
Roan Antelope
Hippotragus equinus

2025 Red list status
Vulnerable
Regional Population Trend
Declining
Change compared
to 2016
Downlisted
Overview
Hippotragus equinus – (É. Geoffroy Saint-Hilaire, 1803)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Hippotragus – equinus
Common Names: Roan Antelope (English), Bastergemsbok (Afrikaans), iMpofu (Zulu), Phofu (Sesotho), Tholo (Venda), Antilope chevaline, Antilope rouane, Hippotrague (French)
Synonyms: No Synonyms
Taxonomic Note:
Six subspecies have been described, largely defined by range gaps, geopolitical borders, or geographical features; however recent molecular evidence supports five evolutionary lineages (Gonçalves et al. 2021). A single evolutionary significant unit (ESU) is present within the assessment region and extends from South Africa to Zimbabwe, Botswana, Namibia and Angola (Alpers et al. 2004). Management authorities therefore work with ESUs and use H. e. subsp. equinus as the putative indigenous subspecies within the assessment region. Hippotragus e. subsp. cottoni should also be seen as an indigenous subspecies. The equinus/cottoni complex is treated as one by nature conservation authorities as there may be no significant genetic differences between the two. Many of the Roan Antelope in South Africa are H. e. subsp. cottoni or equinus x cottoni hybrids (especially on private properties). West African Roan Antelope (H. e. subsp. koba), native to North-West Africa in Senegal, Guinea-Bissau, Nigeria and Benin, is considered a divergent ESU from H. e. subsp. equinus (Havemann et al. 2016; Gonçalves et al. 2021), therefore any import or trade of this ESU into the assessment region is prohibited.
Red List Status: VU – Vulnerable, C1; D1 (IUCN version 3.1)
Assessment Information
Assessors: Muller, K.L.1, Kruger, J.2, da Silva, J.,1 & Selier, S.A.J.1
Reviewers: Nel, L.3 & Ferreira, S.M.4
Institutions: 1South African National Biodiversity Institute (SANBI); 2Department of Economic Development, Environment and Tourism Limpopo (LEDET), 3SA Hunters and Game Conservation Association, 4SANParks
Previous Assessors: Koen, J., Collins, K. & Nel, L.
Previous Reviewers: Parrini, F., Child, M.F. & Mallon, D.P.
Previous Contributors: Blignaut, C. & Havemann, C.
Assessment Rationale
This charismatic antelope exists at low densities within the assessment region, occurring in savannah woodlands and grasslands. The current wild population within the natural distribution range is estimated at 860 individuals, of which 516 – 602 are considered as mature individuals. The population estimate does not include state-owned protected and privately managed subpopulations outside the natural distribution range. State-owned protected areas in South Africa hold the legal mandate to conserve the country’s biodiversity, yet many are failing to effectively fulfil this role due to limited resources, capacity constraints, and increasing external pressures (Patel et al. 2023). As a result, there is growing recognition of the need to involve the private sector in broader conservation efforts. However, while private land can offer valuable opportunities for biodiversity protection, it cannot be expected to uphold the national conservation mandate. This is because the primary objective of most private landowners is to manage land for productivity—whether through tourism, hunting, or agriculture—rather than long-term ecological sustainability. However, these commercial activities often create valuable secondary conservation outcomes, demonstrating that economic use and ecological stewardship can be mutually reinforcing (Shumba et al. 2020; Taylor et al. 2021).
Currently (2020–2024), there are an observed 197 individuals (118 – 138 mature individuals) existing on eight state-owned protected areas within the natural distribution range. While there was a historical crash of 90% in the largest subpopulation in Kruger National Park (KNP) between 1986 and 1993, the subpopulation has since stabilised, with a reported total of 50 individuals in 2012, and currently the total is estimated at 58 (35 – 41 mature individuals). Overall, over the past three generations (2000 – 2024), based on available data for the remaining eight state-owned protected areas, there has been a net population reduction of 43%, which indicates an ongoing decline in state-owned protected areas but not as severe as the historical reduction. Further long-term data are needed to more accurately estimate the national population trend. The species is therefore listed as Vulnerable C1. Subpopulations of Roan Antelope in the private sector within their natural range were evaluated for wildness (SANBI, unpublished data, 2024). The assessment found that 50% of private subpopulations could be considered wild, contributing 297 individuals to the national total eligible for Red List assessment. However, it should be noted that the genetic integrity of such subpopulations remains uncertain. Nevertheless, assessed wild subpopulations in the private sector were included in this assessment until genetic protocols have been established to test and confirm the genetic integrity of privately managed Roan Antelope. While privately managed subpopulations appear to be increasing overall, the number and distribution of truly wild subpopulations in the private sector remain uncertain. This highlights the need for future evaluations to determine the wildness status of these populations more accurately. Therefore, this species warrants a delisting from Endangered D to Vulnerable D1, as the minimum confirmed wild mature population within the natural range has increased from < 250 individuals to < 1,000 individuals.
The main threats to this species are a reduction in habitat quality and lack of resources and capacity for effective management of state-owned protected areas. In the private sector, the loss of genetic diversity due to hybridisation with West African Roan Antelope (H. e. subsp. koba) remains a concern. Additionally, the lack of suitable incentives for the private sector to conserve wild subpopulations may result in a shift from extensive ranching to intensive breeding practices, which threatens the overall conservation efforts of Roan Antelope. Key interventions for this species should include drastic improvement in the management effectiveness of state-owned protected areas that keep Roan Antelope. Increasing the area of suitable habitat available including correct habitat management (for example managing for low-density species and de-stocking competing herbivores) within the natural distribution range under formal protection is also recommended. Incentives should be provided to the private sector for managing Roan Antelope subpopulations in a way that contribute to its long-term conservation of the species. A national metapopulation plan should be developed for sustaining the genetic diversity and resilience of the species and reducing the threat of further genetic contamination with divergent ESUs such as H. e. subsp. koba. Such interventions rely on partnerships with the private sector.
Regional population effects: Although this species is on the edge of its range within the assessment region, its range is not continuous. Private and state-owned subpopulations are isolated by fencing. The only natural dispersal routes that might exist are between the KNP, Zimbabwe and Mozambique (the Greater Limpopo Transfrontier Park) and perhaps through Botswana, but Roan Antelope are mostly restricted to northern Botswana (C. Havemann pers. comm. 2015). Similarly, Roan Antelope has not been observed in either the 2010 or 2013 aerial census of the Limpopo National Park, Mozambique, suggesting they have declined or are locally extinct (Stephenson 2013). Translocations of Roan Antelope within the private sector are primarily carried out to enhance genetic diversity and maintain population fitness, however it is uncertain whether state-owned protected areas follow similar management practises. Thus, immigration appears to be negligible and there is no confirmation that it takes place, and so no rescue effect is possible. It should be noted that the absence of a structured meta-population management framework inhibits the effective assessment of regional population effects.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: There is a change in the previous Red List category from Endangered D1 to Vulnerable D1 as the minimum confirmed wild mature population within the natural range has increased from < 250 individuals to < 1,000 individuals. The total number of Roan Antelope included in this assessment is 860 individuals, with an estimated 516 – 602 mature individuals. This figure encompasses both state-owned protected subpopulations and a larger sample of privately owned wild subpopulations not included in the previous assessment. Therefore, the perceived increase in the overall population estimate compared to the previous assessment is because of the larger sample of private subpopulations included. The number of Roan Antelope in state-owned protected areas has declined, dropping from 333 individuals across nine protected areas in 2016 to just 197 individuals in eight protected areas in 2024. Currently, 77% of the total estimated population consists of wild subpopulations on private land within the species’ natural distribution range.
