
Egyptian Free-tailed Bat
Tadarida aegyptiaca

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Tadarida aegyptiaca – (É. Geoffroy, 1818)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – MOLOSSIDAE – Tadarida – aegyptiaca
Common Names: Egyptian Free-tailed Bat, Egyptian Guano Bat, Egyptian Nyctinome (English), Egiptiese Losstertvlermuis (Afrikaans)
Synonyms: Nyctinomus aegyptiaca (A. Geoffroy Saint-Hilaire, 1818), N. geoffroyi (Temminck, 1827), tragatus (Dobson, 1874), talpinus (Heuglin, 1877), anchietae (Seabra, 1900), bocagei (Seabra, 1900), brunneus (Seabra, 1900), tongaensis (Wettstein, 1916), gossei Wroughton, 1919, sindica Wroughton, 1919, thomasi Wroughton, 1919
Taxonomic Note: Two subspecies of Tadarida aegyptiaca have been recorded from the assessment region (Hayman & Hill 1971). These include T. a. aegyptiaca, which extends from North Africa (Algeria and Egypt) southwards through East Africa and into the Western Cape of South Africa, as well as the smaller and darker T. a. bocagei (Seabra 1900) from central and western Africa (Hayman & Hill 1971; Skinner and Chimimba 2005). Monadjem et al. (2020) recommend further studies to address the taxonomic status of the widespread African populations.
In some databases (e.g. https://www.mammaldiversity.org/taxon/1005261) this species is listed as Nyctinomus aegyptiacus, while in other databases (e.g. www.gbif.org/species/2433016) Nyctinomus is listed as a basionym. In the absence of taxonomic clarity about the status and distribution of the genus, species, and subspecies we have chosen to use Tadarida until the situation is better resolved.
Red List Status: LC – Least Concern (IUCN version 3.1)
Assessment Information
Assessors: Richards, L.R.1, Howard, A.2, Lötter, C.A.3 & Richardson, E.J.4
Reviewer: de Villiers, M.5
Institutions: 1Durban Natural Science Museum, 2University of the Free State, 3Inkululeko Wildlife Services (Pty) Ltd, 4Independent Consultant at Richardson & Peplow Environmental, 5CapeNature
Previous Assessors and Reviewers: MacEwan, K., Jacobs, D., Schoeman, C., Richards, L.R., Cohen, L., Monadjem, A., Sethusa, T. & Taylor, P.
Previous Contributors: Nicholson, S.K., Relton, C., & Raimondo, D.
Assessment Rationale
The species is very widely distributed (with an estimated extent of occurrence of 1,519,832 km2), locally common and recorded from many formally protected areas within the assessment region. Craniodental morphometric and genetic data indicate the taxon represents a species complex, with a diminutive central-western form and a larger eastern form, recorded from southern Africa (Reddy 2015; Reddy et al. 2019). These forms loosely correspond to T. a. aegyptiaca and T. a. bocagei. More detailed investigations, including comparison to type or topotypic specimen material, are required to ascertain whether the two forms represent valid species. It predominantly forages above canopy height and is therefore very susceptible to mortality from wind turbine blade collisions and possibly barotrauma (Doty & Martin 2013; Aronson 2022). Barotrauma is tissue damage caused by rapid excessive changes in air pressure, conditions which result from turbine blade movement (Baerwald et al. 2008; Cryan & Barclay 2009; Rydell et al. 2010). While previously not considered to face significant threats, there is a current confirmed, severe threat posed by wind farms. Reports from most operational wind farms indicate that of all turbine-related bat mortalities, this species is killed in the highest numbers, with males seemingly impacted more often than females (L. Richards, unpublished data). It is not certain, however, whether this species is declining at a rate fast enough to qualify for a threatened status. Certainly, this species needs close monitoring and substantially improved fatality mitigation at wind farms across the region.
