Black-backed Jackal
Lupulella mesomelas

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Lupulella mesomelas – Schreber, 1775
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – CANIDAE – Lupulella- mesomelas
Common Names: Black-backed Jackal, Silver-backed Jackal (English), Rooijakkals, Swartrugjakkals (Afrikaans), Phukubjê (Pedi), Phokobjê (Pedi, Tswana), Phokobje, Phokojoe, Phokojwe (Sotho), Imphungushe, Jakalasi, Mpungutje, Impungutjee (Swazi), Hungudzwa, Jajaja, Mhungubya, Phungubya (Tsonga), Phokobyê, Phokojê, Phokojwê, Sekgêê (Tswana), Phungubwe, Phunguhwe, Phunguhwe i re na Mutana Mutswu (Venda), Impungutye (Xhosa), Ikhanka, Impungushe, Inkanka (Zulu)
Synonyms: achrotes, arenarum, elgonae, mcmillani, schmidti, variegatoides
Taxonomic Note:
Meester et al. (1986) assigned all southern African material to the nominate subspecies, Canis mesomelas mesomelas. However, several authors have now adopted a provisional taxonomy, with the Black-backed Jackal and the Side-striped Jackal (Lupulella adustus) falling outside the clade of the African wolf group, and therefore needing a separate genus, that is Lupulella for the African jackals (Geraads 2011; Dinets 2015; Viranta et al. 2017; Atickem et al. 2018).
|
Red List Status |
|
LC – Least Concern, (IUCN version 3.1) |
Assessment Information
Assessors: Drouilly, M.1, Nicholson, S.K.2 & da Silva, J.M.3
Reviewers: Power, J.4 & Do Linh San, E.5
Contributor: Patel, T.2
Institutions: 1Panthera,2Endangered Wildlife Trust, 3South African National Biodiversity Institute, 4North West Provincial Government, 5Sol Plaatjie University
Previous Assessors & Reviewers: Minnie, L., Avenant, N., Kamler, J.F., Butler, H., Parker, D., Drouilly, M., du Plessis, J. & Do Linh San, E.
Previous Contributors: Relton, C., Page-Nicholson, S., Child, M.F. & Schulze, E.
Assessment Rationale
The Black-backed Jackal is endemic to sub-Saharan Africa and is widespread throughout the region. It is considered a generalist canid with an opportunistic lifestyle and occupies most habitats within the assessment region. Black-backed Jackals are the dominant predators of livestock in the assessment region and are thus widely persecuted in an attempt to reduce population size and associated livestock losses. However, despite centuries of population reduction efforts, Black-backed Jackals still persist, and even thrive, on farmlands and in most conservation areas. Therefore, these population control efforts appear ineffective (Nattrass et al. 2020) but may result in temporary and localised reductions in population size. Although no accurate population estimates are available, we estimate that there are more than 10,000 individuals in the assessment region, and anecdotal evidence suggests that Black-backed Jackal population size and distribution may have increased over the last 15 years (see for example Drouilly et al. 2019c). Therefore, due to this resilience, a lack of range-wide decline, irrespective of concerted control efforts, we infer that this species will persist, and list it as Least Concern.
Regional population effects: The Black-backed Jackal’s range within the assessment region is continuous with the rest of its southern African range and we suspect that dispersal across geo-political boundaries occurs. Black-backed Jackals appear to have few dispersal barriers (Ferguson et al. 1983; Minnie 2016; Tensen et al. 2019) and may disperse over long distances, exceeding 100 km (Bothma 1971; Ferguson et al. 1983; Humphries et al. 2016; Minnie 2016; Nattrass et al. 2020). Thus, there is no reason to believe that trans-regional and trans-boundary movements will decrease in the future.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Drouilly M, Nicholson, SK & da Silva JM. 2025. A conservation assessment of Lupulella mesomelas. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Distribution
Geographic Range
The Black-backed Jackal is endemic to sub-Saharan Africa (Loveridge & Nel 2004). It occurs in two geographically isolated disjunct populations: one in East Africa and another in southern Africa (Coe & Skinner 1993). The two populations are separated by the Mozambican Gap (from the Zambezi River to Tanzania; Kingdon 1977). According to Ansell (1960), Black-backed Jackals are absent from much of equatorial Africa. This disjunct distributional pattern occurs in other arid-adapted African endemics (e.g. Aardwolf Proteles cristatus and Bat-eared Fox Otocyon megalotis; Allio et al. 2021) and suggests that these two populations were once connected during the drier conditions of the Pleistocene (Loveridge & Nel 2004, 2013; Skinner & Chimimba 2005). The northern subspecies, C. m. schmidti, occupies southern Ethiopia, southern Sudan, Somalia, Kenya, Uganda, and northern Tanzania. The range of the southern subspecies, C. m. mesomelas, extends from the southern part of South Africa to the southwestern part of Angola, northern Botswana, central Zimbabwe, and southwestern Mozambique (Hoffmann 2014). Recent molecular research suggests that these two subspecies may in fact warrant species status owing to divergence in mitochondrial lineages, but genome-wide data with adequate geographical sampling is required in support of this hypothesis (Atickem et al. 2018). However, if this hypothesis is supported and the current and “new” species exhibit reduced distributional ranges and lower population densities, it may require a revision of their conservation status (Atickem et al. 2017).
