Black Wildebeest
Connochaetes gnou

2025 Red list status
Least Concern
Regional Population Trend
Increasing
Change compared
to 2016
No Change
Overview
Connochaetes gnou – (Zimmermann, 1780)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Connochaetes – gnou
Common Names: Black Wildebeest, White-tailed Gnu (English), Swartwildebees (Afrikaans), Gnu (Khoikhoi), Imbudumo (Ndebele), Podumö (Sepedi), Mmamononwane (Sesotho), Ingongoni (Swati), Mbutuma (Tsonga), Khongoni (Venda), Inqu (Xhosa), Inkonkoni (Zulu)
Synonyms: No Synonyms
Taxonomic Note:
The two lineages leading from a Blue Wildebeest-like ancestor to the two modern Wildebeest species diverged approximately one million years ago. The two species share the same chromosome number of 2n=58 and interspecific hybrids are fertile. First generation (F1) hybrids can be distinguished from pure animals based on external phenotypic characters, but hybrids are not distinguishable from pure animals following unidirectional backcrosses with pure Black Wildebeest over two or more generations. Levels of genetic diversity in Connochaetes gnou are lower compared to the Blue Wildebeest (C. taurinus) (Grobler et al. 2005 and ongoing work at the University of the Free State and the National Zoological Gardens), which is most likely due to the historic bottlenecks experienced by C. gnou in the late 1800s. The evolution of a distinct southern endemic Black Wildebeest in the Pleistocene was associated with, and possibly driven by, a shift towards a more specialised kind of territorial breeding behaviour, which can only function in open habitat. Thus, the evolution of the Black Wildebeest was directly associated with the emergence of Highveld-type open grasslands in the central interior of South Africa (Ackermann et al. 2010).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Shrader, A.M.1 & da Silva, J.2
Reviewers: Mallon, D.3
Institutions: 1Department of Zoology & Entomology, University of Pretoria, South Africa, 2South African National Biodiversity Institute, 3IUCN SSC Antelope Specialist Group
Previous Assessors: Vrahimis, S., Grobler, P., Brink, J., Viljoen, P. & Schulze, E.
Previous Reviewers: Child, M.F. & Mallon, D.P.
Previous Contributor: Selier, J.
Assessment Rationale
This is an endemic species occurring in open grasslands in the central interior of the assessment region. There are at least 15,655 individuals (counts conducted between 2016 and 2023) on protected areas across the Free State, Gauteng, North West, Northern Cape, Eastern Cape, and Mpumalanga provinces (mostly within the natural distribution range). Black Wildebeest are also found in KwaZulu-Natal (KZN), but population estimates were not available for this assessment. This yields a minimum number of mature individuals of 9,393-10,959 (using a 60-70% mature population structure). The largest subpopulation of 2,773 individuals (2021 count) is found in Bloemhof Dam Nature Reserve in the North West province. The total population estimate is slightly lower than the previous assessment. However, this is likely due to missing data (i.e. KwaZulu-Natal) and a lack of reliable data from other protected areas, rather than a reflection of a population decline. As such, this is an underestimate as the data are not complete for all the protected areas in these provinces plus there are many more subpopulations on private wildlife ranches for which comprehensive data are unavailable. Most subpopulations in protected areas are stable or increasing. For example, the subpopulation in the Eastern Cape provincial protected areas has remained relatively stable with a 0.4% annual increase from 2012-2022 (i.e., 4,708 to 4,922) individuals), while there was a 7.5% annual increase in the subpopulation in the North West provincial parks from 2012 to 2021 (i.e., 2,764 to 4,845 individuals).
