Giant Rat
Cricetomys ansorgei

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Cricetomys ansorgei – Thomas, 1904
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – NESOMYIDAE – Cricetomys ansorgei
Common Names: Giant Rat, Southern Giant Pouched Rat (English), Reuse Rot (Afrikaans), Ndovhi (Venda)
Synonyms: C. gambianus adventor Thomas & Wroughton 1907; C. g. viator Thomas 1904; C. g. cunctator Thomans & Wroughton 1908; elongis Thomas 1910; C. g. enguvi Heller 1912; C. g. osgoodi Heller 1912; C. g. consensi Hinton 1919; C. g. haagneri Roberts 1926; C. g. vaughan-jonesi St Leger 1937; C. g. selindensis Roberts 1946.
Taxonomic status: Species
Taxonomic Note: While previously included under C. gambianus, it has been elevated to species status (Musser & Carleton 2005; Olayemi et al. 2012). Olayemi et al. (2012) used mitochondrial cytochrome b phylogeny in combination with cranial measurement data to separate this African genus into six species (C. gambianus, C. ansorgei, C. emini, C. kivuensis and two undescribed taxa). Currently the precise distributional limits of the species are undefined. However, they can be broadly differentiated based on their geographic ranges (Monadjem et al. 2015). Cricetomys ansorgei is distributed in the savannahs of East Africa and the northern regions of southern Africa. This species cannot be distinguished from other Cricetomys species based on morphological characteristics but is dissimilar on a molecular level (Olayemi et al. 2012) and is the only species of this genus occurring within the assessment region.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Richards, L.R.1 & da Silva, J.M.2
Reviewers: Taylor, P.3, Smith, C.4 & Patel, T.4
Institutions:1Durban Natural Science Museum,2South African National Biodiversity Institute,3University of the Free State,4Endangered Wildlife Trust
Previous Assessors and Reviewers: MacFayden, D., Richards, L. & Relton, C.
Previous Contributors: Child, M.F., Avenant, N.L., Avery, M., Baxter, R., Monadjem, A., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
Although the species is on the edge of its southerly range within South Africa, it occurs in a number of protected areas, tolerates habitat modification (commensal with humans), and is expanding its range in parallel with human settlements. The added availability of food associated with these settlements has resulted in increased numbers in these areas. There are currently no major threats that could result in population decline. Although heavily hunted for bushmeat in some areas, their numbers seem to be increasing as they are regularly detected on camera traps in local communities and are often considered a crop pest. While Friedmann and Daly (2004) listed this species as Vulnerable C1 based on suspected small population size and the assumption that the population was isolated, combined with the assumption that it was threatened by agricultural expansion, we know that the species is resilient to anthropogenic disturbance and is likely to be expanding with human settlements. Furthermore, contrary to what was reported in 2004, this species is connected with the rest of its range through rural settlements and rescue effects are possible. The species remains as Least Concern.
Regional population effects: The population is not isolated, and its range appears to be continuous throughout the savannah regions of southern Africa. It is a large species, capable of dispersal, and is not hampered by urban and peri-urban environments. Rescue effects are possible through dispersal from Botswana, Zimbabwe and Mozambique.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Richards LR & da Silva JM. 2025. A conservation assessment of Cricetomys ansorgei. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The range of C. ansorgei is widespread in the savannahs of southern and East Africa; it appears to be replaced by an undescribed taxon from Central Africa (Cricetomys sp 2 sensu Olayemi et al. 2012), whose range extends into the Congo Basin where the forest has been cleared (Monadjem et al. 2015). The northern limits of this species are unknown, particularly in East Africa, where its relationship to C. gambianus is not resolved (Monadjem et al. 2015).