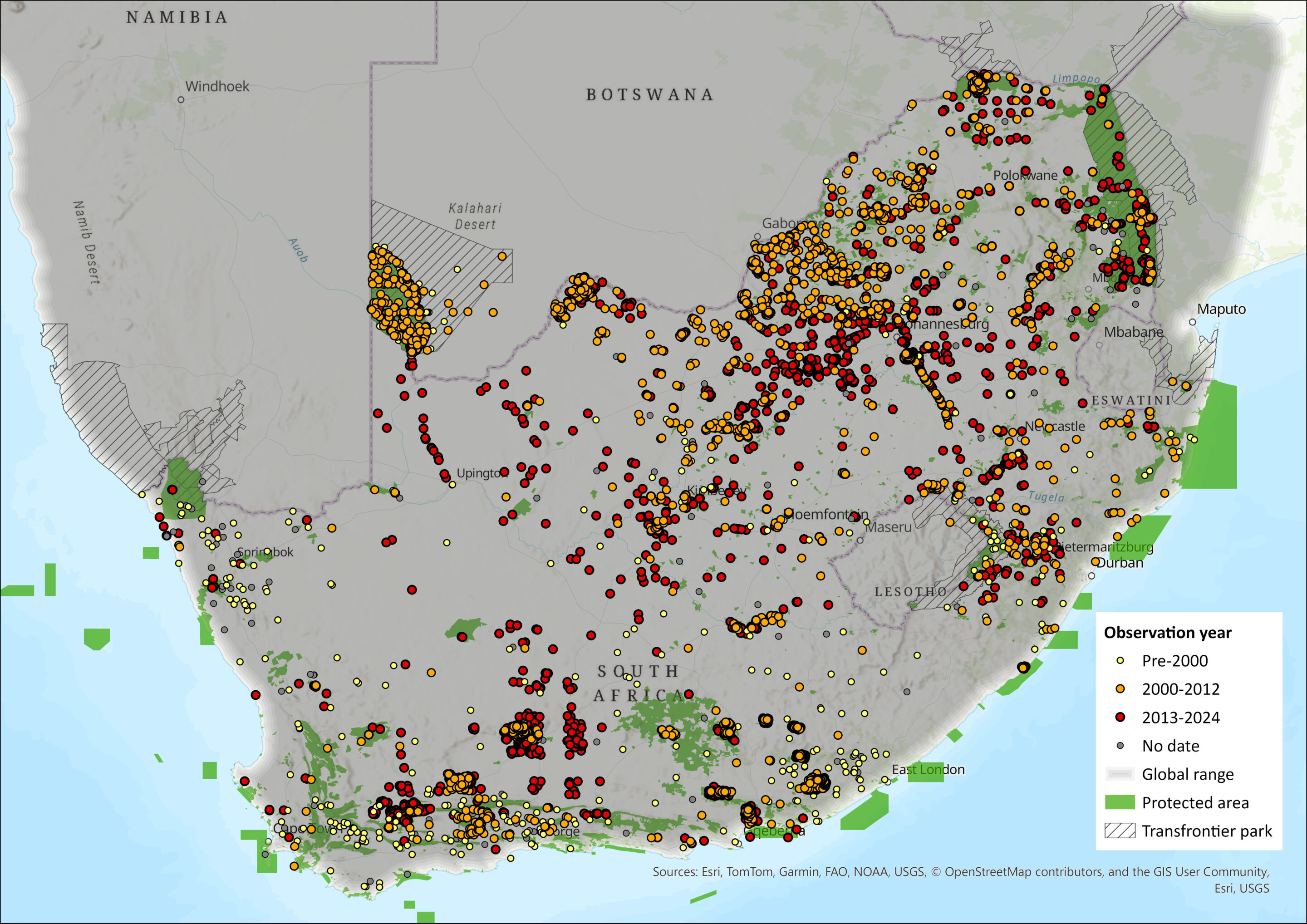
In the assessment region, Black-backed Jackals historically occurred throughout Eswatini, in several locations in Lesotho, and throughout South Africa – except for the Southern Coast Forests around Knysna, Western Cape Province (Skinner & Chimimba 2005), and the Highveld of the former Transvaal Province until the early 1950s (van der Merwe 1953). They are regionally absent and rare from north-eastern KwaZulu-Natal, as is evidenced from none recorded in the Phinda area (Side-striped Jackal were recorded there) in an intensive camera trap study (see Da Rosa 2019), and only 3 of 7 Zululand reserves recording the species, with notable absences from the eastern coastal areas (see Pretorius 2019). It seems over time there has been a decline in reporting from this north-eastern part of KwaZulu-Natal, South Africa (see Map, supporting these mentioned studies).
They have been known to occur throughout most of Lesotho (Lynch 1994), Eswatini and South Africa (see Map). They are widespread across most conservation and livestock farming areas (Loveridge & Nel 2004). The previous assessment indicated that they did not occur along the coast and the immediate interior between Storms River and Port Elizabeth. However, based on predator–livestock conflict reports from small-stock farmers (Minnie 2009; DEDEA problem animal control register 2011 – G. Ferreira pers. comm. 2012), it seems that the population is now expanding into this area (also see map). They appear to be absent from highly developed cities, towns and settlements. However, vocalisations have been recorded in residential areas in Midrand, Gauteng Province (Z.J.K. Madikiza pers. comm. 2014), so they may persist in the peripheries of human settled areas. Similarly, other generalist canids (e.g. Red Foxes Vulpes vulpes: Soulsbury et al. 2007; Coyotes Canis latrans: Gese & Bekoff 2004) persist successfully in urban areas (i.e. they are urban exploiters). This could also become the case for Black-backed Jackals as, similar to several other canids, they are dietary generalists with a high reproductive output and are behaviourally flexible. These attributes may assist them in becoming successful urban exploiters, even if less extensively as others elsewhere.
