
Egyptian Tomb Bat
Taphozous perforatus

2025 Red list status
Near threatened
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Taphozous perforatus – É. Geoffroy, 1818
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – EMBALLONURIDAE – Taphozous – perforatus
Common Names: Egyptian Tomb Bat, African Taphozous, Geoffroy’s Tomb Bat, Lesser Tomb Bat, Perforated Taphozous Bat (English), Egiptiese Witlyfvlermuis (Afrikaans)
Synonyms: Taphozous sundani Thomas, 1915; Taphozous perforatus sudani Thomas, 1915; Taphozous perforatus haedinus Thomas, 1915; Taphozous perforatus australis Harrison, 1962; Taphozous perforatus rhodesiae Harrison, 1964
Taxonomic Note:
Meester et al. (1986) recognised three subspecies, including the nominate subspecies (Taphozous perforatus perforatus) from Egypt (where this species was initially described), T. p. sudani Thomas, 1915 from Sudan, and T. p. haedinus Thomas, 1915 from Kenya. However, it remains uncertain how the geographically isolated southern African population (named T. p. rhodesiae, and described from the Shashi-Limpopo confluence; Harrison 1964) is related to these subspecies (Monadjem et al. 2020). T. p. rhodesiae was originally recognised as a subspecies of T. sudani (T. sudani australis), which was previously identified as a distinct species from T. perforatus (ACR 2024).
Red List Status: NT – Near Threatened D2 (IUCN version 3.1)
Assessment Information
Assessors: Richards, L.R.1, da Silva, J.M.2
Reviewer: Taylor, P.3
Institutions: 1Durban Natural Science Museum, 2South African National Biodiversity Institute, 3University of the Free State
Previous Assessors and Reviewers: Richards, L.R., Cohen, L., Jacobs, D., MacEwan, K., Monadjem, A., Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Raimondo, D., Nicholson, S.K, & Relton, C.
Assessment Rationale
Although widespread throughout sub-Saharan Africa, the Arabian Peninsula and the Indian subcontinent, the species is only recorded from two isolated subpopulations in the assessment region (the Border Cave along the Lebombo escarpment on the South Africa-Eswatini border, and the Limpopo and Shashi confluence along the borders of South Africa and southern Zimbabwe, with no evidence of more localities within the assessment region); this is likely the southernmost limit of the range of the species in sub-Saharan Africa. The nearest known colonies are in southern Zimbabwe and adjacent Mozambique. Specific threats and population trends are unknown. However, monitoring data from the Border Cave colony indicates a stable or increasing subpopulation between 2011 and 2014 (L. Richards, unpublished data). Although this species qualifies for Vulnerable D2 based on two locations, it is uncertain whether there are any plausible threats (although see below), and thus we list as Near Threatened D2 due to the regional criterion.
Regional population effects: It has high wing loading (Norberg & Rayner 1987) and thus dispersal capacity is assumed to be good. However, the population is sparsely distributed in southern Africa, and it is unclear whether significant rescue effects are possible. For the purpose of this assessment, we assume rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Richards LR & da Silva JM. 2025. A conservation assessment of Taphozous perforatus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Please scroll horizontal on mobile to view tables
Regional Distribution and occurrence
Geographic Range
The Egyptian Tomb Bat occurs widely throughout northern and sub-Saharan Africa, the Arabian Peninsula, and east to the Indian subcontinent. In sub-Saharan Africa, records extend along the Nile and east to Ethiopia and northern Somalia, and west to Mauritania, Senegal, Gambia, Guinea-Bissau, Ghana, Burkina Faso, Benin, Niger, and northern Nigeria, and south to Kenya (including Lamu Island), Tanzania, Democratic Republic of the Congo, Zambia, Zimbabwe, Mozambique and Botswana (Kock et al. 2008; Monadjem et al. 2020; ACR 2024). However, it is patchily distributed in southern Africa, occurring in southern Zimbabwe, Mozambique and an isolated subpopulation in the Okavango Delta of Botswana (Monadjem et al. 2020).