Regional population effects: This species is present within South Africa’s neighbouring countries and is distributed along the country’s borders. Its high wing-loading (Schoeman & Jacobs 2008) means dispersal and thus recolonisation of extinct or declining populations are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Richards LR, Howard A, Lötter CA & Richardson EJ. 2025. A conservation assessment of Tadarida aegyptiaca. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The Egyptian Free-tailed Bat is found throughout Africa, and in the Arabian Peninsula through to India, Sri Lanka, Bangladesh and south Asia (Bates & Harrison 1997). It is widespread and may still be abundant throughout most of southern Africa, occurring from the Western Cape of South Africa north through to Namibia and southern Angola, and through Zimbabwe to central and northern Mozambique (Monadjem et al. 2020). It is widely distributed in the assessment region, occurring in all nine provinces of South Africa as well as in Lesotho and Eswatini (Skinner & Chimimba 2005; Monadjem et al. 2016, 2020). Its estimated extent of occurrence is 1,519,832 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 2100
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
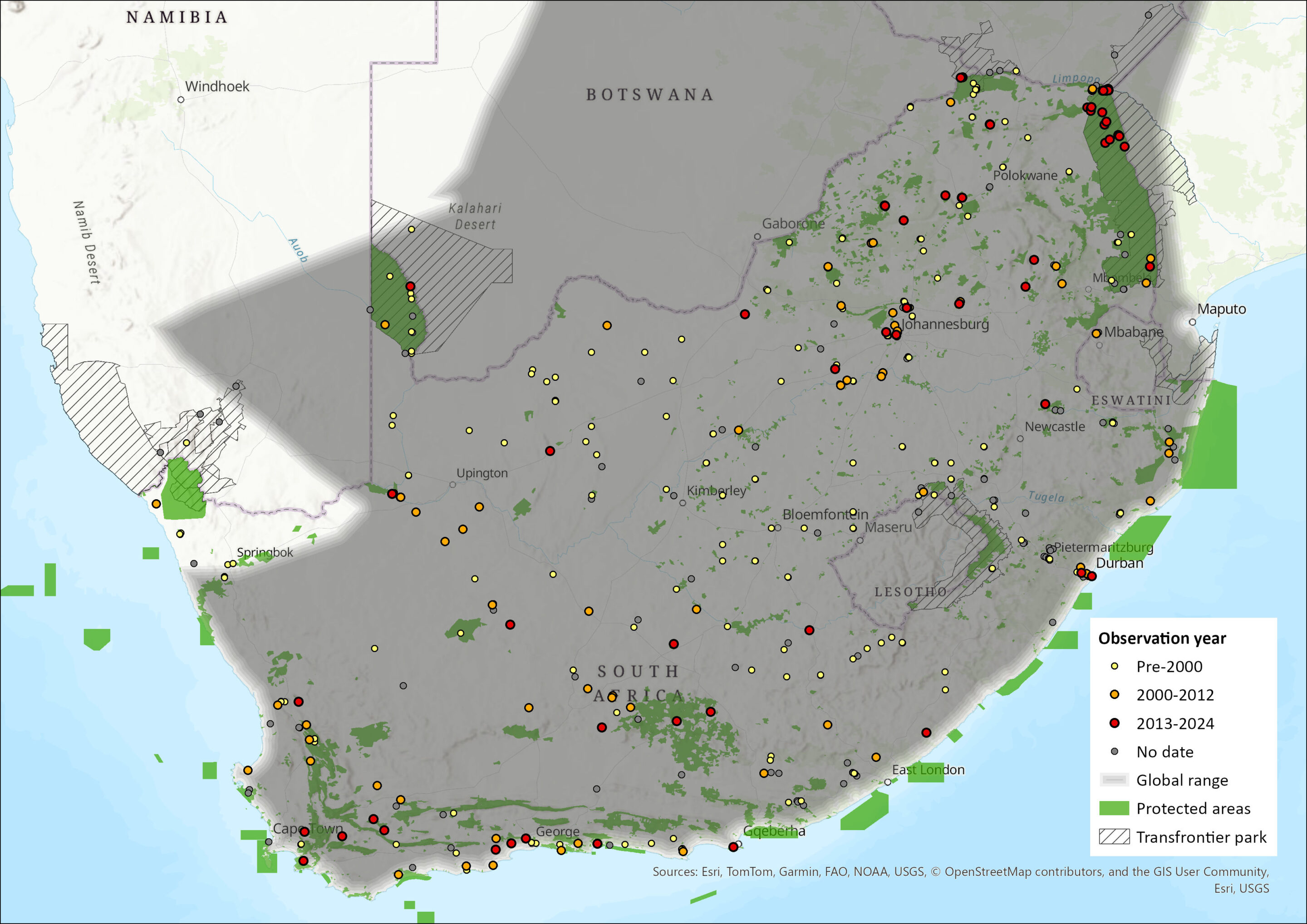 Figure 1. Distribution records for Egyptian Free-tailed Bat (Tadarida aegyptiaca) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Egyptian Free-tailed Bat (Tadarida aegyptiaca) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Indomalayan
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Afghanistan | Extant | Native | – | – |
| Algeria | Extant | Native | ||
| Angola | Extant | Native | – | – |
| Bangladesh | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Egypt | Extant | Native | ||
| Eswatini | Extant | Native | – | Resident |
| Ethiopia | Extant | Native | – | – |
| India | Extant | Native | – | – |
| Iran, Islamic Republic of | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Morocco | Extant | Native | ||
| Mozambique | Extant | Native | – | Resident |
| Namibia | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Oman | Extant | Native | – | – |
| Pakistan | Extant | Native | – | – |
| Saudi Arabia | Extant | Native | – | – |
| South Africa | Extant | Native | – | Resident |
| Sri Lanka | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| South Sudan | Extant | Native | ||
| Tanzania, United Republic of | Extant | Native | – | – |
| Tunisia | Extant | Native | ||
| Uganda | Extant | Native | – | – |
| Yemen | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