Black-backed Jackals experience local fluctuations in density with the possibility of local extirpations based on the intensity of predator control programmes (Beinart 1998; Nattrass et al. 2020a). Their local densities may also fluctuate according to the presence of other sympatric predators such as Caracal (Caracal caracal; Ferreira 1988) and Lynx (Felis caracal; Pringle & Pringle 1979), but Black-backed Jackal occupancy was shown to not be affected by Caracal occupancy in the Karoo, but hypothetically more by the presence of apex predators (Woodgate et al. 2023). This may result in fluctuations of their distributional range (Nattrass et al. 2020b), which is highlighted by the expansion of the population into previously extirpated areas such as the Baviaanskloof Mega-Reserve, the mountains east of Cradock, and the areas south of the N2 between Humansdorp and Cape St Francis in the Eastern Cape Province.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 3660
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Black-backed Jackal, (Lupulella mesomelas) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
|
Country |
Presence |
Origin |
Formerly Bred |
Seasonality |
|
Angola |
Extant |
Native |
– |
– |
|
Botswana |
Extant |
Native |
– |
– |
|
Djibouti |
Extant |
Native |
– |
– |
|
Eritrea |
Extant |
Native |
– |
– |
|
Eswatini |
Extant |
Native |
– |
– |
|
Ethiopia |
Extant |
Native |
– |
– |
|
Kenya |
Extant |
Native |
– |
– |
|
Lesotho |
Extant |
Native |
– |
– |
|
Mozambique |
Extant |
Native |
– |
– |
|
Namibia |
Extant |
Native |
– |
– |
|
Somalia |
Extant |
Native |
– |
– |
|
South Africa |
Extant |
Native |
– |
– |
|
Sudan |
Extant |
Native |
– |
– |
|
Tanzania, United Republic of |
Extant |
Native |
– |
– |
|
Uganda |
Extant |
Native |
– |
– |
|
Zimbabwe |
Extant |
Native |
– |
– |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Fuller et al. (2021) conducted a review to try and understand how drylands mammals will respond to climate change. They showed that mammals inhabiting arid regions are confronting not only escalating heat stress but also decreasing water and food resources due to climate change. Insufficient water resources prevent evaporative cooling, resulting in heightened core body temperatures during hot periods, while lack of food decreases the capacity to regulate core temperature during cold nights. The combination of limited food and water will narrow the temperature range within which core body temperature is maintained relatively constant, thereby escalating the risk of physiological dysfunction and mortality. Adaptations in behaviour, such as adjusting activity patterns between day and night or seeking out thermally stable microclimates, may enable individuals to remain within a suitable temperature range for their body to function normally, but may incur costs such as decreased foraging efficiency, heightened competition or increased risks of conflicts with people and their activities, ultimately affecting fitness. Black-backed Jackals are mostly living in drylands and depend, like some of their prey, on waterholes maintained by farmers and reserve managers (especially lactating females; M. Drouilly, pers. obs. 2013-2015). With rising temperatures, vast landscapes currently occupied by Black-backed Jackals might be abandoned by struggling farmers, having a direct effect on water availability and food resources for Black-backed Jackals and their prey. However, to comprehensively evaluate how dryland mammals such as Black-backed Jackals will cope with rising heat stress amidst water and food shortages, it is imperative to gain a deeper understanding of the sensitivities and reactions of these mammals to both the direct and indirect impacts of climate change, through more research. Conversely, because they are arid adapted, they may expand into wetter areas and thrive there too.
Population
Several authors in southern Africa have estimated local population size. For example, in the Drakensberg Mountains, KwaZulu-Natal Province, Rowe-Rowe (1982) estimated densities of 0.34–0.40 individual / km². In the Free State and Northern Cape provinces, estimated densities were 0.02 individual / km² on game farms that actively managed Black-backed Jackals compared to 0.33 to 0.43 individual / km² on game farms that did not (Klare et al. 2010; Kamler et al. 2013). Furthermore, on the game farms that did not actively manage jackals, jackal density varied depending on prey diversity and abundance (Klare et al. 2010). Thus, Black-backed Jackal densities likely vary depending on the dispersion and abundance of resources, as well as the intensity and frequency of predator management. Local population density may also be influenced by the density and composition of the carnivore community (e.g. African Lion Panthera leo, Leopard Panthera pardus, Spotted Hyaena Crocuta crocuta, and Caracal). Apex predators may facilitate scavenging opportunities for Black-backed Jackals (Van de Ven at el. 2013) resulting in increased local densities but may also increase the risk of predation (i.e. interspecific competition, Kamler et al. 2020) resulting in reduced local densities (Brassine & Parker 2012; Minnie 2016). Consequently, it is expected that Black-backed Jackal population density should vary greatly in the assessment region (Nattrass et al. 2020).
The regional variation in population density relative to local resource abundance (amongst other factors) is exemplified by the Black-backed Jackal populations on the Namibian coast. Nel et al. (2013) compared jackal densities between three areas that differ markedly in resource availability. Densities varied from 0.07 individual / km² along the Skeleton coast (low food resources), to 2.91 individuals / km² at Sandwich harbour (intermediate), reaching a maximum of 13.05 individuals / km² at Cape Cross (high). Due to a localised increase in resource abundance in the latter area, jackals were non-territorial with widely overlapping home ranges.
To date, no accurate population estimates for the Black-backed Jackal within the assessment region exist, and extrapolating local population densities to the entire assessment region is inappropriate and may result in extremely inaccurate population estimations. However, owing to the minimum densities mentioned above and the wide distribution of this species in the assessment region, and a tentative extrapolation, there are likely well over 10,000 mature individuals in the population. Anecdotal evidence suggests that numbers have increased over the last 15 years in some areas (Avenant & du Plessis 2008; du Plessis 2013; Drouilly et al. 2019c). Thus, owing to this species’ wide distributional range and persistence in the face of persecution, it is expected that the population will not decline in the future. At finer scales, individuals from neighbouring areas will re-establish territories in the vacant areas (i.e. compensatory immigration; Minnie et al. 2016). However, lethal control in combination with other management practices may result in local extirpations (Walton & Joly 2003; Blaum et al. 2009; Kamler et al. 2013), for most areas of the species range, there is likely more chance of recolonisations happening (Natrass etc one).