In the assessment region, the species appears to be peripheral, recorded from Border Cave along the Lebombo escarpment on the South Africa-Eswatini border (White 2011; specimen records of the Durban Natural Science Museum) and from the northern borders of the Limpopo and Shashi River confluence (adjacent to the Mapungubwe National Park) along the borders of South Africa and southern Zimbabwe (Harrison 1962; Skinner & Chimimba 2005). It was recently documented from the Ratho Bush Camp along the southern bank of the Limpopo River and ca. 35 km west of the Limpopo and Shashi River confluence (Gauteng and Northen Regions Bat Interest Group record). The species may occur or utilise areas of similar topography to Border Cave within the greater Jozini area and the immediate low-lying regions of Eswatini. Despite the lack of vouchered specimen records, predictive species distribution models support species occurrence within Eswatini (Monadjem et al. 2021). The species has an EOO of 95,948 km2 within the assessment region.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): Unknown
Elevation Upper Limit (in metres above sea level): Unknown
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
Map
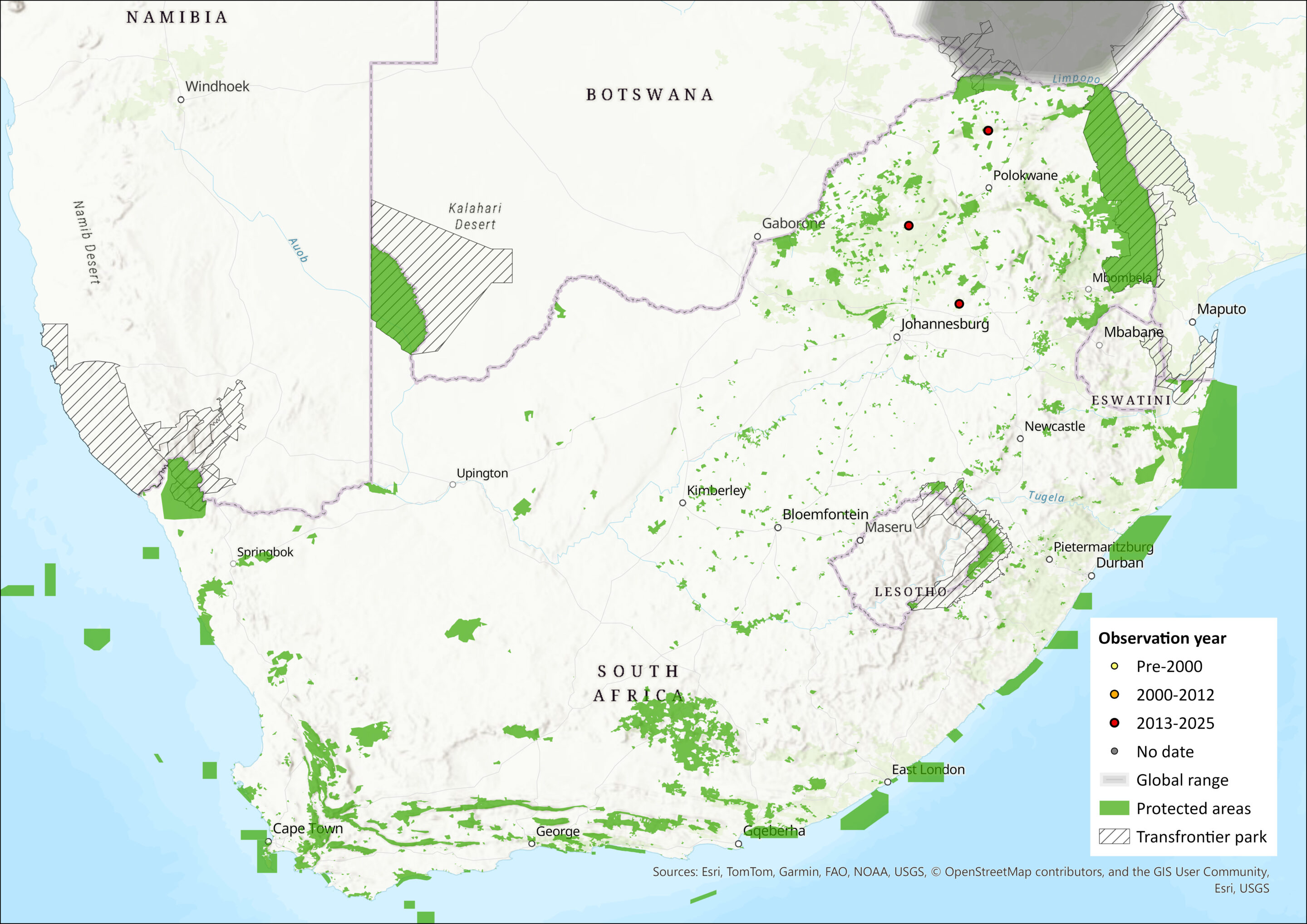 Figure 1. Distribution records for Egyptian Tomb Bat (Taphozous perforatus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Egyptian Tomb Bat (Taphozous perforatus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Indomalayan, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Presence Uncertain | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Presence Uncertain | Native | – | – |
| Djibouti | Extant | Native | – | – |
| Egypt | Extant | Native | – | – |
| Eritrea | Presence Uncertain | Native | – | – |
| Eswatini | Possibly Extant | Native | – | Resident |
| Ethiopia | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Presence Uncertain | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| India | Extant | Native | – | – |
| Iran, Islamic Republic of | Extant | Native | – | – |
| Israel | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Mozambique | Presence Uncertain | Native | – | – |
| Namibia | Presence Uncertain | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Oman | Extant | Native | – | – |
| Pakistan | Extant | Native | – | – |
| Palestine, State of | Presence Uncertain | Native | – | – |
| Saudi Arabia | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | Resident |
| South Sudan | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| United Arab Emirates | Extant | Native | – | – |