While grassland habitat loss remains a threat to this species, it continues to be well represented in protected areas and the expansion of wildlife ranching may be conserving additional habitat. The only major identified threat is the risk of hybridisation with Blue Wildebeest, a threat enhanced by the fenced and fragmented nature of existing subpopulations. Although it is likely that Black and Blue Wildebeest hybridised occasionally, they are naturally spatially separated through ecological specialisation. However, translocation of Black Wildebeest outside their historical range has increased contact between the two species. This, coupled with historical habitat reduction has restricted the two species to pockets of reserves and may have artificially induced hybridisation. There is worrying evidence that some formally protected reserves may contain hybrid individuals and further research is in progress to assess the extent of hybridisation in the population (Grobler et al. 2018). This species will need reassessment once comprehensive data on the extent of hybridisation is produced. Currently, the hybridisation threat is being managed through implementing management practices such as separating Black and Blue Wildebeest in protected areas and through enforcing strict translocation policies. In line with this, we limited the data we used in this assessment to reserves that did not contain Blue Wildebeest. The minimum number of mature individuals is between 9,393-10,959, which would justify retaining a Least Concern listing. Currently, these reserves are generally considered to contain genetically pure animals based on management history include Benfontein Game Farm (Northern Cape), SA Lombard Nature Reserve (North West), Suikerbosrand Nature Reserve (Gauteng), possibly Geluk Farm (Free State), and extra-limitally in Grootte Schuur Estate (Western Cape). Conservation efforts should be concentrated on sustaining these pure animals and on establishing additional pure subpopulations through a coordinated translocation programme and metapopulation plan. The role of incidental and deliberate back-crossing in reducing the frequency of introgressed alleles in putative hybrid population should also be investigated.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Shrader AM & da Silva JM. 2025. A conservation assessment of Connochaetes gnou. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
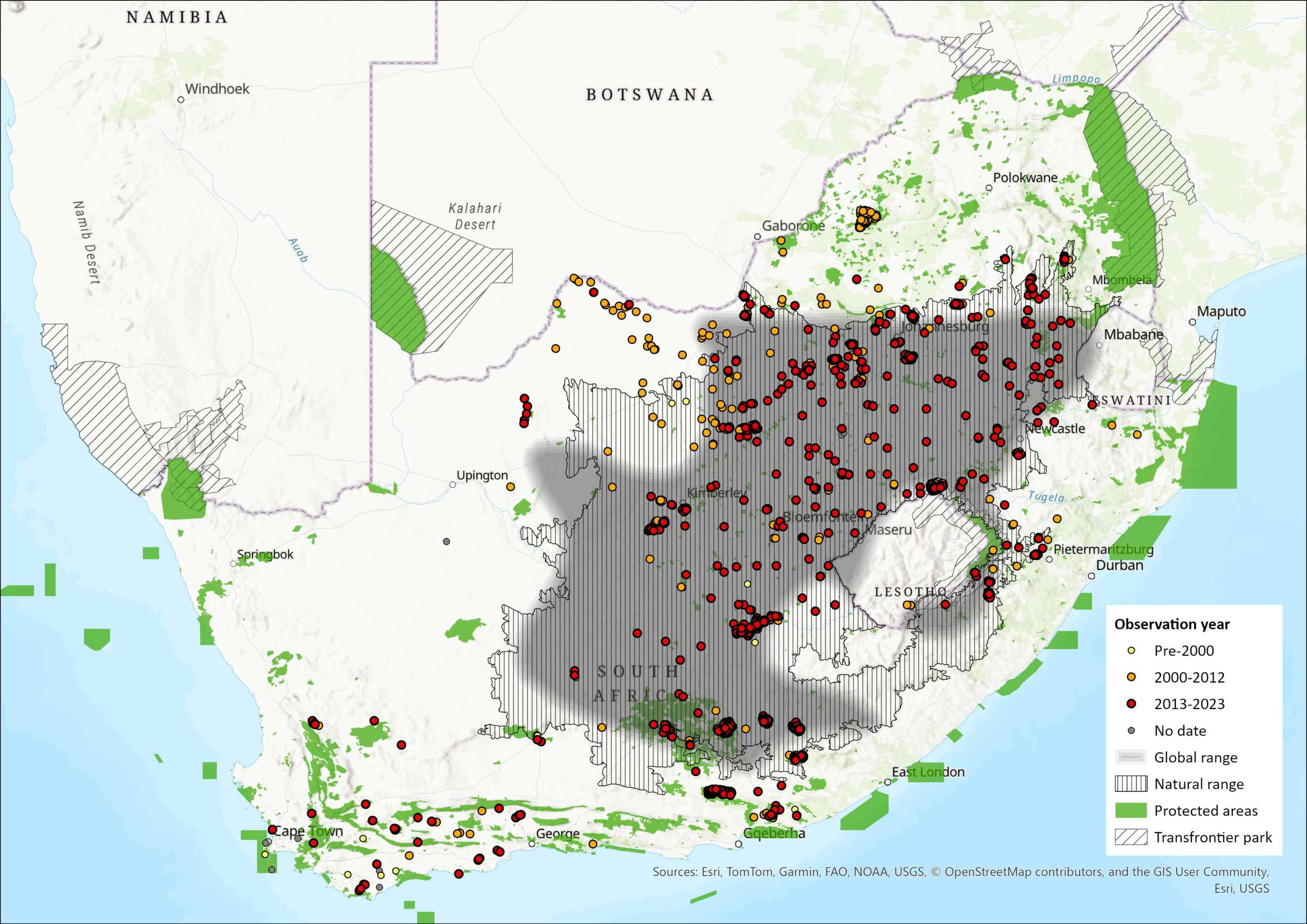
Geographic Range
The Black Wildebeest occurs in South Africa, Eswatini, and Lesotho. Essentially, the species was found in the Grassland and Karoo regions of the central and Northern Cape, the whole of the Free State and the southern highveld regions of the former Transvaal (von Richter 1971a, 1974a). It was also recorded in western Lesotho (where they had become locally extinct through over-hunting) and the Grassland areas of western Eswatini) (Lynch 1994, Monadjem 1998) and have subsequently been reintroduced to both countries (Skinner and Chimimba 2005). In KZN, there are reports of Black Wildebeest having occurred in the open Grassland areas below the Drakensberg range. This species attracted much attention from most early explorers in South Africa. Vivid descriptions of vast herds with animals performing curious prancing movements can be read in the diaries of these travellers. This peculiar behaviour resulted in Black Wildebeest often being called the “clowns of the veld”. The strange appearance of the animal was apparently a cause of embarrassment for early naturalists who were confused with the classification of an animal which has “the mane and tail of the horse; the form of the head and the horns resemble the ox; and in the legs and delicate make of the body it appears of the antelope species” (Lichtenstein 1930).