Population Information
Current population trend: Stable, based on extent of occurrence, wide habitat tolerance and persistence despite concerted population reduction efforts.
Continuing decline in mature individuals? No
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: It is not currently possible to determine the extent or number of subpopulations. Black-backed Jackal distribution is regarded to be continuous in southern Africa.
All individuals in one subpopulation: Unknown
Number of mature individuals in population: The exact number is unknown but is expected to be more than 10,000 mature individuals.
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Severely fragmented: No. Black-backed Jackals have a broad habitat tolerance and can exist in agricultural and rural landscapes.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Few population genetic studies have been conducted on Black-backed Jackals, and those that have were conducted in South Africa, were done with samples originating primarily from the Western Cape (Tensen et al. 2018, Tensen 2019) and the Northern Cape province (Slater et al. 2022). Tensen et al. (2018, 2019) focused their study on Black-backed Jackals in the Central Karoo of the Western Cape (n=25), although they had samples collected from a broader geographic region (see Tensen et al. 2019: Limpopo, n=3; Mpumalanga, n=7; North West, n=6; Northern Cape, n=14; Eastern Cape, n=24). Slater et al. (2022) focused their study on Black-backed Jackals on small-livestock farms in the Karoo.
The study on the genetic structure and diversity of the lethally managed population of Black-backed Jackals in the Central Karoo (Tensen et al. 2018) used mitochondrial markers to assess dispersal across the landscape, while nuclear microsatellite markers were used to investigate genetic diversity and relatedness among individuals. Black-backed Jackals in the region were found to comprise two genetic clusters, but the weak differentiation between the clusters (FST=0.08) and the high level of inbreeding (FIS=0.112) in the Central Karoo cluster suggests that they likely represent endemic populations. The study also found high levels of genetic diversity, which may result from compensatory dispersal as a result of high population turnover consequent of intensive lethal management in the region (Drouilly et al. 2023). Relatedness was low and did not differ among males and females, indicating no sex-biased dispersal (Tensen et al. 2018), which may be the result of lethal management of the population interfering with demographic structure and social stability in the population. Using mitochondrial markers across a broader scale that included several samples from a broader geographic region of South Africa (Limpopo, n=3; Mpumalanga, n=7; North West, n=6; Northern Cape, n=14; Eastern Cape, n=24), Tensen et al. (2019) found that individuals from the south, central and northern parts of South Africa show no genetic distinction, despite a geographic distance of approximately 1500 km. Previous studies found population structure for jackals on a local scale (James, Scott & Yarnell, 2017; Minnie et al. 2018b; Tensen et al. 2018), which may point to a hierarchical population structure based on stepwise dispersal. Hence, gene flow may be limited between certain regions, while occasional long-distance dispersal maintains the overall metapopulation structure (Minnie et al. 2018). The little genetic structure we observed in Black-backed Jackals is likely explained by a lack of dispersal barriers, and the highly vagile nature and ecological flexibility of jackals (Nattrass et al. 2020b). Similarly, through the use of 13 microsatellites, Slater et al. (2022) showed an absence of genetic differentiation in the jackal population they studied in the Karoo. They found relatedness to be significantly higher within
farms than between farms and documented 18 occurrences of dispersal events where full siblings were detected on different farms (range: 0.78–42.93 km). Distance between identified parent–offspring varied from 0 to 36.49 km and no evidence for sex-biased dispersal was found. Once again, these results suggest that in response to ongoing lethal management, this jackal population is most likely able to maintain genetic diversity through physiological and behavioural compensation mechanisms.
Additional studies on the genetics of Black-backed Jackals in landscapes where they are not lethally managed would be interesting to compare results with lethally managed environments and to obtain estimates of effective population size.
Habitats and ecology
Black-backed Jackals are relatively unspecialised and well-suited for an opportunistic lifestyle in a wide variety of habitats (Loveridge & Nel 2013). They have a wide habitat tolerance, occupying Highveld grasslands (Ferguson et al. 1983; Morwe 2013), montane grasslands (Rowe-Rowe 1982), scrublands (Skinner & Chimimba 2005), savannahs (Wyman 1967; Kingdon 1977; Lamprecht 1978; Moehlman 1983; Fuller et al. 1989; Estes 1991), woodland savannah mosaics (Smithers 1971; Loveridge & Macdonald 2002) and farmlands (Humphries et al. 2016; Drouilly et al. 2019a). The Black-backed Jackal has long been perceived as an arid-adapted species (Loveridge & Nel 2013). However, it also occurs in more mesic areas (mean annual precipitation > 1,000 mm; Rowe-Rowe 1982, 1992; Loveridge & Nel 2013; Humphries et al. 2016) with recent expansions into the more mesic South Coast area (Western Cape), where it was previously absent (see Distribution above). It shows a preference for open habitats but will occupy dense vegetation (Hoffmann 2014). On farmland in the arid Central Karoo, jackals have been shown to favour flat plains at low elevation with vegetation thick enough to provide hiding places (Drouilly et al. 2018a).
