Common Duiker
Sylvicapra grimmia

2025 Red list status
Least Concern
Regional Population Trend
Stable
change compared
to 2016
No Change
Overview
Sylvicapra grimmia – (Linnaeus, 1758)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Sylvicapra – grimmia
Common Names: Common Duiker, Bush Duiker, Grey Duiker, Grimm’s Duiker (English), Gewone Duiker, Duiker (Afrikaans), Ipunzi, Ejayelekileko (Ndebele), Impunzi (Ndebele, Xhosa, Zulu), Phuthi (Sepedi, Sesotho), Phuti, Ntsha (Sepedi), Photi, Phôti, Phothi (Setswana), Imphunzi (Swati), Mhunti (Tsonga), Ndsa, Ntsa (Venda), Céphalophe de Grimm (French), Céphalophe du Cap (French), Kronenducker (German)
Synonyms: Capra grimmia Linnaeus, 1758
Taxonomic Note:
As many as fourteen subspecies of Sylvicapra grimmia have been recognised in Africa (Grubb & Groves 2001; Wilson 2013), however it is difficult to identify the precise geographic limits of each subspecies as the Common Duiker has a continuous distribution across sub-Saharan Africa (Wilson 2013). Some of these subspecies can be distinguished based on colouration and size characteristics (Wilson 2013). Within the assessment region, S. g. grimmia occurs in the extreme south of the species’ range (Western, Eastern and Northern Cape provinces; Skinner & Chimimba 2005), and is substantially greyer in colour compared to the other subspecies, hence the alternate name, Grey Duiker in the Cape (Wilson 2013). Sylvicapra g. caffa is found further north of S. g. grimmia within the KwaZulu-Natal, North West, Gauteng, Mpumalanga and Limpopo provinces (Skinner & Chimimba 2005), extending into southern Mozambique and eastern Zimbabwe (Wilson 2013). The sandy-coloured S. g. steinhardti ranges throughout Namibia into Angola, Botswana and marginally into the Northern Cape Province to Port Nolloth (Wilson 2013). While distribution is continuous, there are many cases of intergradation but geographical boundaries between forms have not been delineated accurately (IUCN SSC Antelope Specialist Group 2016). Hence only the species is assessed here.
| Red List Status |
| LC – Least Concern, (IUCN version 3.1) |
Assessment Information
Assessors: Smith, C.1 & da Silva, J.2
Reviewer: Venter, J.3
Institutions: 1Endangered Wildlife Trust, 2South African National Biodiversity Institute, 3Nelson Mandela University, South Africa
Previous Assessors: Birss, C. & Selier, J.
Previous Reviewer: Child, M.F.
Previous Contributor: Relton, C.
Assessment Rationale
Listed as Least Concern as the species is widespread and abundant within the assessment region, with a global total population size in the millions. Although bushmeat hunting may cause localised declines, the Common Duiker is resilient and should continue to exist in large numbers over its range. This species is also able to exist in agricultural landscapes, providing that the natural peripheral vegetation cover remains intact. It is a key prey species and subpopulations should be sustained by improving habitat condition and installing permeable fences on land outside protected areas as part of holistic management strategies to reduce potential livestock/game damage from predators.
Regional population effects: There are numerous routes for dispersal into the assessment region through transfrontier areas, such as the Great Limpopo Transfrontier Park, Kgalagadi Transfrontier Park and the Greater Mapungubwe Transfrontier Conservation Area.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Smith C & da Silva JM. 2025. A conservation assessment of Sylvicapra grimmia. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The Common Duiker is one of the most widely distributed antelopes throughout sub-Saharan Africa (IUCN SSC Antelope Specialist Group 2016), with an almost continuous distribution across the continent (Apio et al. 2015). They occur within savannah woodland and scrub habitats. Although they may utilise the shelter of forest fringes when disturbed, they are generally absent from forests (Skinner & Chimimba 2005). They do not occur within desert regions unless they are following vegetated watercourses, such as parts of the Namib Desert (Skinner & Chimimba 2005). Similarly, they avoid open grasslands where tree cover is limited, aside from the very long grassland habitats of Nyika Plateau in Malawi (Skinner & Chimimba 2005). Despite the large-scale anthropogenic habitat conversion, much of their historical geographic range has remained stable as they can persist despite dense human populations (IUCN SSC Antelope Specialist Group 2016). This species is considerably adaptable to land transformation, as it is known to persist in peri-urban and urban areas, and on the fringes of agricultural areas, where natural vegetation remains predominantly undisturbed.