By the end of the 19th century, excessive hunting had reduced the formerly vast population to a few individuals surviving on two farms in the Free State Province of South Africa (Skinner and Chimimba 2005). Trade in game skins had become a flourishing business and it was reported that a single farm in Kroonstad exported 157,000 Black Wildebeest and Blesbok (Damaliscus pygargus phillipsi) skins in 1866 alone (Garson-Steyn and Garson-Steyn 1964). Since then, protection by farmers and conservation agencies has allowed the species to recover (East 1999, Vrahimis 2013). This achievement is a rare conservation success in Africa and can be attributed to a few conservation-minded farmers. It has now been reintroduced to parts of its former range (western Eswatini and western Lesotho) and introduced into farmland areas outside of its natural range, including Namibia (East 1999). Recently, Black Wildebeest have also been introduced to private farms in Botswana (Grobler et al. 2011). Within the assessment region, they have been introduced far outside of the natural range into both Limpopo (however, management agencies on protected areas are removing such extra-limital subpopulations) and Western Cape provinces. One of the subpopulations in the latter province, located on Grootte Schuur Estate, is generally considered to be a “pure” subpopulation.
In the past, the Blue and Black Wildebeest ranges barely overlapped (Estes and East 2009), although this may have been different in the Pleistocene (Brink et al. 1999). One of the known wildebeest range overlap areas was in the vicinity of the confluence of the Vaal and Orange rivers where thornveld and Karoo veld types converged. There are, however, indications that Blue and Black Wildebeest herds never utilised the same area in this region at the same time. Arguably the best example of a separation between the two wildebeest species was reported for the south-eastern Mpumalanga region in the vicinity of Amsterdam during the late 1800s. In this area Blue Wildebeest and Black Wildebeest were reportedly separated seasonally and apparently also never occurred simultaneously in that specific area (Forbes Diaries, National Archives).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): NA
Depth Upper Limit (in metres below sea level): NA
Depth Zone: NA
Biogeographic Realms
Biogeographic Realm: Afrotropical
Map
Figure 1. Distribution records for Black Wildebeest (Connochaetes gnou) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Botswana | Extant | Introduced | – | – |
| Eswatini | Extant | Reintroduced | – | – |
| Lesotho | Extant | Reintroduced | – | – |
| Namibia | Extant | Introduced | – | – |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: NA
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Black Wildebeest are found within open grasslands of the central plateau of the region. As such, the main threat that climate change may pose to them is via impacts on these grasslands. Impacts may include changes in forage quality (Morris et al. 2022) and quantity, increasing temperatures (Hulme et al. 2001), and changes in rainfall patterns (Hulme et al. 2001, Dore 2005, Ayanlade et al. 2018). Increasing temperature and CO2 stimulates plant production. However, in the sourveld grasslands associated with poor-quality soils in high-rainfall areas, forage quality may decline as the poor-quality soil may not be able to support increased plant productivity (Morris et al. 2022). As such, overall food quality in some protected areas my decline to a point where they are unable to maintain large numbers of Black Wildebeest. By contrast, high-quality sweetveld grasslands found on nutrient rich soils experiencing low rainfall will likely remain high in response to the increasing CO2 and the warmer conditions associated with climate change (Morris et al. 2022). Nevertheless, climate driven changes in rainfall patterns may impact the quality and distribution of high-quality grasses. In response, Black Wildebeest may experience a reduction in food quality in some of the protected areas where they are currently found. In addition, changes in temperature and rainfall patterns may increase the likelihood of extreme droughts (Ayanlade et al. 2018) that could negatively affect food resources for Black Wildebeest across their range.
Population information
The last population estimate for the species prior to this assessment was 16,260 (9,765-11,382 mature individuals) (Vrahiminis 2017). Black Wildebeest sex and ratio data for two Free State protected areas over a period of two years revealed average mature subpopulations of 66% and 68% (E. Schulze unpubl. data). Within the assessment region, there is a minimum estimated of 15,655 individuals (counts conducted between 2012 and 2021) on formally and privately protected areas across the Free State, Gauteng, North West, Northern Cape, Eastern Cape, and Mpumalanga provinces. Black Wildebeest are also found in KwaZulu-Natal, but population estimates were not available for this assessment. This yields a minimum mature population size of 9,393-10,959 (using a 60-70% mature population structure). This is likely to be an underestimate as not all private sector data are available.
