Cape Hare
Lepus capensis

2025 Red list status
Least Concern
Regional Population Trend
Declining
Change compared
to 2016
No Change
Overview
ANIMALIA – CHORDATA – MAMMALIA – LAGOMORPHA – LEPORIDAE – Lepus – capensis
Common Names: Cape Hare, Arabian Hare, Brown Hare, Desert Hare (English), Vlakhaas (Afrikaans), Umvundla (Ndebele, Xhosa), Mofuli (Sesotho), Matshwaratsela(na), Mmutla wamatshwaratselana, Matsaatsela, Mmutla wamatsaatsela, Mmutlê wamatsaatsela, Ditshêtlhane, Moduôlô (Setswana), Logwatja (Swati), Mpfundla (Tsonga), Muvhuda, Khomu (Venda), Unogwaja (Zulu), Sungura (Swahili)
Synonyms: 38 listed in Africa by Hoffmann and Smith (2005)
Taxonomic Note:
The entire taxonomy of Lepus capensis throughout its range is unclear. Taxonomic review of the species is urgently needed; otherwise, it is possible that some forms may go extinct before they are formally identified.
Hoffmann and Smith (2005) restricted L. capensis to the South African distribution, citing no evidence of gene flow between the southern and northern ranges. This has been supported by Lado et al. (2019) who reported that a deep phylogenetic break dating to the Early Pleistocene occurred between the African and Asian L. capensis groups resulting in the latter being more closely related to Eurasian hare species. A list of synonyms is provided based on four geographic locations (South Africa, East Africa, Arabia and Near East, and northwest Africa), which are informal subdivisions of L. capensis sensu lato. The authors suggested that these four groups might represent distinct species, but further research is needed to verify this. In the Near East and Arabia L. c. arabicus; in South Africa L. c. capensis; L. c. aquilo, L. c. carpi, L. c. granti; East Africa L. c. aegyptius, L. c. hawkeri, L. c. isabellinus, L. c. sinaiticus; and L. c. atlanticus, L. c. schulmbergeri, L. c. whitakeri in northwest Africa (Hoffmann and Smith 2005, Schai-Braun and Hackländer 2018).
According to Harrison and Bates (1991) there are eight subspecies in Arabia: L. c. syriacus (Syria, Lebanon, northern Israel); L. c. sinaiticus (southern Israel, Sinai); L. c. connori (east of the Euphrates in Iraq); L. c. arabicus (western Iraq, Kuwait, Saudi Arabia, Yemen); L. c. cheesmani (Saudi Arabia, Qatar, UAE, Yemen, Oman); L. c. omanensis (UAE, Oman); L. c. atallahi (Bahrain, Qatar?); L. c. jefferyi (Masirah Island, Oman). The taxonomic status of L. c. jefferyi needs clarification, as it may represent a good species. Hares present in Qatar also require taxonomic investigation.
The taxonomic position of the Sardinian Hare is unresolved. Hoffmann and Smith (2005) include the Sardinian Hare as one of the unassigned synonyms of L. mediterraneus Wagner, 1841 or typicus Hilzheimer, 1906, in L. granatensis. Analysis of the mtCR-1 sequence indicated that Sardinian Hares form a monophyletic clade with North African Hares (Scandura et al. 2007). A genetic and morphometric analysis supports the hypothesis that the Sardinian Hare was introduced from North Africa (Canu et al. 2012). A phylogenetic analysis of mtCR-1 sequences from Tunisian and Egyptian Hares characterised them as monophyletic and separate from L. capensis (Ben Slimen et al. 2006). However, a study of the nuclear gene pool of L. capensis, L. europaeus and the North African Hare indicated that the North African Hare as well as L. europaeus belong to L. capensis (Ben Slimen et al. 2005), supporting Petter’s (1959, 1961) hypothesis of the inclusion of L. europaeus in capensis. Ben Slimen et al. (2008a) suggest that in a case such as the genus Lepus, where evolution is “rapid and to some extent reticulate”, species designation based solely on mtDNA is misleading without examination of the nuclear gene pool. Ben Slimen et al. (2008a) has shown that genetic differentiation between L. capensis and L. europaeus could be attributed to geographic distance rather than divergence. They speculate that gene flow may be occurring in the Near East where distributions meet resulting in the potential for intergraded populations. However, Ben Slimen et al. (2008b) propose that “a combined phylogenetic, phylogeographic, and population genetic approach, based on various nuclear and mitochondrial markers and including other biological characters, such as phenotypic and morphometric data,” is needed for conclusive evidence of a single species complex. A recent study looking at a partial transferrin nuclear gene and phylogenetic relationship of hares in Tunisia showed “shared ancestral polymorphism between North African and Chinese hares” (Awadi et al. 2016). It also concluded that the Tunisian Hare is “well differentiated from hares considered belonging to brown hares L. europaeus from central Europe” (Awadi et al. 2016). In light of the continuing uncertainty regarding the taxonomic status of the Sardinian and North African Hares, both will remain included in capensis and L. europaeus retains its taxonomic status as a distinct species.
