However, more recent literature suggested that vernayi may be applicable to all specimens from southern Africa and should include P. leucomelas Monard 1933 as a synonym (Monadjem et al. 2020a). The validity of these species is uncertain until further research is conducted. It is evident that further integrative taxonomic studies on African pipistrelloids are required to disentangle within and between species relationships.
Red List Status: NT – Near Threatened, D2 (IUCN version 3.1)
Assessment Information
Assessors: Richards, L.R.1 & da Silva, J.M.12
Reviewer: Balona, J.3
Contributors: Patel, T.4 & Roxburgh, L.4
Institutions: 1Durban Natural Science Museum, 2South African National Biodiversity Institute, 3Gauteng and Northern Regions Bat Interest Group, 4Endangered Wildlife Trust
Previous Assessors and Reviewers: Monadjem, A., Jacobs, D., Cohen, L., MacEwan, K., Richards, L.R., Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Relton, C., Raimondo, D. & Nicholson, S.K.
Assessment Rationale
Within the assessment region, this species is historically known from two major protected areas: Augrabies Falls National Park and Great Limpopo Transfrontier Park. It was recently recorded within the De Beers Venetia diamond mining footprint (Cory-Touissant 2021). Further field surveys and research are needed to more accurately delimit its distribution within the assessment region and to resolve its taxonomy. A reassessment may be warranted once more comprehensive data become available.
Regional population effects: Although records of this species in southern Africa are scattered, its habitat is connected through transfrontier conservation areas between South Africa and Zimbabwe. However, it has low wing loading (Norberg & Rayner 1987) so recruitment and rescue effects are uncertain.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: Although this slow-flying species occurs within at least two large protected areas, it has been uplisted to Near Threatened D2 due to its restricted distribution within the assessment region, uncertainty regarding population size (possibly fewer than 1,000 individuals), and concerns about the persistence of the local population at Augrabies Falls National Park. Additionally, potential threats exist in anthropogenically modified landscapes, such as open-cast mine tailings areas, where the species is closely associated with water bodies that may contain pollutants (e.g., Cory-Touissant et al. 2021).
Red List Index
Red List Index: Uplisted
Recommended citation: Richards LR & da Silva JM. 2025. A conservation assessment of Vansonia rueppellii. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species is widespread across Africa. It has a disjunct distribution through North Africa, with records from Morocco to Algeria, Tunisia and Libya (Benda et al. 2004). Its range extends along the Nile River through Egypt to the northern reaches of the Red Sea and the western regions of Sinai (ACR 2025). In West Africa, it has been recorded along the coast of Senegal and at the border between Senegal and Mauritania. Its range is continuous from northern Sudan southwards to northern Zimbabwe, and extends eastwards from western Zambia across to western Mozambique. In southern Africa, it occurs widely across the northern parts of the region from the Kruger National Park through to Zimbabwe, Zambia, Malawi, southern Democratic Republic of Congo, northern Botswana and west to Angola and the extreme north of Namibia (Monadjem et al. 2020; ACR 2025).