In the North West Province, the subpopulations are thriving to the extent that hunting alone is no longer an effective control measure and large–scale translocation and culling are required for effective population management (Power 2014). The provincial population is stable or increasing and face no severe threats (Nel 2015). There are an estimated additional 3,980 Black Wildebeest on private game farms in the province.
Overall, the population size is increasing. For example, the mean annual population growth rate from 2012 to 2018 across the 12 sites in provincial nature reserves in the Free State Province was 8.1% (i.e., 2,228 to 4,960), while the subpopulation in Golden Gate National Park showed an annual decline of 5.4% from 2016 to 2020. In the North West Province, the average annual growth rate for the seven provincial protected areas was 21.1% (i.e., 2,764 to 4,845 individuals) from 2012 to 2021. In the Eastern Cape the subpopulation in Mountain Zebra National Park has remained relatively stable with an annual growth of 0.5% from 2012 to 2023 (922 to 973 individuals). In KZN, the subpopulations in Chelmsford, Coleford, Impendle, Midmar, Ntsikeni, and Wagendrift Nature Reserves are all increasing or stable. Generation length for this species has been calculated as 7.8 years (Pacifici et al. 2013). Over the period from 2012 to 2021 (the period with the most reliable data), there was an estimated population increase of 55% (3,686 to 5,707 individuals) using a sample of eight protected areas from the Eastern Cape and North West provinces for which long-term data were available.
Population Information
Continuing decline in mature individuals? No
Current population trend: Increasing
Number of mature individuals in population: 9,393-10,959
Extreme fluctuations in the number of subpopulations: No
Continuing decline in number of subpopulations: No
All individuals in one subpopulation: No
Severely fragmented: Yes. All subpopulations exist in fenced protected areas or ranches.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Low
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Low
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
There is no evidence of population genetic structure within the geographical range of this species. Consequently, all individuals are part of a single metapopulation. Whether there was any historical genetic structure cannot be known, as the species was hunted to the point of extinction, leaving only a few small groups remaining on a few farms in a small part of its original range (in the Free State). These remaining animals acted as the founding population for all animals today. To minimise the further loss of genetic diversity within the species, it is critical that all sites with Black Wildebeest apply a metapopulation approach and move animals between farms and reserves to try to resemble natural gene flow between fenced populations of the species.
While natural movement of individuals between sites is limited by fences, the constant trade in live animals of this species ensures gene flow is maintained. Considering this and that the overall population is relatively large with the number of viable, growing and stable populations across the species range increasing, the genetic health of the species is assumed to be stable. The current census size for the species in the assessment region is estimated at 9,393-10,959 individuals, which could suggest an Ne between 970 and 11,00 (based on a conversion ratio of 0.1-0.3). However, the actual estimates might vary given that the animals are part of a greater metapopulation outside of the assessment region (possibly increasing Ne), while the possible effects of inbreeding due to the previous bottleneck might still be apparent (possibly decreasing Ne).
A key risk to Black Wildebeest is hybridisation with Blue Wildebeest. Within the North West province, some private game farms stock Blue and Black Wildebeest together, thereby presenting the risk of hybridisation (Power 2014). Similarly, in the Eastern Cape Province, the conservation value of both Oviston and Mpofu Nature Reserves has been undermined by exposure to C. taurinus, but 718 individuals from Oviston Nature Reserve have since been kept separate from the rest of the population. This highlights the need to keep Black and Blue Wildebeest separate within the natural range of Black Wildebeest and to ensure the conservation value of formally protected areas. From a genetic perspective, the ex-situ population of Black Wildebeest in European zoos may potentially be important, since some of these animals are descendent from animals exported in the 1950s and 1960s, and are thus representative of the genetic make-up of the species before large-scale translocations started in southern Africa. For example, populations generally considered pure based on management history include Benfontein Game Farm (Northern Cape), SA Lombard Nature Reserve (North West), Suikerbosrand Nature Reserve (Gauteng), possibly Geluk Farm (Free State), and extra-limitally in Grootte Schuur Estate (Western Cape). This yields at least 1,347 individuals in total (808–943 mature). Closer, uniform monitoring of the genetic integrity of animals in all formally protected areas is in progress and the number of pure populations may rise. This species must be reassessed as soon as more data are available.