In southern Africa, Common Duiker occur extensively through Namibia, Botswana (although they are uncommon in the region of the Okavango Delta), Zimbabwe and Mozambique (south of the Zambezi River). Within the assessment region, it is widespread through all provinces and through all biomes in South Africa, is widespread in Eswatini, but is considered rare in Lesotho (Lynch 1994), which may be a result of its lack of sufficient vegetation cover and browse resources (Skinner & Chimimba 2005).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realms
Biogeographic Realm: Afrotropical
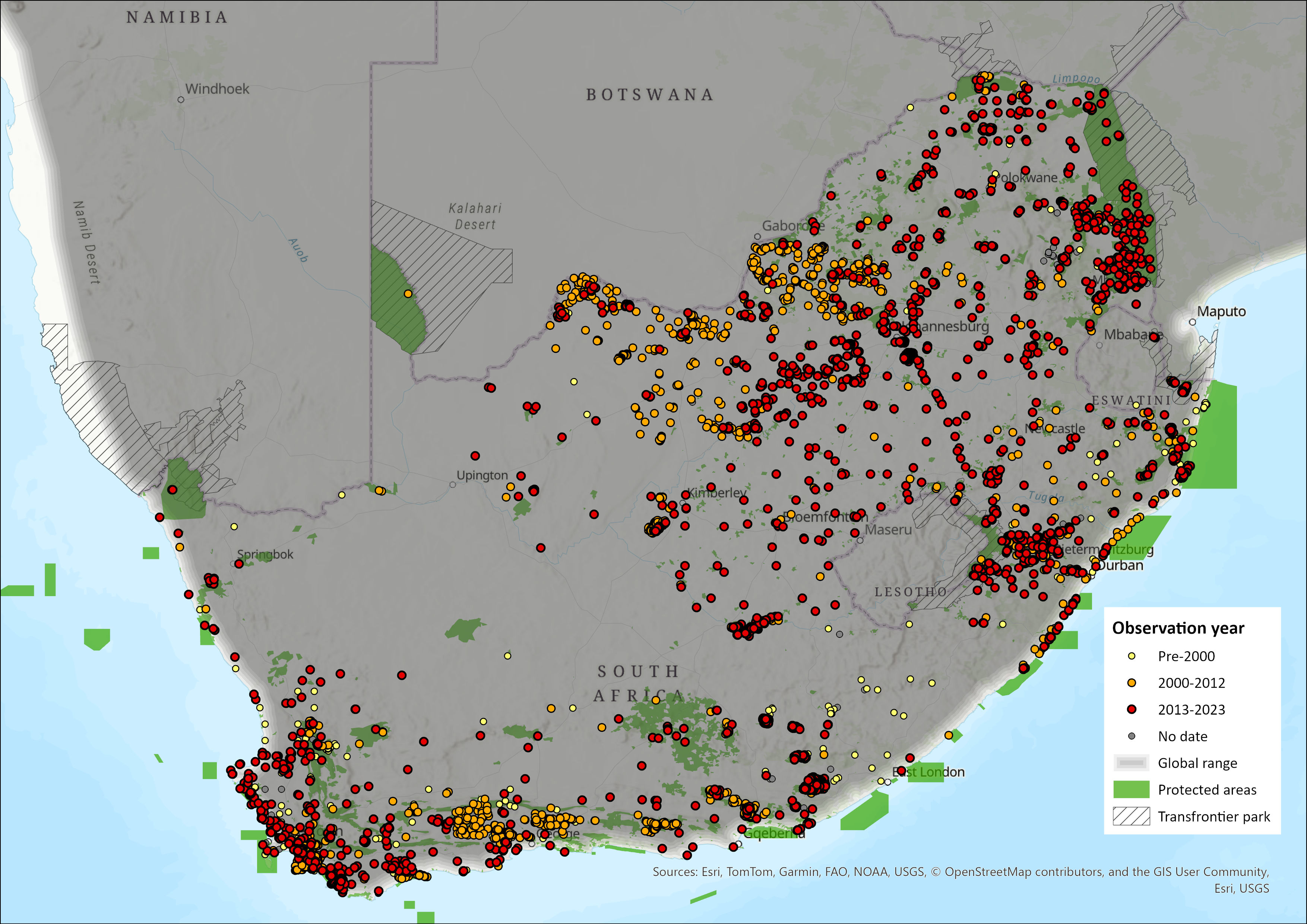
Map
Click on map to view/enlarge
Figure 1. Distribution records for Common Duiker (Sylvicapra grimmia) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Djibouti | Possibly Extinct | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Presence Uncertain | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
No recent research or literature has focused on how climate change might affect this species in the future.