Red List Index
Red List Index: Downlisted
Recommended citation: Muller KL, Kruger J, da Silva JM & Selier SAJ. 2025. A conservation assessment of Hippotragus equinus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The Roan Antelope formerly occurred widely in the savannah woodlands and grasslands of sub-Saharan Africa but has been eliminated from large parts of its former range (Havemann et al. 2016). For example, it has declined dramatically over the past two decades in Botswana, Namibia and Zimbabwe and has been almost entirely eradicated in Angola and Mozambique (summarised in Havemann et al. 2016). However, small remnant subpopulations may exist in Mozambique primarily for hunters (K. Collins pers. comm. 2016). It was also eliminated from Swaziland and later reintroduced to the privately owned Mkhaya Nature Reserve (East 1999; Chardonnet & Crosmary 2013). Roan Antelope have always been the scarcest of the antelope species in the Lowveld and higher lying bushveld areas, numbering just 100 in the former Transvaal (outside of KNP) in the early 1970s (Lambrechts 1974). Within South Africa, by the mid-twentieth century, Roan Antelope consisted of a subpopulation in KNP and a subpopulation in the New Belgium Block and its immediate environs in the Waterberg, Limpopo Province. In an attempt to save these last free roaming Roan Antelope from extinction, the former Transvaal Provincial Administration embarked on a mass capture operation during the period 1968–1972 to capture these individuals to provide formal protection in a proclaimed provincial protected area, Percy Fyfe Nature Reserve.
Roan Antelope naturally occur in Limpopo (and marginally Mpumalanga) bushveld areas through to the open savannahs in certain areas of North West, Northern Cape and Free State provinces. Although the type specimen for this species originates from the Northern Cape, it was historically extirpated from the region (Skinner & Chimimba 2005). However, subpopulations have since been reintroduced in the Northern Cape Province and have bred successfully. Two subpopulations were introduced into KwaZulu-Natal Province at Ithala and Weenen Nature Reserves but were removed prior to 2000. Although the area of occupancy (AOO) for Roan Antelope has been declining in key protected areas, such as KNP (Harrington et al. 1999), its overall occupancy in South Africa is expanding, driven by its status as a high-value game species within the private wildlife ranching sector, which has led to increased breeding and trade.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 750
Elevation Upper Limit (in metres above sea level): 1500
Depth Lower Limit (in metres below sea level): NA
Depth Upper Limit (in metres below sea level): NA
Map
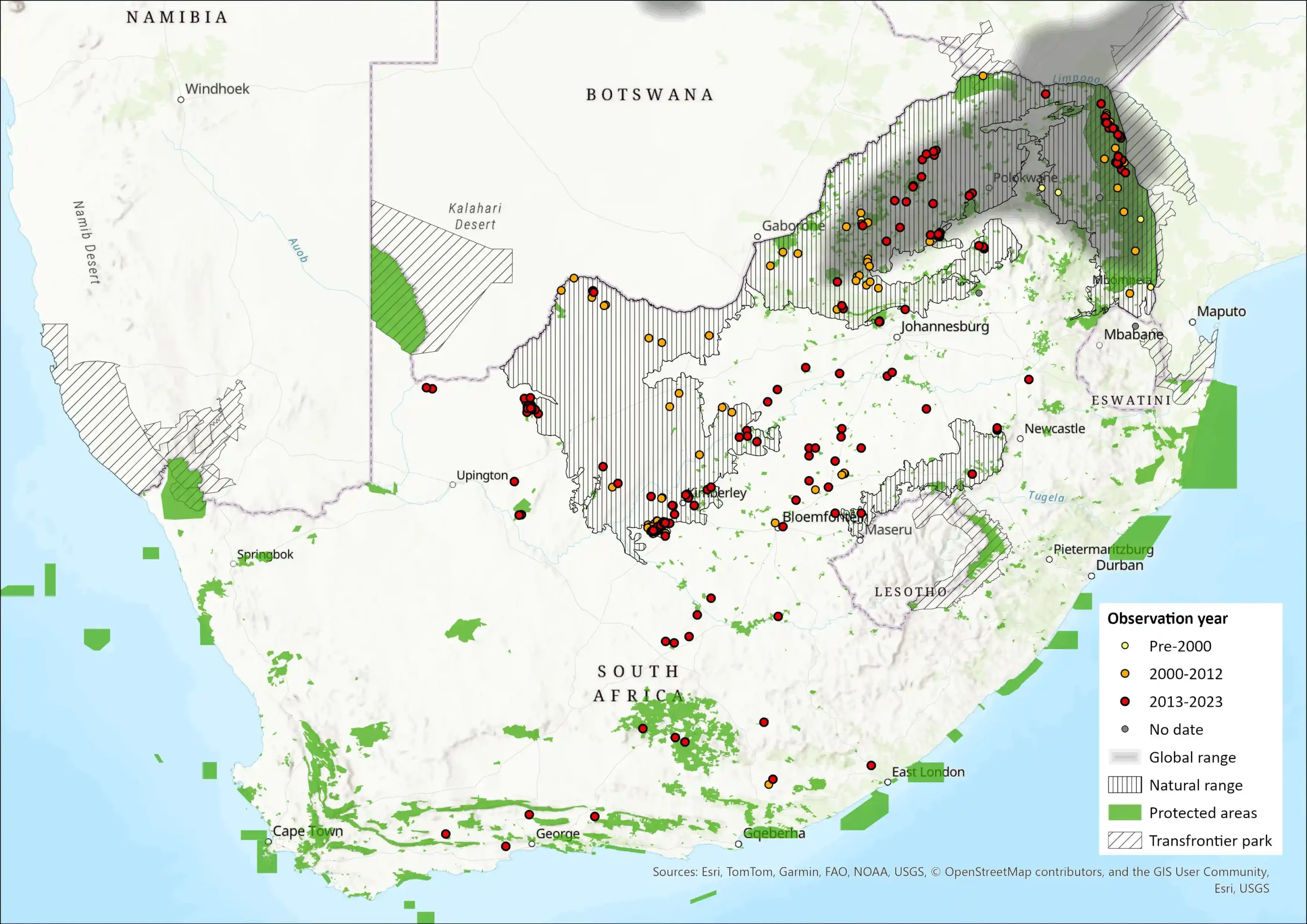 Figure 1. Distribution records for Roan Antelope (Hippotragus equinus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Roan Antelope (Hippotragus equinus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extinct Post-1500 | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Eritrea | Extinct Post-1500 | Native | – | – |
| Eswatini | Extant | Reintroduced | – | – |
| Ethiopia | Extant | Native | – | – |
| Gambia | Possibly Extinct | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Mozambique | Possibly Extinct | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: No
FAO Area Occurrence
FAO Marine Areas: No
Climate change
Southern Africa is largely a semi-arid region characterized by highly unpredictable rainfall patterns, with frequent occurrences of droughts and floods (Washington & Preston 2006). It is widely regarded as one of the regions that is most at risk from climate change due to its limited ability to adapt, especially in rural communities, alongside a strong reliance on rain-fed agriculture (IPCC 2014). Projections indicate that Africa will become progressively hotter and drier, with arid and semi-arid lands expected to expand by 5–8% by 2080 (Boko et al. 2007). Average seasonal temperatures are anticipated to rise, while extreme weather events such as El Niño-related disruptions, wildfires, and severe storms are likely to become more frequent in southern Africa (Davis 2011; IPCC 2014). Key climate change factors—including prolonged droughts, flooding, and rising temperatures—are expected to have significant impacts on the region’s wildlife (Chidumayo et al. 2011). Roan Antelope populations may be indirectly affected by increased temperatures and shifting rainfall patterns, which influence plant productivity through bottom-up ecological processes. These mechanisms suggest that herbivore populations are regulated by the availability and quality of forage (Gandiwa 2014). As a result, subpopulations may decline due to habitat loss within fenced areas, where shifts in suitable habitat occur but the animals’ ability to disperse is restricted by physical barriers.
Temperature and rainfall pattern changes:
Climate change is expected to make the western parts of South Africa drier, reducing the availability of suitable reintroduction sites and making in situ conservation more challenging (Sandler 2012). Roan Antelope exist at the edge of its range within South Africa, preferring high-rainfall dystrophic woodlands (>1000 mm/year) further north (Heitkönig & Owen-Smith 1998). Harrington et al. (1999) found that the general patterns of change in ungulate populations in northern KNP are related to rainfall patterns. Populations of most species expanded during the high rainfall period of the late 1970s and early 1980s, then declined during the later 1980s when rainfall was generally lower, especially during the severe drought of 1991/2. Following improved rainfall, Roan Antelope herds bred successfully and increased in numbers in the area where artificial water-points were closed. As temperatures rise and rainfall decreases (Boko et al. 2007), the species will face increasing habitat constraints, further limiting viable populations. Maruping-Mzileni et al. (2024) found that annual mean daily temperature range, temperature seasonality, minimum temperatures of the coldest month, and precipitation of the wettest month affects Roan Antelope distribution in Mokala National Park, Northern Cape Province.