No formal study on the direct effects of climate change in this species has yet been conducted. However, due to the increasing temperatures across most of the species’ distribution (Mbokodo et al. 2020), the roosting preferences and foraging areas are predicted to shift. Most studies on the effects of climate change on bats are based on predictive species distribution modelling, thus there is a lack of empirical studies measuring behavioural, physiological, phenological or genetic responses to extreme and seasonal climatic changes, especially in the Global South (Festa et al. 2023; Pio et al. 2014). Globally, there have been documented declines in bat populations, species richness and distributions in relation to water availability with increasing global aridity which may become a growing concern as heat waves and maximum temperatures are expected to increase over much of South Africa (Adams and Hayes 2021; Mbokodo et al. 2020). Cory-Toussaint et al. (2010) found this species can enter prolonged periods of heterothermy for multiple days, suggesting hibernation capabilities.
Population Information
Although there are no accurate population estimates, this species is widespread and common within the assessment region, as well as within the rest of its range. In general, Egyptian free-tailed bats are capable of utilising synanthropic roosts (roof spaces) was well as natural roosts such as crevices in rocks. It is unknown whether the two identified forms vary in their roosting ecology. Individuals roost communally in small to medium-sized groups, which may number in the dozens to hundreds (Herselman & Norton 1985). Additionally, the species is well represented in museums, with over 450 specimens examined in Monadjem et al. (2020). In Renewable Energy Development Zones and other areas where there are clusters of operational wind farms, however, the cumulative impact of these may result in population declines especially in the absence of effective fatality mitigation for this species.
Current population trend: Stable
Continuing decline in mature individuals? Unknown
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: Unknown
All individuals in one subpopulation: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
No population genetic studies exist on the species. Molecular mitochondrial DNA and morphometric data indicate that T. aegyptica represents a species complex (Reddy 2015; Reddy et al. 2019). Further phylogenetic studies, in combination with phylogeographic investigations, are necessary to shed light on the taxonomic status of the southern African populations.
Habitats and ecology
The Egyptian Free-tailed Bat occurs across a range of habitats, foraging high above the vegetation in desert, semi-arid scrub, savannah and grassland habitats, and also in urbanised areas and agricultural land (Skinner & Chimimba 2005; Monadjem et al. 2020). It is known to be common in arid scrub and open grassland regions (Skinner & Chimimba 2005). In arid areas, its presence is commonly associated with surface water (Sirami et al. 2013), which provides a source of drinking water (Skinner & Chimimba 2005), and usually has concentrated densities of insect prey (Monadjem et al. 2020). As such, it is especially important to ensure that wind turbines in arid areas are developed as far as possible from natural and man-made water supplies.