Until recently, Black-backed Jackals were considered generalist omnivores (cf. Hayward et al. 2017), consuming whatever food resource is readily available (Loveridge & Macdonald 2003; Kok & Nel 2004; Klare et al. 2010; Fourie et al. 2015; Pohl 2015; Drouilly et al. 2018b) and scavenging when the opportunity arises (Drouilly et al. 2019b). However, they actively hunt small- to medium-sized mammalian prey (Kamler et al. 2010, 2012a; Klare et al. 2010) but also prey on the fawns/lambs of larger ungulates (Klare et al. 2010), including domestic ones (Drouilly et al. 2018b, 2019b). Although their diet is dominated by mammals, they supplement it with fruit, birds, reptiles, amphibians, insects, and fish (Do Linh San et al. 2009; Brassine & Parker 2012; Kamler et al. 2012a; Morwe 2013; van de Ven et al. 2013; Minnie 2016; Drouilly et al. 2018b; Favaretto et al. 2024). The predominance of small- to medium-sized mammals may reflect an optimal foraging strategy, as Black-backed Jackals prefer to prey on mammals with a body mass of 14–26 kg (Hayward et al. 2017). Additionally, they significantly prefer small ungulates (< 30 kg) which hide their young (e.g. Springbok Antidorcas marsupialis and Common Duiker Sylvicapra grimmia; Hayward et al. 2017), but that’s not always the case, especially if non-guarded small livestock is available (Drouilly et al. 2018b). Consequently, they have a wide dietary niche that expands and contracts in relation to the local availability and dispersion of resources (Kaunda & Skinner 2003; Klare et al. 2010; Kamler et al. 2012b; Morwe 2013; Pohl 2015; Drouilly et al. 2018b). Additionally, in the presence of larger carnivores, such as Cheetah (Acinonyx jubatus), Lions, and Leopards, scavenged food sources may contribute substantially to their diet (e.g. Van de Ven et al. 2013; Fourie et al. 2015; Minnie 2016), but incongruences in the literature indicate that this might not be the case throughout the assessment region (e.g. Brassine & Parker 2012; Yarnell et al. 2013; Drouilly et al. 2018b).
In a stable social system, Black-backed Jackals are monogamous (Moehlman 1987), with the dominant mated pair defending a mutually exclusive breeding territory (Loveridge & Nel 2004). Pair formation may increase hunting success (Lamprecht 1978) and is critical for territorial defence and successful pup rearing (Moehlman 1987). Social structure varies and may consist of family groups (1–8 individuals; Rowe-Rowe 1978, 1984) incorporating the dominant pair and their offspring (Ferguson et al. 1983; Loveridge & Macdonald 2001; Kamler et al. 2019), as well as subadults that have delayed dispersal to assist in raising their siblings (i.e. helpers; Moehlman 1979; Rowe-Rowe 1982; Ferguson et al. 1983). Additionally, the territorial pair may tolerate subordinate individuals on the fringe of its territory (i.e. floaters; Ferguson et al. 1983). Territorial relaxation/collapse may occur during periods of high resource abundance (e.g. at seal colonies in Namibia; Jenner et al. 2011; Nel et al. 2013), where the dominant pair allows other individuals into its territory (Macdonald 1979; Rowe-Rowe 1982; Ferguson et al. 1983; Hiscocks & Perrin 1988; McKenzie 1990; Loveridge & Macdonald 2001, 2003).
Black-backed Jackal mating peaks during winter (Skead 1973) and gestation lasts for 60–70 days (Bernard & Stuart 1992; McKenzie 1993; Walton & Joly 2003). Parturition usually occurs from winter to early spring (Bothma 1971; Bernard & Stuart 1992; McKenzie 1993). Importantly, temporal variation in this reproductive pattern may occur in response to resource availability and abundance (Fairall 1968; Rowe-Rowe 1978; Bernard & Stuart 1992; McKenzie 1993; Bingham & Purchase 2002; Walton & Joly 2003). Females usually have one litter per year, with a litter size of 1–9, depending on the female’s body condition (Minnie et al. 2016) and social status (Loveridge & Nel 2013). Additionally, populations experiencing high levels of anthropogenic mortality may produce larger litters relative to populations that are lightly managed or unmanaged (i.e. compensatory reproduction; Minnie et al. 2016).
Pups usually remain in the den from August to November, emerge after 3 weeks, and are weaned at 8–9 weeks of age (Ferguson et al. 1983). They start foraging with their parents at 3 months of age but remain in close proximity (≤ 2 km) to the den until 6 months of age (Ferguson et al. 1983; Moehlman 1987). When they are approximately 7 months old, they start moving further from the den.
They become sexually mature at 11 months but only start reproducing at 2 years of age (Ferguson et al. 1983; Moehlman 1987). However, populations that are lethally managed may compensate for increased mortality and reproduce at younger ages (i.e. compensatory reproduction; Minnie et al. 2016). Young individuals can stay in the natal territory and assist with rearing the next litter (i.e. helpers) or disperse in search of mates and a territory (Ferguson et al. 1983; Moehlman 1987). According to Rowe-Rowe (1992), adults seldom live beyond 7 years.
Home range size varies considerably, with ranges between 4 and 33 km² being reported. Humphries et al (2016) estimated mean resident home ranges (95% FK) across seasons for adult males, adult females and juvenile males; these were 11.4 ± 4.3 km², 5.6 ± 0.36 km² and 2.15 ± 0.45 km², respectively. While in the Kalahari Gemsbok National Park (now Kgalagadi Transfrontier Park in Northern Cape) home ranges averaged between 2.56 and 8.8 km² (Ferguson et al 1983). Kamler et al. (2019) also calculated home range size of Black-backed Jackals on Benfontein Game Farm in the Northern Cape according to their social status and found that mean (±SE) home-range size of alphas (11.0 ± 0.8 km², n = 4) did not significantly differ from that of beta jackals (9.8 ± 0.7 km², n = 8). Variations in home range size may be attributed to variation in food availability, as individuals occupying areas with higher prey density tend to have smaller home ranges (e.g. Ferguson et al. 1983). Further, seasonal variation is apparent and home range size decreases during the whelping season (Loveridge & Macdonald 2001).