Riparian fringes along the Limpopo and Zambezi rivers may explain outlying records of this species in semi-arid savannahs of southern and northern Zimbabwe (Monadjem et al. 2020a). Within the assessment region, there is an isolated record from the Augrabies Falls National Park, Northern Cape, which may represent a vagrant or an overlooked population (Monadjem et al. 2020a). Ecological niche models indicate that suitable habitats are largely confined to the extreme northern regions of South Africa, the border between the Northern Cape and Namibia, and select areas of Eswatini, primarily associated with the Lebombo mountain range (Monadjem et al. 2010). Habitat models suggest the species may occur more widely in Namibia and adjoining parts of southern Angola, and along the Zambezi River in Mozambique (Monadjem et al. 2010).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 2 m asl (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 1900 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
Map
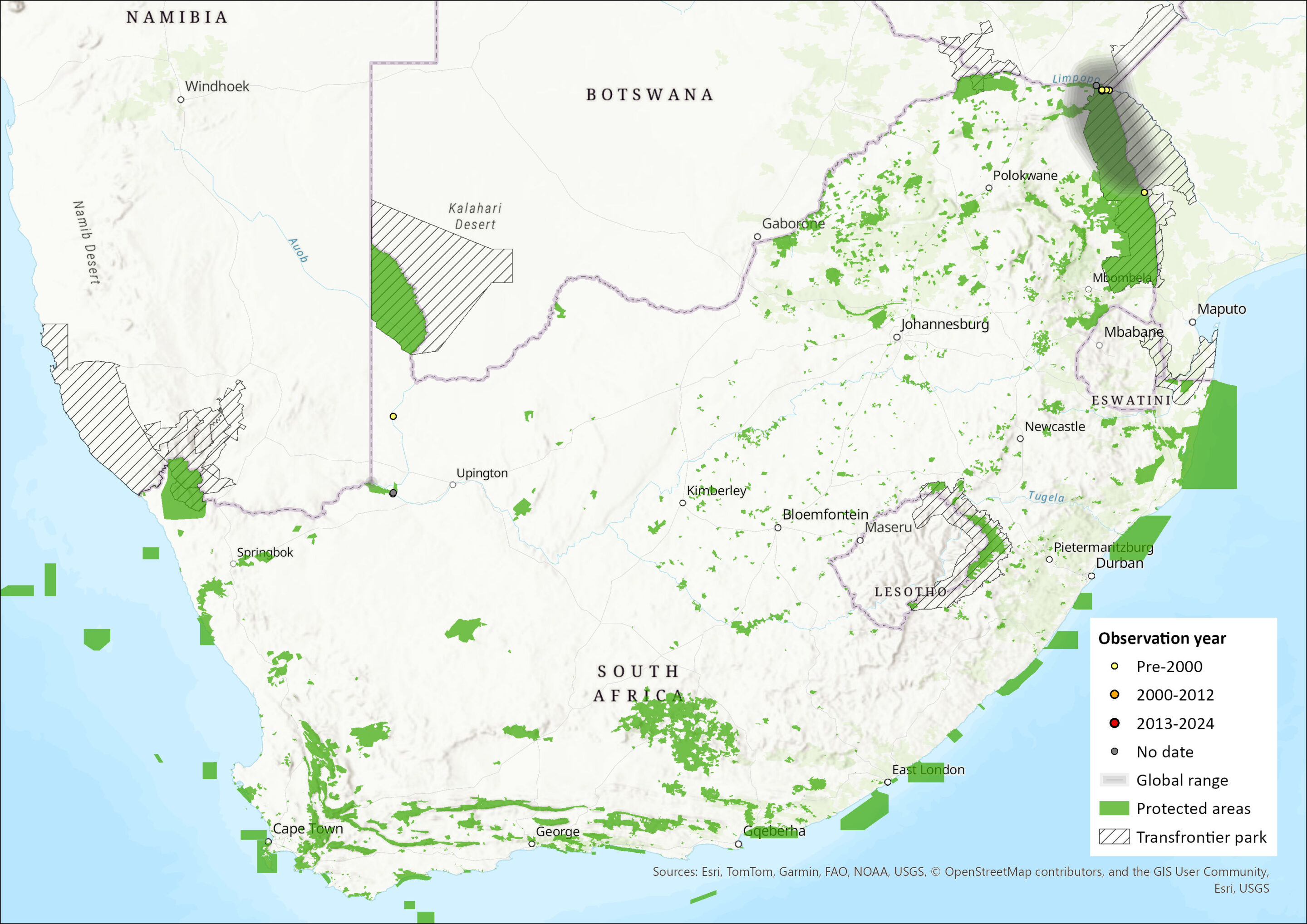 Figure 1. Distribution records for Rüppel’s Pipistrelle (Vansonia rueppellii) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Rüppel’s Pipistrelle (Vansonia rueppellii) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Algeria | Extant | Native | – | – |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Egypt | Extant | Native | – | – |
| Egypt -> Sinai | Extant | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Iraq | Extant | Native | – | – |
| Israel | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Kuwait | Presence Uncertain | Native | – | – |
| Liberia | Extant | Native | – | – |
| Libya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mauritania | Extant | Native | ||
| Morocco | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Tunisia | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Yemen | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
To date, no empirical studies have investigated the potential impacts of climate change on this species. Vansonia rueppellii appears to be highly dependent on water bodies where it forages by slow-hawking over water (Happold 2013). Sherwin et al. (2012) identified water stress and the aquatic aerial hawking feeding strategy as potential risk factors associated with climate change. This species may therefore be vulnerable to shifts in hydrological conditions induced by climate change.
Population information
This species is predominantly known from captures in mist nets in the assessment region, thus no data are available for population size or trends (ACR 2025). However, it is considered uncommon as it is not well represented in museums, with only 60 specimens examined in Monadjem et al. (2020a).
Current population trend: Unknown
Continuing decline in mature individuals? Unknown
Number of subpopulations: One
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: Unknown
All individuals in one subpopulation: Presumed to belong to one subpopulation in the assessment region
Number of mature individuals in largest subpopulation: Unknown
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Unknown
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Unknown
Probability of extinction in the wild within 100 years: Unknown
Population genetics
No population genetic work has been conducted on this species. Within the assessment region, P. rueppellii has been recorded from three localities, which could represent distinct subpopulations (Bushmanland, the Lowveld and the Mopane Bioregion). While classified as uncommon, no real measure of population size can be quantified; thereby limiting an estimate of the effective population size (Ne) for each subpopulation.