Forage quantity and quality:
Roan Antelope are predominantly low-density grazers and reportedly favour open savanna or open grassland, with medium to tall grass (Heitkönig 1994). Shifts in temperature and rainfall patterns may reduce the quantity and quality of available forage. With lower rainfall, grasslands may become less productive, leading to nutritional stress for the species. Warmer temperatures could also accelerate plant senescence, reducing the availability of high-quality grazing material, which is crucial for sustaining populations in already marginal habitats, such as Roan Antelope within the assessment region.
Fire management:
Climate change may alter fire regimes by increasing the frequency and intensity of wildfires due to drier conditions. While fire plays a natural role in maintaining grassland ecosystems, more frequent or severe fires could reduce the availability of suitable grazing areas and alter habitat structure in ways that negatively impact Roan Antelope populations. Pacifici et al. (2015) investigated how fire management affects Roan, Sable Antelope (Hippotragus niger) and Tsessebe (Damaliscus lunatus) occurrence and density in KNP. The study found that a 20% increase in precipitation led to a 13-14% increase in the density of the Roan Antelope, while a 20% decrease in precipitation led to a decrease in the density of the species by roughly 15%. At the same time, the authors noted that the amount of yearly precipitation can increase the inconsistency in the density of the Roan Antelope. Overall, Pacifici et al. (2015) concluded that Roan Antelope is the species most resilient to climate change among the three species of antelope studied. However, a fire management plan that involves spatial alternation between the areas preferred by the Roan Antelope as a means of maintaining the quality of pastures is recommended.
Invasive species:
Drier conditions may also favour the spread of invasive plant species, which can outcompete native vegetation and reduce the quality of grazing resources. Invasive plants often have lower nutritional value and can alter ecosystem dynamics (Milton 2004; O’Connor & van Wilgen 2020), making it harder for Roan Antelope to find suitable forage.
Population information
Overall, the total number of Roan Antelope considered for this assessment is 860 individuals, representing 516 – 602 mature individuals, which includes both state-owned and privately owned wild subpopulations. This is only a perceived increase from the previous assessment (estimating a total of 218 – 294 mature individuals in 2016), mainly due to a larger sample of private subpopulations included in this assessment. However, a continued decline in subpopulation numbers is observed in state-owned protected areas. Roan Antelope subpopulation estimates in state-owned protected areas have declined from approximately 333 individuals in nine state-owned protected areas (2016) to 197 individuals in eight state-owned protected areas at the end of 2024. Wild Roan Antelope in private ownership within the natural distribution range make up 77% of the current total population estimate.
State-owned: Approximately 197 Roan Antelope currently (2020 – 2024) occur on eight state-owned protected areas that fall within the natural distribution range (Table 1). Mature population structure is inferred to be 60–70% based on a 63% mature herd structure as suggested by Perrin and Taolo (1998). This yields a minimum mature population size of 118 – 138 individuals in state-owned protected areas. Generation length is calculated as 8.4 years (Pacifici et al. 2013). Over three generations (2000 to 2024), based on available data for eight state-owned protected areas, there has been a net population reduction of 43%, which indicates a continuing decline of the species in state-owned protected areas. The subpopulation in KNP declined from 450 to approximately45 individuals between 1986 and 1993, a 90% decline over less than a decade (Harrington et al. 1999). However, subpopulation numbers in KNP increased from 22 to 50 individuals between 2002 and 2012 (Ferreira et al. 2013) and are currently (2023) estimated at 58 individuals (37 – 41 mature individuals; Greaver et al. 2024). A total of four nature reserves in the Limpopo Province currently (2020-2024) keep Roan Antelope subpopulations. As of 2024, the subpopulations at Nylsvlei Nature Reserve and Schuinsdraai Nature Reserve are functionally extinct, with only a single individual remaining in each reserve. A total of 22 individuals are present in Percy Fyfe Nature Reserve (in 2024) and 14 in Wonderkop Nature Reserve (estimated in 2020). Between 2016 and 2024, subpopulations in Percy Fyfe Nature Reserve and Wonderkop Nature Reserve have declined by 74% and 59%, respectively.
The subpopulation on Mokala National Park, Northern Cape Province, was reintroduced in 2006 with a founder size of six and subsequent reintroductions of 37 individuals (2007) and eight individuals (2009/2010). In 2016, the subpopulation was estimated at 60 individuals after some were lost to drought (C. Bissett, unpubl. data). Currently (2024), the subpopulation in Mokala National Park is estimated at 44 individuals based on an aerial census and 50 individuals from ground counts. Therefore, a slight decline in this subpopulation is observed between 2016 and 2024. Dronfield Nature Reserve, Northern Cape Province, currently (2023) keeps 49 Roan Antelope. The subpopulation on Sandveld Nature Reserve, Free State Province, has experienced a decline from 32 individuals in 2018 to only two remaining in 2025 (B. Qekwana, DESTEA pers. comm. 2025).
At least two state-owned protected subpopulations of Roan Antelope have become locally extinct since the previous Red List assessment in 2016. Kgaswane Mountain Nature Reserve is the only state-owned protected area in North West Province that once kept Roan Antelope. However, since 1999, the subpopulation has fluctuated between three and five individuals (Nel 2015) and is currently (2023) considered as locally extinct within the reserve. Seekoeivlei Nature Reserve, Free State Province, had two individuals in 2018, however, no Roan Antelope were recorded in the reserve in 2025.
Table 1. Summary of population size estimates for Roan Antelope (Hippotragus equinus) in state-owned protected areas. This is based on available data only and thus may underestimate total numbers.
| Province | Number of state owned protected areas | Subpopulation total (2020 – 2024) |
| Free State | 1 | 2 |
| Limpopo | 4 | 38 |
| Limpopo/Mpumalanga | 1 | 58 |
| North West | 0 | 0 |
| Northern Cape | 2 | 99 |
| Total | 8 | 197 |
Private: There are many more Roan Antelope present on private land across the assessment region, however, exact numbers in all provinces remain uncertain, mainly due to a lack of available data from provincial authorities. Rough population estimates in 2013 suggested that at least 3,500 individuals are managed on private wildlife ranches (Bezuidenhout 2013). The North West Department of Economic Development, Environment, Conservation & Tourism (DEDECT) reported a total number of 1,490 Roan Antelope present on 66 private wildlife ranches at the end of 2024 based on permit data (J. Power, DEDECT, pers. comm. 2025). Between January 2024 and March 2025, a total of 975 Roan Antelope are present on a number of private wildlife ranches in the Free State Province (B. Qekwana, DESTEA, pers. comm. 2025).
However, there is ongoing debate about whether subpopulations on private wildlife reserves and wildlife ranches can be considered as wild, given the way they are managed. Subpopulations dependent on direct intervention are not considered wild if they would go extinct within 10 years without intensive management (IUCN Standards and Petitions Subcommittee 2014). A preliminary analysis in 2016 (N = 26 subpopulations nationwide; EWT unpubl. data) into the proportion of private subpopulations that can be considered wild indicated that only 0.8 – 5% of individuals (14 – 88 individuals) could be considered wild (Child et al. 2019), and therefore eligible for inclusion in the Red List, as most were kept in areas smaller than reported home range sizes and managed intensively. In 2024, a separate follow-up assessment of the wildness of subpopulations (N = 25) on a total of 17 private wildlife ranches was conducted using the framework developed by Child et al. (2019). One property indicated that they manage West African Roan Antelope (H. e. subsp. koba), therefore, due to the uncertainty regarding hybridisation and introgression of this specific subpopulation managed on the property, it was excluded from the wildness assessment. Subsequently, a total of 24 Roan Antelope subpopulations in the private sector were assessed (Table 2). The results indicated that 50% of the assessed subpopulations could be considered wild, with 83% occurring within their natural distribution range (SANBI, unpubl. data, 2024). This assessment contributes an additional 297 individuals within the natural distribution range to the total population count in state-owned protected areas. Within the Northern Cape there are two private reserves that manages Roan Antelope in extensive systems and can thus be considered wild and eligible for inclusion in the Red List. These properties (500 km2 and 900 km2 in size) manages 257 and 109 Roan Antelope respectively. Thus, in total, 860 individuals (516 – 602 mature individuals) qualify for inclusion in the Red List.
