Mozambique Woodland Mouse
Grammomys cometes

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Grammomys cometes – (Thomas & Wroughton, 1908)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Grammomys – cometes
Common Names: Mozambique Thicket Rat, Coastal Forest Mouse, Mozambique Woodland Mouse (English), Mosambiek-woudmuis, Kusmuis (Afrikaans)
Synonyms: Thamnomys cometes Thomas & Wroughton, 1908
Thamnomys dolichurus littoralis Heller, 1912
Grammomys silindensis Roberts, 1938
Grammomys selousi Denys et al, 2011
Taxonomic Note: A recent comprehensive revision by Bryja et al. (2025) showed G. cometes (described from Inhambane, Mozambique) to be the correct name applied to specimens occurring from the Eastern Cape of South Africa (Pirie Forest) through KwaZulu-Natal into eastern Zimbabwe and Mozambique (e.g., Mt. Gorongosa and Mt. Namuli), Mt. Mulanje in Malawi, and the Chimanimani Mountains on the Zimbabwe–Mozambique border), the southern Rift of northern Malawi, Eastern Arc Mountains, and coastal forests of Tanzania (including Mafia Island) and southern Kenya.
Grammonys cometes and G. dolichurus are easily distinguished in body size (larger in G. cometes) and karyotype (2n=44-50 in G. cometes, 2n=52 in G. dolichurus), and both may occur sympatrically, e.g. in KwaZulu-Natal (Taylor 1998). According to Bryja et al. (2017; 2025), specimens from the Eastern Cape referred to G. cometes by Kryštufek et al. (2008) are in fact, G. dolichurus.
Red List Status: LC – Least Concern
Assessment Information
Assessors: Taylor, P.1 & da Silva, J. M.2
Reviewer: Kraai, M.3
Institutions:1University of the Free State,2South African National Biodiversity Institute,3Sol Plaatje University
Previous Assessors & Reviewers: Madikiza, Z., Gebe, Z., Kryštufek, B., Baxter, R. & Do Linh San, E.
Previous Contributor: Relton, C., Child, M.F., Avenant, N., Avery, M., MacFadyen, D., Monadjem, A., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
Although the Mozambique Thicket Rat was listed as Data Deficient in the 2004 assessment, we continue to list it as Least Concern as field surveys and taxonomic reviews have revealed its range to extend west beyond the Amathole mountains with several records from coastal and Afromontane forests in KwaZulu-Natal (Taylor 1998). This species thus has a wide extent of occurrence within the assessment region with potential rescue effects from Mozambique and Zimbabwe. Although deforestation is a threat, much of its habitat is protected within the assessment region and it is not suspected to be declining. However, further surveys are needed to clarify population status and research is needed to resolve its taxonomy. This species should be reassessed as new data emerge.