Habitats and ecology
This species is a selective short grass grazer and inhabits the open plains grasslands and karoo shrublands of South Africa and Lesotho (von Richter 1971b, 1974b; Codron and Brink 2007; Codron et al. 2011). The high central plateau grasslands are characterised by flat to rolling plains, and mountainous areas with altitudes ranging from 1,350-2,150 m asl (Vrahimis 2013). Open habitats are essential for the reproductive behaviour of the Black Wildebeest because territorial males require an unobstructed view of their territories in order to breed. The specialised territorial breeding behaviour of the Black Wildebeest is the reason why the Black Wildebeest is historically confined to the Highveld and Karoo areas and why it is reproductively isolated from the sympatric Blue Wildebeest. Ecological separation between the two species is incomplete although habitat heterogeneity is a key factor keeping the two wildebeest species separated (Brink 2005, 2016; Helm 2006). Given the functional meaning of Black Wildebeest horn and cranial shape (Brink 1993, 2016), the evolutionary process appears to have been linked to, or possibly caused by, a shift in mating behaviour towards permanent territoriality in males, which contrasts to Blue Wildebeest that have both territorial and non-territorial mating systems. The Black Wildebeest can be distinguished from the Blue Wildebeest by its white rather than black tail. The alternative name of these two species, “gnu”, comes from the male’s characteristic nasal call, described as “ge-nu”.
Ecosystem and cultural services: The Black Wildebeest is a feisty, gregarious species that often occurs in high densities with other selective short grass grazers such as Springbok (Antidorcas marsupialis) and Blesbok (von Richter 1974a). Unfortunately, concentrations of these species often lead to grassland degradation and the establishment of largely homogeneous grazing lawns in higher rainfall areas and areas with a poor basal cover in lower rainfall areas. Due to this tendency, many game farmers prefer to rather keep Blue Wildebeest than Black Wildebeest in order to prevent veld deterioration. Despite its reputation as a habitat degrader, the Black Wildebeest is still considered a flagship species of the central grasslands, mainly due to their unique, spirited behaviour, and endemic status.
A prancing Black Wildebeest appears on the South African five Rand coin and the animal has in the past been displayed on South African postage stamps (von Richter 1974a).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.4. Shrubland -> Shrubland – Temperate | – | Suitable | – |
| 4.4. Grassland -> Grassland – Temperate | – | Suitable | – |
Life History
Generation Length: 7.8 years
Age at Maturity: Female or unspecified: 16 months
Age at Maturity: Male: 4 years
Size at Maturity (in cms): Female: Shoulder height: 115.7 cms
Size at Maturity (in cms): Male: Shoulder height: 120.6 cms
Longevity: 19 years 7 months
Average Reproductive Age: Males: 4 years; Females: 16 months
Maximum Size (in cms): Shoulder height: 120.6 cms
Size at Birth: 14 kg
Gestation Time: 8.5 months
Reproductive Periodicity: Annual
Average Annual Fecundity or Litter Size: single calf
Natural Mortality: Disease
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Historically, it is likely that Black Wildebeest moved extensively in search of high-quality food. However, due to extensive fencing, these movements are restricted to within protected areas.
Congregatory: Gregarious with lone territorial males, female herds (mean 28.4 individuals), and bachelor herds (mean 20.6 individuals) (Skinner and Chimimba 2005).
Systems
System: Terrestrial
General Use and Trade Information
The level of trade is domestic, commercial, and international. The trade in live animals at game auctions, trophy hunting, and culling for the venison market are the main uses of the species for economic gain. The numbers taken by trophy hunts are unlikely to have a detrimental effect on the population. Presently, with the large-scale increase in the number of game ranches being developed throughout South Africa, landowners are keeping a wider range of species on their properties, primarily to cater for local and overseas hunters. This has resulted in an increasing number of farms keeping both wildebeest species together. Wildlife ranching may also lead to bush encroachment, which may facilitate hybridisation as Black Wildebeest prefer open plains (Brink 2005). This has led to several confirmed cases of hybridisation in some provinces, and the genetic integrity of especially the Black Wildebeest is being threatened by this activity.
While the extensive distribution that the Blue Wildebeest has in Africa means that this species is not similarly threatened, the genetic integrity of the South African population is at risk, a threat that could seriously impact on the credibility of the local hunting industry. It goes without saying that this would also have serious economic implications for the value of both these species. In order to remedy this situation, it is of utmost importance to impose stricter translocation regulations, on a national level, to address this serious problem.
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
No |
– |
Yes |
Extensive use in trophy hunting, live animal sales and venison. Hides provide good quality leather. |
National Commercial Value: Yes
International Commercial Value: Yes
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
1. Food – human |
– |
true |
true |
– |
|
15. Sport hunting/specimen collecting |
– |
true |
true |
– |
|
16. Establishing ex-situ production * |
– |
– |
– |
– |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Mostly live animal sales and culling for the venison market. Majority of harvesting is from extensive wildlife ranches.
Threats
Historically, the main threat to this species was hunting pressure, habitat loss, and periodic outbreak of disease (Vrahimis 2013). However, now that the species has recovered and numbers are increasing, the only significant threats are hybridisation with the Blue Wildebeest, which can occur when the two species are mixed unnaturally on fenced land (East 1999, Grobler et al. 2011, Grobler et al. 2018); and loss of genetic diversity from existing in isolated fenced areas, leading to isolation from the wider gene pool of the species. The effects of fragmentation and isolation may be increased by small founder population size. Both threats require a Biodiversity Management Plan and the formulation of a metapopulation strategy.
