Impala
Aepyceros melampus

2025 Red list status
Least Concern
Regional Population Trend
Increasing
Change compared
to 2016
No Change
Overview
Aepyceros melampus – (Lichtenstein, 1812)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Aepyceros – melampus
Common Names: Common Impala, Impala (English), Rooibok (Afrikaans), Mhara (Shona), Phala (Sotho,Tswana, Venda), Impala (Zulu)
Synonyms: No Synonyms
Taxonomic Note:
Two subspecies are recognised, supported by molecular data (Nersting & Arctander 2001; Lorenzen et al. 2006): the Common Impala (A. m. melampus) and the Black-faced Impala (A. m. petersi). While there is no genetic structuring of Impala in East and southern Africa, microsatellite analysis does suggest some degree of isolation along the east-south gradient, which results in some distinct genetic clades, particularly in northern Kenya (Lorenzen et al. 2006, 2012).
Red List Status: LC – Least Concern
Assessment Information
Assessors: Shrader, A.M1. & da Silva, J.2
Reviewer: Patel, T.3
Institutions: 1Department of Zoology & Entomology, University of Pretoria, South Africa; 2South African National Biodiversity Institute, Kirstenbosch Research Centre, Cape Town, 8000, South Africa; 3Conservation Planning & Science Unit, Endangered Wildlife Trust, South Africa.
Previous assessors: Hoffmann, L. & Castley, G.
Previous Reviewer: Selier, J.
Previous Contributors: Ferreira, S.M., Child, M.F. & IUCN SSC Antelope Specialist Group
Assessment Rationale
This species is widespread, common, and abundant in numerous protected areas within the assessment region. There are well over 10,000 mature individuals (an estimated 123,998–168,451 individuals in 2023 in Kruger National Park alone), and nearly all (if not all) subpopulations are stable or increasing to the extent that large-scale culls are necessary for many areas, which may form the foundation of a sustainable wildlife-based economy in these areas if managed properly. Similarly, the global population is estimated at almost 2 million, of which about 50% are on private land (stable or increasing) and 25% in protected areas (stable). There are no immediate threats to this species, although artificial selection for desired colour traits and/or deliberate and accidental hybridisation with Black-faced Impala (A. m. petersi) may compromise the integrity of the wild population if these subpopulations are used in translocations or to establish new subpopulations (Miller et al. 2020). However, the future of the species is secure if it continues to occur in large, adequately protected populations.
Regional population effects: The species range is generally continuous through East and southern Africa, with only the Black-faced Impala being isolated from these former populations. There is likely to be movement within extensive wildlife regions to facilitate genetic mixing and there is the potential for dispersal across transfrontier boundaries.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Shrader AM & da Silva JM. 2025. A conservation assessment of Aepyceros melampus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The Impala’s current distribution range remains largely unchanged from their historical range. The species is native to Angola, Botswana, Kenya, Malawi, Mozambique, Namibia, Rwanda, South Africa, Eswatini, Tanzania, Uganda, Zambia, and Zimbabwe. It has become locally extinct in parts of Uganda, now only occurring in south-west Uganda in the Lake Mburo National Park (Averbeck 2002), and has been extirpated from Burundi, but has also been introduced to Gabon (East 1999; Fritz & Bourgarel 2013). Although the Common Impala occurs widely in southern and East Africa, from central and southern Kenya to northern KwaZulu-Natal, west to Namibia and southern Angola, the Black-faced Impala (A. m. petersi) is naturally confined to the Kaokoland in the north-west parts of Namibia, and neighbouring south-western Angola (Lorenzen et al. 2006). To guard against its extinction, Black-faced Impala were translocated to south-western Etosha, Namibia on the edge of the historic Black-faced Impala range (Green & Rothstein 1998). Today, this subspecies occurs in Namibia between the Otjimborombonga area (ca 12°45’E) and Swartbooisdrift on the Cunene River, southward to the Kaoko Otavi area in the south-western part of the Etosha National Park, and the Kamanjab District just south of the Park (Fritz and Bourgarel 2013). There is no information on the current status of this subspecies in Angola.