This species roosts communally during the day in small to medium-sized groups (Herselman & Norton 1985; Monadjem et al. 2020). Roost habitats include, but are not limited to, rock crevices, under exfoliating rock sheets, tree hollows, caves, behind the bark of dead trees, building crevices and roofs of houses (Herselman & Norton 1985; Taylor 1998; Skinner & Chimimba 2005; Monadjem et al. 2020). It is considered an open-air forager, foraging from ground level to well above the canopy of the vegetation in the varied habitat types and, therefore, has a “High” risk of fatality from wind turbines (MacEwan et al. 2020). It feeds mainly on Diptera, Hemiptera and Coleoptera and, to a lesser degree, Lepidoptera (Monadjem et al. 2020). Definite seasonal patterns in activity levels of this species are emerging from long-term monitoring studies for proposed and operational wind farms in the assessment region, with a typical peak at most wind farm sites in late summer and autumn (February, March, and April; Aronson 2022). Gestation is approximately four months and typically, a single young is born once a year in November or December (Bernard & Tsita 1995; Monadjem et al. 2020).
Ecosystem and cultural services: As this species is one of the most widespread and common insectivorous bat species in the assessment region, it may play an especially important role in controlling insect populations including agricultural pest species (Boyles et al. 2011; Kunz et al. 2011). Bats often prey on the insect species that destroy crops, such as macadamia orchards in northern Limpopo (Boyles et al. 2011; Kunz et al. 2011; Taylor et al. 2018). Ensuring a healthy population of insectivorous bats can thus result in a decrease in the use of pesticides which may be detrimental to human and ecosystem health (Frank 2024).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | -Yes |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | -Yes |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: Mean forearm length = 4.71 ± 0.20 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Morearm length = 4.59 ± 0.17 cm (Monadjem et al. 2020)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: Four months/16 weeks (Monadjem et al. 2020)
Reproductive Periodicity: Once a year (Monadjem et al. 2020)
Average Annual Fecundity or Litter Size: 1 offspring per mature female per year (Monadjem et al. 2020)
Natural Mortality: (Not specified)
Breeding Strategy
| Does the species lay eggs? |
| No |
| Does the species give birth to live young |
| Yes |
| Does the species exhibit parthenogenesis |
| No |
| Does the species have a free-living larval stage? |
| No |
| Does the species require water for breeding? |
| No |
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: Roosts communally in small to medium-sized groups, which may number in the dozens to hundreds (Herselman and Norton 1985).
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence to suggest that this species is traded or harvested within the assessment region. However recent study by Tarango et al. (2025) listed this species as imported into the U.S.A so online e-commerce platforms should be monitored for illegal trade in bat taxidermy and specimens from the assessment region.
The species has an economic value as an agricultural pest control. It controls insect populations including agricultural pest species (Boyles et al. 2011; Kunz et al. 2011). Bats often prey on the insect species that destroy crops, such as macadamia orchards in northern Limpopo (Boyles et al. 2011; Kunz et al. 2011; Taylor et al. 2018).
National Commercial Value: Yes
International Commercial Value: No
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Of all bat fatalities reported by Aronson (2022) from data that were collated from 25 operational wind farms in South Africa for the period 2011-2020, T. aegyptiaca was the most commonly recorded. The potential cumulative impact of multiple wind farms in certain Renewable Energy Development Zones (such as the Cookhouse and Komsberg REDZ) is especially concerning, given that multiple wind farms in these areas have exceeded their bat fatality threshold for several years (SABAA unpubl. data).
The situation needs to be monitored very carefully, and mitigation measures applied to avoid unsustainable losses, as the reproductive capacity of this species is limited to one pup per adult female per year (Monadjem et al. 2020).