Population information
Aerial surveys produced population density estimates of 0.01–0.15 individuals / km², however, due to the secretive nature of this species, this is likely to be an underestimate (East 1999). Within favourable habitats where this species is common, ground surveys revealed population density estimates of 0.3–1.7 individuals / km² (IUCN SSC Antelope Specialist Group 2016). Wilson (2001) summarises some recorded densities of Common Duiker from various localities in Africa in different vegetation types using line transects. Within the assessment region, in the Greater Addo Elephant National Park, Common Duiker occur across a substantial range of habitats, and densities ranged from 0.44 individuals / km² in the less favourable Dunefield habitat class, to 17 individuals / km² in the more suitable Riparian Woodland habitat class (Boshoff et al. 2002). A global population estimate of 1,660,000 was recommended by East (1999), however, more recently Wilson (2013) suggested that this may in fact be an underestimate, and the overall population may be more in the range of 10 million individuals. Generally, the population trend of the Common Duiker is considered to be stable, although some localised declines, as a result of hunting pressure, have been identified in Gabon and Niger (IUCN SSC Antelope Specialist Group 2016).
Within the assessment region, while no comprehensive count data are available, we infer from their wide distribution and high densities that there are well over 10,000 mature individuals with a stable or increasing population trend, albeit with localised declines in some areas from severe hunting pressure. The Common Duiker is one of the few African antelope species that has demonstrated adaptation to urbanisation and human settlements and exhibits continuous distribution within semi-urbanised environments. Its wide range of tolerance for different habitats enables its continuous distribution across the bioregional variation within South Africa, although it has been proposed that this species exhibits clinal variation across bioregions which would need to be supported by genetic research.
Population Information
Current population trend: Stable. Although the population may be increasing in some areas, in other regions increased bushmeat demand and hunting is causing local declines.
Continuing decline in mature individuals: Yes, from severe hunting in some areas.
Number of mature individuals in population: >10,000 individuals
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Phylogenetics studies examining the relationships within Cephalophinae have been undertaken (van Vuuren & Robinson 2001; Johnston & Anthony 2012) and report the genus Philantomba to be separate from Sylvicapra and Cephalophus). No fine-scale examination of the population genetic structure and diversity for this species has taken place. However, based on its current distribution, and presence inside and outside of protected areas, it is expected to disperse between sites, and exist as a large metapopulation throughout the assessment region including neighboring countries. While no census data exists for the species in the region, it is predicted that there are well over 10,000 mature individuals, with the population generally being stable or increasing. As such, it is expected that the effective population size for the species in the assessment region exceeds the 500 threshold.