Common Impala have been introduced to numerous privately-owned game ranches and small reserves throughout southern Africa. Within the assessment region, this species now occurs in all provinces, although it is extra-limital within the Western, Northern and Eastern Cape as well as the western regions of the North West Province (Castley et al. 2001), throughout the Free State, and also in the Hluhluwe-Umfolozi Park in the province of KwaZulu-Natal . Common Impala were introduced into the Tussen-die-Riviere Nature Reserve (Plug & Badenhorst 2001) in the Free State Province, mainly for hunting purposes (Watson 2006). Black-faced Impala have been introduced into South Africa for wildlife ranching and hunting purposes (G. Castley unpubl. data).
Elevation / Depth / Depth Zones
levation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): NA
Depth Upper Limit (in metres below sea level): NA
Depth Zone: NA
Biogeographic Realms
Biogeographic Realm: Afrotropical
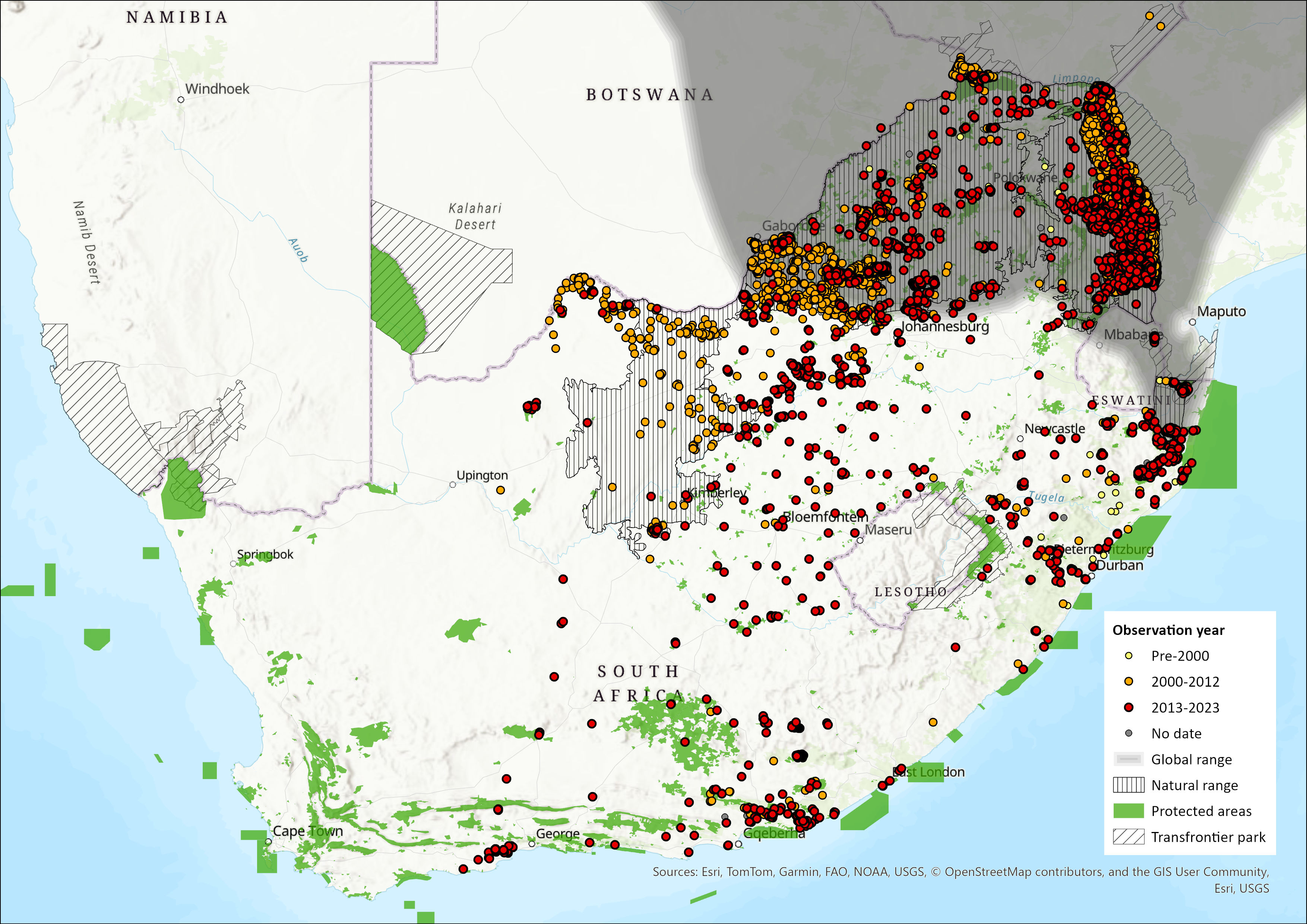
Map
Figure 1. Distribution records for Impala (Aepyceros melampus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burundi | Extinct Post-1500 | Native | – | – |
| Congo, The Democratic Republic of the | Presence Uncertain | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Gabon | Extant | Introduced | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
To date, the only study to explore the potential implication of climate change on impala is that of Hunninck et al. (2020). Using normalised difference vegetation index (NDVI) as a measure of vegetation quality, Hunninck et al. (2020) suggested that climate-induced declines in forage quality could negatively impact impala populations irrespective conservation measures and low levels of human disturbance. However, as impala are mixed feeders (i.e., feed on both grass and woody vegetation), they may be buffered against changes in the availability of specific food types caused by changes in vegetation dynamics and habitat shifts. For example, an increase in woody