
Rhodesia Horseshoe Bat
Rhinolophus rhodesiae

2025 Red list status
Vulnerable
Regional Population Trend
Unknown
Change compared
to 2016
New Assessment
Overview
Rhinolophus rhodesiae – Roberts, 1946
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – RHINOLOPHIDAE – Rhinolophus – swinnyi
Common Names: Rhodesia Horseshoe Bat (English)
Synonyms: No Synonyms
Taxonomic Note:
This species was previously grouped within R. swinnyi. However, based on bacular morphology, molecular sequencing and echolocation calls, it has been shown that the northeastern populations are a separate species, R. rhodesiae, first described by Roberts (1946) from Bezwe River in southern Zimbabwe, and clearly distinct from R. swinnyi, which is limited to the southeastern portion of the assessment region (Dool et al. 2016; Taylor et. al. 2018; 2019; Demos et al. 2019; Taylor et al. 2024). Most importantly, R. rhodesiae has an echolocation peak frequency of 100 kHz compared to 107 kHz in R. swinnyi (Dool et al. 2016) and it has a larger, more robust baculum compared to a fine and delicate baculum in swinnyi (Taylor et al. 2018; 2024). Species delimitations are not fully resolved in the genus Rhinolophus and some publications show discrepancies in phylogenetic relationships, disputing the species-level for R. rhodesiae (Demos et al. 2019). The main source of phylogenetic discrepancies is likely the type of marker(s) used. Specifically, the use of mitochondrial DNA has shown to reflect introgression between lineages (Puechmaille et al. 2011; Dool et al. 2016; Taylor et al. 2018; Demos et al. 2019; Benda et al. 2024). Indeed, mitochondrial DNA introgression has occurred between R. rhodesiae and R. simulator, resulting in near-identical cyt-b sequences in spite of clear phenotypical differences between these two species (Dool et al. 2016; Taylor et al. 2018). Nuclear introns have provided useful reconstructions of phylogenetic relationships specifically if used in sets and in combination with additional independent markers (Dool et al. 2016), however, they have thus not supported species-level (Dool et al. 2016; Demos et al. 2019). Phenotypic traits such as baculum morphology and acoustic data should be part of a comprehensive approach to resolve species/lineage delimitations in this genus and for this taxon.
Red List Status: VU – Vulnerable, C2a(i) (IUCN version 3.1)
Assessment Information
Assessors: Taylor, P.1, Richards, L.R.2, Balona, J.3, Howard, A.1, Richardson, E.4, van der Meulen, K.1 & da Silva, J.M.5
Reviewers: Bastian, A.6 & Smith, C.7
Contributors: Raimondo, D.5 & Patel, T.7
Institutions: 1University of the Free State, 2Durban Natural Science Museum, 3Gauteng and Northern Regions Bat Interest Group, 4Independent Consultant at Richardson & Peplow Environmental, 5South African National Biodiversity Institute, 6University of KwaZulu-Natal, 7Endangered Wildlife Trust
Assessment Rationale
Based on its currently defined range within the assessment region (Taylor et al. 2024), this species qualifies as Vulnerable C2a(i) as the regional population is suspected to consist of fewer than 10,000 mature individuals, with no subpopulation having more than 1,000 mature individuals. The species occurs fairly widely but patchily, associated with mountains in Zimbabwe, Mozambique, Malawi and Tanzania.