Habitats and ecology
This species occurs extensively across a variety of habitats, with the exception of deserts and rainforests. The availability of woody vegetation or tall grass cover is an integral habitat requirement for this species for shelter, protection, shade and food resources, thus the Common Duiker is most characteristically a savannah woodland species. They occasionally occur in open tall grasslands, extending into mountainous habitats, such as Mt. Kilimanjaro and Mt. Kenya (Wilson 2013), as well as the Fynbos Biome in the southwestern extent of their range; but are generally absent from short grasslands (Skinner & Chimimba 2005). The Common Duiker adapts successfully to habitat conversion and fragmentation resulting from agricultural expansion, remaining along the fringes of cultivated areas (Photo 1), occasionally extending into agricultural lands once crops have reached sufficient heights to provide cover (Skinner & Chimimba 2005; Wilson 2013). They also survive in areas where there is low secondary growth (IUCN SSC Antelope Specialist Group 2016).
A study conducted in the Soutpansberg Mountains showed that the Common Duiker preferred feeding in areas with tall grass and scattered fern (sufficient cover and escape routes), followed by wooded islands and thick fern (lack of sightlines/escape routes and presence of predator ambush sites), whereas little foraging occurred at the edges and rocky areas (hard substrate that impede escape potential) (Baker & Brown 2013). Within the Grants Valley, Eastern Cape chicory provided more than one third of the Common Duiker’s winter diet and a substantial proportion (14.4%) of the spring diet (Kigozi 2003). Its wide range of tolerance for different habitats enables its continuous distribution across the bioregional variation within South Africa, although it has been proposed that this species exhibits clinal variation across bioregions, which would need to be supported by genetic research. Habitat connectivity across different vegetation types is essential to maintain gene-flow and clinal variation within this species. Its distribution is continuous and widespread throughout South Africa with lower densities in areas without suitable cover.
The Common Duiker is a selective feeder or concentrate selector with a varied diet, but is predominantly a browser, which does not concentrate its feeding on one of a few species (Prins et al. 2006). This species is known to consume a variety of trees, shrubs, flowers, foliage, fruit, seeds, bark, roots, herbs, and occasionally, cultivated crops (Wilson 2013, Jansen van Vuuren et al. 2021). In the Overberg, the dominant species in the Common Duikers diet was Hermannia, and Hordeum vulgare as the second most abundant (Jansen van Vuuren et al. 2021). Gagnon and Chew (2000) reported the percentage of monocots in the diet of the Common Duiker as 12%, whereas a study conducted in southern Mozambique found 14-30% inclusion of monocots in the Common Duiker diet (Prins et al. 2006).
Generally solitary, the Common Duiker is only found in female-young pairs, or male-female pairs while the female is in oestrus (Skinner & Chimimba 2005). This species is an aseasonal breeder, and young may be born at any time of the year following a gestation period of 191 days (Bowland 1997). Young matures rapidly, and females are able to conceive at just 8–9 months old (Bowland 1997; Skinner & Chimimba 2005). Usually one lamb is born, weighing 1.5 kg (Bowland 1997). Due to their small size, the common duiker is vulnerable to all predators like the Caracal (Caracal caracal), Black-backed Jackal (Lupulella mesomelas), and their young are vulnerable to eagles (Jansen van Vuuren et al. 2021). They require dense vegetation cover from predators.
Ecosystem and cultural services: This species forms a valuable prey component of the diet of a number of predators, such as Leopard (Panthera pardus) (Hayward et al. 2006), Lion (Panthera leo) and Spotted Hyaena (Crocuta crocuta) (Hayward 2006).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Marginal | – |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Marginal | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.4. Shrubland -> Shrubland – Temperate | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 14.1. Artificial/Terrestrial -> Artificial/Terrestrial – Arable Land | – | Suitable | – |
| 14.2. Artificial/Terrestrial -> Artificial/Terrestrial – Pastureland | – | Suitable | – |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Suitable | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Suitable | – |
| 14.6. Artificial/Terrestrial -> Artificial/Terrestrial – Subtropical/Tropical Heavily Degraded Former Forest | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is used for traditional and trophy hunting. The trade is properly controlled and is considered to be sustainable. It is also hunted as bushmeat, particularly in rural areas, the effects of which may cause local declines. Due to the abundance and low commercial value, interest in captive breeding and keeping of this species is insignificant.