Many treatments indicate that the range of L. capensis extends into China, Mongolia and Russia; however, recognition of L. tibetanus and L. tolai as distinct species removes consideration of L. capensis as occurring in this region (Hoffmann and Smith 2005). Recent chromosome-level genome assembly by Dong et al. (2024) suggests between seven and nine hare species may exist in China alone.
| Red List Status |
| LC – Least Concern, (IUCN version 3.1) |
Assessment Information
Assessors: Wilson-Hartmann, B.1 & da Silva, J.2
Reviewer: Moodley, S.3
Institutions: 1McGregor Museum, Kimberley, 2South African National Biodiversity Institute, 3Private
Previous Assessors & Reviewers: Robinson, T.J., Relton, C. & Child, M.F.
Previous Contributors: Roxburgh, L. & Smith, A.
Assessment Rationale
The Lepus species in South Africa occur widely within the assessment region in many habitats that both protected and transformed. Ongoing molecular research is indicating that the northern and southern populations are distinct which in theory raises the potential threat severity to the southern populations. These species may require reassessment when such data becomes available. The Cape Hare (L. capensis) is common in suitable habitats, preferring a wide variety of grassland and open habitat, while avoiding only bushy or closed habitats. For example, on Benfontein Game Farm, Kimberley, Northern Cape, density has been estimated as 16.5 individuals / km2. Although local declines may be occurring due to overhunting for bushmeat and fur in some areas, there was no evidence to suggest a widespread population decline during previous assessment periods. However, with the outbreak of Rabbit Haemorrhagic Disease Virus strain 2 (RHDV-2) in November 2022 in the Northern Cape Province, South Africa which subsequently spread to all provinces (DALRRD 2024), urgent investigations are needed to ascertain the effect on populations and subpopulations.
Regional population effects: Taxonomic revision is ongoing to differentiate the geographical range limits of Lepus species in southern Africa.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Wilson-Hartmann B & da Silva JM. 2025. A conservation assessment of Lepus capensis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional distribution and occurence
Geographic Range
Lepus capensis has an extensive but scattered distribution across southern, eastern and northern Africa, occurring in Mediterranean, Coastal, Sahel, Savannah and southern African biotic zones (Ellerman & Morrison-Scott 1951; Ellerman et al. 1953; Palacios et al. 2008; Happold 2013). However, recent taxonomic delineation (Lado et al. 2019; Dong et al. 2024) is improving our understanding of its geographical distribution. Currently, there is a gap in its distribution as it does not occur in Malawi, southern Tanzania, northern Zimbabwe and parts of Mozambique (Happold 2013). It is restricted to non-forested regions (Boitani et al. 1999). There appears to be an isolated population in Botswana near the Makagadikadi Pans (Boitani et al. 1999; Skinner & Chimimba 2005). Within the assessment region, the Cape Hare occurs extensively across the Northern and Western Cape, into the western Northwest Province, across much of the Free State, western Lesotho, Eswatini and marginally into the northwestern parts of KwaZulu-Natal and Eastern Cape provinces (Skinner & Chimimba 2005). Since this species moves into burnt areas when grasses begin to sprout, its range has expanded with increased levels of bush clearance and fires across Africa (Happold 2013).