Regional population effects: The species occurs in several countries north of South Africa.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: New Assessment
Red List Index
Red List Index: New Assessment
Recommended citation: Taylor P, Richards LR, Balona J, Howard A, Richardson E, van der Meulen K & da Silva JM. 2025. A conservation assessment of Rhinolophus rhodesiae. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species has been recorded from the eastern parts of South Africa, much of Zimbabwe, and Mozambique, with additional scattered records further north in Malawi, Zambia, Democratic Republic of the Congo and Tanzania (including the island of Zanzibar) (Skinner & Chimimba 2005). Within the assessment region this species has been found in Limpopo, Mpumalanga and KwaZulu-Natal provinces of South Africa and in Eswatini (Monadjem et al. 2020). There are two known locations in the midlands of KwaZulu-Natal province, Hlabisa Forest and Ferncliff Cave, where both R. swinnyi and R. rhodesiae co-occur, emphasising the reproduction isolation between the two species. The extent of occurrence is 118,354 km2 and area of occupancy, according to the IUCN method of using a 2x2km grid, is 92km2, with the species occurring in 23 grid cells. Using a larger grid size of 10×10 km gives an AOO of 2,300 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 13 m asl (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 1,536 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: (Not specified)
Map
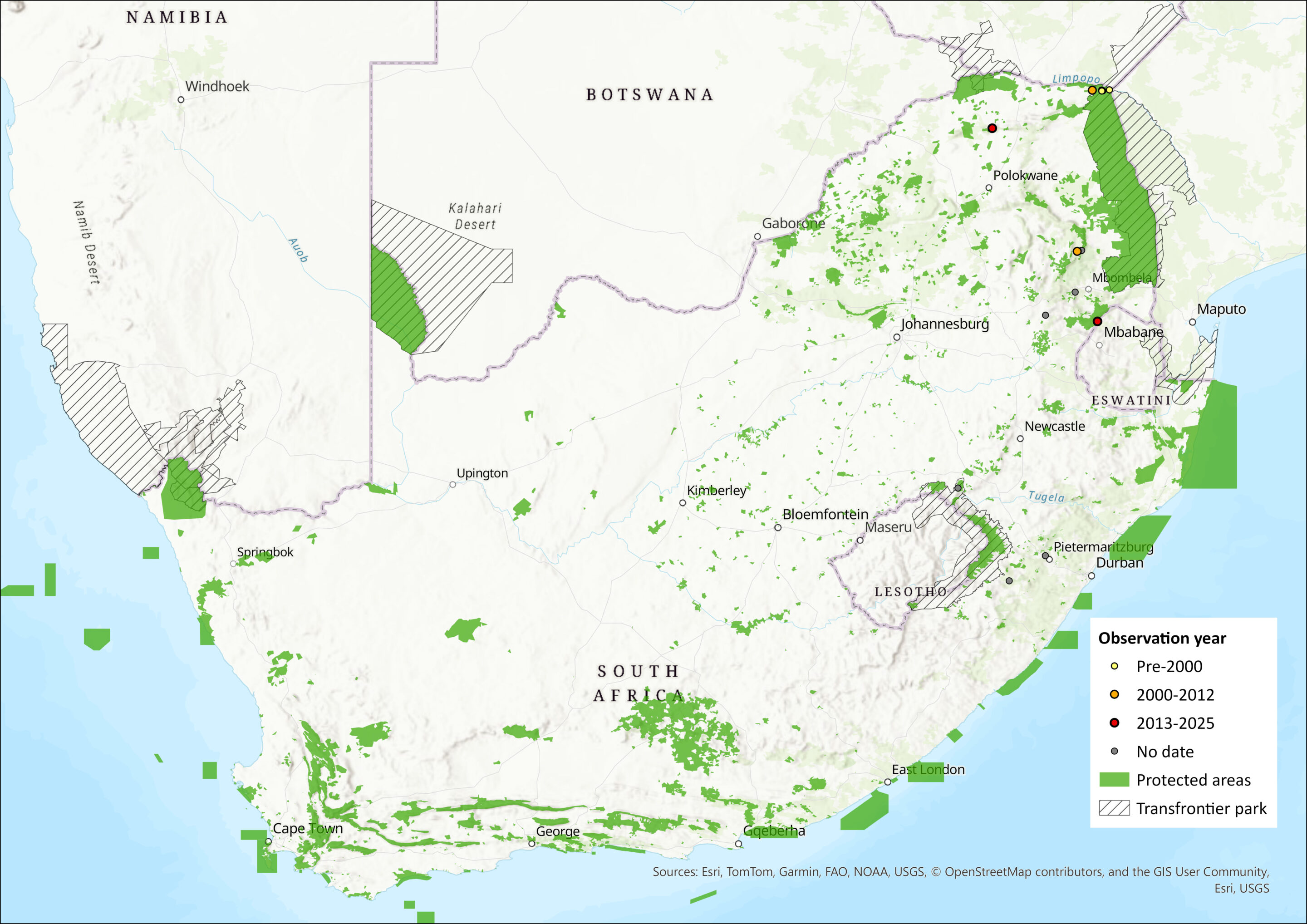 Figure 1. Distribution records for Rhodesia Horseshoe Bat (Rhinolophus rhodesiae) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Rhodesia Horseshoe Bat (Rhinolophus rhodesiae) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Eswatini | Extant | Native | ||
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Based on the worst-case scenario (SSP5-8.5) and HADGEM3 projection of CPIM6, Maxent models for this species did not predict substantial changes due to climate change by 2070 (Taylor et al. 2024). However, due to the increasing temperatures across most of the species’ distribution (Archer et al. 2018; Mapungwa et al. 2023), the roosting preferences and foraging areas are predicted to shift. Most studies on the effects of climate change on bats are based on predictive species distribution modelling, thus there is a lack of empirical studies measuring behavioural, physiological, phenological or genetic responses to extreme and seasonal climatic changes, especially in the Global South (Festa et al. 2023; Pio et al. 2014). Globally, there have been documented declines in bat populations, species richness and distributions in relation to water availability with increasing global aridity which may become a growing concern as heat waves and maximum temperatures are expected to increase over Sub-Saharan Africa (Adams & Hayes 2021).
