Natal Multimammate Mouse
Mastomys natalensis

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Mastomys natalensis – (Smith, 1834)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Mastomys – natalensis
Common Names: Natal Multimammate Mouse, Natal Mastomys (English), Natalse Vaalveldmuis (Afrikaans), Lehomo (Sesotho)
Synonyms: Mastomys hildebrandtii (Peters, 1878); Myomys fumatus (Peters, 1878)
Taxonomic Note: A good review of the systematics of Mastomys is provided by Granjon et al. (1997). Mastomys spp. are cryptic and difficult to distinguish morphologically but clearly separable by molecular and chromosomal markers (Britton-Davidian et al. 1995; Lecompte et al. 2005). For example, within the assessment region, M. coucha and M. natalensis can be distinguished only through chromosome number (in M. coucha 2n = 36; in M. natalensis 2n = 32) and molecular markers (Colangelo et al. 2013) but not on cranio-dental features, nor a multivariate analysis (Dippenaar et al. 1993).
Red List Status: LC – Least Concern
Assessment Information
Assessors: Russo, I.M.1 & da Silva, J.M.2
Reviewer: Smith, C.3
Institutions:1Cardiff University, 2South African National Biodiversity Institute,3Endangered Wildlife Trust
Previous Assessors & Reviewers: du Plessis, J., Russo, I.M. & Child, M.F.
Previous Contributors: Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Monadjem, A., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
This species is listed as Least Concern as it has a wide distribution within the assessment region, where it likely occurs in most protected areas. Mastomys natalensis are abundant in human-transformed areas, including agricultural areas and areas affected by human disturbances. There are currently no significant threats that could cause range-wide decline. Additionally, both species of Mastomys are known as prolific breeders with population numbers likely to recover quickly after a decline. Because of their reproductive characteristics, population eruptions often occur under favourable conditions. Landowners and managers should pursue ecologically-based rodent management strategies and biocontrol instead of rodenticides to regulate population explosions of this species.