Table 2. Summary of assessed subpopulations (N = 24) of Roan Antelope (Hippotragus equinus) in the private wildlife industry. This is based on available data only and thus may underestimate total numbers.
| Prov. | Inside natural distrib. range | No. assessed subpop. | Total | No. subpop. considered wild | Wild subpop. total |
| Eastern Cape | No | 4 | 53 | 2 | 15 |
| Free State | Yes | 3 | 59 | 3 | 59 |
| Limpopo | Yes | 7 | 242 | 6 | 213 |
| North West | Yes | 1 | 40 | 0 | – |
| Northen Cape | Yes | 9 | 226 | 1 | 25 |
| Total | 24 | 620 | 12 | 312 |
Roan Antelope have been introduced into a number of reserves, both state-owned and private, outside of the species natural distribution range. Based on current data, at least 103 individuals occur collectively on state-owned and private reserves, that could be considered wild (Table 3). However, these subpopulations do not fall within the natural distribution range of the species and thus cannot be included in the assessment (IUCN Standards and Petitions Subcommittee 2014). A total of 15 individuals from two subpopulations considered wild occur in the Eastern Cape Province. However, further assessment is required to determine the wildness extent of privately owned subpopulations in other provinces. A reported total of 87 Roan Antelope was managed on eight private wildlife ranches between 2020 and 2022 in the Western Cape Province, however, the extent of subpopulation wildness on these properties remains unknown. Only one state-owned protected area, located in the Eastern Cape Province reported to house one remaining Roan Antelope in 2022. However, it is important to note that this identified subpopulation, consisting of a single individual, does not contribute to the effective population size, as it cannot reproduce in isolation unless additional individuals are introduced.
Table 3. Summary of population size estimates for Roan Antelope (Hippotragus equinus) outside the natural distribution range. This is based on available data only and thus may underestimate total numbers.
| Province | Inside natural distribution range | Type | Number of protected areas/properties | Subpopulation total (2020-2024) |
| Eastern Cape | No | Private | 2 | 15 |
| Eastern Cape | No | State-owned | 1 | 1 |
| Western Cape | No | Private | 8 | 87 |
| Total (Private) | 10 | 102 | ||
| Total (State-owned) | 1 | 1 | ||
| Overall total outside natural distribution range | 11 | 103 |
Overall, Roan Antelope numbers have declined drastically in state-owned protected areas, from an estimated 333 individuals in nine protected areas in 2016, to a total of 197 in eight remaining state-owned protected areas. In contrast, numbers are increasing on private wildlife ranches, though they are likely underestimated due to limited accurate data. Since the initial wildness assessment was performed in 2016, the proportion of privately managed subpopulations that can be considered wild has increased from between 0.8 – 5% to 50%. Speculative reasons for this stark increase either may be from overall shifts in management regimes in the private sector, or from differences in data collection and analysis methods between the wildness assessments performed in 2016 and 2024. More information is needed to determine the cause for the increase in the proportion of wild subpopulations in the private sector. Moreover, uncertainty persists regarding the national wild status of managed subpopulations and their potential inclusion in future Red List assessments, highlighting the need for private sector involvement in wildness evaluations.
Population Information
Continuing decline in mature individuals?: Unknown
Qualifier: Estimated
Justification: Based on uncertainty of subpopulations included in the previous assessment and some subpopulations added in the current assessment that were not included in the previous assessment. Lack of resources, capacity and expertise for efficient management in state-owned protected areas are projected and suspected to cause continuing decline of mature individuals. In contrast, subpopulations within the private sector are suspected to have increased since the previous assessment.
Extreme fluctuations in the number of subpopulations: No
Justification: The number of subpopulations in total increased since the previous assessment, mainly due to assessed wild subpopulations in the private sector which were not included in the previous assessment. Number of subpopulations within state-owned protected areas seem stable.
Continuing decline in number of subpopulations: Unknown
Qualifier: Estimated
Justification: The subpopulations occur in KNP, provincial nature reserves and on private wildlife ranches. Number of subpopulations on state-owned protected areas have declined from nine (2016) to six (2020 – 2024) if subpopulations that are considered functionally extinct are excluded. Subpopulations on private land increased because of an increased demand for Roan Antelope on private wildlife ranches and the number of subpopulations that can be considered wild are also increasing.
All individuals in one subpopulation: No
Number of mature individuals in largest subpopulation: 38
Number of subpopulations: 20
Justification: Subpopulations are defined as any fenced area, as there is little exchange between these areas and no natural dispersal. Many more subpopulations exist on private protected areas and wildlife ranches.
Subpopulation details: A total of eight state-owned protected areas house subpopulations, while a total of 12 subpopulations assessed in the private sector are considered wild.
Number of mature individuals: 516 – 602
Subpopulation trend: Increasing
Qualifier: Estimated
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: 10%
Population genetics
A recent phylogeographic study conducted across the full range of H. equinus uncovered five evolutionary lineages (or evolutionarily significant units; ESUs), with a single Southern ESU comprised of individuals from Zimbabwe, South Africa, Botswana, Namibia and Angola (Gonçalves et al. 2021). Consequently, a single ESU is naturally present within the assessment region. However, evidence of hybridisation between Roan Antelope naturally occurring in South Africa (H. e. subsp. equinus) and those of West African decent (H. e. subsp. koba) has been found (van Wyk et al. 2019), which is problematic. The full extent of this problem is not fully known, hence more extensive molecular studies are needed incorporating individuals from private and protected areas across South Africa. Additionally, an estimate of effective population size for the metapopulation is needed (excluding hybrid individuals which could skew the results) to get a better understanding of the genetic health of the species in the assessment region.
Habitats and ecology
Roan Antelope inhabit savanna woodlands and grasslands within the bushveld and Lowveld of southern Africa and prefer habitats with a cover of high grasses and woody plants (Dorgeloh 1998; Knoop & Owen-Smith 2006), which play an important role for both grazing and calving (Chardonnet & Crosmary 2013). As such, they may be especially sensitive to changes in grass height and composition, as Roan Antelope rely on grass to camouflage their young and for foraging (Havemann et al. 2016). Roan Antelope are most abundant in moist or dystrophic savannas and sandveld woodlands where soils are predominantly infertile (Heitkönig & Owen-Smith 1998), such as Terminalia sericea and Philenoptera nelsii woodlands. They are water-dependent grazer/browsers, foraging at the boundary between ephemeral wetland and savannas, for example, in the northern plains of KNP (Kröger & Rogers 2005). Roan Antelope currently only occur in the northern plains of KNP (Owen-Smith et al. 2012) but previously occurred throughout the national park. Vlei grasslands are a key resource area in certain areas like the KNP, where Themeda triandra and Panicum maximum are important key resource grasses used during the dry season (Knoop & Owen-Smith 2006). Roan Antelope also show a preference for sandveld woodlands with predominantly infertile soils (Heitkönig & Owen-Smith 1998). Habitats that have low densities of competitor and predator species appear to be crucial for their survival (Havemann et al. 2016).
Roan Antelope are considered a low-density species, naturally occurring in small, scattered populations across large home ranges. Their survival and reproductive success are closely linked to maintaining sufficient interspecies distance, as they are highly sensitive to the presence of other large herbivores. McLoughlin & Owen-Smith (2003) analysed the decline of Roan Antelope populations in KNP. The authors found that increased predation pressure, potentially exacerbated by the presence of other herbivore species, was a significant factor in the population decline. The research suggests that the proximity of other species may disrupt the spatial behaviour of Roan Antelope, making them more susceptible to predators and affecting their access to resources.