Black and Blue Wildebeest are two distinct species and do not interbreed naturally because they occupy different habitats (for example, Black Wildebeest cannot reproduce in habitats with extensive tree cover), although there may have historically been hybrid zones with flux in movement patterns. Fossil evidence and historical records suggest that the two species have a long history of sympatric occurrence in central southern Africa (Brink et al. 1999, Skead 2011). It is likely that reproductive isolation would have been in effect following the speciation of Black Wildebeest and that it remained intact until extensive habitat loss in the central interior of southern Africa (Brink 2005). This started in the early 19th century, but the skeletal evidence for hybridisation is seen only very recently, since the 1990s. This may be linked to increased levels of habitat loss and disturbance as these two species have been forced into sustained sympatry which may have led to artificial inflation of hybridisation frequency. The occurrence of Black x Blue Wildebeest hybrids (cross-breeds) was first reported in KZN in the early sixties. First generation hybrids are easily identified, but the offspring of hybrids that have interbred with pure Black Wildebeest are difficult to recognise on appearance alone. Hybrids are fertile (Fabricius et al. 1988, Ackermann et al. 2010). In several cases it appears that a disruption of the normal demographic/social structure has been involved as well. An example of this was reported for Spioenkop Nature Reserve (Langley 1995). A morphometric analysis of skeletal materials housed in the Florisbad Quaternary Research Department, which were collected from Free State provincial nature reserves over the last decade, revealed a high incidence of skull and tarsus abnormalities which might be linked to historical incidences of hybridisation (J. Brink unpubl. data). Further data is required to corroborate the extent and severity of hybridisation in formally protected areas. The threat of hybridisation, however, can be managed. As long as hybridisation is not allowed to occur on national or provincial protected areas, and the establishment of private protected areas continues to counter the loss of habitat through agriculture and mining, this species should continue to prosper.
Habitat trend: Declining in area and quality, mainly due to expanding agricultural activities, a rapid increase in opencast mining activities and bush encroachment. In the Free State, loss of natural vegetation during the period 1994–2009 amounted to 6% (7,871 km2) of the surface area, while the period 2000–2009 experienced a 2.4% loss (N. Collins, unpubl. data), where the greatest loss occurred in the Grassland biome (6.4%). It remains to be evaluated whether the expansion of wildlife ranching is mitigating the net loss of pristine habitat. Climate change is also projected to be a threat as it may influence the extent and quality of grasslands across the Black Wildebeest range. Black Wildebeest are not particularly sensitive to drought as their natural range also includes the Karoo and many semi-arid areas, which indicates a high tolerance to drier conditions. The same applies to the quality of grasslands as they can survive in degraded grassland.
Conservation
In 2008, about 20% of the population occurred in protected areas and around 80% occurred on private farmland and conservancies. The current situation is unknown and should be reassessed through a population survey. Formally protected areas demonstrate the potential for rapid population growth of Black Wildebeest. Conservationists should focus on adequately protecting these reserves and sustaining their habitat quality. The most immediate conservation intervention necessary is separating C. taurinus and C. gnou within the natural range of C. gnou. This is especially important in formally protected areas, which should be maintained as source pools of genetically diverse Black Wildebeest.
The deliberate mixing of Blue and Black Wildebeest on any property would be a contravention of the SA National Biodiversity Act (Environmental Management: Biodiversity Act). Furthermore, it would also not be in line with the original IUCN guidelines for the reintroduction of species (IUCN SSC 2013) as it could diminish the already reduced genetic diversity within both species. All provincial nature conservation agencies have taken action to avoid wildebeest hybridisation by attempting to keep the two species separate. The 2008 NEMBA (National Environmental Act) TOPS (Threatened or Protected Species) regulations, in terms of the National Environmental Management: Biodiversity Act, 2004 (Act 10 of 2004), prohibits the translocation of species to an extensive wildlife system where a possibility of transmitting disease or hybridisation exists. In the Free State, legislation exists for private landowners too. The Free State’s Department of Economic, Small Business Development, Tourism and Environmental Affairs (DESTEA) Standard Conditions on Adequate Fencing Permits policy does not allow for the live relocation of any wildebeest from an area where both species are present because of possible hybridisation concerns, unless purity is proven by means of DNA testing. Culling of hybrid subpopulations have already occurred on Maria Moroka Nature Reserve in the Free State, Spioenkop Nature Reserve in KZN and Malolotja Nature Reserve in Eswatini and on some private properties such as Laohu Valley Reserve in the south-western Free State.