Regional population effects: Dispersal is highly possible through contiguous habitat along north and northeastern borders and because they utilise transformed habitats.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Russo IM & da Silva JM. 2025. A conservation assessment of Mastomys natalensis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
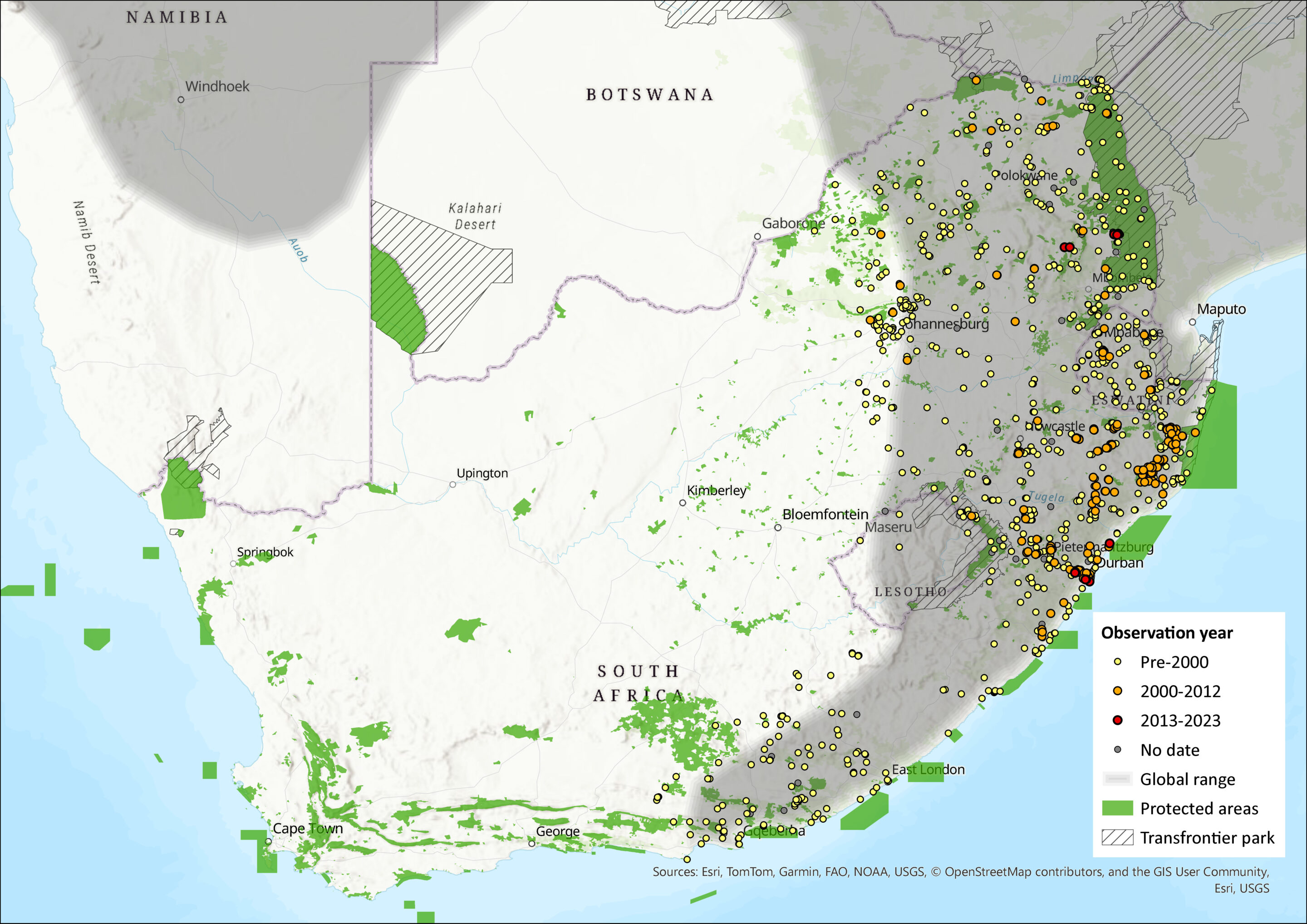
Regional Distribution and occurrence
Geographic Range
They have a very wide distribution across the savannahs, grasslands and agricultural landscapes of sub- Saharan Africa (Monadjem et al. 2015). Mastomys natalensis has the widest distribution of all African rodents (Colangelo et al. 2013) and are almost ubiquitously distributed across the African continent (van Hooft et al. 2008).
Mastomys coucha co-occurs only marginally with M. natalensis in South Africa (Venturi et al. 2004), with a possible zone of overlap along the eastern escarpment. It overlaps more extensively in southern Zimbabwe (Gordon 1978) and northern Namibia (Monadjem et al. 2015). Additional research is still needed to determine the precise zone of parapatry (Venturi et al. 2004).
Within the assessment region, M. natalensis is predominantly found in the wetter, eastern regions (east of the Drakensberg escarpment), or in the low altitude/high rainfall eastern coastal region, extending to northeastern South Africa (Venturi et al. 2004). This habitat preference appears to apply at small spatial scales too. For example, in Roan Camp, Kruger National Park, M. natalensis dominated in wetter areas, whereas M. coucha was found in relatively more high-altitude, low-rainfall areas (Kneidinger et al. 2014). Mastomys natalensis has been documented in Gauteng Province (Venturi et al. 2004), but as yet, not in the North West Province (Power 2014). A recent landscape genetics study of M. natalensis from the Hluhluwe-iMfolozi Park, KwaZulu-Natal Province, showed that the most significant landscape features shaping gene flow are slope aspect, vegetation cover, topographic complexity and rivers (Russo et al. 2016). Eastern facing slopes and thicket vegetation promote gene flow/movement throughout the landscape, whereas topographic complexity and rivers act as barriers to gene flow (Russo et al. 2016).