As this species roosts in caves, it may also be somewhat vulnerable to roost disturbance. Many traditional ceremonies and tourism activities take place in caves, but the extent of disturbance and extermination is currently unknown. Cory-Toussaint et al. (2022) reported significantly higher levels of zinc and mercury in the blood and fur of T.aegyptiaca from opencast diamond mines than reference areas in northern Limpopo.
Roof-roosting individuals along the KwaZulu-Natal coastal region may be subject to persecution, displacement, or incidental poisoning from building fumigation activities (Bats KZN, L. Richards, pers. obs.)
The potential impact of agricultural pesticides on this species is not known and requires further investigation.
Conservation
This species is found in many protected areas in the assessment region, including large reserves such as Vhembe Biosphere Reserve, Mapungubwe National Park (MNP), Venetia Limpopo Nature Reserve (VNR), Kruger National Park, iSimangaliso Wetland Park, Ithala Game Reserve, Maloti-Drakensberg Transfrontier Conservation and Development Area, Addo Elephant Park, Madikwe Game Reserve, Pilanesberg National Park, Augrabies Falls National Park, and the Kgalagadi Transfrontier Park.
To mitigate mortalities of this species from turbine collisions on wind farms, development of wind farms must avoid encroachment into the prescribed buffers around water supplies and confirmed and potential roosts of this species (for buffer recommendations see MacEwan et al. 2020 or later). Turbine-fatalities of this species must be reduced below fatality threshold values (as calculated according to MacEwan et al. 2018 or later) with interventions such as: i) ultrasound to deter bats (Weaver et al. 2020); ii) blanket curtailment of turbines at low wind speeds during seasons and hours of the night when this species is most active (Berthinussen et al. 2010; Arnett et al. 2011; Hayes et al. 2019); iii) acoustic smart curtailment of turbines in response to real-time bat activity (https://www.wildlifeacoustics.com/smart-system); and/or optimised smart curtailment of turbines during predicted periods of high bat activity based on statistical modelling (https://west-inc.com/wp-content/uploads/2024/04/Optimized-Smart-Curtailment-SOQ_FINAL.pdf). As high bat fatalities at wind farms cannot be easily offset (Aronson et al. 2018; Mark Botha pers. comm.), avoidance and minimisation of bat fatalities is critical.
Recommendations for land managers and practitioners:
- Development of wind farms must avoid encroachment into the prescribed buffers around waterbodies and confirmed and potential roosts of this species.
- Turbine-fatalities of this species must be reduced with interventions such as ultrasound to deter bats and curtailing turbines during low wind speeds.
- Data sharing by wind farm managers into a national database is essential in order to calculate cumulative impacts and thereafter implement collaborative mitigation and management efforts, and to inform decisions regarding the authorization of new wind farms in key areas.
- Better post-construction compliance monitoring at WEFs.
Research priorities:
- An integrative taxonomic approach, utilising multiple data sets in combination with comparison of topotypic material, to resolve the taxonomic status of the two identified southern African forms. This will inform whether the above Red Data List status requires amendment or not.
- A meta-analysis of all recorded T. aegyptiaca fatalities at wind farms in South Africa to date, to assess the cumulative impact of this industry on this species, and to identify any spatial, temporal, or turbine-specific trends, which may assist in devising effective fatality mitigation for future implementation.
- Research to assess the efficacy of different mitigation methods to reduce bat mortality at wind farms.
- Wind farm carcasses of this species should be used to obtain greater insight into the biogeography, genetics, reproductive biology, ecology (e.g. diet), and ecosystem services of this species throughout southern Africa.
- Surveys should be performed to investigate the activity of this species at sea (inshore and offshore), ahead of emerging plans to develop offshore wind farms around southern Africa.
- Research in identifying possible migratory routes of this species.
- Investigation on the sub population sizes and trends, and population size estimate.
- Physiological effects of agrochemicals and pesticides.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
- If bats are excluded from buildings, use bat-friendly methods (timing of removals relative to breeding season, use of “one-way valves” to prevent trapping, no fumigation, etc).