Taxonomic resolution and vetting of museum records through molecular research is needed urgently to resolve the tentative geographical ranges.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 1500
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realms
Biogeographic Realm: Afrotropical, Palearctic
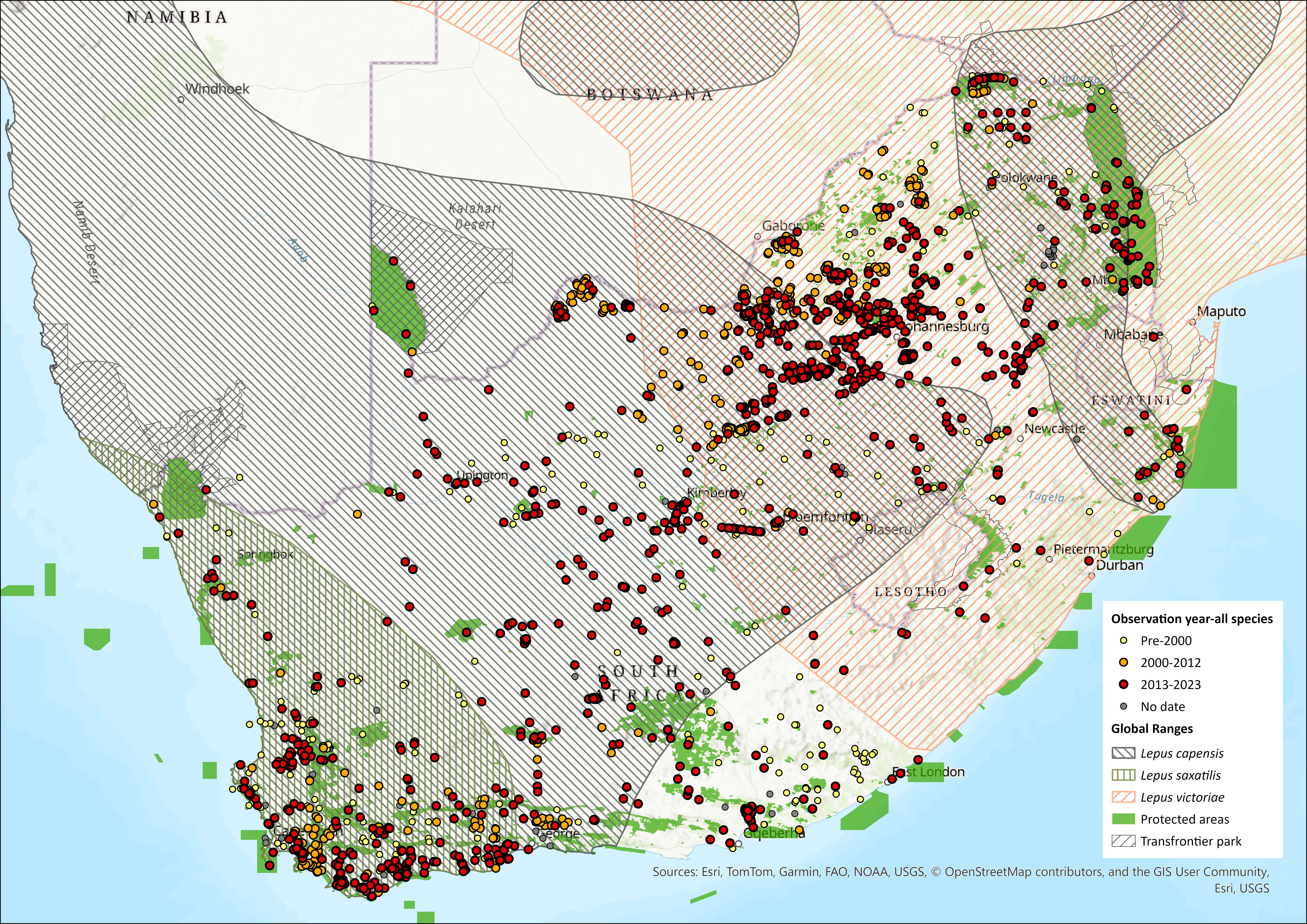
Map
Figure 1. Distribution records for Cape Hare (Lepus capensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Algeria | Extant | Native | – | – |
| Bahrain | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Cyprus | Extant | Native | – | – |
| Egypt | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| India | Extant | Native | – | – |
| Iran, Islamic Republic of | Extant | Native | – | – |
| Iraq | Extant | Native | – | – |
| Israel | Extant | Native | – | – |
| Italy | Extant | Native | – | – |
| Italy -> Sardegna | Extant | Native | – | – |
| Jordan | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Kuwait | Extant | Native | – | – |
| Lebanon | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Libya | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Morocco | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Oman | Extant | Native | – | – |
| Pakistan | Extant | Native | – | – |
| Palestine, State of | Extant | Native | – | – |
| Qatar | Extant | Native | – | – |
| Saudi Arabia | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Syrian Arab Republic | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Tunisia | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| United Arab Emirates | Extant | Native | – | – |
| Western Sahara | Extant | Native | – | – |
| Yemen | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Whilst climate change per se is unlikely to have a dramatic effect on this species during this assessment period, climate change is already affecting the spread of infectious diseases beyond their typical geographic reach. Whilst this may not be the cause of the outbreak of Rabbit Haemorrhagic Disease Virus strain 2 (RHDV-2) in South Africa at the end of 2022, it can assist in the spread of the virus and increase the susceptibility of hares and rabbits to the effects of droughts and periods of flooding. Often, the cross-border spread of infectious diseases is further exacerbated by the lack of global governance (that led to the introduction of this disease into South Africa and allowed it to spread to neighbouring countries), policies or a consensus to mitigate climate change. As a result, the current and future burden on humans, animals and plants is significant, especially if these infectious diseases cause large scale outbreaks such as RHDV-2.
Population information
Lepus capensis is probably the most abundant Lepus species in Africa, with densities ranging from 4.7–24.8 hares / km2 in South Africa alone (Happold 2013c). For example, on Benfontein Game Farm, Northern Cape, density has been estimated as 16.5 individuals / km2 (Stenkewitz et al. 2010).