Population information
In parts of its range, it is considered to be uncommon, however, Taylor (2000) records that it is fairly common in Zimbabwe (albeit listed as R. swinnyi). It generally forms small colonies of fewer than ten animals (ACR 2024). Within the assessment region, this species is known from fewer than ten records (some of which may be colonies). Small numbers of calls of this species were recorded at several localities in the Blouberg and western and eastern Soutpansberg Mountain range in Limpopo (Taylor et al. 2013a; Linden et al. 2014; Weier et al. 2017, 2021) but in macadamia orchards in the Soutpansberg foothills they are not recorded, or recorded in very low numbers (Taylor et al. 2013b; Weier et al. 2021), suggesting that they are highly sensitive to agricultural intensification. Quite notably, considerable acoustic surveying efforts recently (2017-2020) has not detected this species in the northern Kruger (>120,000 call sequences from 24-point localities; Brinkley et al. 2020), or Kruger generally (> 130,000 call sequences from 60-point localities; Stankova et al. in press). The last record of R. rhodesiae from Kruger was in 1985 (Brinkley et al. 2020). The overall population in the assessment area is suspected to be less than 10,000 mature individuals.
Current population trend: Unknown
Continuing decline in mature individuals: Yes, inferred.
Number of Subpopulations: Four, see below
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: Suspected
All individuals in one subpopulation: Unknown
Number of mature individuals in largest subpopulation: 1000
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Mitochondrial DNA introgression has presumably occurred between this species and the morphologically distinct R. simulator (Taylor et al. 2018; 2024).
Given the presumed limited dispersal capability for the species and their occurrence in low numbers within colonies, it is expected that at least four subpopulations exist within the assessment region (Limpopo; Mpumalanga; KwaZulu-Natal; Eswatini). It is possible the Limpopo subpopulation likely forms a metapopulation with Zimbabwe and the overall subpopulation may be estimated at greater than 1000 individuals; however, they are highly reliant on suitable habitat to maintain this connectivity. The other three subpopulations are estimated at have far fewer than 1000 individuals. A nuclear assessment would be beneficial at uncovering the contemporary population genetic structure and diversity within the species.
Based on the available information, two genetic indicators can be quantified for this species. The Convention on Biological Diversity’s complementary indicator – the proportion of populations maintained within species – would receive a score of 1.0, with no populations expected to have gone extinction, and all four subpopulations remaining (4/4=1.0). For the CBD’s headline indicator – the proportion of populations with an effective population size greater than 500 – conversion ratios between 0.1-0.3 can be applied to population estimates to get a general indication of the effective population size (Ne) of each. Three of the four subpopulations have fewer than 1000 individuals, translating to an Ne less than 100-300 for each. While the Limpopo population is generally estimated at being greater than 1,000 individuals overall, applying the conversion ratio still has this subpopulation below the 500 threshold. As such the overall indicator value for the species would be 0 (0/4 subpopulations greater than Ne 500).
It is recommended that a population genetic/genomic study be undertaken on this species across all of its subpopulations, to get a better idea of the relationship between Nc and Ne.
Habitats and ecology
This species is found in temperate Afromontane scarp forests similar to R. swinnyi in KwaZulu-Natal where the two species co-occur. In the Soutpansberg and Pafuri section of the Kruger National Park, they seem to occur in riparian or mistbelt forests as well as mesic savanna woodlands (Linden et al. 2014; Weier et al. 2016).