Benfontein Game Reserve in the Northern Cape and SA Lombard Nature Reserve in the North-West Province have pure subpopulations of Black Wildebeest (established in 1954 partly to protect Transvaal’s last herds) that can re-stock potentially contaminated populations. Suikerbosrand Nature Reserve in Gauteng is also likely to contain a pure subpopulation. Groote Schuur Estate in the Western Cape also has a pure subpopulation that can be used to re-stock other reserves, but this subpopulation is not counted in this assessment as it is outside the natural distribution range.
Recommendations for land managers and practitioners:
The highest priority with regards to Black Wildebeest conservation is to develop genetic markers for determining hybrid Black Wildebeest populations. This work is well advanced at the University of the Free State and the National Zoological Gardens, but consensus on parameters for purity still need to be agreed on by stakeholders. Until these markers are in general use, translocations from formally protected areas to private reserves and amongst private reserves should be continued (but not from private reserves to formally protected reserves). Standardised genetic testing and monitoring should be encouraged across all provinces and in Namibia. Live removal from areas that previously or currently house both species should be prohibited. Habitat management aimed at the preservation and maintenance of grasslands should be a priority to ensure population growth.
Research priorities:
The determination of the genetic integrity of all subpopulations and the resulting identification of “clean” subpopulations is a priority. Ongoing research aims to achieve this, to establish robust genetic markers and thresholds for purity to detect hybrids. Once this is done and all subpopulations have been tested (by means of a standardised procedure), the replacement of hybrid subpopulations should commence. Therefore, priority research projects should be:
- Continued genetic studies to establish robust genetic markers to detect hybrids and develop a standardised genetic testing procedure.
- Determine the genetic integrity of all Black Wildebeest populations and identify pure subpopulations using the standardised genetic testing procedure.
- Remove all hybrid subpopulations and replace them with stock from pure subpopulations. Also to consider the role of backcrossing in diluting the effects of hybridisation.
- Impacts of the establishment of this species outside it former range should be ascertained.
Encouraged citizen actions:
- Submit photos of Black Wildebeest showing any abnormalities to your local conservation agency.
- Do not stock both Blue and Black Wildebeest on the same property.
Bibliography
Ackermann, R.R., Brink, J.S., Vrahimis, S. and de Klerk B. 2010. Hybrid wildebeest (Artiodactyla: Bovidae) provide further evidence for shared signatures of admixture in mammalian crania. South African Journal of Science 106: 1-4.
Ayanlade, A. Radeny, M., Morton, J.F. and Muchaba, T. 2018. Rainfall variability and drought characteristics in two ago-climatic zones: An assessment of climate change challenges in Africa. Science of the Total Environment 630: 728-737.
Bissett, C., Ferreira, S., Bezuidenhout, H., Smit, I., Daemane, E., Mokoena, V. and Sikhosana, T. 2016. Golden Gate Highlands National Park herbivore off-take recommendations 2016: An integrated approach combining local knowledge with data derived from animal census, herbivore models, vegetation field monitoring and satellite imagery. Scientific Services, South African National Parks.
Brink, J.S. 1993. Postcranial evidence for the evolution of the black wildebeest, Connochaetes gnou: an exploratory study. Palaeontologia Africana 30: 61-69.
Brink, J.S. 2005. The evolution of the black wildebeest (Connochaetes gnou) and modern large mammal faunas of central southern Africa. University of Stellenbosch.
Brink, J.S. 2016. Faunal evidence for mid- and late Quaternary environmental change in southern Africa. In: J. Knight & S.W. Grab (eds), Quaternary environmental change in southern Africa: physical and human dimensions, pp. 286-307. Cambridge University Press, Cambridge, UK.
Brink JS, Berger LR, Churchill SE. 1999. Mammalian fossils from erosional gullies (dongas) in the Doring River drainage, Central Free State Province, South Africa. Pages 79–90 in Becker C, Manhart H, Peters J, Schibler J, editors. Historia Animalium ex Ossibus. Rahden/Westf, Germany.
Codron, D. and Brink, J.S. 2007. Trophic ecology of two savanna grazers, Blue Wildebeest Connochaetes taurinus and black wildebeest Connochaetes gnou. European Journal of Wildlife Research 53: 90-99.
Codron, D., Hull, J., Brink, J.S., Codron, J., Ward, D. and Clauss, M. 2011. Effect of competition on niche dynamics of syntopic grazing ungulates: contrasting the predictions of habitat selection models using stable isotope analysis. Evolutionary Ecology Research 13: 217-235.
Dore, M.H.I. 2005. Climate change and changes in global precipitation patterns: What do we know? Environment International 31: 1167-1181.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
East, R. 1999. African Antelope Database 1999. Gland, Switzerland and Cambridge, UK.
Estes R.D. and East, R. 2009. Status of the wildebeest (Connochaetes taurinus) in the wild 1967-2005. WCS Working Paper 37. Wildlife Conservation Society, New York.