There are likely to be errors in the distribution maps due to the inability of being able to separate the two species on morphological evidence. Even sperm morphology is very similar between M. natalensis and M. coucha (Breed 1995). The use of molecular research to vet and reclassify museum records should be used to more accurately delineate the areas of sympatry of these two species.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Natal Multimammate Mouse (Mastomys natalensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
There is a possibility that it might be affected by the 1.5-4°C increase in temperature predicted under various climate change scenarios (Engelbrecht et al. 2024). Rainfall is predicted to decrease in parts of its range and increases in the Kwa-Zulu Natal area. These decreases will result in a more arid and drought-prone habitat which could affect food availability in the species distribution range. While the increases could result in wetter areas and flooding which could.
Population
Mastomys is often the most abundant genus in an area. M. natalensis often sharply increases in abundance after some form of disturbance, such as fire (Monadjem et al. 2015). When M. natalensis numbers decrease in a population it may be associated with an increase in general small mammal species diversity (Monadjem & Perrin 2003). Rautenbach et al. (2014) found that M. natalensis was the most frequently captured rodent species at Phinda Private Game Reserve, KwaZulu-Natal Province, where abundance differed significantly amongst vegetation types but not amongst seasons, and it was most common in Acacia karroo and Combretum apiculatum woodlands. Dispersal rates and dispersal distances per generation in M. natalensis have been shown to be relatively high (van Hooft et al. 2008). Given these dispersal dynamics, M. natalensis exhibits a pattern of kin clustering at smaller geographic scales (van Hooft et al. 2008). It has also been recorded that individuals can move over distances larger than 400 m.
Population Information
Current population trend: Stable/increasing, based on no net decline in habitat and possible range expansions in the assessment region.
Continuing decline in mature individuals: No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No, occurs extensively in agricultural and disturbed areas and has high dispersal rates (van Hooft et al. 2008).
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Based on phylogenetic evidence from mitochondrial DNA, animals within the assessment region fall within a single clade showing high haplotype diversity (Colangelo et al. 2013; Hánová et al. 2021). This could suggest they form part of a single metapopulation. However, a landscape genetic study focusing on individuals within the Hluhluwe-iMfolozi Park, KwaZulu-Natal Province revealed fine scale structure within M. natalensis (Russo et al. 2016). Four genetic clusters were identified: one cluster was predominantly restricted to locations south of the Black iMfolozi River and another cluster north of the White iMfolozi River. The other two clusters were associated with grassland or locations in the south of the park (Russo et al. 2016). Given the potential for substantial substructure within the assessment region, a more comprehensive population genetic study is needed before adequate inferences can be made on the genetic structure and diversity within this species. As such, the Kunming-Montreal Global Biodiversity Framework’s population genetic indicators (the proportion of maintained populations within a species (PM indicator); populations within a species with an effective population size (Ne) greater than 500 (Ne 500 indicator) cannot be calculated at this time.
Habitats and ecology
As the common and generic names suggest, there is a large number of mammae present, between eight and 12 pairs from the sternum to inguinal region. Both Mastomys species are terrestrial and nocturnal with a diet that varies from granivorous to omnivorous, sometimes including arthropods and carrion (Monadjem et al. 2015). For example, in Umvoti Vlei Conservancy, KwaZulu-Natal Province, while Rhabdomys pumilio was primarily granivorous, M. natalensis preferred green plant foods (Fuller & Perrin 2001), where its diet changed from predominantly plant material in summer to mainly seeds in winter. They are to some extent dependent on water but occur in areas where water is only seasonally available, such as along the Orange River valley in the Northern Cape (Skinner & Chimimba 2005). They have also been observed to swim (Hickman & Machiné 1986; Power 2014).
Mastomys natalensis is typically associated with agricultural fields and homes but also occurs in natural savannahs and grasslands (Leirs 2013b). It tolerates disturbance well and can be abundant in Aristida and disturbed grasslands (Fuller & Perrin 2001). It is a generalist species that rapidly colonises areas following disturbance, such as fire, overgrazing and cultivation (Meester et al. 1979; Monadjem 1997). Home ranges of M. natalensis in Swaziland were 626 ± 86 m2 for males and 718 ± 88 m2 for females (Monadjem & Perrin 1998). Leirs et al. (1996) reported home ranges of > 1,000 m2 for M. natalensis.