Roan Antelope are susceptible to a range of diseases, with theileriosis posing a significant threat. Theileria is a tick-borne protozoan parasite transmitted by vectors such as Rhipicephalus appendiculatus and R. evertsi evertsi. Theileriosis can cause severe illness or death in susceptible hosts, especially when animals are moved into areas where they have no prior exposure and, therefore, no immunity. In Roan Antelope, Theileria infections can lead to symptoms such as high fever, lethargy, and anaemia, and are often fatal if not promptly diagnosed and treated (Clift et al. 2022). The risk is heightened in areas where tick populations are high or where other livestock or wildlife serve as reservoirs for the parasite (Steyl et al. 2012). As such, careful disease screening, tick control, and quarantine protocols are essential components of responsible Roan Antelope management, particularly during translocation or in intensive breeding operations. In the private sector, managing theileriosis in Roan Antelope involves a combination of preventative measures and prompt therapeutic interventions. Regular acaricide treatments and habitat management to reduce tick populations are standard practices. Therapeutically, antiprotozoal agents such as buparvaquone are commonly used, with effectiveness closely tied to early detection and administration. However, while these interventions can significantly improve survival in managed settings, there is concern that chemically treated individuals may have reduced resilience in natural ecosystems. Animals that survive due to medical intervention may lack natural immunity, potentially increasing their vulnerability upon release or in areas with high disease pressure. As noted by Steyl et al. (2012), treatment may save individuals in the short term but does not replace the role of natural selection in developing population-level resistance. This has implications for the long-term viability of released or free-ranging Roan Antelope populations managed through intensive veterinary care.
Ecosystem and cultural services: Roan Antelope is a flagship species in the Waterberg region. It is also a valuable international hunting species.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 4.7. Grassland -> Grassland – Subtropical/Tropical High Altitude | – | Suitable | – |
Life History
Generation Length: 8.4 years
Age at Maturity: Female or unspecified: 2 – 3 years
Age at Maturity: Male: 3 – 4 years
Size at Maturity (in cms): Female: 130 – 150
Size at Maturity (in cms): Male: 140 – 160
Longevity: 15 – 17 years (wild); 20 – 25 years (captivity)
Average Reproductive Age: 2 – 3 years (female); 5 – 6 years (male)
Maximum Size (in cms): 160
Size at Birth (in cms): 65 – 70
Gestation Time: 276 – 287 days
Reproductive Periodicity: Calving interval of 10 to 10.5 months
Average Annual Fecundity or Litter Size: 1
Natural Mortality: 15 – 17 years
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Roan Antelope exhibit sedentary to semi-nomadic movement patterns, typically staying within a well-defined home range but moving in response to resource availability. Their movement patterns are influenced by factors such as seasonal changes, water availability, and habitat quality (Skinner & Chimimba 2005).
Aggregatory: Yes
Systems
System: Terrestrial
General Use and Trade Information
Roan Antelope is a high-value game species that is commercially bred and traded in South Africa, primarily for international hunting and live sales (Taylor et al. 2016). As a result, a significant portion of the private sector manages Roan Antelope in intensive to semi-extensive systems across the country (Taylor et al. 2016). Private sector management typically separates subpopulations into breeding and wild stock, often on the same property. Breeding stock is generally kept in camp systems, which range in size from less than 1 km2 to over 5 km2 (based on 24 subpopulations; SANBI, unpubl. data 2024). These systems may involve disease control and genetic selection for specific traits, with animals frequently sold at auctions. Genetic selection entails pairing bulls with desired traits with a herd to produce offspring with similar characteristics; however, it does not involve genetic manipulation or engineering, which refers to altering an organism’s DNA using laboratory technologies (IFOAM 2014). Wild stock, on the other hand, consists of free-ranging Roan Antelope that roam across the entire wildlife ranching property without disease management or genetic selection. These populations are primarily managed for international hunting.
Between 2016 and 2024, a total of 2,304 Roan Antelope were sold live on formal auctions across South Africa. At the end of 2024, a total of 316 Roan Antelope were sold on auction, with an estimated value of R19 million, making up 3% of the total rare game sold on auction that year. There is a moderate demand for Roan Antelope bulls/hunting animals. Of all bulls/hunting animals sold on auction, the demand for Roan Antelope is the lowest of all species. Roan Antelope bulls are currently (2024) selling for between R30,000 and R60,000, with prices varying based on horn size and age of the animals (AWA 2024). Between 2016 and 2023, a total of 1,670 Roan Antelope were hunted by international hunters. Number of international hunts declined in 2020 due to the COVID-19 pandemic, but subsequently increased, reaching a total 258 by the end of 2023. International hunting market prices have declined from 2018 onwards, followed by somewhat increasing market trends between 2022 and 2023 (DFFE Professional Hunting Registers, unpubl. data; Fig. 1).
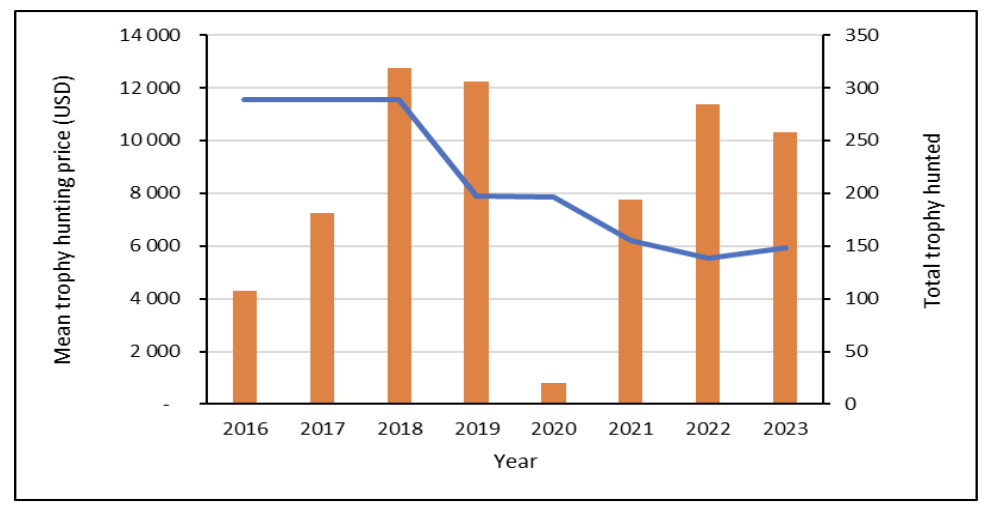 Figure 1. Mean international hunting price (blue line) and total international hunts of (orange bars) Roan Antelope between 2016 and 2023.
Figure 1. Mean international hunting price (blue line) and total international hunts of (orange bars) Roan Antelope between 2016 and 2023.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| No | Poaching for bushmeat may be occurring on a small scale but is not a major threat. | Yes | Hunting and live animal sales. Hunting on local, national and international scale. |
National Commercial Value: Yes
International Commercial Value: Yes
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | – | true | – | – |
| 15. Hunting/specimen collecting | – | true | true | – |
| 17. Other (free text) | true | Live animal sales at auctions |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Roan Antelope subpopulations managed in the private wildlife ranching sector are frequently harvested mainly for international hunting purposes due to its significant economic value. Offtake/harvest in the form of hunting from subpopulations in state-owned protected areas does not occur.
Threats
Throughout Africa, the Roan Antelope has been eliminated from large parts of its former range because of poaching and loss of habitat due to the expansion of human settlements and now survives mainly in protected areas. Within the assessment region, all wild Roan Antelope subpopulations exist in fenced protected areas, with additional stock existing in private wildlife ranches and wildlife reserves. Habitat loss and degradation through ecological mismanagement within state-owned protected areas is the greatest ongoing threat to the species, however, the loss of genetic integrity of the native subspecies remains a concern mainly in the private sector that needs to be addressed.