Within savannah regions, the wildlife and ranching industry has probably had a positive influence on this species, as areas of suitable habitat have increased. However, in arid and fynbos regions the conversion from livestock to wildlife ranching is likely to have resulted in increased competition for food resources, and resultantly local abundance declines of Common Duiker, due to the stocking of additional species that did not naturally occur within those bioregions. Additionally, game farms with high quality impermeable fences may pose a threat to gene flow.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Bushmeat and traditional hunting | – | Trophy hunting |
National Commercial Value: Yes
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | – | – |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Bushmeat and traditional hunting, also trophy hunting
Threats
No major threats have been identified for the Common Duiker, however, this species is vulnerable to localised intensive hunting, which may result in local subpopulation declines or extinctions. This species is generally resilient and highly adaptable to habitat conversion and fragmentation, and is often able to persist within human-modified habitats (IUCN SSC Antelope Specialist Group 2016), such as on the fringes of agricultural areas and in close proximity to settlements, provided that suitable vegetation cover is available. However, the erection of impermeable fences is likely to inhibit gene flow. The Common Duiker is fairly resilient to disturbance from human settlements, even where feral dogs pose a risk, demonstrating well developed predator avoidance behaviour. However, young may be especially vulnerable to mortality, due to the increasing presence of stray dogs outside of protected areas. Similarly, increased hunting with dogs in rural areas is responsible for local declines (sensu Grey-Ross et al. 2010).
Current habitat trend: Stable. Human development and agriculture throughout the country have reduced habitat for native wildlife species. However, the Common Duiker shows wide habitat tolerance, and is able to withstand human induced transformation in most areas of its range. Extensive wildlife ranching in savannah habitats, too, may be generally improving veld condition for this species or conserving land that would otherwise be overgrazed by livestock. However, in other bioregions, such as arid and fynbos habitats, where larger herbivores were historically less diverse and numerous, and ecological niches are rather narrow, this species is less tolerant of increased competition by other locally introduced herbivores. Wildlife ranching is often reliant on erecting high quality impermeable fences which causes habitat fragmentation and impedes gene flow particularly in the smaller antelope and other similar sized animals.
Conservation
Although this species is present within numerous protected areas, including Kruger National Park and Kgalagadi Transfrontier Park, as well as on private lands, its survival is not currently dependent on protected areas as they are abundant in landscapes outside protected areas. However, the maintenance of gene flow and clinal variation will depend on the establishment and maintenance of landscape and biodiversity corridors, such as by installing permeable fences. Population persistence and abundance should be monitored on private lands, and localised threats associated with feral dogs and bushmeat hunting should be controlled as much as possible. The conservation of natural habitat, especially Renostveld is important as natural vegetation forms a significant part of the Common Duiker’s diet (Jansen van Vuuren et al. 2021).
Recommendations for land managers and practitioners:
- Monitor persistence through the collection of sightings records.
- Monitor density where species is hunted.
- Determine level of clinal variation at landscape scale through genetic research: collect and bank genetic samples to support genetic analysis.
- Promote this species as a natural forage species for indigenous predators as part of the “holistic approach” to damage-causing animal management. Apply genetic conservation principles in the management of ecotypic species. Where feasible and practical, ensure that fences are permeable particularly on conservation/stewardship sites.
Research priorities: The collection and storage of distribution information to monitor persistence of Common Duiker in the landscape is being conducted by CapeNature in the Western Cape. This information is used in bioregional planning and to inform gaps in data for the Western Cape. Research priorities include:
- Quantifying the effects of wildlife ranching on this species across bioregions.
- Quantifying the effects of habitat fragmentation on gene flow and clinal variation within this species.
- Quantifying the level of bushmeat hunting and illegal hunting with dogs.
Encouraged citizen actions:
- Report sightings and roadkills on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Landowners should ensure that disturbance of this species and its young is kept to a minimum, particularly with regards to domestic dogs.
- Report feral dogs and illegal hunting to the local municipality or conservation agency for follow-up actions.
- Landowners should monitor persistence and population densities.