vegetation via bush encroachment may reduce grass availability, but it increases woody plant resources. However, the acceptability and quality of emerging woody vegetation would determine the full extent to which climate-induced vegetation changes impact impala. Nevertheless, an overall reduction in food availability would be concerning.
Population information
Population estimates are available for most of the Common Impala’s current range. East (1999) estimated the total numbers of Common Impala at ~ 2 million. Although accurate estimates are not available, the subspecies is considered very common in the assessment region and occurs in almost all of the protected areas. For example, it is estimated that 123,998–168,451 individuals occur within the Kruger National Park alone (2023 estimate) and the subpopulation is increasing (South African National Parks unpublished data). The species further occurs on several private game ranches throughout the assessment region. There are > 100 subpopulations within the assessment region. Moreover, within these subpopulations, conservation efforts can positively impact population dynamics. For example, after 15 years of improved conservation efforts in two game reserves adjacent to the Serengeti National Park, Tanzania, the impala density doubled in one of the protected areas, while group sizes increased in both areas (Flølo et al. 2021). The only real isolation is between the KwaZulu-Natal subpopulations and those in the Lowveld (Mpumalanga, Limpopo) areas. Subpopulations are also fragmented by fencing but the constant trade in live animals of this species ensures gene flow is maintained. However, artificial selection for desired colour traits and/or deliberate and accidental hybridisation with Black-faced Impala (A. m. petersi) does pose a threat (Miller et al. 2020). Moreover, increased human disturbance (e.g., pastoralism and hunting) have been found to negatively impact sex ratios, reproduction, and recruitment of impala in the Serengeti ecosystem (Setsaas et al. 2018).