Home range size also varies between social classes, with territorial adults generally having smaller home ranges than subadults (e.g. Ferguson et al. 1983). This may be due to subordinate individuals dispersing in search of mates and territories, whereas dominant pairs are resident (Ferguson et al. 1983; Humphries et al. 2016). In general, the home ranges of dominant pairs do not overlap (< 10 %) with other mated pairs (Ferguson et al. 1983; Kamler et al. 2019). However, the home ranges of subordinate individuals may overlap extensively with both subordinate and dominant individuals (Ferguson et al. 1983; Rowe-Rowe 1982).
Ecosystem and cultural services: Black-backed Jackals play a vital role in predator–prey interactions and ecosystem functioning (Tambling et al. 2018). This role may be even more pronounced in areas where large carnivores have been extirpated (e.g. farmlands) leaving Black-backed Jackals (and Caracals) to fulfil the role of apex predators. Consequently, they may regulate populations of smaller carnivores and prey (du Plessis 2013; Bagniewska & Kamler 2014). Given their preference for small- to medium-sized ungulates, they may regulate the populations of species falling within this preferred prey weight range (e.g. Springbok: Klare et al. 2010; Morwe 2013; Blesbok Damaliscus pygargus phillipsi: du Plessis 1972), though this is often under appreciated by landowners owing to commercial interests, though at least it can negate the need to cull these animals by having jackals do it for management.
Black-backed Jackals consume small rodents; thus, it is conceivable that they may regulate these populations. For example, Bagniewska and Kamler (2014) showed that they may suppress prey populations such as Cape Ground Squirrels (Xerus inauris) and hares (Lepus spp.). However, little evidence in support of this prey regulation hypothesis exists (Swanepoel et al. 2017). Thus, the role of this species in regulating crop pests and the spread of diseases by these pests (many of these rodent species act as disease vectors), as well as other herbivores which may compete with livestock for grazing (e.g. Hyrax Procavia capensis: Pohl 2015) is unclear.
Black-backed Jackals may also suppress smaller carnivore populations via lethal (intraguild predation, e.g. Yellow Mongoose Cynictis penicillata, Small-spotted Genet Genetta genetta, and Bat-eared Fox: Klare et al. 2010; Kamler et al. 2012a; Bagniewska & Kamler 2014; Drouilly et al. 2018b) and non-lethal (competition, e.g. Cape Fox Vulpes chama: Kamler et al. 2013) interactions.
Further, anecdotal evidence suggests that the competitive interactions between Black-backed Jackals and Caracals on livestock farms may produce concurrent fluctuations in population densities (du Plessis 2013). When Black-backed Jackal densities are reduced due to human persecution, Caracal densities apparently increase (Pringle & Pringle 1979; Ferreira 1988), but that was not the case when looking at co-occupancy probabilities of both species in the Karoo (Woodgate et al. 2023). In the northeastern part of the assessment area (northern Limpopo, Mpumalanga, and KwaZulu-Natal provinces), Black-backed Jackals occur in sympatry with Side-striped Jackals (Lupulella adustus). There, Black-backed Jackals may aggressively exclude Side-striped Jackals from preferred habitats, as observed in Zimbabwe (Loveridge 1999).
Black-backed Jackals have also been shown to consume fruits and their seeds, and a recent study confirmed that jackals play a role in endozoochory, helping with the dispersion of Grewia occidentalis, Pappea capensis, Diospyros lycioides, and Vachellia karroo. Although those seed viability and germination were significantly lower than uningested seeds, many seeds were able to survive ingestion by jackal and germinate successfully (Favaretto et al. 2024).
Historically in South Africa, the Khoi were the first people to give cultural and aesthetic values to the Black-backed Jackal. They depicted the species in their storytelling as a sympathetic character that was “attractive and entertaining”, able to “consistently manage[s] to outwit the powerful and mighty by exploiting their vulnerabilities” (Wittenberg 2014). Both the Koi and the Afrikaner sheep farmers were socially and politically marginalised, although in different ways, and the jackal became a reflection and symbol of that marginalisation. Today, Karoo farmers’ perceptions of jackals are also consistent with and likely embedded in anthropomorphic characteristics attributed to the species by the hunter-gatherer |Xam (Wittenberg 2014) and in Afrikaans folktales. In both these linguistic traditions, jackals are consistently portrayed negatively and associated with cunningness and dishonesty and with terms such as “sly”, “cunning and crafty” and “deceitful and greedy trickster” (Stewart 2004; Drouilly et al. 2021). Character traits such as vanity, greed, naivety, selfishness, and cruelty are often synonymous with jackals (Stewart 2004). In most cases, the trickster is a small creature that poses no threat to larger animals and often outwits enemies that have a competitive advantage (Stewart 2004). Additionally, the Black-backed Jackal is also seen as an important ancestral spirit manifestation for both isiXhosa and isiZulu people (P. Bernard pers. comm. 2015). Traditional healers have spirit animal guides, and these are shown in a dream to the healer. It is then incumbent on the healer to incorporate the skin of the animal guides into their traditional garb. Black-backed Jackal skins are often incorporated into the headgear (known as isidlokolo) of a traditional healer if the animal is that healer’s animal guide (P. Bernard pers. comm. 2015).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.7. Shrubland -> Shrubland – Subtropical/Tropical High Altitude | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 4.7. Grassland -> Grassland – Subtropical/Tropical High Altitude | – | Suitable | – |
| 8.1. Desert -> Desert – Hot | – | Suitable | – |
| 12.3. Marine Intertidal -> Marine Intertidal – Shingle and/or Pebble Shoreline and/or Beaches | – | Suitable | – |
| 14.1. Artificial/Terrestrial -> Artificial/Terrestrial – Arable Land | – | Suitable | – |
| 14.2. Artificial/Terrestrial -> Artificial/Terrestrial – Pastureland | – | Suitable | – |
Life History
Generation Length: 2.72 years (95% CI: 2.50–3.18) in a lethally-managed population in the Karoo, but probably around 5 years otherwise across the rest of its range
Age at maturity: female or unspecified: Around 11 months
Age at Maturity: Male: Around 11 months
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 60-65 days
Reproductive Periodicity: Females may produce two litters per year, but it depends on the locality.