Lepus species generally exhibit a fast population turnover and a high rate of reproduction (Kryger et al. 2004b). While a slow decline has been predicted due to habitat loss and hunting pressure (Kryger et al. 2004a), it is expected that land conversion from livestock to wildlife may benefit Lepus species and counteract population declines by creating and reconnecting suitable habitat patches. However, the status of scattered subpopulations and isolated subspecies is largely unknown and may be severely threatened by the recent RHDV-2 outbreak that is affecting all Lepus and Pronolagus populations throughout southern Africa. For example, populations are fragmented within South Africa’s central grassland regions. Further research is required to assess the impact of this.
Population Information
Continuing decline in mature individuals? Yes
Extreme fluctuations in the number of subpopulations: Yes
Continuing decline in number of subpopulations: Yes
All individuals in one subpopulation: No
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Unknown
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Unknown
Probability of extinction in the wild within 100 years: Unknown
Population genetics
Phylogenetic analyses using mitochondrial DNA and microsatellite data identified two clades within L. capensis corresponding to northern and southern individuals (Lado et al. 2019). However, given the small sample size of the Southern (South African) population, further analyses with larger sample sets are needed to clarify its degree of differentiation and divergence.
No population genetic studies have been conducted on this species to infer further substructure within the assessment region or to quantify effective population size.
Habitats and ecology
Lepus capensis is very adaptable and lives in a wide variety of grassland and open habitat, avoiding only bushy or closed habitats (Happold 2013). Preferring dry, open habitats (Boitani et al. 1999), the species is widespread throughout Nama-Karoo and Succulent Karoo biomes and occurs in parts of the Grassland and Savannah Biome in southern Mozambique (Skinner & Chimimba 2005). Cape Hares in the Free State were found to have home ranges of 6.459 ha and 8.25 ha for males and females, respectively, which are defended in some portions, but may overlap at the fringes (Wessels 1978). However, home range size is known to vary depending on habitat type (Flux & Angermann 1990). It may reach densities of 25 hares/1km2 in some areas (Stuart & Stuart 2015). This species feeds both by browsing and grazing and can survive successfully without a continued supply of surface water by relying on forage as a source of moisture (Skinner & Chimimba 2005). Usually preferring short, green grasses, the diet of Cape Hares in East Africa was found to consist mostly of Digitaria spp. and Eragrostis spp. (Stewart 1971). In the Karoo, however, Kerley (1990) documented Cape Hares browsing on various species, favouring Galenia fruticose and Osteospermum sinuatum and were not recorded feeding on grasses. Predominantly nocturnal, the Cape Hare is only occasionally seen during the day during overcast conditions (Skinner & Chimimba 2005). They are usually solitary, except when females (who are larger than the males) are in oestrus, and although breeding can take place all year round, most births occur between July and December (Wessels 1978). The gestation period lasts approximately 42 days and females may produce between one and three young (Smithers 1971). Wessels (1978) found that they may have up to four litters per year, while Flux (1981) noted that up to eight litters per year are possible with between 1.3 and 2.0 individuals per litter.