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| No | – | – |
Current population trend: Increasing
Extreme fluctuations in the number of subpopulations: No
Continuing decline in number of subpopulations: No
All individuals in one subpopulation: No
Number of mature individuals in largest subpopulation: 86,715 to 101,168 (using a mean of 144525 individuals and 60-70% mature population structure)
Number of Subpopulations: Unknown but >100
Severely fragmented: Uncertain. Restricted by fences over most of their range but frequently traded at game auctions.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Low
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Low
Probability of extinction in the wild within 100 years: Low
Population genetics
The two recognised subspecies (the Common Impala, A. m. melampus, and the Black-faced Impala, A. m. petersi), are supported by molecular data and have been confirmed to be separate Evolutionarily Significant Units (ESUs) (Nersting & Arctander 2001; Lorenzen et al. 2006). Within the ESU of A. m. melampus strong genetic structure has not been detected; however, microsatellite analysis does suggest some degree of isolation along the east-south gradient, resulting in some distinct genetic clades (Lorenzen et al. 2006, 2012). These are currently referred to as Management Units (MUs), of which five are recognised: 1) northern KwaZulu-Natal, South Africa; 2) Limpopo Province, South Africa; 3) southern Africa: north-eastern Namibia, south-eastern Angola, Botswana, Zimbabwe, Mozambique, Zambia, southern Democratic Republic of the Congo and Malawi; 4) East Africa: central Tanzania, Kenya and Uganda; and 5) Samburu: Samburu National Reserve, Kenya.
Effective population sizes (Ne) for each ESU and MU are not available; however, considering the overall population is estimated to be greater than 2,000,000 for A. m. melampus with the number of mature individuals estimated at>10,000, it is expected that the Ne for each should exceed the Ne 500 threshold. Natural movement of individuals between ESUs and MUs is limited by human modified landscapes (e.g., farms, urban sprawl) and fences. However, the constant trade in live animals of this species ensures gene flow is maintained. Nevertheless, artificial selection for desired colour traits (e.g., melanistic/black impala) and/or deliberate and accidental hybridisation between the Common Impala and Black-faced Impala (A. m. petersi) does pose a threat to the genetic integrity of the species (Miller et al. 2020). Consequently, it is recommended that no mixing of individuals from the different ESUs and MUs takes place until additional population genetic research is done incorporating genes across the genome to verify the genetic structure within this species.
Habitats and ecology
While the natural range of the subspecies comprises predominantly savannah communities, the Common Impala is a generalist and adapts well to other vegetation types. This has contributed to its success as a wildlife ranching species. While this may improve the survival of Common Impala in a variety of habitats, it may also be difficult to remove the species once it has been introduced to these areas if retaining natural species composition is a management objective. The Common Impala is an edge (ecotone) species which throughout its distribution range is associated with woodland which, preferring light woodland with little undergrowth and grassland of low to medium height. While the subspecies generally avoids open grassland and floodplains, it occurs on the ecotone between the two and will graze on open grassland with a flush of fresh green grass. It is absent from montane areas. Cover and the availability of surface water are essential habitat requirements.
Ecosystem and cultural services: The Common Impala is one of the most common antelope within the assessment region and could become the keystone species for a sustainable wildlife-based rural economy and low-carbon food supply. This venison market economy would be determined by sustainable harvest of populations within the assessment region but would still require further investigation to determine specific sustainable yields and associated benefits to local communities and the wildlife industry. Suggestions of how such wildlife driven systems might support both subsistence and commercial ventures are not known (Féron 1995). However, while the commercial farming of certain wildlife species can deliver economic benefits to rural communities it is important to consider the broader implications of such management objectives, particularly in relation to population demographics and impacts on vegetation communities (Gordon et al. 2004). Common Impala have previously been the focus of game cropping initiatives in Kenya and Zambia, although these were generally small-scale operations (Ntiamoa-Baidu 1997). More recently, Averbeck (2002) assessed the potential of cropping and safari hunting of Common Impala in Uganda and concluded that cropping was not feasible given current levels of poaching and the required investment. Should the wildlife industry contemplate the intensive farming of Common Impala to facilitate sustainable cropping it will also be necessary to consider health regulations associated with human consumption (Ramrajh 2012) as well as the condition and welfare of the animals. Lewis et al. (1997) concluded that night cropping of wild impala was a satisfactory method to harvest Common Impala and resulted in better meat quality when compared to diurnal (Kritzinger et al. 2004). Wildlife health risks may compromise the success of cropping operations as Ezenwa (2004) has shown that parasite infection rates in Common Impala are higher in small reserves, as well as those with a high diversity of other bovids.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 4.4. Grassland -> Grassland – Temperate | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
Life History
Generation Length: 9 years
Age at maturity: female or unspecified: 2 years
Age at Maturity: Male: 2 years
Size at Maturity (in cms): Female: Total length: 156.3 cm, Shoulder height: 89.3 cm
Size at Maturity (in cms): Male: Total length: 158.1 cm, Shoulder height: 91.3 cm
Longevity: 12-15 years
Average Reproductive Age: Males: 3 years; Females: 2 years
Maximum Size (in cms): Total length: 162.5 cm, Shoulder height: 91.3 cm
Size at Birth: 5 kg
Gestation Time: 194-200 days
Reproductive Periodicity: Annual
Average Annual Fecundity or Litter Size: 1 lamb
Natural Mortality: 2.3% (Ogutu et al. 2012)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Males move within territories (5-8 ha) during the rut otherwise herds move within home ranges (80-180 ha).