Due to its high abundance in agricultural landscapes, M. natalensis is a significant crop pest and frequently digs up newly-planted maize seeds or climbs maize stalks to feed on the seeds (Leirs 2013b), as well as feeding on stored grains inside dwellings. Although the specific impact of M. coucha on agricultural crops has not yet been assessed, it is widely accepted that it may cause extensive losses similar to that observed for M. natalensis (Skinner & Chimimba 2005; Leirs 2013a). According to Mulungu et al. (2011), damages to maize by M. natalensis during population outbreaks may exceed 80% of the harvest in some areas.
Both M. coucha and M. natalensis are opportunistic breeders that have the ability to breed throughout the year whenever conditions are favourable, and breeding is strongly correlated with rainfall. In most areas, however, reproduction does not occur during winter. They are known as prolific breeders and, although this rarely happens, they can carry up to 24 foetuses at once, under favourable conditions. Their gestational periods and the interval between litters are also relatively short, with litter sizes varying from 1–27 young (Monadjem et al. 2015). Due to their reproductive characteristics, multimammate mice populations are known to erupt under favourable conditions (Skinner & Chimimba 2005; Leirs 2013a, 2013b).
Ecosystem and cultural services: Mastomys spp. are indicators of poor ecosystem integrity as they become the dominant small mammals in a community during and after a disturbance (Avenant & Kuyler 2002; Avenant et al. 2008; Avenant 2011). They are also vectors of disease, where M. coucha is more susceptible to experimental plague infection than M. natalensis, and thus more implicated in plague epidemiology (Isaäcson et al. 1981; Venturi et al. 2004). Both species may act as seed dispersers, pollinators, and form a forage resource for carnivores, especially in post-fire landscapes, as they do not vacate the area following fires.
IUCN Habitats Classification Scheme
|
Habitat |
Season |
Suitability |
Major Importance? |
|
2.1. Savanna -> Savanna – Dry |
– |
Suitable |
– |
|
2.2. Savanna -> Savanna – Moist |
– |
Suitable |
– |
|
3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry |
– |
Suitable |
– |
|
3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist |
– |
Suitable |
– |
|
14.1. Artificial/Terrestrial -> Artificial/Terrestrial – Arable Land |
– |
Suitable |
– |
|
14.2. Artificial/Terrestrial -> Artificial/Terrestrial – Pastureland |
– |
Suitable |
– |
|
14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens |
– |
Suitable |
– |
|
14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas |
– |
Suitable |
– |
|
15.7. Artificial/Aquatic & Marine -> Artificial/Aquatic – Irrigated Land (includes irrigation channels) |
– |
Marginal |
– |
|
15.8. Artificial/Aquatic & Marine -> Artificial/Aquatic – Seasonally Flooded Agricultural Land |
– |
Marginal |
– |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Both species are used for the pet industry. However, this is not expected to impact the populations.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
These species are important from a human health purview because they are a reservoir host for a number of organisms that cause human diseases (Keogh & Price 1981; Venturi et al. 2004; Skinner & Chimimba 2005; Leirs 2013a), and because their distributions are closely related to the outbreak of plague in some areas (Isaäcson et al. 1981). They may also be considered an agricultural pest in some areas, especially during population outbreaks (Monadjem et al. 2011). Due to these threats, rodenticides are often used to control these species (Makundi & Massawe 2011). It is, however, envisaged that poisoning will only have a short-term impact on Mastomys population numbers (Makundi & Massawe 2011), with populations likely to recover due to their reproductive characteristics (Skinner & Chimimba 2005). In Limpopo Province, for example, subsistence farmers who experienced damage to staple crops from Rattus rattus, R. norvegicus and Mastomys spp. reported low success from rodenticides and kill-traps to control the damages (von Maltitz et al. 2003). Perhaps more important are the knock-on effects such poisons may have within the broader ecosystem through bioaccumulation or unintentional poisoning of non-target species, thus incentivising the use of ecologically-based management methods. It also is uncertain how the diseases associated with Mastomys species affect the rodents themselves (Leirs 2013b).