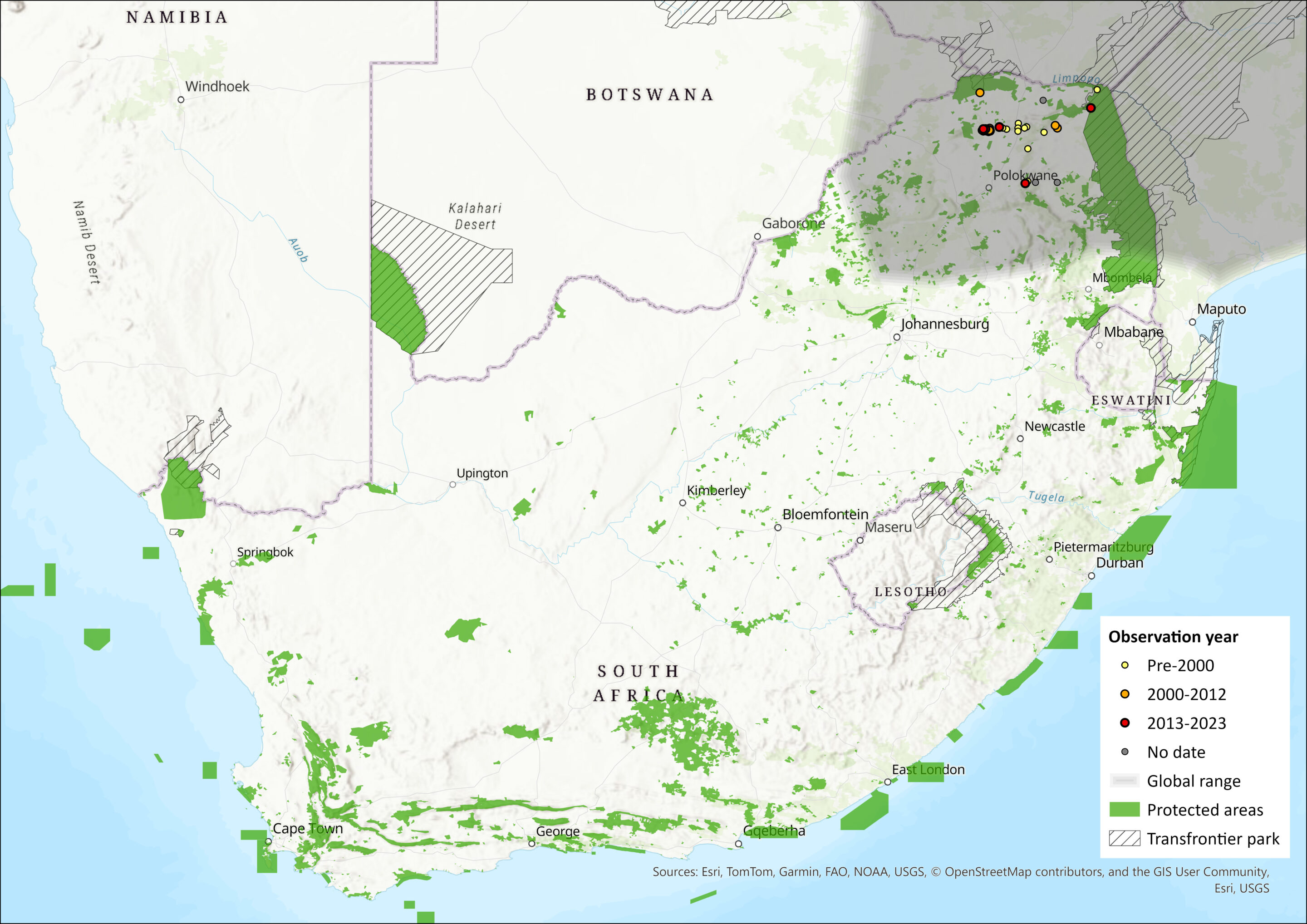
Within the assessment region, they occur in the Limpopo Province of South Africa, including Punda Maria in the northern Kruger National Park and within the Thohoyandou district, inclusive of the Vhembe Biosphere and associated reserves. De Graaff (1981) suggested that they occur in St Lucia, KwaZulu-Natal. However, subsequent surveys discount its presence in this area (Taylor 1998). Although Friedmann and Daly (2004) suggested the South African population was isolated in the Limpopo Province limited almost entirely to the Soutpansberg Mountains, new information suggests a more extensive distribution and a range expansion along with human settlements, which are expanding on the boundaries of protected areas (sensu Wittemyer et al. 2008). This species is relatively common in Punda Maria rest camp in the Kruger National (KNP), with occurrence records published to iNaturalist and GBIF (GBIF.org 2024; Vos and iNaturalist 2024). The species also occurs at Pafuri in the north of the KNP (D. MacFadyen unpubl. data).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not Specified)
Elevation Upper Limit (in metres above sea level): (Not Specified)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
Map
Figure 1. Distribution records for Giant Rat (Cricetomys ansorgei) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence within southern Africa
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
To date, no empirical research has examined the impact of climate change on this species. However, physiological studies on the thermoregulatory traits of Cricetomys ansorgei may offer insights into the potential effects of global climatic variability on southern African populations.