Congregatory: Move in small herds of 6-20 individuals and larger congregations up to 100 individuals.
Systems
System: Terrestrial
General Use and Trade Information
The trade in this species is local subsistence and local, national and international commercial trade in meat, live sales, and trophy hunts. There is no anticipated negative effect on the population. Trade has had a positive effect through the reintroduction of the species into former parts of its range. Even though it is not a high-value species in the ecotourism industry, it is still utilised within this sector. However, where this subspecies has been introduced into areas beyond its natural range, it can subsequently prove difficult to remove, resulting in possible competition with other species and may ultimately become a threat to the indigenous fauna in these areas.
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
Yes |
Species is used locally as a meat source. |
Yes |
Used nationally and internationally for meat, live sales and trophy hunts. |
National Commercial Value: Yes
International Commercial Value: Yes
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
1. Food – human |
true |
– |
– |
– |
|
15. Sport hunting/specimen collecting |
– |
true |
true |
– |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Extensive ranching occurs. Harvested for meat, trophy hunts, and live sales. Small proportion of subpopulations harvested sustainably as part of hunting packages or to control herbivore numbers. Species is intensively bred for colour variants and sold to other breeders.
Threats
There are currently no major threats to the species. However, private landowners within the assessment region may be hybridising Common Impala with Black-faced Impala, which would compromise the genetic integrity of the Common Impala population. This has been identified as a severe threat to the Black-faced Impala (Green & Rothstein 1998). Moreover, Miller et al. (2020) recorded evidence of natural hybridisation between Black-faced Impala and introduced Common Impala Southern Cross Private Game Reserve, Namibia and two potential hybrids in Etosha National Park, Namibia. Similarly, selected breeding for colour variants by game ranchers may pose a threat to the genetic diversity of the Common Impala population though increased rates of inbreeding. Such threats should be quantified and monitored. Expanding human settlements, especially along protected area boundaries (Wittemyer et al. 2008), or around conservancies and game farms, may also result in increased poaching of the Common Impala within the assessment region (Lindsey et al. 2013).
Current habitat trend: Increasing. Game ranching continues to increase the area of occupancy and habitat quality for this generalist species. However, its presence may also ultimately pose a threat to populations of other native wildlife that are unable to compete with this habitat generalist.
Conservation
The Common Impala is one of the most abundant antelopes in Africa, with about one-quarter of the population occurring in protected areas. Its future is secure as long as it continues to occur in large, adequately protected, and managed populations in protected areas, private farms and conservancies. Landowners should continue to form conservancies to sustain wild and free-roaming herds and to share the economic and cultural benefits of this species with local communities. However, regulation of translocation is required to prevent inbreeding and hybridisation with the extra-limital Black-faced Impala and to prevent Common Impala causing habitat degradation outside of its natural range. Similarly, reintroduction efforts should consider whether it is within the natural range of the subspecies and should reintroduce a suitable founder size. For example, the success of Black-faced Impala translocations was heavily influenced by the size of the founder population as well as the presence of predators in the recipient area (Matson et al. 2004). Reintroductions should follow the IUCN guidelines (IUCN/SSC 2013).