- Promote fence permeability.
- Submit hunting returns (this enables higher confidences in calculating impacts of hunting and evaluating bag limit size).
- Create conservancies and maintain green corridors in urban landscapes.
- Understand and support the concept of genetic conservation with particular regard to ecotypic species and their management.
Bibliography
Apio, A., Umuntunundi, P., Lerp, H., Bierbach, D., Plath, M. and Wronski, T., 2015. Persistence of two small antelope species in the degraded Mutara Rangelands (Akagera Ecosystem) based on pastoralists’ and farmers’ perceptions. Human Ecology, 43, pp.613-620.
Baker MAA, Brown JS. 2013. Foraging and habitat use of Common Duikers, Sylvicapra grimmia, in a heterogenous environment within the Soutpansberg, South Africa. African Journal of Ecology 52:318–327.
Boshoff AF, Kerley GIH, Cowling RM, Wilson SL. 2002. The potential distributions, and estimated spatial requirements and population sizes, of the medium to large-sized mammals in the planning domain of the Greater Addo Elephant National Park. Koedoe 45:85–116.
Bowland AE. 1997. Common Duiker Sylvicapra grimmia. Page 261 in Mills G, Hes L, editors. The Complete Book of Southern African Mammals. Struik Publishers, Cape Town, South Africa.
East, R. 1999. African Antelope Database 1998. IUCN, Gland, Switzerland and Cambridge, UK.
Gagnon M, Chew AE. 2000. Dietary preferences in extant African Bovidae. Journal of Mammalogy 81:490–511.
Grey-Ross R, Downs CT, Kirkman K. 2010. An assessment of illegal hunting on farmland in KwaZulu-Natal, South Africa: implications for Oribi (Ourebia ourebi) conservation. South African Journal of Wildlife Research 40:43–52.
Grubb P, Groves CP. 2001. Revision and classification of the Cephalophinae. Pages 703–728 in Wilson VJ, editor. Duikers of Africa: Masters of the African Floor. Chipangali Wildlife Trust, Bulawayo, Zimbabwe.
Hayward MW. 2006. Prey preferences of the Spotted Hyaena (Crocuta crocuta) and degree of dietary overlap with the Lion (Panthera leo). Journal of Zoology 270:606–614.
Hayward MW, Henschel P, O’Brien J, Hofmeyr M, Balme G, Kerley GIH. 2006. Prey preferences of the Leopard (Panthera pardus). Journal of Zoology 270:298–313.
IUCN SSC Antelope Specialist Group. 2016. Sylvicapra grimmia. The IUCN Red List of Threatened Species 2016: e.T21203A50194717.
Jansen van Vuuren, A., Fritz, H. and Venter, J.A., 2022. Five small antelope species diets indicate different levels of anthrodependence in the Overberg Renosterveld, South Africa. African Journal of Ecology, 60(4), pp.917-925.
Kigozi F. 2003. The significance of chicory to the diet of common duiker at Grants valley, Eastern Cape Province, South Africa. African Journal of Ecology 41:289–293.
Lynch CD. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10:177–241.
Prins HHT, de Boer WF, van Oeveren H, Correia A, Mafuca J, Olff H. 2006. Co-existence and niche segregation of three small bovid species in southern Mozambique. African Journal of Ecology 44:186–198.
Skinner JD, Chimimba CT. 2005. The Mammals of the Southern African Subregion. Third edition. Cambridge University Press, Cambridge, UK. Wilson VJ. 2001. Duikers of Africa: Masters of the African Forest Floor. Directory Publishers, Bulawayo, Zimbabwe.
Wilson, V.J. 2001. Duikers of Africa: Masters of the African Forest Floor. Directory Publishers, Bulawayo, Zimbabwe.
Wilson VJ. 2013. Sylvicapra grimmia Common Duiker. Pages 235243 in Kingdon JS, Hoffmann M, editors. The Mammals of Africa. Volume VI: Pigs, Hippopotamuses, Chevrotain, Giraffes, Deer and Bovids. Bloomsbury Publishing, London, UK.