Habitats and ecology
It appears to be associated with large rivers and wetlands in dry savannah or woodland habitats (Skinner & Chimimba 2005; Monadjem et al. 2020a), often within arid regions. Within the assessment region, the species is recorded from Bushmanland, the Lowveld and the Mopane Bioregion. The single record from the Northern Cape was collected from Augrabies Falls National Park in a mist net set over a small rock pool located approximately 200 m from the main falls (Skinner & Chimimba 2005). Nearly all the southern African specimens were netted, so its roosting habits are not known. The species is presumed to exhibit plasticity in its roosting behaviour, as individuals from other parts of its range have been observed roosting under rocks, inside buildings, under peeling wall plaster, and behind notice boards (Allen and Loveridge 1933; Ansell, 1960; Hoogstraal, 1962). It often hunts over open water. For example, in the Okavango Delta, individuals were observed gleaning floating insects off the water surface in Xugana Lagoon and some individuals also landed in the water (Monadjem et al. 2020a). It is a clutter-edge forager and exhibits variability in its diet between regions (Monadjem et al. 2020a). For example, in the Kruger National Park, its diet included predominantly Coleoptera, while at Sengwa in Zimbabwe, its diet comprised Coleoptera, Lepidoptera, Trichoptera and Diptera (Monadjem et al. 2020a).
Ecosystem and cultural services: As this species is insectivorous, it may contribute to controlling insect populations that damage crops (Boyles et al. 2011; Kunz et al. 2011). Ensuring a healthy population of insectivorous bats can thus decrease the need for pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 8.1. Desert -> Desert – Hot | – | Suitable | – |
Life History
Generation Length: Unknown
Age at Maturity: Female or unspecified: Unknown
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Female: Mean forearm length (sexes combined) = 3.49 ± 0.24 cm (Monadjem et al. 2020b)
Longevity: Unknown
Average Reproductive Age: Unknown
Maximum Size (in cms): Maximum forearm length (sexes combined) = 3.93 cm (Monadjem et al. 2020a)
Size at Birth (in cms): Unknown
Gestation Time: Unknown
Reproductive Periodicity: Unknown
Average Annual Fecundity or Litter Size: Unknown
Natural Mortality: Unknown
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Unknown
Congregatory: Congregates for social foraging over water bodies elsewhere in its range Happold 2013). Also reported to roost in pairs under rocks in Egypt (Hoogstraal 1962), although it is uncertain whether this roosting behaviours presents in the population/s from the assessment region.
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence to suggest that this species is harvested or traded within the assessment region.
Local Livelihood: N/A
National Commercial Value: T This insectivorous species has not been recorded in any agroecosystems within the assessment region. Nevertheless, it likely plays a role in the control of general insect populations.
International Commercial Value: Unknown
End Use: None reported
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: N/A
Threats
This rarely encountered bat species is known from only a few localities within the assessment region. Its small body size and low wing loading (Monadjem et al. 2020a) suggest limited dispersal capability, potentially rendering it vulnerable to rapid environmental changes in its local habitat. The population size cannot be accurately determined, as no known roosts have been identified and only single individuals have been captured during surveys. However, it is unlikely that the total population exceeds 1,000 individuals given the sparse occurrence records despite intensive surveys in the northern regions of South Africa. Furthermore, the status of individuals from Augrabies Falls National Park remains uncertain, as no recent reports of the species from the area have been documented.
No specific major threats were identified for this species within the assessment region, as it was previously known only from protected areas. However, it has been recorded during recent field surveys at an open cast mine in the Limpopo province (Cory-Toussaint 2021). The extent of the population in anthropogenically modified areas remains uncertain. However, as in natural habitats, the species was closely associated with water bodies, specifically those linked to tailings dams. The species appears to be highly dependent on water sources for foraging; therefore, water pollutants and hydrological changes linked to climate change pose a threat to the species. Climate change has been identified as an increasing global threat to other bat species (Sherwin et al. 2012) and may similarly impact the food availability and energetic expenditure this species. If this species is found to occur more extensively outside protected areas, agricultural transformation and the subsequent loss of an insect prey base may be a potential threat. Further research is required to delineate the distributional limits, taxonomic status, roosting behaviour and other threats to this species.