Modified landscapes, such as those overgrazed by livestock, are suitable habitats for Lepus capensis (Flux & Angermann 1990). Similarly, Lepus species are attracted to cultivated areas and gardens (Happold 2013). The distributional limits of L. capensis and L. saxatilis overlap somewhat (Skinner & Chimimba 2005). The former extends into arid, open regions while the latter is confined to areas of grass cover within savannah woodland and scrub habitats and adapts easily to agricultural landscapes (Kryger et al. 2004b). Similarly, where L. capensis and L. victoriae co-occur, the latter prefers scrubbier and more montane habitats (Flux & Angermann 1990). All Lepus species have a preference for green grasses (Skinner & Chimimba 2005).
Ecosystem and cultural services: Lepus species form a valuable prey component for a broad array of small carnivores, such as Black-backed Jackals (Lupulella mesomelas; Kamler et al. 2012; Bagniewska & Kamler 2014) and Verreaux’s Eagles (Aquila verreauxii; Cruz-Uribe & Klein 1998). Hares were found to be the dominant prey taxon for Martial Eagles (Polemaetus bellicosus) in the Cape region of South Africa, making up 50% of the total diet (Boshoff et al. 1990). Lepus species were also found to constitute a small component of the diet of Cape Foxes (Vulpes chama) in the Free State (Kamler et al. 2012).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 8.1. Desert -> Desert – Hot | – | Suitable | – |
| 8.2. Desert -> Desert – Temperate | – | Suitable | – |
| 14.1. Artificial/Terrestrial -> Artificial/Terrestrial – Arable Land | – | Suitable | – |
| 14.2. Artificial/Terrestrial -> Artificial/Terrestrial – Pastureland | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: 1 year
Age at Maturity: Male: -1 year
Size at Maturity (in cms): Female: 55cm
Size at Maturity (in cms): Male: 51cm
Longevity: 3-5 years
Average Reproductive Age: (Not specified)
Maximum Size (in cms): 59cm
Size at Birth (in cms): (Not specified)
| Gestation Time |
| 42 Days |
Reproductive Periodicity: Year-round
Average Annual Fecundity or Litter Size: 1-3 (4 litters/year)
Natural Mortality: (Not specified)
Breeding Strategy
| Does the species lay eggs? |
| No |
| Does the species give birth to live young |
| Yes |
| Does the species exhibit parthenogenesis |
| No |
| Does the species have a free-living larval stage? |
| No |
| Does the species require water for breeding? |
| No |
Movement Patterns
Movement Patterns: Unknown
Congregatory: Unknown
Systems
System: Terrestrial
General Use and Trade Information
Lepus species are hunted recreationally for sport, bushmeat and fur at a subsistence level in the assessment region. However, this is not expected to have a substantial effect on the population. L. capensis is utilised for traditional medicine in South Africa, as they are believed to have medicinal or curative properties (Maliehe 1993; Ntiamoa-Baidu 1997).
|
Subsistence |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
Yes |
Bushmeat and fur |
– |
– |
National Commercial Value: No
International Commercial Value: No
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
1. Food – human |
true |
– |
– |
– |
|
3. Medicine – human & veterinary |
– |
|
– |
– |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: (Not specified)
Threats
Across their range, as well as within the assessment region, Lepus species are threatened by habitat loss and fragmentation due to urban sprawl, agricultural encroachment, commercial plantations, and infrastructure development for tourism (Drew et al. 2004; Kryger et al. 2004a). Additionally, these species are threatened by hunting pressure through both recreational sport hunting, as well as subsistence hunting for bushmeat and fur (Kryger et al. 2004a). While agricultural and urban expansion may not necessarily cause direct declines, they may increase hunting pressures. Hunting pressure is likely to cause local subpopulation declines. For example, drastic population declines have been observed in KwaZulu-Natal, specifically in Harding and Port Shepstone in southern KwaZulu-Natal (Kryger et al. 2004a). Lepus species may also be vulnerable to predation by domestic dogs associated with farming areas.