Average Annual Fecundity or Litter Size: Litter size can be 1-9 but usually 3-6.
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
No significant trade in Black-backed Jackal products exists (Hoffmann 2014). However, infrequent trade has been documented (Williams & Whiting 2016; Nieman et al. 2018), including trophy hunting (L. Minnie pers. obs. 2013) and the sale of skins in the curio trade, but this should have little effect on population persistence. The extent of use of Black-backed Jackal products is unknown, but it has been recorded. A recent study used interviews with traditional healers operating from 17 impoverished, rural communities in the Western Cape Province of South Africa to provide a comprehensive inventory of the number and frequency of animals used and sold. Results showed that Black-backed Jackals were one of the species most used in the traditional medicine trade. Their body parts (i.e., fat, bone, skin, fur, liver, oil) are used to resolve court cases, to reduce prison sentences, or to cure mental illnesses and increase cognitive ability (Nieman et al. 2018). In certain areas, jackal meat is consumed (Avenant 2004; L. Minnie pers. obs. 2013). The impacts of use and trade on Black-backed Jackal populations is presumed to be negligible but large-scale studies are missing. Additionally, several researchers have used or are currently using dead Black-backed Jackals for research purposes (e.g. Bingham & Purchase 2002; Brassine 2011; Minnie 2016). These specimens are collected from farm and reserve managers who killed them, presumably to reduce predation impacts on valued ungulate species and livestock. This retaliatory killing of Black-backed Jackals is probably the largest threat to population persistence.
The species is less charismatic than all the larger carnivores, but they can be aesthetically pleasing, especially during whelping when jackal pups provide endearing photographic opportunities on reserves.
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
Yes |
A source of meat in some areas. Body parts used for cultural purposes and traditional medicine. |
Yes |
Hunted for trophies and skin. |
National Commercial Value: No
International Commercial Value: No
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
3. Medicine – human & veterinary |
true |
– |
– |
– |
Is there harvest from captive/cultivated sources of this species? Unknown
Harvest Trend Comments: Unknown
Threats
Within the assessment region, Black-backed Jackals are hunted, trapped and illegally poisoned for their role as livestock and ungulate predators. For example, in the Central Karoo, interviews with 77 small-livestock farmers showed that 97.4% reported using lethal methods to prevent predation of their livestock by Caracal and Black-Backed Jackal. Half of them also reported using poison either regularly (17%) or as a last resort (33%) for dealing with the most elusive and difficult-to-catch individuals (Drouilly et al. 2023). Persecution occurs in most livestock farming areas and in some game farming areas (Beinart 1998; van Niekerk 2010; du Plessis 2013; Badenhorst 2014; Minnie 2016; Schepers 2016; Pirie et al. 2017; Tensen et al. 2018) and is widespread across the assessment region. Historically, hunting clubs and jackal-proof fencing was subsidised by the government to assist farmers with Black-backed Jackal population control (Stadler 2006; Bergman et al. 2013; Nattrass & Conradie 2015; Carruthers & Nattrass 2018; Nattrass et al. 2020b). These hunting clubs killed a large number of individuals (e.g. Gunter 2008; Bothma 2012; Conradie 2012). For example, Oranjejag killed an average of 786 Black-backed Jackals per year in the Free State Province between 1959 and 1991 (Bothma 2012). Culling subsequently declined after the abolishment of government subsidies, but a recent (2009 onwards) resurgence in culling operations over large areas has occurred (van Niekerk 2010; du Plessis 2013). Full-time damage-causing animal hunters are now commonplace (du Plessis 2013). The intensity of lethal management presumably fluctuates with actual and perceived predation rates, but currently we suspect that it is increasing. In the Karoo, between April 2014 and April 2015, professional hunters and small-livestock farmers killed 299 Black-backed Jackal during five blanket hunts that lasted on average 8.6 nights, which represents an annual persecution rate of 1.9 jackals/100 km², which is not taking into account other individuals killed through routine predator control, such as trapping (Drouilly, unpublished data; Drouilly et al. 2023). In their study on jackal survival on farmland, Kamler et al. (2020) showed that jackals were killed both by humans and apex carnivores and that those mortalities produced similar effects on jackal survival.
While it is still unknown how many Black-backed Jackals are killed annually within the assessment region, population control efforts appear largely ineffective (Nattrass et al. 2020a) and probably only succeed in producing a temporary reduction in local population size (Avenant & du Plessis 2008; Minnie et al. 2016). In fact, despite concerted population reduction efforts for more than 350 years, Black-backed Jackals still persist (Stadler 2006; Bergman et al. 2013; Kerley et al. 2017) and their range and impacts are even reported to be increasing (Drouilly et al. 2019c). This persistence may be ascribed to their highly adaptable nature and their high reproductive potential and dispersal abilities (du Plessis et al. 2015; Minnie et al. 2016). Black-backed Jackal populations that are subject to high levels of anthropogenic mortality may start reproducing at younger ages and produce larger litters (i.e. compensatory reproduction; Minnie et al. 2016). This is attributed to a disruption in the normal, mutually exclusive, territorial structure – due to high levels of lethal management – which allows younger individuals to reproduce (Minnie et al. 2016). Further, the variation in management intensity within the assessment region results in the formation of a source-sink system, which allows for compensatory immigration (Minnie 2016; Tensen et al. 2018, 2019; Slater et al. 2022). Here, compensatory immigration occurs when individuals from unmanaged or lightly managed areas (e.g. conservation areas) disperse into the vacant territories in managed areas (e.g. livestock farms; Minnie 2016; Minnie et al. 2016). The combination of compensatory reproduction and dispersal is likely the reason for their persistence in the face of severe anthropogenic mortality and may continue to negate population reduction efforts.