Fabricius, C., Lowry, D. and van den Berg, P. 1988. Fecund black wildebeest x blue wildebeest hybrids. South African Journal of Wildlife Research 18: 35-37.
Garson-Steyn, F. and Garson-Steyn, H. 1964. The Black Wildebeest. African Wildlife 18: 103-104.
Grobler, J.P., Hartl, G.B., Grobler, N., Kotze, A., Botha, K. and Tiedemann, R. 2005. The genetic status of an isolated Black Wildebeest (Connochaetes gnou) population from the Abe Bailey Nature Reserve, South Africa: Microsatellite data on a putative past hybridization with blue wildebeest (C. taurinus). Mammalian Biology 70: 35-45.
Grobler, J.P., Rushworth, I., Brink, J.S., Bloomer, P., Kotze, A., Reilly, B. and Vrahimis, S. 2011. Management of hybridization in an endemic species: decision making in the face of imperfect information in the case of the Black Wildebeest—Connochaetes gnou. European Journal of Wildlife Research 57: 997-1006.
Grobler, P., van Wyk, A.M., Dalton, D.L., Jansen van Vuuren, B. and Kotzé, A. 2018. Assessing introgressive hybridization between blue wildebeest (Connochaetes taurinus) and black wildebeest (Connochaetes gnou) from South Africa. Conservation Genetics 19: 981-993.
Helm, C.V. 2006. Ecological separation of the black and blue wildebeest on Ezemvelo Nature Reserve in the highveld grasslands of South Africa. University of Pretoria.
Hulme, M., Doherty, R., Ngara, T., New, M. and Lister, D. 2001. African climate change: 1900-2100. Climate Research 17: 145-168.
IUCN SSC Antelope Specialist Group. 2008. Connochaetes gnou. The IUCN Red List of Threatened Species 2008: e.T5228A11120065. Available from http://dx.doi.org/10.2305/IUCN.UK.2008.RLTS.T5228A11120065.en.
IUCN. 2017. The IUCN Red List of Threatened Species. Version 2017-2. Available at: www.iucnredlist.org. (Accessed: 14 September 2017).
IUCN/SSC. 2013. Guidelines for Reintroductions and Other Conservation Translocations. Version 1.0. Page viiii + 57 pp. IUCN Species Survival Commission, Gland, Switzerland.
Langley, N. 1995. Black wildebeest x blue wildebeest hybrid at Spioenkop Nature Reserve. Natal Parks Board.
Lichtenstein, H. 1930. Travels in Southern Africa in the years 1803–1806. London 1812–1815. Van Riebeeck Society Numbers 10 and 11.
Lichtenstein H. 1930. Travels in southern Africa in the years 1803, 1804, 1805, 1806. Volumes I and II. Van Riebeeck Society, Cape Town, South Africa.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsing van die Nasionale Museum Bloemfontein 10: 177-241.
Monadjem, A. 1998. Distributional patterns and conservation status of mammals of Swaziland, southern Africa. Koedoe 41: 45-59.
Monadjem, A. 1998. Distributional patterns and conservation status of mammals of Swaziland, southern Africa. Koedoe 41:4559.
Morris, C. D., Kirkman, K.P. and Zacharias, P.J.K. 2022. Will the grass be greener on the other side of climate change? South African Journal of Science 118: 13844.
Nel, P. 2015. Population estimates for large herbivores and predators in protected areas in the North West Parks Board November 2015. North West Parks Board.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Skead, C.J. 2011. Historical incidence of the larger land mammals in the broader Western and Northern Cape provinces.In: Boshoff A.F., Kerley G.I.H, Lloyd P. (ed.), Centre for African Conservation Ecology, Nelson Mandela Metropolitan University, Port Elizabeth.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Von Richter, W. 1971a. Past and present distribution of the black wildebeest, Connochaetes gnou Zimmermann (Artiodactyla: Bovidae) with special reference to the history of some herds in South Africa. Annals of the Transvaal Museum 27: 35-57.
von Richter, W. 1971b. Observations on the biology and ecology of the Black Wildebeest (Connochaetes gnou). South African Journal of Wildlife Research 1: 3-16.
Von Richter, W. 1974a. Connochaetes gnou. Mammalian Species 50: 1-6.
von Richter, W. 1974b. Survey of the adequacy of existing conserved areas in relation to wild animal species. Koedoe 17: 39-69.
Vrahimis, S. 2013. Connochaetes gnou. In: J.S. Kingdon, D. Happold, T.M. Butynskyi, M. Hoffmann, M. Happold & J. Kalina (ed.), The Mammals of Africa. Vol VI. Hippopotamuses, Pigs, Deer, Giraffe and Bovids, Academic Press, Amsterdam, The Netherlands.
Vrahimis, S., Grobler, P., Brink, J., Viljoen, P. & Schulze, E. 2017. Connochaetes gnou. The IUCN Red List of Threatened Species 2017: e.T5228A50184962.