However, a more urgent threat has recently emerged. In October 2022, reports of unusual Lepus deaths were received from the Middelpos, Springbok and Fraserburg areas in the Northern Cape. In November 2022 affected carcasses tested by Onderstepoort Veterinary Research Laboratory, the deaths were confirmed to as the result of the highly contagious and acute fatal hepatitis of Leporids called Rabbit Haemorrhagic Disease Virus 2 strain (RHDV-2). The new variant RHDV-2 affects rabbits, but also causes fatal RHD in various Lepus species, including Sardinian Cape Hares (L. capensis mediterraneus), Italian Hares (L. corsicanus), and Mountain Hares (L. timidus) (Rocchi & Dagleish 2018). This disease, which originated in China in 1984, primarily affects all rabbit and hares. Caused by a calicivirus, it was previously absent in South Africa. Infection typically occurs by the oral route. It spreads very easily, normally through direct contact between individuals but is also carried by insects, birds, scavengers feeding on carcasses, contaminated food or urine and faeces, and by humans on clothes, shoes, tools and car tyres. The virus can persist in infected meat (even if frozen) and for long periods in decomposing carcasses (Gleeson & Petritz 2020). Incubation varies from one to three days, and death usually occurs 12-36 hours after the onset of fever.
The main clinical manifestations of the acute infection are nervous and respiratory signs, apathy and anorexia. In RHDV-2, clinical signs and mortality are observed even in young animals from 7 to 15 days of age onwards. Indirect control of the disease is easily achieved by vaccination. RHDV has never been reported in humans and other mammals. Outbreaks tend to be seasonal (typically peak breeding seasons) in wild rabbit and hare populations where adults have survived infection and are immune (Kerr & Donnelly 2013). As young kits grow up and stop nursing, they no longer receive the antibodies in their mother’s milk and become susceptible to infection. Mortality rates are high and up to 90% of infected individuals die (WOAH 2025) not all rabbits exposed to RHDV-2 become overtly ill. A small proportion of infected rabbits clears the virus without developing signs of disease (Kerr & Donnelly 2013). Asymptomatic carriers also occur, and can continue to shed virus for months, thereby infecting other animals. Surviving rabbits develop a strong immunity to the specific viral variant with which they were infected (Gleeson & Petritz 2020).
This outbreak is of concern for indigenous rabbit and hare species due to their historic naivety to the virus and thus susceptibility to the virus. Whilst a vaccine is available for captive situations overseas, there is no specific treatment for RHDV-2 and management is limited to preventing further spread.
Often, the cross-border spread of infectious diseases is further exacerbated by the lack of global governance, a factor that led to the introduction of this disease into South Africa and allowed it to spread to neighbouring countries.
Conservation
Lepus species are widely distributed across the assessment region, occurring within numerous protected areas, including both formally and privately protected areas (Kryger et al. 2004a). A call for sustainable utilisation of these species is recommended (Kryger et al. 2004a), as they may constitute a low-carbon source of protein and may economically benefit local communities and landowners (Asibey 1974). The development of conservancies to protect appropriate habitats for local subspecies and forms is recommended.
Recommendations for land managers and practitioners:
- This species would benefit from suitable land management: landowners should retain corridors of grassland between grazed areas and decrease stocking rates to avoid overgrazing and the loss of grass cover.
Research priorities:
- Taxonomic revision of the Lepus capensis genus, including the identification of isolated populations and localised subspecies.
- Survey studies to effectively document the distributional limits and geographical overlap of species and subspecies.
- Population size and trends.
- Effects of wildlife ranching on Lepus species within the assessment region.
- Determination of the impact of RHDV-2 on the southern African population, particularly at subpopulation level.
Encouraged citizen actions:
- Landowners should create conservancies to utilise this species sustainably.
- Refrain from having too many dogs on farms.
- Report sightings of free-roaming individuals on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas to enhance the distribution maps.
- Report unusual death outbreaks potentially linked to RHDV-2 and prevent the spread of the disease.