Regional population effects: Rescue effect is possible through dispersal from Mozambique or Zimbabwe in the Great Limpopo Transfrontier Conservation Area, and from Mozambique into northern KwaZulu-Natal. It is restricted to the forest biotic zone and requires dense and developed forests regardless of the elevation. This type of ecosystem is naturally fragmented in southern Africa, and not all forest patches are inhabited although they seem suitable for the species. The range is therefore fragmented but also imperfectly known. For example, it is not possible to claim whether a gap of 500 km, between KwaZulu-Natal and the Amathole complex, where no G. cometes have been found, is a gap in distribution or in our knowledge.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Taylor P & da Silva JM. 2025. A conservation assessment of Grammomys cometes. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
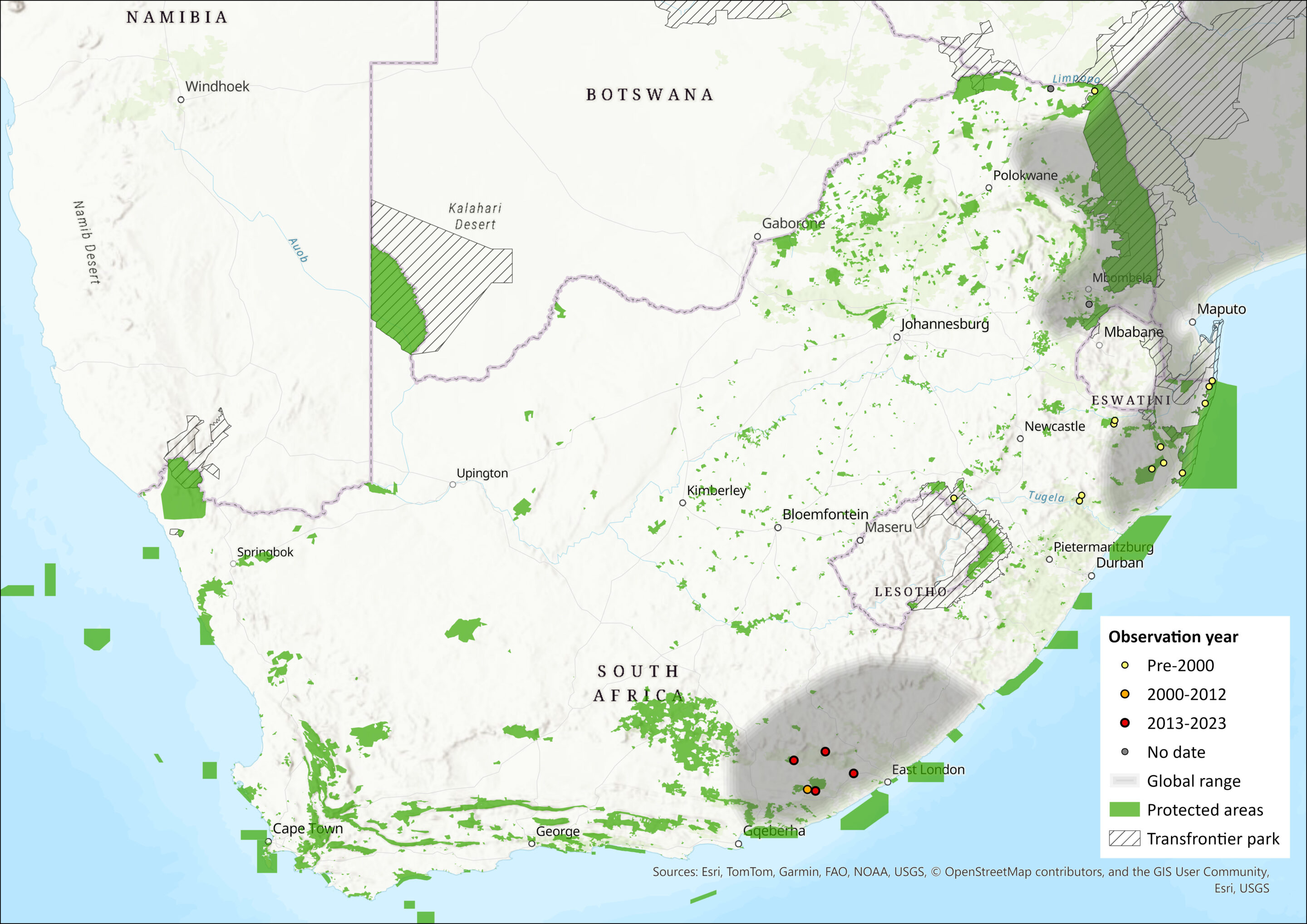
This species is endemic to the eastern parts of southern Africa, south of the Zambezi River in southern Mozambique, eastern Zimbabwe and South Africa, including Limpopo, KwaZulu-Natal and Eastern Cape provinces. There is a single record from Mpumalanga, but its validity is doubtful (Skinner & Chimimba 2005). It is possibly present in Eswatini and Lesotho, although this requires confirmation.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Mozambique Woodland Mouse (Grammomys cometes) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Eswatini | Presence Uncertain | Native | – | – |
| Malawi | Extant | |||
| Mozambique | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania | Extant | |||
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Drying trends may affect the suitability of moist Afromontane forests where this species is found.
Population
The species is traditionally reported as rare and the great majority of known individuals were collected or trapped since 2002 (Kryštufek et al. 2008; Arnolds 2009; Gebe 2014). It has a strong subpopulation in the Amathole forest complex, and it is suspected that the extent of occurrence might increase extensively if the entire forest complex was sampled. Between 2002 and 2003, relative densities were described for three sites in the Eastern Cape: 5.1 specimens were caught per 100 trap nights in Hobbiton (Hogsback Forest); 0.8 specimen per 100 trap nights were caught in Fort Fordyce Nature Reserve; and, in Great Fish River Nature Reserve (GFRNR), 2.2 specimens per 100 trap nights were caught (Kryštufek et al. 2008). At one specific site in GFRNR – a riverine Combretum forest – Gebe (2014) similarly recorded a yearly trapping success of 2.2 captures per 100 trap nights, with a minimum of 0.7 capture in spring and a maximum of 4.2 in autumn. Gebe (2014) recorded a minimum of 38 different individuals over the whole year in a 6 ha forest stretch, which corresponds to a density of 6.3 individuals/ha. However, some populations of this species can fluctuate locally with rainfall, in a “boom and bust” fashion.