| Yemen | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Of the 19 South African national parks, Mapungubwe National Park is deemed the most susceptible to climate change impacts (Coldrey et al. 2022). It is predicted that by 2050, the semi-arid Mapungubwe region, will no longer support 43% of the current known mammalian taxa (Coldrey et al. 2022). Furthermore, there has been an overall decrease in the number of days of rainfall and a noted increase in minimum and maximum temperatures at both coastal and inland regions of the KwaZulu-Natal province (Ndlovu et al. 2021), where the species occurs at a single site. These trends are likely linked to climate change and indicate to drier conditions in future (Ndlovu et al. 2021). Climate change is recognised as a potential threat to most bat species, as the climate is known to influence the biogeography of bats, the availability of food, energetic expenditure, as well as the timing and duration of important processes such as reproduction, hibernation, development and torpor (Sherwin et al. 2013). However, this remains to be verified as a threat to the species within the assessment region.
Population information
The Egyptian Tomb Bat is common in parts of its African range, but is less common elsewhere. It is found in small colonies (between six to eight individuals) in the southern African subregion (Harrison 1962; Skinner & Chimimba 2005; Monadjem et al. 2020) but can number in the hundreds in southern Zimbabwe (Monadjem et al. 2020). In South Asia the abundance, population size and trends for this species are not known, and the species has only been recorded from a few localities (Bates & Harrison 1997). Direct observation and counting of individuals at Border Cave (border between South Africa and Eswatini over a three period, suggests that the population may be increasing. In 2014, it contained between 20-25 individuals (L.R. Richards unpubl. data), from an estimated 10–15 individuals originally observed in 2011 (White 2011). The southern African population of this species is poorly represented in museums, with just 28 records used in Monadjem et al. (2020).
Current population trend: Unknown
Continuing decline in mature individuals? Unknown
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: Unknown
All individuals in one subpopulation: Unknown)
Number of mature individuals in largest subpopulation: > 20 based on known numbers of individuals at Border Cave and netted individuals from the Limpopo province
Number of Subpopulations: One
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Cannot be determined
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Cannot be determined
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
No population genetic studies have been conducted on the species; however, based on the ecomorphology of the species, it is capable of long-distance dispersal and hence is thought to exist as a single population within the assessment region, possibly also connecting to neighbouring countries in southern Africa. A population genetic/genomic study is required to confirm this. Population size estimates are unknown for the species within the assessment region; therefore, a measure of effective population size (Ne) is not possible to quantify at this time.