Conservation
All occurrence records in the assessment region occur within the protected Kruger National Park and Augrabies Falls National Park (Monadjem et al. 2020), apart from the Venetia mine footprint (Cory-Touissant 2019). As such, no specific conservation interventions are necessary at present, however preservation of natural habitats and conservation of water courses associated with anthropogenically-modified habitats may be key to ensuring the species persistence in these areas. Globally, a study on the impacts of pesticides is required, especially ways in which the impact could be minimised (ACR 2025). Bat species would benefit from holistic land management that reduces pesticide use and conserves buffer strips of natural vegetation to sustain insect biomass.
Recommendations for land managers and practitioners:
- Promote and conserve natural habitats, especially riparian systems.
- Report newly identified synanthropic roosts/roosting sites to relevant authorities.
- The impacts of agricultural pesticides to bats, especially ways in which these threats may be minimised
Research priorities:
- Identify further occurrence records within the assessment region, through intensified sampling in areas predicted to harbour suitable habitats.
- Further field surveys to more accurately delimit its distribution within the assessment region.
- Identification of key roosting sites in the assessment region and investigate roosting behaviour.
- Integrative taxonomic revision to resolve the status of the putative subspecies.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution. This species is small and easily distinguished from other pipistrelle bats by its pure white underparts.
- Grounded bats should be reported to the nearest bat interest group, and any deceased individuals should be submitted to a reputable museum for documentation and research purposes.
Bibliography
ACR. 2024. African Chiroptera Report. 2024. AfricanBats NPC, Pretoria. i – xv; 1 – 1397.
Allen, G.M. and Loveridge, A. 1933. Reports on the scientific results of an expedition to the southwestern highlands of Tanganyika Territory. II. Mammals. Bulletin of the Museum of Comparative Zoology at Harvard College, 75(2), pp.45-140.
Benda, P., Hanak, V., Andreas, M., Reiter, A. and Uhrin, M. 2004. Two new species of bats (Chiroptera) for the fauna of Libya: Rhinopoma hardwickii and Pipistrellus rueppellii. Myotis 41–42: 109-124.
Boyles JG, Cryan PM, McCracken GF, Kunz TH. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Cory-Toussaint, D. 2019. The potential of bats as bioindicators for areas currently transformed by opencast mining within the Vhembe Biosphere Reserve, Limpopo Province, South Africa. PhD Thesis, University of Venda.
Cory-Toussaint, D., Taylor, P.J. and Barnhoorn, I.E. 2022b. Non-invasive sampling of bats reflects their potential as ecological indicators of elemental exposure in a diamond mining area, northern Limpopo Province, South Africa. Environmental Science and Pollution Research, 29(9), pp.13647-13660.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Happold, M. 2013. Pipistrellus rueppellii Rüeppell’s Pipistrelle; pp 647-649. In Happold, M and Happold, D.C.D. Happold (eds.). 2013. Mammals of Africa. Volume IV: Hedgehogs, Shrews and Bats. Bloomsbury Publishing, London, United Kingdom.
Helbig MT, Datzmann T, Mayer F, Fahr J. 2010. Molecular phylogeny of African “pipistrelle” bats (Vespertilionidae) suggests new clades, rearrangement of genera, and extensive cryptic diversity within species. Pages 168–169 in Horácek I, Benda P, editors. 15th IBRC – the Conference Manual: Programme, abstracts, list of participants. Volume of abstracts of the 15th International Bat Research Conference. Prague, Czech Republic.
Hoogstraal H. 1962. A brief review of the contemporary land mammals of Egypt (including Sinai. 1: Insectivora and Chiroptera. Journal of the Egyptian Public Health Association 37: 143-162.
Koubínová D, Irwin N, Hulva P, Koubek P, Zima J. 2013. Hidden diversity in Senegalese bats and associated findings in the systematics of the family Vespertilionidae. Frontiers in Zoology 10: 48.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2010. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020a. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis, Second Edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Demos, T.C., Dalton, D.L., Webala, P.W., Musila, S., Kerbis Peterhans, J.C. and Patterson, B.D., 2020b. A revision of pipistrelle-like bats (Mammalia: Chiroptera: Vespertilionidae) in East Africa with the description of new genera and species. Zoological Journal of the Linnean Society, 191(4), pp.1114-1146.
Monadjem, A., Richards, L.R., Decher, J., Hutterer, R., Mamba, M.L., Guyton, J., Naskrecki, P., Markotter, W., Wipfler, B., Kropff, A.S. and Dalton, D.L., 202. A phylogeny for African Pipistrellus species with the description of a new species from West Africa (Mammalia: Chiroptera). Zoological Journal of the Linnean Society, 191(2), pp.548-574.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J., 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Sherwin HA, Montgomery WI, Lundy MG. 2013. The impact and implications of climate change for bats. Mammal Review 43: 171–182.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