Conservation
These species are associated with a wide range of habitats, varying from disturbed areas to areas with more pristine habitat (Avenant et al. 2008; MacFadyen et al. 2012), and thus likely occur in most protected areas throughout their distribution range. As such, no specific interventions are necessary at present. However, the use of ecologically-based rodent management (EBRM) should be encouraged over the use of pesticides to limit population explosions (Makundi & Massawe 2011). Overall, EBRM relies on a strong ecological understanding of the target species and the development of species-specific management strategies at the farming level. It may include the reduction of key resources, such as food and nesting sites, at critical times of the year through habitat modification and the selective use of techniques for culling rodents at specific times of the year and in specific habitats (Singleton et al. 2004, 2007). For example, the use of owl nest boxes has been suggested as an important bio-control method in both small mammal ecosystem services (pollinators and seed dispersers) and management (Russo et al. 2016). In a recent study, no difference in M. natalensis population dynamics was observed within monocultures or mosaic agricultural lands, meaning that management in both agricultural systems could focus on the same aspects of the species’ ecology (Sluydts et al. 2009).
Bio-control should also be encouraged as an alternative single control method, although Vibe-Peterson et al. (2006) demonstrated that the introduction of more predators into an area may not have a clear impact on Mastomys population densities due to the influence of compensatory breeding.
Recommendations for land managers and practitioners:
- Development and implementation of EBRM strategies suitable to Mastomys and applicable to specific areas (Makundi & Massawe 2011). For example, as has been trialled in Limpopo Province (von Maltitz et al. 2003).
- The use of bio-control, such as owl boxes, to mitigate the threat of Mastomys as an agricultural pest and as a threat to human health.
Research priorities:
- Accurate distributions of M. natalensis and M. coucha, including areas of sympatry, need to be determined using molecular markers (for example, Kneidinger et al. 2014).
- Applied ecological studies need to be conducted that can inform and form the basis of EBRM strategies (Makundi & Massawe 2011).
- The contribution of Mastomys spp. to the distribution and transfer of human diseases is also an important research area.
Encouraged citizen actions:
- Farmers could contribute to the development and implementation of EBRM strategies.
- Promotion of bio-control to regulate population explosions by attracting predators to an area. One method is to erect perches and install owl nest boxes in urban and rural green spaces.
Bibliography
Avenant NL, Kuyler P. 2002. Small mammal diversity in the Maguga Dam inundation area, Swaziland. South African Journal of Wildlife Research 32: 101-108.
Avenant NL, Watson JP, Schulze E. 2008. Correlating small mammal community characteristics and habitat integrity in the Caledon Nature Reserve, South Africa. Mammalia 72: 186–191.
Avenant, N.L. 2011. The potential utility of rodents and other small mammals as indicators of ecosystem “integrity” of South African grasslands. Wildlife Research 38: 626–639.
Breed W. 1995. Variation in sperm head morphology of muroid rodents of Africa: phylogenetic implications. In: Jamieson GBM, Ausio J, Justine J-L (ed.), Advances in Spermatozoal Phylogeny and Taxonomy., pp. 409–420. Paris, France.
Britton-Davidian J, Catalan J, Granjon L, Duplantier J-M. 1995. Chromosomal phylogeny and evolution in the genus Mastomys (Mammalia, Rodentia). Journal of Mammalogy 76: 248–262.
Colangelo P, Verheyen E, Leirs H, Tatard C, Denys C, Dobigny G, Duplantier J-M, Brouat C, Granjon L, Lecompte E. 2013. A mitochondrial phylogeographic scenario for the most widespread African rodent, Mastomys natalensis. Biological Journal of the Linnean Society 108: 901–916.