Bibliography
Angermann, R. 1965. Revision der Palaarkitschen und Athiopischen Arten der Gattung Lepus (Leporidae, Lagomorpha). Humboldt University of Berlin, Germany.
Asibey, E.O.A. 1974. Wildlife as a source of protein in Africa south of the Sahara. Biological Conservation 6: 32–39.
Bagniewska, J.M. and Kamler, J.F. 2014. Do black-backed jackals affect numbers of smaller carnivores and prey? African Journal of Ecologyvvvv 52: 546–567.
Ben Slimen, H., Suchentrunk, F., Stamatis, C., Mamuris, Z., Sert, H., Alves, P.C., Kryger, U., Shahin, A.B., and Ben Ammar Elgaaied, A. 2008. Population genetics of cape and brown hares (Lepus capensis and L. europaeus): A test of Petter’s hypothesis of conspecificity. Biochemical Systematics and Ecology 36: 22-39.
Boitani, L., Corsi, F., De Biase, A. 1999. A Databank for the Conservation and Management of the African Mammals. Istituto di Ecologia Applicata, Rome, Italy.
Collins, K., Kryger, U., Matthee, C., Keith, M. and van Jaarsveld A. 2008. Lepus saxatilis.
Cruz-Uribe, K. and Klein, R.G. 1998. Hyrax and hare bones from modern South African eagle roosts and the detection of eagle involvement in fossil bone assemblages. Journal of Archaeological Science 25: 135–147.
Department Agriculture, Land Reform and Rural Development. 2024. Rabbit haemorrhagic disease outbreak update report. Directorate Animal Health, 27 February 2024.
Dong, X., Liu, Y., Chen, Y. et al. A chromosome-level genome assembly of Cape hare (Lepus capensis). Sci Data 11, 1081 (2024). https://doi.org/10.1038/s41597-024-03953-2
Drew, C., Barcelo, I. and Saji, A. 2004. A Report on the Findings of an Ecological and Environmental Survey of the Sweihan Region. Environmental Research and Wildlife Development Agency, Abu Dhabi, UAE.
Ellerman, J.R. and Morrison-Scott, T.C.S. 1951. Checklist of Palaearctic and Indian Mammals 1758 to 1946. British Museum (Natural History), London, UK.
Ellerman, J.R., Morrison-Scott, T.C.S. and Hayman, R.W. 1953. Southern African Mammals 1758 to 1951: A Reclassification. British Museum (Natural History), London, UK.
Flux, J. C. 1981. Reproductive strategies in the genus Lepus. University of Guelph, Guelph.
Flux, J.E.C. and Angermann, R. 1990. The hares and jackrabbits. In: J.A. Chapman and J.E.C. Flux (eds), Rabbits, Hares and Pikas: Status Survey and Conservation Action Plan, pp. 61-94. IUCN, Gland, Switzerland.
Gleeson, M. and Petritz, O.A. 2020. Emerging Infectious Diseases of Rabbits. Veterinary Clinics of North America: Exotic Animal Practice. 23 (2): 249–261.
Happold, D.C.D. 2013. Lepus capensis Cape Hare. In: Happold DCD (ed.), Mammals of Africa Volume III: Rodents, Hares and Rabbits, pp. 699–701. Bloomsbury Publishing, London, UK.
Hoffmann, R.S. and Smith, A.T. 2005. Order Lagomorpha. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World, pp. 185-211. Johns Hopkins University Press, Baltimore, Maryland, USA.
Kamler, J.F., Stenkewitz, U., Klare, U. Jacobsen, N. F., and Macdonald, D. W. 2012b. Resource partitioning among cape foxes, bat-eared foxes, and black-backed jackals in South Africa. Journal of Wildlife Management 76: 1241-1253.
Kerley, G.I.H. 1990. Browsing by Lepus capensis in the Karoo. South African Journal of Zoology 25: 199–200.
Kerr, P.J. and Donnelly, T.M. 2013. Veterinary Clinics of North America: Exotic Animal Practice. 16 (2): 437–468.
Kryger, U. 2002. Genetic variation among southern African hares (Lepus spec.) as inferred from mitochondrial DNA and microsatellites. Ph.D. Thesis. University of Pretoria.