Poor habitat management: Effective habitat management in state-owned protected areas is limited due to insufficient resources such as funding and human capacity (Clements et al. 2022). Roan Antelope may be highly sensitive to habitat changes resulting from both natural disturbances, such as high densities of elephants (Loxodonta africana) and other large mammals, and human activities (Codron et al. 2009). This sensitivity is likely due to Roan Antelope being low-density species, their patchy distribution, specialised vegetation preferences, and feeding behaviour. Although, further research is needed to confirm these effects. Their ability to adapt to such disturbances may be constrained by their mating systems and feeding strategies. In particular, they may be vulnerable to changes in grass height and composition, as they depend on grass for both foraging and concealing their young (Havemann et al. 2016). Elephant populations within KNP are showing increasing trends, likely negatively affecting other species such as Roan Antelope in the park (Louw et al. 2021). Additionally, Roan Antelope are sensitive to competition and cannot co-exist with high densities of game or cattle. In KNP, the construction of artificial water-points in semi-arid regions increased predation rates by lions (Panthera leo), following the influx of zebra (Equus quagga) and blue wildebeest (Connochaetes taurinus; Harrington et al. 1999); and also due to the associated habitat degradation and competition with other herbivores (Harrington et al. 1999; Grant & van der Walt 2000; Grant et al. 2002). However, the closing of artificial water-points in these areas has not led to a recovery of Roan Antelope numbers (Ferreira et al. 2010), possibly due to an Allee effect where reduced herd vigilance causes increased juvenile mortality from predation and declining population numbers (Owen-Smith et al. 2012). This demonstrates that correct long-term habitat management of existing protected subpopulations are crucial in conserving Roan Antelope. Natural habitat of Roan Antelope in the private sector is fragmented by anthropogenic factors such as agricultural expansion, human settlements and impermeable game fences (Selier et al. 2018). There has been a loss of habitat quality in the private sector due to previous overstocking and areas being much smaller than the natural home ranges of the species (including some state-owned protected areas). However, a total of 50% of assessed breeding subpopulations in the private sector (based on 24 subpopulations; SANBI, unpubl. data) are housed in camp systems larger than the reported home range sizes for the species (>2 km2). This finding underscores the potential for including privately managed subpopulations in future Red List assessments.
Poor genetic management: The high economic value of Roan Antelope makes them desirable in the private wildlife industry. To mitigate losses from poaching and predation, they were often managed in breeding camps smaller than their natural home ranges and subjected to intensive management. This included genetic selection for specific traits such as horn length, supplementary feeding, and veterinary care. Such management regimes may compromise the genetic integrity of Roan Antelope, potentially reducing their suitability for reintroduction into less intensively managed systems (as seen with oribi Ourebia ourebi; Grey-Ross et al. 2009). In South Africa, several private wildlife ranches host subpopulations of West African Roan Antelope (H. e. subsp. koba), raising concerns about hybridisation with the native southern African Roan Antelope (H. e. subsp. equinus; van Wyk et al. 2019; Gonçalves et al. 2021). Indeed, a recent study performed by van Wyk et al. (2019) provided evidence of hybridisation and introgression between southern African Roan Antelope and West African Roan Antelope. The loss of genetic integrity poses a severe threat to the subspecies in South Africa (Barry 2003). For instance, in 2000, of the estimated 1,237 Roan Antelope in the country, only 520 belonged to the indigenous H. e. subsp. equinus, while the remainder were either exotic subspecies or hybrids (Barry 2003). However, the current genetic status of most privately managed populations remains uncertain, complicating permit allocations and hindering key enforcement interventions of strict translocation protocols. Further survey work and ground-truthing is needed to establish genetic integrity of private subpopulations and identify private subpopulations that can enhance the resilience of the overall wild population. Hybridisation among depleted subpopulations is a major biodiversity concern, as outbreeding depression can hinder recovery. For example, camera-trapping and molecular surveys recently documented introgressive hybridisation between the Giant sable antelope (Hippotragus niger subsp. variani) and sympatric Roan Antelope in Angola following severe wartime poaching (Vaz Pinto et al. 2016).
However, it should be noted that assessments of private sector subpopulations indicate that the proportion of wild subpopulations has increased since initial assessments in 2019 (see the “Population” section). Additionally, while a typical herd is 6–10 individuals (based on herd dynamics in KNP; Joubert 1970), recent assessment of subpopulations managed in the private sector indicate that 25% of subpopulations contain 10 or more individuals, and 75% contain 20 or more individuals (N = 24; SANBI, unpubl. data). While specific studies directly correlating herd size with genetic diversity in Roan Antelope are limited, van Wyk et al. (2019) and Gonçalves et al. (2021) highlight the importance of management practices and population dynamics in maintaining genetic variation in Roan Antelope. Further research focusing directly on herd size would be necessary to draw definitive conclusions. Consequently, these wild subpopulations may thus contribute to the overall conservation of Roan Antelope, although the underlying causes of this shift in management practices remain understudied. Moreover, costs incurred by the private sector that manages Roan Antelope subpopulations extensively are not currently incentivised. This may inhibit the shift from intensive to extensive management regimes and prevent the private sector from contributing to Roan Antelope conservation.
Conservation
Roan Antelope currently only remain in eight state-owned protected areas within the assessment region. The largest subpopulation that occurs in the Kruger National Park has remained stable since 2008 with a reported total of 50 individuals to a total of 58 individuals in 2023. Additionally, Mokala National Park and Dronfield Nature Reserve in the Northern Cape Province each house subpopulations of approximately 50 individuals. Subpopulations within remaining nature reserves in the Limpopo, Free State and North West provinces have all experienced stark declines since the previous Red List assessment in 2016 (see the “Population” section). Conservationists should therefore focus on conserving Roan Antelope in situ within national and provincial nature reserves and national parks within the natural distribution range of Roan Antelope. Contrastingly, the subpopulations considered wild within the private wildlife ranching sector seem to have grown (see the “Population” section). However, additional analysis is required to determine the factors driving this increase and to accurately evaluate the level of wildness in these private sector subpopulations. This highlights the importance of applying the wildness framework developed by Child et al. (2019) to incorporate wild subpopulations managed by the private sector into the regional Red List.
The establishment of new protected areas (or expansion of existing protected areas) with suitable habitat and improved management of such protected areas has been proposed as a key intervention to meet conservation goals. Conservation areas need to be large enough to support resilient subpopulations and provide a buffer to increasing human populations at reserve edges (Newmark 2008; Wittemyer et al. 2008). However, concerns have been raised regarding the lack of necessary resources, including funding and human capacity, to implement proposed expansion strategies in state-owned protected areas across Africa (Clements et al. 2022). As a result, there is growing acknowledgment of the potential for privately-owned land to play a significant role in meeting both international and regional conservation objectives (Kremen & Merenlender 2018; Taylor et al. 2021). One mechanism to achieve protected area expansion is to coordinate with the private sector adjoining protected areas in forming conservancies to expand the conservation estate or incentivising the restoration of key habitats through the transition from intensive to extensive management of the landscape and Roan Antelope subpopulations. This includes interventions such as decreasing artificial water-point provision, reducing grazing pressure and implementing an ecological fire regime (Dorgeloh et al. 1996; Owen-Smith 1996; Dorgeloh 1998) as well as implementing correct harvest management to maintain effective social units (Caro et al. 2009).
Additionally, climate change may ultimately undermine conservation efforts for this species, as it makes the western parts of the country drier (thus reducing the suitability of benign reintroduction sites), which makes in situ conservation more difficult (Sandler 2012). It must be remembered that this species exists at the edge of its range in the assessment region: it reaches its highest densities north of South Africa in high-rainfall (> 1000 mm/annum) dystrophic woodlands (Heitkönig & Owen-Smith 1998), so any drier areas are marginal habitats. Since southern Africa is expected to get drier as a consequence of global change (Boko et al. 2007), suitable natural habitats will decrease. Thus, conservation efforts for this species should be prioritised in parts of its core natural range to combat the effects of climate change. To achieve this, conservationists must understand both the extent and distribution of available habitat, requiring close collaboration with the private sector (as mentioned above).