Recommendations for land managers and practitioners:
- Sustainable utilisation of the Common Impala should be a priority for private landowners and communities who want to galvanise a wildlife-based economy. Conservationists should provide incentives for landowners to provide affordable, low-carbon protein to local communities and to create conservancies where the benefits of this subspecies are shared.
- An important consideration for wildlife managers is the possible role that Common Impala may play in propagating foot and mouth disease in other wildlife and domestic livestock populations. Impala are known carriers of foot and mouth disease and have transmitted the disease to cattle in the past (Vosloo et al. 2006). Consequently, control measures may need to be put in place in situations where wildlife and livestock may interact (Vosloo et al. 2009), or where Common Impala are sourced from potential foot and mouth disease areas for reintroduction to other areas.
- There are no requirements for future supplementation from captive stocks, and captive breeding for conservation is not recommended.
Research priorities:
- The extent of artificial selection and hybridisation within the wildlife ranching industry. Detailed information related to the trends in Impala populations within private reserves.
- Further exploration of potential hybridisation with Black-faced impala in Namibia.
- The feasibility of this species as alternative and sustainable source of protein in rural communities.
- Methods of creating wildlife-based economies from this species and its efficacy as a source of protein for local communities.
- More robust harvesting models are required that will help managers calculate off-take rates that will ensure sustainable populations. Currently, no research is being conducted on such models.
- Evaluation of this species impact on the environment where it has been introduced/or applications to introduce it is pending approval.
Encouraged citizen actions:
- Raise awareness among the hunting community of the risks to wildlife populations from proliferation of selectively-bred colour variants.
Bibliography
Averbeck C. 2002. Fakultät für Ernährung, Landnutzung und Umwelt, Technische Universität München.
Castley JG, Boshoff AF, Kerley GIH. 2001. Compromising South Africa’s natural biodiversity: inappropriate herbivore introductions. South African Journal of Science 96: 365–378.
East, R. (compiler). 1999. African Antelope Database 1998. IUCN, Gland, Switzerland and Cambridge, UK.
Ezenwa, V.O. 2004. Parasite infection rates of impala (Aepyceros melampus) in fenced game reserves in relation to reserve characteristics. Biological Conservation 118: 397-401.
Ferreira S, Gaylard, A, Greaver, C, Hayes, J, Cowell C, Ellis G. 2013. Animal abundances in Parks 2012/2013. Scientific Services, SANParks, Skukuza, South Africa.
Flølo, L.M., Hunninck, L., May, R., Jackson, C.R., Setsaas, T.H., Holmern, T. and Røskaft, E. 2021. Behavioural and demographic changes in impala populations after 15 years of improved conservation management. Global Ecology and Conservation 27: e01586.
Fritz, H. and Bourgarel, M. 2013. Aepyceros melampus Impala. In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. VI. Pigs, Hippopoyamuses, Chevrotain, Giraffes, Deer, and Bovids, pp. 480-487. Bloomsbury Publishing, London, UK.
Féron, E.M. 1995. New food sources, conservation of biodiversity and sustainable development: can unconventional animal species contribute to feeding the world? Biodiversity and Conservation 4: 233-240.
Gordon IJ, Hester AJ, Festa-Bianchet M. 2004. The management of wild large herbivores to meet economic, conservation and environmental objectives. Journal of Applied Ecology 41: 1021-1031.
Green, W.C.H. and Rothstein, A. 1998. Translocation, hybridization, and the endangered Black-Faced Impala. Conservation Biology 12(2): 475-480.