Kryger, U., Keith, M., Collins, K. and van Jaarsveld, A. 2004. Lepus saxatilis. In: Y. Friedmann and B. Daly (ed.), Red Data Book of the Mammals of South Africa: A Conservation Assessment, pp. 416-417. Endangered Wildlife Trust and CBSGCAMP Programme and Database, South Africa.
Kryger, U., Matthee, C., Keith, M., Collins, K. and van Jaarsveld, A. 2004. Lepus capensis. In: Y. Friedman and B. Daly (eds), Red Data Book of Mammals of South Africa: A Conservation Assessment, pp. 414-415. Endangered Wildlife Trust and CBSG (IUCN/SSC).
Kryger, U., Robinson, T. J. and Bloomer, P. 2004. Population structure and history of southern African scrub hares, Lepus saxatilis. Journal of Zoology (London) 263: 121-133.
Lado, S., Alves, P.C., Islam, M.Z. et al. 2019. The evolutionary history of the Cape hare (Lepus capensis sensu lato): insights for systematics and biogeography. Heredity 123, 634–646.
Maliehe, T.M. 1993. Non-wood forest products in South Africa. Paper prepared for the Regional Expert Consultation on non-wood forest products for English-speaking African countries. Commonwealth Science Council, Arusha, Tanzania.
Ntiamoa-Baidu, Y. 1997. Wildlife and food security in Africa. FAO Conservation Guide 33, Food and Agriculture Organization of the United Nations, Rome. Available at: http://www.fao.org/docrep/w7540e/w7540e00.htm#Contents.
Palacios, F., Angelone, C., Alonso, G. andReig, S. 2008. Morphological evidence of species differentiation within Lepus capensis Linnaeus, 1758 (Leporidae, Lagomorpha) in Cape Province, South Africa. Mammalian Biology-Zeitschrift für Säugetierkunde 73: 358–370.
Petter, F. 1959. Elements d’une revision des lievres africains du sous-genre Lepus. Mammalia 23: 41-67.
Rautenbach, I. L. 1982. Mammals of the Transvaal. Ecoplan monograph, N.S. Supplemento 1: 111-211.
Rocchi, M.S. and Dagleish, M.P. 2018. Diagnosis and prevention of rabbit viral haemorrhagic disease 2. In Practice. 40 (1): 11–16.
Robinson, T.J., Matthee, C.A. 2005. Phylogeny and evolutionary origins of the Leporidae: a review of cytogenetics, molecular analyses and a supermatrix analysis. Mammal Review 35: 231–247.
Robinson, T.J. 1986. Incisor morphology as an aid in the systematics of the South African Leporidae (Mammalia: Lagomorpha). African Zoology 21: 297–302.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smithers, R.H.N. 1971. The mammals of Botswana. National Museums of Rhodesia, Museum Memoir 4: 1-340.
Smithers, R.H.N. and Wilson, V.J. 1979. Check List and Atlas of the Mammals of Zimbabwe Rhodesia. Trustees of the National Museums and Monuments of Rhodesia, Salisbury, Rhodesia.
Stenkewitz, U., Herrmann E, Kamler JF. 2010. Distance sampling for estimating springhare, cape hare and steenbok densities in South Africa. South African Journal of Wildlife Research 40: 87-92.
Stewart, D.R.M. 1971. Diet of Lepus capensis and L. crawshayi. African Journal of Ecology 9: 161–162.
Stuart, C. and Stuart, M. 2015. Stuarts’ Field Guide to Mammals of Southern Africa: including Angola, Zambia & Malawi. Penguin Random House, South Africa.
Suchentrunk, F., Ben Slimen, H. and Kryger, U. 2009. Molecular evidence of conspecificity of South African hares conventionally considered Lepus capensis L., 1758, Mammalian Biology, 74(5):325-343.
Taylor, P.J. 1998. Regional patterns of small mammal abundance and community composition in protected areas in KwaZulu-Natal. Durban Museum Novitates 23: 42–51.
Taylor, P.J. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.
Wessels, T.C. 1978. Bevolkingsdinamika van die vlakhaas Lepus capensis L., 1758 in die Willem Pretorius-wildtuin. M.Sc. Thesis. University of Pretoria.
World Organisation for Animal Health. Rabbit Haemorrhagic Disease.