Bibliography
Adams, R.A. and Hayes, M.A. 2021. The importance of water availability to bats: climate warming and increasing global aridity. 50 years of bat research: foundations and new frontiers, pp.105-120.
Arnett, E.B., Huso, M.M., Schirmacher, M.R. and Hayes, J.P. 2011. Altering turbine speed reduces bat mortality at wind-energy facilities. Frontiers in Ecology and the Environment 9: 209–214.
Aronson, J. 2022. Current state of knowledge of wind energy impacts on bats in South Africa. Acta Chiropterologica 24: 221-238.
Aronson, J., Sowler, S. and MacEwan, K. 2018. Mitigation guidance for bats at wind energy facilities in South Africa. Edition 2. South African Bat Assessment Association, South Africa.
Baerwald, E.F., D’Amours, G.H., Klug, B.J. and Barclay, R.M. 2008. Barotrauma is a significant cause of bat fatalities at wind turbines. Current Biology 18: 695–696.
Bendjeddou, M.L., Loumassine, H.E., Metallaoui, W., Chiheb, K., Farfar, A., Bounaceur, F., Boukheroufa, F., Bouslama, Z. and Dietz, C. 2016. First record of Nyctinomus aegyptiacus for Tunisia. Vespertilio 18:23-27.
Bernard, R.T.F. and Tsita, J.N. 1995. Seasonally monoestrous reproduction in the molossid bat, Tadarida aegyptiaca from low temperate latitudes (33 S) in South Africa. South African Journal of Zoology, 30(1), pp.18-22.
Bates, P.J.J. and Harrison, D.L. 1997. Bats of the Indian Subcontinent. Harrison Zoological Museum, Sevenoaks, England, UK.
Berthinussen, A., Richardson, O.C. and Altringham, J.D. 2010. Bat Conservation: Global Evidence for the Effects of Interventions. Synopses of Conservation Evidence. Pelagic Publishing, Exeter.Boyles JG, Cryan PM, McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Cory-Toussaint, D., McKechnie, A.E. and Van der Merwe, M. 2010. Heterothermy in free-ranging male Egyptian free-tailed bats (Tadarida aegyptiaca) in a subtropical climate. Mammal Biology, 75 pp. 466-470.
Cory-Toussaint, D. and Taylor, P.J. 2022. Anthropogenic light, noise, and vegetation cover differentially impact different foraging guilds of bat on an opencast mine in South Africa. Frontiers in Ecology and Evolution, 10, p.752665.
Cory-Toussaint, D., Taylor, P.J. and Barnhoorn, I.E. 2022. Non-invasive sampling of bats reflects their potential as ecological indicators of elemental exposure in a diamond mining area, northern Limpopo Province, South Africa. Environmental Science and Pollution Research, pp.1-14.
Cryan, P.M. and Barclay, R.M. 2009. Causes of bat fatalities at wind turbines: hypotheses and predictions. Journal of Mammalogy 90: 1330–1340.
Doty, A.C. and Martin, A.P. 2013. Assessment of bat and avian mortality at a pilot wind turbine at Coega, Port Elizabeth, Eastern Cape, South Africa. New Zealand Journal of Zoology 40: 75–80.
Festa, F., Ancillotto, L., Santini, L., Pacifici, M., Rocha, R., Toshkova, N., Amorim, F., Benítez‐López, A., Domer, A., Hamidović, D. and Kramer‐Schadt, S. 2023. Bat responses to climate change: a systematic review. Biological Reviews, 98(1), pp.19-33.
Frank, E.G. 2024. The economic impacts of ecosystem disruptions: Costs from substituting biological pest control. Science 385,0344.
Hayman, R.W. and Hill, J.E. 1971. Order Chiroptera. Pages 1–73 in Meester J, Setzer HW, editors. The Mammals of Africa: An Identification Manual. Part 2. Smithsonian Institution Press, Washington, DC, USA.