The population trend in the assessment region is unknown, but it is unlikely to be declining. It is probably under-represented in the literature, as although the species can sometimes be detected when traps are exclusively set on the ground (Arnolds 2009), 75–82% of captures in Afromontane forest (Malinga 2003; Dali 2006) and 92–100% of captures in Combretum lowland forest were made with traps set in trees (Kryštufek et al. 2008; Gebe 2014).
Population Information
Current population trend: Unknown, but probably stable as there is no evidence of decline.
Continuing decline in mature individuals: Unknown, but probably not.
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: It is not currently possible to determine the extent or number of subpopulations.
Severely fragmented: Yes, it is assumed that the population is severely fragmented, as a consequence of fragmentation of their natural forest habitat. This species does not occupy pine plantations.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
No population genetic study has been undertaken on this species and it is uncertain whether the 500 km gap between the KwaZulu-Natal and the Amathole observations is indicative of a real gap in the distribution of the species or simply a gap in our knowledge. A fine scale molecular study is highly recommended to help fill these gaps in our understanding. Until such time, it is not possible to quantify the Convention on Biological Diversity’s Global Biodiversity Framework’s two genetic indicators – proportion of populations maintained and proportion of populations with an effective population size greater than 500.
Habitats and ecology
Mozambique Thicket Rats are found in dense evergreen forests in southern Africa (de Graaff 1981). They have also been reported from drier bushveld or coastal forest (Skinner & Chimimba 2005). In KwaZulu-Natal, they have been recorded in montane forests and Podocarpus forests; and are thought to favour denser, more well-developed forests compared to G. dolichurus (Taylor 1998). In the Eastern Cape, they are generally restricted to Podocarpus-dominated Afromontane forest and riverine Combretum forest (Kryštufek et al. 2008; Gebe 2014). In the GFRNR, however, during periods of boom, individuals will move out of the forest and venture into the thicket (R. Baxter pers. obs. 2003–2007). This explains why this species can be caught, albeit locally in small numbers, in nearly all habitat types of the reserve (Do Linh San et al. 2011). In Mozambique, this species has been recorded from forest fringes and thickets (Smithers & Lobão-Tello 1976).
Mozambique Thicket Rats are predominantly nocturnal, arboreal and prefer dense canopy cover, possibly because this decreases predation risk at the microhabitat level (Gebe 2014). During the day rats essentially sleep inside branches and trunks, and rarely in burrows, dense bushes or dead logs (Gebe 2014). In GFRNR, Cape Bushwillows (Combretum caffrum) are the predominant trees used for the resting sites, probably due to their abundance in the forest, and their propensity to rot from the inside and provide natural cavities. Mozambique Thicket Rats use several resting sites throughout the year, but site fidelity on consecutive days is very high (Gebe 2014). Specific resting sites are used by several different rats, although simultaneous sharing is relatively rare, and generally only takes place between males. Based on extensive trapping and radio-tracking data, Gebe (2014) suggested that females are solitary but not territorial, while at least some males are more social or tolerant towards each other and have overlapping home ranges. These observations could be indicative of a promiscuous mating system.
Very little is known about the reproductive biology of the species. Skinner and Chimimba (2005) stated that G. cometes can have from 2 to 5 young and up to three litters during the warm, wet season, and similar observations have been made at GFRNR (Z.J.K. Madikiza & E. Do Linh San pers. obs. 2006–2012). Similarly, Kryštufek et al. (2008) reported that the animals from different sites used in their study had morphologically heterogeneous reproductive conditions, although a large proportion of specimens of adult G. cometes showed reproductive activity (scrotal testes, presence of placental scars or embryos in the uterus of sacrificed animals). There is virtually no information available on the diet of G. cometes, although we suspect that it is very similar to that of G. dolichurus. The latter is reported to feed on fruits, leaves, stems, invertebrates, green vegetable matter, white plant material and wood fibres (Skinner & Chimimba 2005). It is unclear whether forays on the ground are therefore linked to foraging purposes rather than dispersal events or responses to overpopulation.
Ecosystem and cultural services: Grammomys cometes is one of the core small mammals in the forest ecosystem. In the Eastern Cape, this species represented 18–23.4% of all small mammals captured (Kryštufek et al. 2008), therefore in these habitats it is likely to play a significant ecological role, for example in seed dispersal and/or as a food source for small carnivores (Nqinana 2009; Matolengwe 2010; Mbatyoti 2010; Bizani 2014).