Hunninck, L., May. R., Jackson, C.R., Palme, R., Røskaft, E. and Sheriff, M.J. 2020. Consequences of climate-induced vegetation changes exceed those of human disturbance for wild impala in the Serengeti ecosystem. Conservation Physiology 8: coz117.
IUCN/SSC. 2013. Guidelines for Reintroductions and Other Conservation Translocations. Version 1.0. Page viiii + 57 pp. IUCN Species Survival Commission, Gland, Switzerland.
Kritzinger B, Hoffman LC, Ferreira AV. 2004. A comparison between the effects of two cropping methods on the meat quality of impala (Aepyceros melampus). South African Journal of Animal Science 33: 233-241.
Lorenzen ED, Heller R, Siegismund HR. 2012. Comparative phylogeography of African savannah ungulates. Molecular Ecology 21: 3656-3670.
Lorenzen, E.D. and Siegismund, H.R. 2004. No suggestion of hybridization between the vulnerable black-faced impala (Aepyceros melampus petersi) and the common impala (A. m. melampus) in Etosha National Park, Namibia. Molecular Ecology 13: 3007-3019.
Lorenzen, E.D., Arctander, P. and Siegismund, H.R. 2006. Regional genetic structuring and evolutionary history of the impala Aepyceros melampus. Journal of Heredity 97: 119–132.
Matson TK, Goldizen AW, Jarman PJ. 2004. Factors affecting the success of translocations of the black-faced impala in Namibia. Biological Conservation 116: 359-365.
Miller, S.M., Moeller, C-H, Harper, C.K. and Bloomer, P. 2020. Anthropogenic movement results in hybridisation in impala in southern Africa. Conservation Genetics 21: 653-663.
Nersting L.G. and Arctander, P. 2001. Phylogeography and conservation of impala and greater kudu. Molecular Ecology 10: 711-719.
Nestling, LG, Arctander P. 2001. Phylogeography and conservation of impala and greater kudu. Molecular Ecology 10: 711-719.
Ntiamoa-Baidu Y. 1997. Wildlife and food security in Africa. FAO Conservation Guide 33, Food and Agriculture Organization of the United Nations, Rome. Available at: http://www.fao.org/docrep/w7540e/w7540e00.htm#Contents.
Ogutu, J.O., Piepho, H-P., and Kanga, E. 2012. Dynamics of an insularized and compressed impala population: Rainfall, temperature, and density influences. The Open Ecology Journal 5:1-17.
Plug, I. and Badenhorst, S. 2001. The distribution of macromammals in southern Africa over the past 30,000 years. Transvaal Museum, Pretoria.
Ramrajh S. 2012. Participatory risk assessment for harvesting of impala (Aepyceros melampus) and the distribution of by-products. MMedVet Thesis, University of Pretoria, Pretoria, South Africa.
Setsaas, T., Hunninck, L. Jackson, C.R., May, R. and Røskaft, E. 2018. The impacts of human disturbances on the behaviour and population structure of impala (Aepyceros melampus) in the Serengeti ecosystem, Tanzania. Global Ecology and Conservation 16: e00467.
Vosloo W, Bastos ADS, Boshoff CI. 2006. Retrospective genetic analysis of SAT-1 type foot-and-mouth disease outbreaks in southern Africa. Archives of Virology 151: 285-298.
Vosloo W, Thompson PN, Botha B, Bengis RG, Thomson GR. 2009. Longitudinal Study to Investigate the Role of Impala (Aepyceros melampus) in Foot-and-Mouth Disease Maintenance in the Kruger National Park, South Africa. Transboundary and Emerging Diseases 56: 18-30.
Watson JP. 2006. Check list of the mammals of Tussen-die-Riviere Provincial Nature Reserve, Free State Province, South Africa. Koedoe 49: 111-117.
Wittemyer, G., Elsen, P., Bean, W.T., Burton, A.C.O. and Brashares, J.S. 2008. Accelerated human population growth at protected area edges. Science 321: 123-126.