Habitats and ecology
Throughout its range, this species is commonly associated with open woodland habitats, frequently found along rivers in wooded savannah, but avoids forest, semi-desert and desert areas (Skinner & Chimimba 2005) and prefers open areas where suitable cavernaculous day-roosts are present (Monadjem et al. 2020). It requires the shelter of dark crevices or corners in caves, rock overhangs, stone buildings or rocky outcrops, in which to roost during the day (Skinner & Chimimba 2005; Monadjem et al. 2010). Individuals collected at the Shashi-Limpopo confluence were located near a range of sandstone hills, associated with Acacia woodland, alongside dry river beds (Skinner & Chimimba 2005). Within southern Africa, the species is seldom found in large colonies, although in West Africa colonies can number hundreds of individuals (Skinner & Chimimba 2005). In southern Zimbabwe, colonies have also been found to number in the hundreds (Monadjem et al. 2020). It is an open-air forager with high wing-loading (Norberg & Rayner 1987; Monadjem et al. 2020). Although the diet of individuals within the assessment region has yet to be confirmed, individuals found elsewhere are known to feed predominantly on Lepidoptera, Blattodea, Coleoptera and Orthoptera species (Skinner and Chimimba 2005; Monadjem et al. 2020; Rydell and Yalden 1997).
Ecosystem and cultural services: As this species is an aerial insectivore, it likely plays an important role in controlling insect populations (Boyles et al. 2011; Kunz et al. 2011). Often, bats prey on the insect species that destroy crops including those belonging to the order Lepidoptera, Orthoptera and Coleoptera (Boyles et al. 2011; Kunz et al. 2011). Ensuring a healthy population of insectivorous bats can result in a decrease in the use of pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 5.1. Wetlands (inland) -> Wetlands (inland) – Permanent Rivers/Streams/Creeks (includes waterfalls) | – | Suitable | – |
Life History
Generation Length: Unknown
Age at Maturity: Female or unspecified: Unknown
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Female: Mean total length = 10.3 cm; Mean forearm length = 6.3 cm (Smithers and Wilson 1979)
Size at Maturity (in cms): Male: Mean total length = 10. 77 ±1.25 cm; Mean forearm length = 6.28 ±0.26 cm (Durban Natural Science Museum specimen records)
Longevity: Unknown
Average Reproductive Age: Unknown
Maximum Size (in cms): Females = 10.8 cm (Smithers and Wilson 1979); Males = 10.9 cm (Durban Natural Science Museum specimen records); Harrison (1961) reported maximum size for male specimens from Tanzania as 14.5 cm.
Size at Birth (in cms): Unknown
Gestation Time: Unknown
Reproductive Periodicity: Coincides with austral summer months with pregnant females encountered in November onwards (Smithers 1983).
Average Annual Fecundity or Litter Size: One to two pups per breeding season, based on the reported breeding biology of the Nigerian population (Harrison 1958).
Natural Mortality: Unknown
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: Yes, found in small colonies
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence to suggest that the species is traded or utilised in any form.
Local Livelihood: No evidence
National Commercial Value: N/A
International Commercial Value: N/A
End Use: N/A
Is there harvest from captive/cultivated sources of this species? No evidence
Harvest Trend Comments: N/A
Threats
Human disturbance has been highlighted as a threat to the Egyptian Tomb Bat, but overall, it is unlikely that this species is significantly threatened across its very wide range. In South Asia it is threatened by clearing of thorn forests for agricultural purposes, or for mining and stone quarrying. Roost disturbance due to human interference and development of old buildings for tourism purposes are also considered serious threats (C. Srinivasulu pers. comm.; Molur et al. 2002).
Threats to this species have yet to be quantified within the assessment region. However, climate change is recognised as a potential threat to most bat species (Sherwin et al. 2013). In addition, the species is categorised as “high risk” to renewable wind energy developments; this industry may pose a significant threat should it become established within their area of occurrence. Lastly, Border Cave overlooks an expansive sugar cane production area (interspersed by small reserves) in the Lubombo region of Eswatini. The over-application of agricultural pesticides may threaten the species if such areas are used for foraging.
Conservation
There are no direct interventions necessary for this species within the assessment region. It occurs within Mapungubwe National Park and possibly Venetia Limpopo Nature Reserve (both protected areas occurring in the northern region of the Limpopo Province).
Recommendations for land managers and practitioners:
- Reduce pesticide use in agricultural landscapes.
- Continued protection of the Border Cave archaeological site by the relevant authorities.
- Increased survey effort and the identification of roosts within the greater Mapungubwe region.
Research priorities:
- Further monitoring of the Mapungunbwe and Border Cave populations are needed to establish demographics, population trends, local movement patterns, foraging ecology and specific threats facing the regional population.
- Reference echolocation call data is needed to identify the species from acoustic monitoring data.