Reintroductions and augmentation will only assist in the long term if well-managed and suitable habitat can be conserved. Mokala National Park and Dronfield Nature Reserve, Northern Cape Province, may be used to supplement/augment other state-owned protected subpopulations. Translocations and reintroductions in the private sector and state-owned protected areas should follow the recommendations proposed by Alpers et al. (2004) to avoid crossbreeding between ESUs. Although translocations of Roan Antelope between West and southern Africa is prohibited, it still occurs illegally. Movement of individuals around the remaining regions of the rRoan Antelope’s range is considered less of a conservation concern (Alpers et al. 2004), especially seeing as this species is at the edge of its range. A practical and effective management plan, together with stricter regulations on translocations is needed to optimise the habitat suitability and genetic integrity for free-ranging herds in the assessment region. Greater collaboration between private and public entities is essential, especially on translocation efforts. However, for this to be achieved, the management of state-owned protected areas must be enhanced to enhance the survival of introduced herds. To achieve cooperation with the private sector, the correct incentives and legislation are required for landholders to manage Roan Antelope as free-ranging herds rather than intensively managed animals and ensure the habitat is of good quality. This would also be lucrative for the international hunting industry as hunters would be assured of quality hunting individuals.
Recommendations for land managers and practitioners:
- A Biodiversity Management Plan (BMP) should be developed to inform a national translocation and reintroduction policy. A national translocation policy should then be implemented to iron out the inconsistencies caused by different provincial legislation. The BMP should also include habitat assessments for properties where Roan Antelope are to be translocated.
- Roan Antelope should not be reintroduced to provincial reserves from private properties, due to risk of hybridisation with West African Roan Antelope (H. e. subsp. koba). All translocations should be preceded by genetic testing. A formal risk assessment of the West African Roan Antelope was completed in March 2025, awaiting approval from the Department of Forestry, Fisheries and the Environment (DFFE). This risk assessment considers prohibitions of import and translocations of West African roan antelope on a national scale (SANBI unpublished).
- Protocols to maintain genetic diversity and integrity in Roan Antelope, especially in the private sector, similar to that developed for bontebok (Damaliscus pygargus pygargus), should be prioritised.
- Given the continuing decline of Roan Antelope in state-owned protected areas, it is recommended that a population viability analysis (PVA) be performed to enhance the accuracy of the national population estimate and inform future assessments.
- Wildlife Ranching South Africa (WRSA) set up a Roan Advisory Group to create a national database of Roan Antelope numbers, management practices and genetic origins, including registering private subpopulations, indicating levels of management and genetic status as a precondition to trade. However, this initiative is temporarily on hold, pending development of an information management system that will accommodate and streamline data capture. Provincial departments should collaborate more closely with private initiatives to strengthen their capacity and effectiveness.
- The private wildlife ranching industry should manage Roan Antelope extensively with minimal intervention (for example, no continual supplementary feeding or predator exclusion or veterinary care). To enable this, incentives should be designed to encourage the management of wild subpopulations. This can be achieved through regulatory and ecological benefits such as certification programs recognising sustainable management practices to enhance ecotourism opportunities and increase the value of wildlife-based revenue streams, including international hunting.
- Protected area expansion should be achieved in collaboration with the private sector and local communities to facilitate larger spaces for free-ranging Roan Antelope herds.
- Management effectiveness of state-owned protected areas hosting subpopulations of Roan Antelope must be drastically improved focusing on the specific management requirements such as expertise and funding for this species.
Research priorities:
- Field surveys to determine national population size and management practices for Roan Antelope on private land, including surveying potential suitable habitats for Roan Antelope conservation.
- Genetic studies are a priority to assess the extent of hybridisation between the different subspecies/ESUs in the private sector.
- Research should also focus on assessing the survival of intensively managed individuals upon reintroduction into wild systems and develop best practice guidelines for such reintroductions. Studies on Roan Antelope subpopulations that appear to be stable or increasing and understanding ecological aspects (home range, habitat utilisation, feeding behaviour) are vital for providing important information for use in management and mitigation plans in areas where roan antelope are declining.
Encouraged citizen actions:
Private landholders can contribute to conservation by removing fences, establishing conservancies, or transitioning from intensive to extensive management. This approach allows Roan Antelope to form wild, free-ranging herds within their natural distribution range, promoting their conservation.
Bibliography
African Wildlife Auctions (AWA). 2024. Wildlife industry trends & analysis for 2024. Available at: Wildlife Auctions
Alpers DL, van Vuuren BJ, Arctander P, Robinson TJ. 2004. Population genetics of the roan antelope (Hippotragus equinus) with suggestions for conservation. Molecular Ecology 13: 1771-1784.
Barry A. 2003. Translocation of roan antelope in South Africa and the effect this has had on the genetic diversity of the species. M.Sc. Thesis. University of Johannesburg, Johannesburg, South Africa.
Bezuidenhout R. 2013. High value game farming: How to get started. Farmers Weekly Magazine. Available at: https://www.farmersweekly.co.za/agri-business/agribusinesses/farming-high-value-game-species/.
Boko M, Niang I, Nyong A, Vogel C, Githeko A, Medany M, Osman-Elasha B, Tabo R, Yanda P. 2007. Africa. Climate Change 2007: Impacts, Adaptation and Vulnerability. In: ML Parry, OF Canziani, JP Palutikof, PJ van der Linden, CE Hanson (eds.) Contribution of Working Group II to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, pp. 433–467. Cambridge University Press, Cambridge, UK.
Caro TM, Young CR, Cauldwell AE, Brown DDE. 2009. Animal breeding systems and big game hunting: models and application. Biological Conservation 142: 909-929.
Chardonnet P, Crosmary W. 2013. Hippotragus equinus. In: JS Kingdon, M. Hoffmann (eds.), The Mammals of Africa, Academic Press, Amsterdam, The Netherlands.
Chidumayo E, Okali D, Kowero G, Larwanou M (eds.). 2011. Climate change and African forest and wildlife resources. African Forest Forum, Nairobi, Kenya.
Child MF, Selier SJ, Radloff FG, Taylor WA, Hoffmann M, Nel L, Power RJ, Birss C, Okes NC, Peel MJ, Mallon D, Davies‐Mostert H. 2019. A framework to measure the wildness of managed large vertebrate populations. Conservation Biology 33(5): 1106-1119.
Clements HS, Child MF, Lindeque L, Lunderstedt K, de Vos A. 2022. Lessons from COVID-19 for wildlife ranching in a changing world. Nature Sustainability 5: 1040-1048.
Clift SJ, Martí-Garcia B, Lawrence JA, Mitchell EP, Fehrsen J, Martínez J, Williams JH, Steyl JCA. 2022. Theileriosis in naturally infected roan antelope (Hippotragus equinus). Veterinary Pathology 59(6): 1031-1046.
Codron D, Codron J, Lee-Thorp JA, Sponheimer M, Grant CC, Brink JS. 2009. Stable isotope evidence for nutritional stress, competition, and loss of functional habitat as factors limiting recovery of rare antelope in southern Africa. Journal of Arid Environments 73: 449-457.
Davis CL. 2011. Climate risk and vulnerability: A handbook for southern Africa. Council for Scientific and Industrial Research, Pretoria, South Africa, pp. 92.
Dorgeloh WG. 1998. Habitat selection of a roan antelope (Hippotragus equinus) in mixed bushveld, Nylsvlei Nature Reserve. South African Journal of Wildlife Research 47(2): 47-57.
Dorgeloh WG, van Hoven W, Rethman NG. 1996. Population growth of roan antelope under different management systems. South African Journal of Wildlife Research 26(4): 113-116.
East R. 1999. African Antelope Database 1998. IUCN, Gland, Switzerland and Cambridge, UK.
Ferreira S, Gaylard, A, Greaver, C, Hayes, J, Cowell C, Ellis G. 2013. Summary Report: Animal abundances in Parks 2012/2013. Scientific Services, SANParks, Skukuza, South Africa.
Ferreira S, Gaylard, A, Greaver, C, Herbst M. 2010. Animal abundances in Parks 2009/2010. Scientific Services, SANParks, Skukuza, South Africa.
Gandiwa E. 2014. Vegetation factors influencing density and distribution of wild large herbivores in a southern African savannah. African Journal of Ecology 52: 274-283.
Grey-Ross R, Downs CT, Kirkman K. 2009. Reintroduction failure of captive-bred oribi (Ourebia ourebi). South African Journal of Wildlife Research 39(1): 34-38.