It is probably more dependent on the availability of suitable shelter in which to roost during the day than on specific vegetation types (Skinner & Chimimba 2005). It is dependent on caves, mines and similar habitats for roosting. It appears to be sparsely distributed.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 7.1. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Caves | – | Suitable | – |
| 7.2. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Other Subterranean Habitats | – | Suitable | – |
Life History
Generation Length: Unknown
Age at Maturity: Female or unspecified: Unknown
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Combined sexes: mean forearm length = 4.42 ±0.09 cm; mean total length = 7.48 ±0.54 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Female: Unknown
Size at Maturity (in cms): Male: Unknown
Longevity: Unknown
Average Reproductive Age: Presumably 1-2 years
Maximum Size (in cms): Unknown
Size at Birth (in cms): Unknown
Gestation Time: Unknown
Reproductive Periodicity: Unknown
Average Annual Fecundity or Litter Size: Presumed to be 1 pup per year
Natural Mortality: Unknown
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Unknown, possibly can undergo small-scale migration to different roosts (e.g. maternity roosts), but unlikely capable of large-scale or long-distance migration based on presumed low wing loading.
Congregatory: Yes, but roost in small numbers.
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
Local Livelihood: N/A
National Commercial Value: N/A
International Commercial Value: N/A
End Use: N/A
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: N/A
Threats
Populations may be locally threatened by local harvesting of timber and firewood, and general conversion of land to agricultural use (ACR 2013). Re-mining of old mine adits is a significant threat in the assessment region, as it results in loss of suitable roost habitat for this species. Similarly, the closure (sealing) of old mine tunnels, inter alia, to prevent their exploitation by illegal miners, which the South African government is undertaking at a national level (Government Notice 446, 21 May 2021), could lead to further loss of suitable roosting sites. The absence of recent records in Kruger National Park, despite intense acoustic surveys may be due to damage of woodlands by elephants (Fenton et al. 1997) or destruction of large figs and other riparian trees along the Limpopo Rover by Cyclone Domoina in 1984 (see Taylor et al. 2020 for the importance of this Limpopo riparian strip to horseshoe bats).
Conservation
In the assessment area, the species is present in the protected areas Kruger National Park, Hluhluwe-iMfolozi Game Reserve and various forest reserves in KwaZulu-Natal. Key interventions for this species include protection of known roost sites through appropriate management to limit disturbance, and protected area expansion to incorporate further subpopulations.
Recommendations for land managers and practitioners:
- Limit human disturbance to cave roost sites
- Report discoveries of new roost sites.
Research priorities:
- Further field surveys to discover new roost sites to inform protected area expansion.
- Monitoring of known subpopulations to establish population trends.
- Investigation into the foraging ecology, habitat and roost preferences as well as the reproductive biology of this species is required
- Further genetic studies, using additional nuclear markers, to refine our understanding of the phylogenetic relationships amongst species within the clade of R. rhodesiae and amongst southern Africa taxa in general.
- Landscape genetic studies and the use of microsatellites to gain an understanding of population genetics.
- Further studies on ecomorphological traits (e.g. wing loading and associated wing morphology), may provide additional insights into the distinction of this taxon from similar-sized rhinolophids.
Encouraged citizen actions:
- Limit disturbance to cave and mine roost sites.
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP).
Bibliography
ACR. 2024. African Chiroptera Report. 2024. AfricanBats NPC, Pretoria. i – xv; 1 – 1397.
Archer, E.R., Engelbrecht, F.A., Hänsler, A., Landman, W., Tadross, M. and Helmschrot, J. 2018. Seasonal prediction and regional climate projections for southern Africa. Klaus Hess Publishers.
Benda, P., Uvizl, M., Eiseb, S. J., & Avenant, N. L. 2024. On the systematic position of the horseshoe bats (Mammalia: Chiroptera) from Lesotho. Mammalia, 88, 239–258. https://doi. org/ 10. 1515/ mammalia-2023-0119.
Brinkley, E.R., Weier, S.M., Parker, D.M. and Taylor, P.J. 2021. Three decades later in the northern Kruger National Park: multiple acoustic and capture surveys may underestimate the true local richness of bats based on historical collections. Hystrix, the Italian Journal of Mammalogy 32(2), doi:10.4404/hystrix-00319-2020.
Bronner, G.N. 1990. New distribution records for four mammal species, with notes on their taxonomy and ecology. Koedoe 33: 1–7.