Cricetomys ansorgei has been reported to exhibit a mean body temperature of 35.6 ± 1.11 °C, with a thermoneutral zone ranging from 21 °C to 34 °C (Knight 1988). Knight (1984) proposed that the species selects moisture-rich foods and constructs deep, typically cool burrows to counteract its high thermal conductance and limited capacity to maintain homeostasis in ambient temperatures exceeding 37 °C. These physiological and ecological requirements likely prevent the species from inhabiting more arid environments (Knight 1988). Furthermore, thermal stress resulting from seasonal fluctuations is becoming increasingly severe and frequent (Fuller et al. 2020). Projections suggest that climate change will intensify these conditions, potentially leading to shifts in species distributions and elevational range.
Population Information
This species is relatively common within its range. While no density estimates are available from South Africa, 42 individuals were taken from a 0.5 ha garden in Zimbabwe (Smithers 1983). Its ability to utilise urban and peri-urban environments has allowed population increases in certain areas. Further research is needed to confirm the population size within the assessment region.
Population Information
Current population trend: Stable, with some increases with urban and peri-urban areas across the range.
Continuing decline in mature individuals: No evidence to suggest drastic declines in mature individuals in the wild
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No, this species is known to disperse alongside expanding human settlements.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While no population genetic studies have been undertaken on this species, it is assumed to exist as a single metapopulation. From this, one of the two genetic indicators in the Convention of Biological Diversity’s Global Biodiversity Framework can be quantified – the complementary indicator- proportion of populations maintained within a species – with 1 of 1 populations remaining, resulting in a value of 1.0 for this indicator.
The GBF’s headline indicator – proportion of populations with an effective population size (Ne) greater than 500 – cannot be quantified given limited information on population sizes and densities.
Habitats and ecology
The species occurs in various habitats including savannah, forest and woodland (Mugo et al. 1995; Duplantier and Granjon 2013), as well as farmland, cropland, plantations and rural areas. It is considered to be an adaptable species that is even known to invade sewers. According to Skinner and Chimimba (2005), it has been observed to be expanding its range alongside rural settlements and is not believed to be affected by decline in habitat quality. This would be expected in South Africa, particularly in Thohoyandou. Modified habitats are suitable for this species, including cane fields, subsistence farms and human settlements.
The species is primarily nocturnal but is occasionally active during the late afternoons or early mornings, usually limited to areas of suitable vegetation cover (Skinner & Chimimba 2005). They are omnivorous and hoard food within burrows, generally located at the base of large trees or under boulders, where the environment is moist and well-shaded (Knight 1986). Smithers (1983) noted that these rats are prone to gathering small, often brightly coloured miscellaneous objects, such as bottle caps, string, nails, bones and wire, and storing them within their burrows. Although uncommon, arboreal behaviour is not entirely absent, particularly within agricultural areas with fruit trees.