De Graaff, G. 1981. The Rodents of Southern Africa. Butterworths, Durban, South Africa.
Dippenaar NJ, Swanepoel P, Gordon DH. 1993. Diagnostic morphometrics of two medically important southern African rodents, Mastomys natalensis and M. coucha. South African Journal of Science 89: 300–303.
Engelbrecht, FA., Steinkopf, J., Padavatan, J. & Midgley, GF. 2024. Projections of future climate change in southern Africa and the potential for regional tipping points. In: von Maltitz, G.P., et al. Sustainability of Southern African Ecosystems under Global Change. Ecological Studies, vol 248. Springer, Cham. https://doi.org/10.1007/978-3-031-10948-5_7
Fuller, J.A. and Perrin, M.R. 2001. Habitat assessment of small mammals in the Umvoti Vlei Conservancy, KwaZulu-Natal, South Africa. South African Journal of Wildlife Research 31: 1-12.
Gordon DH. 1978. Distribution of sibling species of the Praomys (Mastomys) natalensis group in Rhodesia (Mammalia: Rodentia). Journal of Zoology 186: 397–401.
Granjon, L., Duplantier, J.-M., Catalan, J. and Britton-Davidian, J. 1997. Systematics of the Genus Mastomys (Thomas,1915) (Rodentia: Muridae) – A Review. Belgian Journal of Zoology 127: 7-18.
Hánová, A., Konečný, A., Mikula, O., Bryjová, A., Šumbera, R. & Bryja, J. 2021. Diversity, distribution, and evolutionary history of the most studied African rodents, multimammate mice of the genus Mastomys: an overview after a quarter of century of using DNA sequencing. J Zool Syst Evol Res 59:2500–2518.
Hickman, G.C. and Machiné, C. 1986. Swimming behaviour in six species of African rodents (Cricetidae, Muridae). Acta Theriologica 31: 449–466.
Isaäcson M, Arntzen L, Taylor P. 1981. Susceptibility of members of the Mastomys natalensis species complex to experimental infection with Yersinia pestis. Journal of Infectious Diseases 144: 80.
Keogh HJ, Price PJ. 1981. The multimammate mouse: a review. South African Journal of Science 77: 484–488.
Kneidinger CM, van Heerden H, MacFadyen D, van der Merwe M, Avenant NL, van der Bank H. 2014. Species identification, habitat preferences and population genetics of Mastomys natalensis (A. Smith, 1834) and M. coucha (A. Smith, 1836) in an enclosed area, Kruger National Park, South Africa. Navorsinge van die Nasionale Museum Bloemfontein 30: 31–45.
Lecompte, É., Brouat, C., Duplantier, J.-M., Galan, M., Granjon, L., Loiseau, A., Mouline, K. and Cosson, J.-F. 2005. Molecular identification of four cryptic species of Mastomys (Rodentia, Murinae). Biochemical Systematics and Ecology 33: 681-689.
Leirs H, Verheyen W, Verhagen R. 1996. Spatial patterns in Mastomys natalensis in Tanzania (Rodentia, Muridae). Mammalia 60: 545–556.
Leirs H. 2013. Mastomys natalensis Natal multimammate mouse (Natal Mastomys). In: Happold DCD (ed.), Mammals of Africa. Volume III: Rodents, Hares and Rabbits, pp. 468–470. Bloomsbury Publishing, London, UK.
Leirs H. 2013a. Mastomys coucha Southern African multimammate mouse (Southern African Mastomys). In: Happold DCD (ed.), Mammals of Africa: Volume III: Rodents, Hares and Rabbits, pp. 463–464. Bloomsbury Publishing, London, UK.
Lynch, C.D. 1983. The mammals of the Orange Free State, South Africa. Navorsinge van die Nasionale Museum Bloemfontein 18: 1-218.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
MacFadyen D. 2007. A comparative study of rodent and shrew diversity and abundance in and outside the N’washitshumbe enclosure site in the Kruger National Park. M.Sc. Thesis. University of Pretoria.