Gonçalves M, Siegismund HR, van Vuuren BJ, Ferrand N, Godinho R. 2021. Evolutionary history of the roan antelope across its African range. Journal of Biogeography, 48: 2812–2827.
Grant CC, van der Walt JL. 2000. Towards an adaptive management approach for the conservation of rare antelope in the Kruger National Park-outcome of a workshop held in May 2000. Koedoe 43: 103-112.
Grant CC, Davidson T, Funston PJ, Pienaar DJ. 2002. Challenges faced in the conservation of rare antelope: A case study on the northern basalt plains of the Kruger National Park. Koedoe 45: 45–66.
Greaver C, Ferreira S, Simms C, Botha J, Crowhurst E. 2024. Estimates of animal species in the Kruger National Park – 2023. Aerial Survey Report – March 2024, South African National Parks (SANParks).
Harrington R, Owen-Smith N, Viljoen PC, Biggs HC, Mason DR. Funston PJ. 1999. Establishing the causes of the roan antelope decline in the Kruger National Park, South Africa. Biological Conservation 90(1): 69-78.
Havemann CP, Retief TA, Tosh CA, Bruyn PJ. 2016. Roan antelope Hippotragus equinus in Africa: A review of abundance, threats and ecology. Mammal Review 46: 144-158.
Heitkönig IMA. 1994. Feeding strategy of roan antelope (Hippotragus equinus) in a low nutrient savanna. Ph.D. Thesis. University of the Witwatersrand, Johannesburg.
Heitkönig IMA, Owen-Smith N. 1998. Seasonal selection of grass swards by roan antelope in a South African savanna. African Journal of Ecology 36: 57-70.
Intergovernmental Panel on Climate Change (IPCC) 2014. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change [Core Writing Team, R.K. Pachauri and L.A. Meyer (eds.)]. IPCC, Geneva, Switzerland, 151 pp.
International Federation of Organic Agriculture Movements (IFOAM). 2014. The IFOAM norms for organic production and processing. Version 2014. IFOAM Organics International, Germany.
IUCN Standards and Petitions Subcommittee. 2014. Guidelines for Using the IUCN Red List Categories and Criteria. Version 11.
Joubert SCJ. 1970. A Study of the Social Behaviour of the Roan Antelope, Hippotragus equinus equinus (Desmarest, 1804), in the Kruger National Park. M.Sc. Thesis. University of Pretoria, Pretoria, South Africa.
Knoop MC, Owen-Smith N. 2006. Foraging ecology of roan antelope: Key resources during critical periods. African Journal of Ecology 44: 228-236.
Kremen C, Merenlender AM. 2018. Landscapes that work for biodiversity and people. Science 362(6412): eaau6020.
Kröger R, Rogers KH. 2005. Roan (Hippotragus equinus) population decline in Kruger National Park, South Africa: Influence of a wetland boundary. European Journal of Wildlife Research 51(1): 25-30.
Lambrechts AW. 1974. The numerical status of sixteen game species in the Transvaal, excluding the Kruger National Park. Journal of the Southern African Wildlife Management Association 64: 1-37.
Louw AS, MacFadyen S, Ferreira S, Hui C. 2021. Elephant population responses to increased density in Kruger National Park. Koedoe 63(1): a1660.
Maruping-Mzileni NT, Bezuidenhout H, Ferreira S, Ramoelo A, Mapuru M, Munyai L, Erusan R. 2024. Implications of ecological drivers on roan antelope populations in Mokala National Park, South Africa. Diversity 16(6): 355.
McLoughlin CA, Owen-Smith N. 2003. Viability of a diminishing roan antelope population: Predation is the threat. Animal Conservation 6(3): 231–236.
Milton SJ. 2004. Grasses as invasive alien plants in South Africa: Working for water. South African Journal of Science 100(1): 69-75.
Nel P. 2015. Population estimates for large herbivores and predators in protected areas in the North West Parks Board November 2015. North West Parks Board.
Newmark WD. 2008. Isolation of African protected areas. Frontiers in Ecology and the Environment 6: 321-328.
O’Connor TG, van Wilgen BW. 2020. The impact of invasive alien plants on rangelands in South Africa In: BW van Wilgen, J Measey, DM Richardson, JR Wilson, TA Zengeya (eds.) Biological Invasions in South Africa. Springer, pp. 459-487.
Owen-Smith N. 1996. Ecological guidelines for waterpoints in extensive protected areas. South African Journal of Wildlife Research 26: 107-112.
Owen-Smith N, Chirimia GJ, Macandza V, Le Roux E. 2012. Shrinking sable antelope numbers in Kruger National Park: what is suppressing population recovery? Animal Conservation 15: 195–204.
Pacifici M, Santini L, Di Marco M, Baisero D, Francucci L, Grottolo Marasini G, Visconti P, Rondinini C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Pacifici M, Visconti, P, Scepi E, Hausmann A, Attorre F, Grant R, Rondinini C. 2015. Fire policy optimization to maximize suitable habitat for locally rare species under different climatic conditions: A case study of antelopes in the Kruger National Park. Biological Conservation 191: 313-321.
Patel T, Cowan O, Little I, Friedmann Y, Blackmore A. 2023. The state of provincial reserves in South Africa: Challenges and recommendations. The Endangered Wildlife Trust, Johannesburg, South Africa.
Perrin MR, Taolo C. 1998. Home range, activity pattern and social structure of an introduced herd of roan antelope in KwaZulu-Natal, South Africa. South African Journal of Wildlife Research 28: 27-32.
Sandler RL. 2012. The Ethics of Species: an Introduction. Cambridge University Press, Cambridge, UK.
Selier J, Nel L, Rushworth I, Kruger J, Coverdale B, Mulqueeny C, Blackmore A. 2018. An assessment of the potential risks of the practice of intensive and selective breeding of game to biodiversity and the biodiversity economy in South Africa. Gland, Switzerland: IUCN.
Shumba T, de Vos A, Biggs R, Esler KJ, Ament JM, Clements HS. 2020. Effectiveness of private land conservation areas in maintaining natural land cover and biodiversity intactness. Global Ecology and Conservation 22: e00935.
Skinner JD. Chimimba CT (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
South African National Biodiversity Institute (SANBI). unpublished. Risk analysis of Hippotragus equinus (É. Geoffroy Saint-Hilaire, 1803) for South Africa as per the risk analysis for alien taxa framework v2.0, approved by the South African Alien Species Risk Analysis Review Panel on 28 March 2025, pp. 30, http://dx.doi.org/10.5281/zenodo.15102314
Stephenson A. 2013. Aerial Wildlife Census Parque Nacionale do Limpopo 2013. Unpublished report.
Steyl, JCA, Prozesky L, Stoltsz WH, Lawrence JA, 2012. Theileriosis (Cytauxzoonosis) in Roan antelope (Hippotragus equinus): Field exposure to infection and identification of potential vectors. Onderstepoort Journal of Veterinary Research 79(1): e1-8.
Taylor WA, Child MF, Lindsey PA, Nicholson SK, Relton C, Davies-Mostert HT. 2021. South Africa’s private wildlife ranches protect globally significant populations of wild ungulates. Biodiversity Conservation 30(30): 4111–4135.
Taylor WA, Lindsey PA, Davies-Mostert H. 2016. An assessment of the economic, social and conservation value of the wildlife ranching industry and its potential to support the green economy in South Africa. The Endangered Wildlife Trust, Johannesburg.
Van Wyk AM, Dalton DL, Kotzé A, Grobler JP, Mokgokong PS, Kropff AS, Jansen van Vuuren B. 2019. Assessing introgressive hybridization in roan antelope (Hippotragus equinus): Lessons from South Africa. PLoS One 14(10): e0213961.
Vaz Pinto P, Beja B, Ferrand N, Godinho R. 2016. Hybridisation following population collapse in a critically endangered antelope. Scientific Reports 6: 18788.
Washington R, Preston A. 2006. Extreme wet years over southern Africa: Role of Indian Ocean sea surface temperatures. Journal of Geophysical Research 111: D15104.
Wittemyer G, Elsen P, Bean WT, Burton ACO, Brashares JS. 2008. Accelerated human population growth at protected area edges. Science 321: 123-126.