As a generalist, and due to slow digestive rates and a complex digestive system, this species is able to forage successfully on poor quality food. These ominovores will consume a variety of food types (such as fruit, bulbs, grains and insects, particularly termites), but primarily vegetable matter. The large cheek pouches are used to transport food and nesting materials. Cultivated foods, such as mangoes, avocados, pecans, macadamias, maize, sorghum, pumpkins and beans, are particular favourites (Skinner & Chimimba 2005). They appear to be directly dependent on the availability of fresh water, or at least their presence in fleshy fruits (Skinner & Chimimba 2005).
They are generally solitary; however, pairs may remain together in a single burrow while breeding (Skinner & Chimimba 2005). Litter size ranges from 2–4 (Knight 1986; Duplantier and Granjon 2013), and this species is known to reproduce well in captivity (Skinner & Chimimba 2005).
Ecosystem and cultural services:
- This species has an excellent sense of smell and is used as a mine-detection animal in Mozambique and Tanzania. In 2009, the rats searched 93,400 m2 of Mozambique, finding 41 mines and 54 other explosive devices, while humans with metal detectors found no additional mines (Poling et al. 2010, 2011; https://www.apopo.org/en/).
- In addition to landmine detection, the species has been trained to detect tuberculosis (https://www.apopo.org/en/)
- It is an important source of protein for some rural communities and the bushmeat trade may be sustainable if regulated, similar to Cane Rat (Thryonomys swinderianus) farming in West and central Africa (Adu et al. 1999).
Life History
Generation Length: Captive-bred individuals typically live for 5 to 8 years; however, some have been documented to live significantly longer, with recorded lifespans of up to 14 years (https://www.apopo.org/en/
Age at Maturity: Female or unspecified: Approximately 5 months (Duplantier and Granjon 2013)
Age at Maturity: Male: c. 20 weeks
Size at Maturity (in cms): Head and body length (sexes combined) = 34.7 cm (Monadjem et al. 2015)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: Testicular descent in males typically occurs at approximately 77 days (Knight 1984)
Maximum Size (in cms): Head and body length (sexes combined) = 41. 3 cm (Monadjem et al. 2015)
Size at Birth (in cms): Birth weight range = 16 – 27 g (Duplantier and Granjon 2013)
Gestation Time: Data from other regions in southern Africa suggest that gestation lasts between 27 and 42 days (Morris 1963; Ewer 1967)
Reproductive Periodicity: Births have been recorded between September and May in Malawi (Duplantier and Granjon 2013)
Average Annual Fecundity or Litter Size: 1- 5 young born per litter (Duplantier and Granjon 2013)
Natural Mortality: A known prey item of various raptors, including eagle owls (Duplantier and Granjon 2013)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Home range is generally 5 hectares but can range between 2.2 to 11 hectares (Skinner and Smithers 1990; Duplantier and Granjon 2013). Currently, there is a lack of information regarding potential long-distance movement patterns.
Congregatory: Within the assessment region, individuals were observed occupying separate burrows (Ewer 1967), however breeding pairs may be found in the same burrow (Skinner & Chimimba 2005). In contrast, instances of multiple individuals sharing a single burrow have been documented in Nigeria (Duplantier & Granjon 2013).
Systems
System: Terrestrial
General Use and Trade Information
Anecdotal evidence suggests that this species is utilised for bushmeat by local communities in Limpopo and in the pet trade by farmers. In Mozambique and Tanzania, this species is used to detect mines, due to its excellent sense of smell (Poling et al. 2010). They are now also being used to detect tuberculosis (Poling et al. 2011). This species is frequently traded and bred as pets (Cooper 2008). They are easily tamed as pets and, until recently, were commonly imported as pets to the United Kingdom and United States. However, in 2003 they were associated with an outbreak of monkeypox in the USA (Witmer et al. 2010) and have since been banned from importation to the USA.
National Commercial Value: None reported, however they may support local communities that rely on them as an occasional food source.