Hayes, M.A., Hooton, L.A., Gilland, K.L., Grandgent, C., Smith, R.L., Lindsay, S.R., Collins, J.D., Schumacher, S.M., Rabie, P.A., Gruver, J.C., and Goodrich-Mahoney, J. 2019. A smart curtailment approach for reducing bat fatalities and curtailment time at wind energy facilities. Ecological Applications, 0(0), 2019, e01881.
Herselman, J. C. and Norton, P. M. 1985. The distribution and status of bats (Mammalia: Chiroptera) in the Cape Province. Annals of the Cape Provincial Museums 16: 74-126.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
MacEwan, K., Aronson, J., Richardson, K., Taylor, P., Coverdale, B., Jacobs, D., Leeuwner, L., Marais, W. and Richards, L. 2018. South African Bat Fatality Threshold Guidelines. Edition 2. South African Bat Assessment Association, South Africa.
MacEwan, K., Sowler, S., Aronson, J. and Lötter, C. 2020. South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities. Edition 5. South African Bat Assessment Association, South Africa.
Mbokodo, I., Bopape, M.J., Chikoore, H., Engelbrecht, F. and Nethengwe, N. 2020. Heatwaves in the future warmer climate of South Africa. Atmosphere, 11(7), p.712.
Monadjem, A., Jacobs, D., Cotterill, W., Hutson, A.M., Mickleburgh, S., Bergmans, W. and Fahr, J. 2016. Tadarida aegyptiaca. The IUCN Red List of Threatened Species 2017: e.T21312A22115459.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis.2nd Edition. University of Witwatersrand Press, Johannesburg.
Pio, D.V., Engler, R., Linder, H.P., Monadjem, A., Cotterill, F.P., Taylor, P.J., Schoeman, M.C., Price, B.W., Villet, M.H., Eick, G. and Salamin, N. 2014. Climate change effects on animal and plant phylogenetic diversity in southern Africa. Global Change Biology, 20(5), pp.1538-1549.
Reddy, S. 2015. Phylogenetic position of, and morphometric variation in Tadarida aegyptiaca (Chiroptera: Molossidae) from southern Africa. Unpublished Master’s thesis. University of KwaZulu-Natal.
Reddy, S., L. R. Richards, J. M. Lamb and A. H. H. Macdonald. 2019. Phylogenetic and morphometric variation in Tadarida aegyptiaca (Chiroptera: Molossidae) from southern Africa. 18th International Bat Research Conference – 28 July – 1 August 2019 – The Slate, Phuket, Thailand: 273.
Rydell, J., Bach, L., Dubourg-Savage, M-J., Green, M., Rodrigues, L. and Hedenström, A. 2010. Mortality of bats at wind turbines links to nocturnal insect migration? European Journal of Wildlife Research 56: 823–827.
Schoeman, M.C. and Jacobs, D.S. 2008. The relative influence of competition and prey defenses on the phenotypic structure of insectivorous bat ensembles in southern Africa. PLoS One 3: e3715.
Sherwin, H.A., Montgomery, W.I. and Lundy, M.G. 2013. The impact and implications of climate change for bats. Mammal Review 43: 171–182.
Sirami, C., Jacobs, D.S. and Cumming G.S. 2013. Artificial wetlands and surrounding habitats provide important foraging habitat for bats in agricultural landscapes in the Western Cape, South Africa. Biological Conservation 164: 30–38.
Skinner, J.D. and Chimimba, C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Taylor, P.J. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.
Tarango, M., Kolby, J.E., Goodman, O.L., Anderson, C.J., Tinsman, J., Kirkey, J., Liew, A., Jones, M., Firth, C. and Reaser, J.K. 2025. Going batty: US bat imports raise concerns for species conservation and human health. One Health, 20, p.100999.
Taylor, P.J., Grass, I., Alberts, A.J., Joubert, E. and Tscharntke, T. 2018. Economic value of bat predation services–A review and new estimates from macadamia orchards. Ecosystem Services, 30, pp.372-381.
Weaver, S.P., Hein, C.D., Simpson, T.R., Evans, J.W., and Castro-Arellano, I. 2020. Ultrasonic acoustic deterrents significantly reduce bat fatalities at wind turbines. Global Ecology and Conservation 24: e01099