IUCN Habitats Classification Scheme
|
Habitat |
Season |
Suitability |
Major Importance? |
|
1.5. Forest -> Forest – Subtropical/Tropical Dry |
– |
Suitable |
– |
|
3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry |
– |
Suitable |
– |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no major threats to this species, however deforestation due to agricultural expansion and resource extraction in forests, as well as expanding residential development in coastal forests, may result in local subpopulation declines or even extinction. Additionally, as this species lives in moist evergreen forests, it may be threatened to a minor extent by increasing aridity, due to climate change.
Current habitat trend: Declining
Conservation
This species occurs in several protected areas within the assessment region, for example Fort Fordyce Nature Reserve, Mpofu Nature Reserve and Great Fish River Nature Reserve. There are no specific interventions currently necessary, but the species would benefit from protected area expansion to include key forest complexes and biodiversity stewardship schemes that could provide corridors for movement.
Recommendations for land managers and practitioners:
- Landowners and city planners can conserve corridors of forest on their properties.
Research priorities:
- A more accurate determination of the geographic distribution of this species is required.
- Taxonomic revision of this species complex is necessary.
- Studies on the reproductive biology and colonisation (dispersal) abilities would provide precious information on the potential resilience of this species to future threats.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas. However, due to morphological similarity and sympatric distributions in some parts, misidentification of this species with G. dolichurus is common.
Bibliography
Arnolds LJ. 2009. Vegetation sampling in correlation with small mammal abundance and richness in the Andries Vosloo Kudu Nature Reserve (Eastern Cape, South Africa). B.Sc. Honours Thesis. University of Fort Hare.
Bizani M. 2014. Diet of the yellow mongoose (Cynictis penicillata) in the Albany Thicket Biome of South Africa. M.Sc. Thesis. University of Fort Hare.
Bryja, J., Peterhans, J.C.K., Lavrenchenko, L.A., Nicolas, V., Denys, C., Bryjová, A., Šumbera, R. & Mikula, O. 2025. Integrative taxonomic revision of the African thicket rats (Murinae: Grammomys): how genomics decreases the number of currently recognized species, Zoological Journal of the Linnean Society. zlae057, https://doi.org/10.1093/zoolinnean/zlae057
Dali LO. 2006. Observations on the ecology of the Mozambique woodland mouse, Grammomys cometes, in Afromontane forest at Hogsback in Eastern Cape province, South Africa. B.Sc. Honours Thesis. University of Fort Hare.
Do Linh San E, Arnolds L, Matolengwe T, Mbatyoti A, Nqinana A, Buwa L, Madikiza Z. 2011. Small mammal abundance, diversity and richness in the Andries Vosloo Kudu Nature Reserve (Eastern Cape, South Africa). 11th African Small Mammal Symposium. Kwaluseni, Swaziland..
Eeley HA, Lawes MJ, Piper SE. 1999. The influence of climate change on the distribution of indigenous forest in KwaZulu-Natal, South Africa. Journal of Biogeography 26: 595–617.
Gebe Z. 2014. Resting site ecology and microhabitat use of the Mozambique thicket rat (Grammomys cometes) in a riverine Combretum forest. M.Sc. Thesis. University of Fort Hare.
Kryštufek B, Baxter RM, Haberl W, Zima J, Bužan EV. 2008. Systematics and biogeography of the Mozambique thicket rat, Grammomys cometes, in Eastern Cape province, South Africa. Journal of Mammalogy 89: 325–335.
Malinga TJ. 2003. Observations on the population ecology of Grammomys cometes in Hogsback Forest, Eastern Cape, South Africa. B.Sc. Honours Thesis. University of Fort Hare.
Matolengwe T. 2010. Diet of the small-spotted genet, Genetta genetta (Carnivora: Viverridae), in the Great Fish River Reserve (South Africa). M.Sc. Thesis. University of Fort Hare.
Mbatyoti OA. 2010. The diet of the Cape grey mongoose Galerella pulverulenta in the Albany Thicket Biome (South Africa). B.Sc. Honours Thesis. University of Fort Hare.
Nqinana A. 2009. Diet of the marsh mongoose Atilax paludinosus in the Andries Vosloo Kudu Nature Reserve (Eastern Cape Province, South Africa) B.Sc. Honours Thesis. University of Fort Hare.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smithers, R.H.N. and Lobao-Tello, J.L.P. 1976. Check list and atlas of the mammals of Mozambique. Trustees of the National Museums and Monuments of Rhodesia, Salisbury, Rhodesia.
Taylor, P. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.
de Graaff, G. 1981. The Rodents of Southern Africa: Notes on their Identification, Distribution, Ecology, and Taxonomy. Butterworths, Durban, Pretoria, South Africa.