Demos, T. C., Webala, P. W., Goodman, S. M., Kerbis Peterhans, J. C., Bartonjo, M., & Patterson, B. D. 2019. Molecular phylogenetics of the African horseshoe bats (Chiroptera: Rhinolophidae): Expanded geographic and taxonomic sampling of the Afrotropics. BMC Evolutionary Biology, 19, 166. https://doi. org/ 10. 1186/ s12862-019-1485-1
Dool, S. E., Puechmaille, S. J., Foley, N. M., Allegrini, B., Bastian, A., Mutumi, G. L., Maluleke, T. G., Odendaal, L. J., Teeling, E. C., & Jacobs, D. S. 2016. Nuclear introns outperform mitochondrial DNA in inter-specific phylogenetic reconstruction: Lessons from horseshoe bats (Rhinolophidae: Chiroptera). Molecular Phylogenetics and Evolution, 97, 196–212. https:// doi. org/ 10. 1016/j. ympev. 2016. 01. 0
Fenton, M.B., Cumming, D.H.M., Rautenbach, I.L., Cumming, G.S., Cumming, M.S., Ford, G.S., Taylor, R.D., Dunlop, J.M., Hovorka, M.D., Johnston, D.S., Portfors, C.V., M.C. Kalcounis and Z. Mahlanga. 1998. Bats and the loss of tree canopy in African woodland. Conservation Biology 12:339-407.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Linden, V. M. G., Weier, S. M.,Gaigher, I., Kuipers, H. I., Weterings, M. J. A. & Taylor, P. J. 2014. Changes of bat activity, species richness, diversity and community composition over an altitudinal gradient in the Soutpansberg range, South Africa. Acta Chiropterologica 16 (1): 27-40, doi: 10.3161/150811014X683246.
Mupangwa, W., Chipindu, L., Ncube, B., Mkuhlani, S., Nhantumbo, N., Masvaya, E., Ngwira, A., Moeletsi, M., Nyagumbo, I. and Liben, F. 2023. Temporal changes in minimum and maximum temperatures at selected locations of southern Africa. Climate 11(4):84.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis, 2nd EdUniversity of Witwatersrand Press, Johannesburg.
Roberts A. 1951. The Mammals of South Africa. The Trustees of the Mammals of South Africa. Central News Agency, Johannesburg, South Africa.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor, P.J. 2000. Bats of Southern Africa: Guide to Biology, Identification, and Conservation. University of Natal Press, Pietermaritzburg, South Africa.
Taylor, P. J., Monadjem, A. and Steyn, J. N. Seasonal patterns of habitat use by insectivorous bats in a subtropical African agro-ecosystem dominated by macadamia orchards. African Journal of Ecology 51: 552-561, https://doi.org/10.1111/aje.12066.
Taylor, P. J., Nelufule, M., Parker, D. M., Cory Toussaint, D., Weier, S. M. 2020. The Limpopo River exerts a powerful but spatially limited effect on bat communities in a semi-arid region of South Africa. Acta Chiropterologica 22(1): 75–86, doi: 10.3161/15081109ACC2020.22.1.007.
Taylor P. J.,MacDonald A., Goodman S. M., Kearney T., Cotterill F. P. D.,Stoffberg S., Mondajem A., Schoeman M. C., Guyton J. Naskrecki P. and Ricahrds L. R. 2018. Integrative taxonomy resolves three new cryptic species of small southern African horseshoe bats (Rhinolophus). Zoological Journal of the Linnean Society, 184: 1249-1
Taylor, P. J., Kearney, T. C., Clark, V. R., Howard, A., Mdluli, M. V., Markotter, W. Geldenhuys, M., Richards, L. R., Rakotoarivelo, A. R., Watson, J., Balona, J., and Monadjem, A. 2024. Southern Africa’s Great Escarpment as an amphitheater of climate-driven diversification and a buffer against future climate change in bats, Global Change Biology, 30:e17344, https://doi.org/10.1111/gcb.17344. 276
Weier, S. M., Linden, V. M. G., Gaigher, I., White, P. J. C. & Taylor, P. J. 2017 Changes of bat species distribution over altitudinal gradients on northern and southern aspects of the Soutpansberg mountain range, South Africa. Mammalia 81: 49-60. DOI 10.1515/mammalia-2015-0055.
Weier, S. M., Linden, V. M. G., Hammer, A., Grass, I. Tscharntke, T., Taylor, P. J. 2021. Bat guilds respond differently to habitat loss and fragmentation at different scales in macadamia orchards in South Africa. Agriculture, Ecosystems and Environment, 320, https://doi.org/10.1016/j.agee.2021.107588.