International Commercial Value: N/A
Is there harvest from captive/cultivated sources of this species? Although no captive breeding colonies established for harvesting purposes are known within the assessment region, such practices have been reported from West Africa (Ajayi 1975).
Harvest Trend Comments: (Not specified)
Threats
There are no major threats to this adaptable species. However, there are a few minor threats to the species within the assessment region:
- It is hunted for bushmeat by local communities in the Limpopo Province, but this is not as prevalent as in other African countries.
- It has potential to be used in the pet trade, but this is considered a minor threat as the species is not regularly recorded in pet-trade syndicates within the assessment region.
- It is considered a pest in certain areas, and the use of poison by farmers is a possible threat.
- Although this species is able to adapt well to modified habitats, the expansion of alien invasive vegetation (especially Eucalyptus spp. and Pinus spp.) may have an effect on habitat suitability for this species.
- Domestic animals, such as dogs, may be a minor threat to this species in urban and peri-urban environments.
However, no population declines have been reported for the above threats, and the species appears resilient to anthropogenic disturbances.
Current habitat trend: Stable
Conservation
The species occurs in several protected areas within the assessment region such as the Kruger National Park, the Greater Mapungubwe Transfrontier Conservation Area, Lajuma Private Nature Reserve and Happy Rest Nature Reserve. Wildlife ranches are expected to make a limited contribution to the conservation status of the species as modified habitats are often utilised (Skinner & Chimimba 2005). No specific interventions are necessary at present, as their numbers are suggested to be increasing, particularly in vegetable gardens and agricultural areas alongside their forest habitats (Mugo et al. 1995).
Recommendations for land managers and practitioners:
- This species could potentially form part of the rural, wildlife-based economy by becoming low-carbon, cheap protein. They have a faster reproductive rate than Cane Rats and could potentially be farmed for sustainable utilisation. In West Africa, Gambian Rat meat is considered a marketable proposition (Mainka & Trivedi 2002).
- Investigate breeding programmes to replace harvesting animals from the wild.
Research priorities:
- Population genetic studies to resolve taxonomic uncertainty concerning species limits of C. ansorgei and C. gambianus.
- Research into the biology of the species in the wild, with special reference to population size, trend estimates, and reproduction and dispersal rates.
- Geographic distribution within the assessment region and patterns of dispersal with rural settlements.
- Efficacy of farming this species as a low-carbon protein source, assessment of demand and supply.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Support the HeroRATS Project where this species is used to detect land-mines and tuberculosis.
- Promote organic methods of problem animal control, without resorting to poisons.
Bibliography
Adu EK, Alhassan WS, Nelson FS. 1999. Smallholder farming of the greater cane rat, Thryonomys swinderianus, Temminck, in southern Ghana: a baseline survey of management practices. Tropical Animal Health and Production 31:223–232.
Ajayi, S.S., 1975. Observations on the biology, domestication and reproductive performance of the African giant rat Cricetomys gambianus Waterhouse in Nigeria. Mammalia 39(3): 343-364.
Cooper RG. 2008. Care, husbandry and diseases of the African giant rat (Cricetomys gambianus). Journal of the South African Veterinary Association 79:62–66.
De Graaff G. 1981. The Rodents of Southern Africa. Butterworths, Durban, South Africa.
Duplantier J-M, Granjon L. 2013. Cricetomys gambianus Gambian giant pouched rat, pp 159-161 in Happold DCD (editor). Mammals of Africa: Volume III. Bloomsbury Publishing, London.
Dzenda T, Ayo JO, Lakpini CA, Adelaiye AB. 2011. Diurnal, seasonal and sex variations in rectal temperature of African Giant rats (Cricetomys gambianus, Waterhouse). Journal of Thermal Biology 36(5):255-63.
Ewer RF. 1967. The behaviour of the African giant rat (Cricetomys gambianus Waterhouse). Zeitschrift für Tierpsychologie 24(1): 6–79
Friedmann Y, Daly B, editors. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. IUCN SSC Conservation Breeding Specialist Group and Endangered Wildlife Trust, South Africa.