MacFadyen DN, Avenant NL, van der Merwe M, Bredenkamp GJ. 2012. The influence of fire on rodent abundance at the N’washitshumbe enclosure site, Kruger National Park, South Africa. African Zoology 47: 138–146.
Makundi RH, Massawe AW. 2011. Ecologically based rodent management in Africa: potential and challenges. Wildlife Research 38: 588–595.
Meester JAJ, Lloyd CNV, Rowe-Rowe DT. 1979. A note on the ecological role of Praomys natalensis. South African Journal of Science 75: 183–184.
Monadjem A, Perrin M. 2003. Population fluctuations and community structure of small mammals in a Swaziland grassland over a three-year period. African Zoology 38: 127-137.
Monadjem A, Perrin MR. 1998. The effect of supplementary food on the home range of the multimammate mouse Mastomys natalensis. South African Journal of Wildlife Research 28: 1–3.
Monadjem A, Themb’a AM, Dlamini N, Eiseb SJ, Belmain SR, Mulungu LS, Massawe AW, Makundi RH, Mohr K, Taylor PJ. 2011. Impact of crop cycle on movement patterns of pest rodent species between fields and houses in Africa. Wildlife Research 38: 603–609.
Monadjem A. 1997. Habitat preferences and biomasses of small mammals in Swaziland. African Journal of Ecology 35: 64-72.
Monadjem, A. 1998. Distributional patterns and conservation status of mammals of Swaziland, southern Africa. Koedoe 41: 45-59.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. . De Gruyter, Berlin, Germany.
Mulungu LS et al. 2011. Dietary differences of the multimammate mouse, Mastomys natalensis (Smith, 1834), across different habitats and seasons in Tanzania and Swaziland. Wildlife Research 38: 640–646.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Rautenbach A, Dickerson T, Schoeman MC. 2014. Diversity of rodent and shrew assemblages in different vegetation types of the savannah biome in South Africa: no evidence for nested subsets or competition. African Journal of Ecology 52: 30-40.
Russo IM, Sole CL, Barbato M, von Bramann U, Bruford MW. 2016. Landscape determinants of fine-scale genetic structure of a small rodent in a heterogeneous landscape (Hluhluwe-iMfolozi Park, South Africa). Scientific Reports 6.
Singleton GR, Brown PR, Jacob J, Aplin KP. 2007. Unwanted and unintended effects of culling: A case for ecologically-based rodent management. Integrative Zoology 2: 247–259.
Singleton GR, Brown PR, Jacob J. 2004. Ecologically-based rodent management: its effectiveness in cropping systems in South-East Asia. NJAS-Wageningen Journal of Life Sciences 52: 163–171.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Sluydts V, Davis S, Mercelis S, Leirs H. 2009. Comparison of multimammate mouse (Mastomys natalensis) demography in monoculture and mosaic agricultural habitat: Implications for pest management. Crop Protection 28: 647–654.
Venturi FP, Chimimba CT, van Aarde RJ, Fairall N. 2004. The distribution of two medically and agriculturally important cryptic rodent species, Mastomys natalensis and M. coucha (Rodentia: Muridae) in South Africa. African Zoology 39: 235–245.
Vibe-Petersen S, Leirs H, Bruyn LD. 2006. Effects of predation and dispersal on Mastomys natalensis population dynamics in Tanzanian maize fields. Journal of Animal Ecology 75: 213–220.
van Hooft P, Cosson J-F, Vibe-Petersen S, Leirs H. 2008. Dispersal in Mastomys natalensis mice: use of fine-scale genetic analyses for pest management. Hereditas 145: 262–273.
von Maltitz EF et al. 2003. Developing a rodent management strategy for South Africa’s Limpopo province. ACIAR Monograph 96, Rats, mice and people: rodent biology and management. Australian Centre for International Agricultural Research, Canberra, Australia.