- Phylogeographic and landscape genetics studies of southern African individuals are needed to establish the extent of gene flow between populations.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution. The Egyptian Tomb Bat is almost identical in body size to that of the Mauritian Tomb Bat (T. mauritianus), but these species can be distinguished by their distinctive colourations. The Egyptian Tomb Bat has a darker back and lacks the greyish-brown (grizzled) colouration of the Mauritian Tomb Bat.
Bibliography
ACR. 2024. African Chiroptera Report 2023. Van Cakenberghe, V. and Seamark, E.C.J. (Eds.) African Chiroptera Project, Pretoria. i – xviii; 1 – 1210 pp.
Bates, P.J.J. and Harrison, D.L. 1997. Bats of the Indian Subcontinent. Harrison Zoological Museum, Sevenoaks, England, UK.
Boyles, J.G., Cryan, P.M., McCracken, G.F., and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Coldrey, K.M., Turpie, J.K., Midgley, G., Scheiter, S., Hannah, L., Roehrdanz, P.R., and Foden, W.B. 2022. Assessing protected area vulnerability to climate change in a case study of South African national parks. Conservation Biology 36(5):e13941. doi: 10.1111/cobi.13941
Driver, A., Sink, K.J., Nel, J.N., Holness, S., van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An Assessment of South Africa’s Biodiversity and Ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Harrison, D.L. 1958. A new race of tomb bat Taphozous perforatus E. Geoffroy, 1818, from northern Nigeria, with some observations on its breeding biology. Durban Museum Novitates 5(11):143-9.
Harrison, D.L. 1962. On bats collected on the Limpopo River, with the description of a new race of the Tomb Bat, Taphozous sudani Thomas 1915. Occasional papers of the Northern Museums of Southern Rhodesia. 26(B): 755-767.
Harrison, D.L. 1964. Notes of some Southern Rhodesian Microchiroptera. Arnoldia 1(3): 1-3.
Kock, D., Sami Amr, Z., Mickleburgh, S., Hutson, A.M., Bergmans, W.M. and Molur, S. 2008. Taphozous perforatus. The IUCN Red List of Threatened Species 2008:e.T21463A9282975.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of southern African mammals. Transvaal Museum Monographs 5: 1–359.
Molur, S., Marimuthu, G., Srinivasulu, C., Mistry, S. Hutson, A.M., Bates, P.J.J., Walker, S., Padmapriya, K. and Binupriya, A.R. 2002. Status of South Asian Chiroptera: Conservation Assessment and Management Plan (C.A.M.P.) Workshop Report. Zoo Outreach Organization/CBSG-South Asia, Coimbatore, India.
Monadjem, A., Simelane, F., Shapiro, J.T., Gumbi, B.C., Mamba, M.L., Sibiya, M.D., Lukhele, S.M. and Mahlaba, T.A. 2021. Using species distribution models to gauge the completeness of the bat checklist of Eswatini. European Journal of Wildlife Research 67(2):21.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. Second Edition. University of Witwatersrand Press, Johannesburg.
Ndlovu, M., Clulow, A.D., Savage, M.J., Nhamo, L., Magidi, J., Mabhaudhi, T. 2021. An Assessment of the Impacts of Climate Variability and Change in KwaZulu-Natal Province, South Africa. Atmosphere 12, 427. https://doi.org/10.3390/atmos12040427
Norberg, U.M. and Rayner, J.M. 1987. Ecological morphology and flight in bats (Mammalia; Chiroptera): wing adaptations, flight performance, foraging strategy and echolocation. Philosophical Transactions of the Royal Society B: Biological Sciences 316: 335–427.
Rydell, J. and Yalden, D.W. 1997. The diets of two high‐flying bats from Africa. Journal of Zoology 242(1):69-76.
Sherwin, H.A., Montgomery, W.I. and Lundy, M.G. 2013. The impact and implications of climate change for bats. Mammal Review 43: 171–182.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smithers, R.H.N. 1983. The mammals of the Southern African subregion University of Pretoria. Pretoria, South Africa.
Smithers, R.H., Wilson V.J. 1979. Checklist and atlas of the mammals of Zimbabwe Rhodesia. National Museums and Monuments of Rhodesia, Salisbury, Rhodesia, Museum Memoir 8:1-47.
White, W. 2011. Observation #12: Possible Taphozous perforatus sighting. African Bat Conservation News 25:2.