Fuller A, Maloney SK, Blache D, Cooper C. 2020. Endocrine and metabolic consequences of climate change for terrestrial mammals. Current Opinion in Endocrine and Metabolic Research 1;11:9-14.
GBIF.org (2024) GBIF Occurrence Download https://doi.org/10.15468/dl.ndmsuw. Accessed on 06 November 2024
Knight MH. The Ecophysiology of the African Giant Rat Cricetomys gambianus (Weterhouse). University of Pretoria (South Africa); 1984.
Knight MH. 1986. Thermoregulation and evaporative water loss in growing African giant rats Cricetomys gambianus. African Zoology 21:289–293.
Knight MH. Thermoregulation in the largest African cricetid, the giant rat Cricetomys gambianus. Comparative Biochemistry and physiology. A, Comparative Physiology 89(4):705-8.
Mainka S, Trivedi M, editors. 2002. Links between Biodiversity Conservation, Livelihoods and Food Security: The sustainable use of wild species for meat. IUCN, Gland, Switzerland and Cambridge, UK.
Monadjem A, Taylor PJ, Denys C, Cotterill FPD. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. De Gruyter, Berlin, Germany.
Morris B. 1963. Notes on the Giant rat (Cricetomys gambianus) in Nyassaland. African Wildlife 17:103-7.
Mugo DN, Lombard AT, Bronner GN, Gelderblom CM. 1995. Distribution and protection of endemic or threatened rodents, lagomorphs and macrosceledids in South Africa. South African Journal of Zoology 30:115–126.
Musser GG, Carleton MD. 2005. Superfamily Muroidea. Pages 894–1531 in Wilson DE, Reeder DA, editors. Mammal Species of the World: a Geographic and Taxonomic Reference. The John Hopkins University Press, Baltimore, USA.
Olayemi A, Nicolas V, Hulselmans JAN, Missoup AD, Fichet- Calvet E, Amundala D, Dudu A, Dierckx T, Wendelen W, Leirs H, Verheyen E. 2012. Taxonomy of the African giant pouched rats (Nesomyidae: Cricetomys): molecular and craniometric evidence support an unexpected high species diversity. Zoological Journal of the Linnean Society 165:700–719.
Poling A, Weetjens B, Cox C, Beyene NW, Bach H, Sully A. 2011. Using trained pouched rats to detect land mines: another victory for operant conditioning. Journal of Applied Behavior Analysis 44:351–355.
Poling A, Weetjens BJ, Cox C, Beyene NW, Sully A. 2010. Using giant African pouched rats (Cricetomys gambianus) to detect landmines. The Psychological Record 60:715.
Skinner JD, Chimimba CT. 2005. The Mammals of the Southern African Subregion. Third edition. Cambridge University Press, Cambridge, UK.
Skinner, J.D. and Smithers, R.H.N. 1990. The Mammals of the Southern African Sub-Region. University of Pretoria Press, Pretoria, 604-606.
Smithers RHN. 1983. The Mammals of the Southern African Subregion. First edition. University of Pretoria Press, Pretoria, South Africa.
Taylor PJ. 1998. Regional patterns of small mammal abundance and community composition in protected areas in KwaZulu-Natal. Durban Museum Novitates 23:42–51.
Vos, P., iNaturalist (2024). iNaturalist Research-grade Observations. iNaturalist.org. Occurrence dataset https://doi.org/10.15468/ab3s5x accessed via GBIF.org on 2024-11-06. https://www.gbif.org/occurrence/3986147786
Witmer GW, Snow NP, Burke PW. 2010. Potential attractants for detecting and removing invading Gambian giant pouched rats (Cricetomys gambianus). Pest Management Science 66:412–416.
Wittemyer G, Elsen P, Bean WT, Burton ACO, Brashares JS. 2008. Accelerated human population growth at protected area edges. Science 321:123–126.