Black-backed Jackals are also persecuted for their role as diseases vectors (e.g. canine distemper virus, rabies virus and anthrax; Bellan et al. 2012), mostly in the central southern African part of their distributional range (Loveridge & Nel 2004). This thus represents a major cause of mortality within these areas. The efficacy of these management programmes is questionable, and McKenzie (1993) suggests that it may actually promote disease transmission via increased dispersal and agonistic interactions associated with a source–sink system. However, the impact of these diseases on Black-backed Jackal populations within the assessment region is unknown. As Black-backed Jackal ranged widely in search of food resources, this resulted in an expansion of a jackal rabies cycle from North West to Gauteng during the dry period of 2016 (Ngoepe et al. 2022).
Research has shown that Black-backed Jackals in the Central Karoo were also vectors of tick-borne pathogens, although with a lower prevalence than jackal populations in more mesic regions. Pathogens such as Hepatozoon canis, a ubiquitous pathogen of domestic and wild canids globally and Theileria sp. most similar to T. ovis, a piroplasm found in small livestock were found in jackals living on small-livestock farms. No Babesia, Ehrlichia or Anaplasma species were identified, although a Sarcocystis sp. sequence was isolated from one jackal. Host-attached ticks comprised three species, Amblyomma marmoreum, Haemaphysalis elliptica/zumpti and Ixodes rubicundus, commonly known ticks in the region (Viljoen et al. 2021).
Urbanisation and human population growth have resulted in habitat transformation, which has been associated with the decline of several species. Although there are no accurate estimates of Black-backed Jackal population densities in the assessment area, it appears that agricultural habitat transformation (except monocultures) does not pose a serious threat to Black-backed Jackals in the assessment area, as they are still widespread in these areas. Conversely, habitat transformations associated with agricultural expansion may contribute to the success of opportunistic species by providing anthropogenic food subsidies (Oro et al. 2013). Anthropogenic food subsidies such as roadkill, naïve livestock and illegal garbage dumps have been related to artificially inflated population sizes of Golden Jackal (Canis aureus; Yom-Tov et al. 1995) and Coyotes (Fedriani et al. 2001). This may also be the case for Black-backed Jackal populations. Thus, anthropogenic habitat transformation may have improved the extent and quality of habitats available to this species. In addition to feeding on roadkill, they themselves do become roadkill (Collinson et al. 2015), as is evidenced by many of the linearly defined occurrence records (see Map).
The recent trend in the conversion of livestock farms to wildlife ranches may represent a land use change that provides a more natural habitat for Black-backed Jackals than livestock farms. This may be ascribed to a more “natural” prey base (e.g. indigenous ungulates vs. introduced livestock) on some ranches. In some cases, ranch managers do not manage Black-backed Jackal populations, but lethal management is ever increasing (e.g. De Waal 2009; Klare et al. 2010; du Plessis 2013; Pirie et al. 2017) as wildlife ranching is in conflict with predators (Thorn et al. 2012; Schepers 2016; Pirie et al. 2017). These populations are managed to reduce predation on valued ungulate species (e.g. high-value colour morphs and trophy animals), and the value of ungulate species may be directly proportional to retaliatory killing (Pirie et al. 2017). The variation in intensity of lethal control between land uses (i.e. game ranch vs. livestock farm) has not been rigorously quantified. However, it appears that in some cases, ranch managers have a more negative attitude towards predators than livestock farmers (Pirie et al. 2017; Drouilly et al. 2021). Thus, it is not possible to discern the net effects of wildlife ranching on the Black-backed Jackal.
Conservation
Black-backed Jackals are widely distributed and occur in most protected areas within the assessment region. Additionally, a large proportion of the population occurs outside protected areas, particularly in livestock farming areas. The Black-backed Jackal is not included in the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) Appendices or in the South African Threatened or Protected Species (TOPS) Regulations. Therefore, it has no legal protection outside protected areas. The largest threat to the Black-backed Jackal is lethal management actions aimed at reducing population size and associated livestock and ungulate losses. Although temporary reduction and local extirpations in response to lethal management actions have been documented (e.g. Blaum et al. 2009; Kamler et al. 2013; Walton & Joly 2003), Black-backed Jackals have persisted in these farming areas despite extensive lethal management (> 350 years; Stadler 2006; Avenant & du Plessis 2008; du Plessis 2013; Kerley et al. 2017; Nattrass & Conradie 2015; Nattrass et al. 2020b) and are expected to persist into the future. Thus, no active conservation efforts are currently required. However, the response of Black-backed Jackal populations to management interventions, as well as the effectiveness of these management interventions in reducing livestock and ungulate predation, is unknown. Thus, it is extremely important to accurately monitor Black-backed Jackal populations throughout the assessment region to determine distribution and abundance, as well as the impact of predator management actions on local populations. This will provide the necessary baseline information to make informed conservation and management decisions. Additionally, conservation efforts aimed at reducing human–wildlife conflict is urgently required.
Recommendations for land managers and practitioners: Several predator and livestock management techniques have been employed to reduce predation on livestock (Drouilly et al. 2020, 2023). However, the efficacy of these techniques in reducing predation as well as the impacts on Black-backed Jackal populations are unknown and likely vary tremendously throughout the assessment region. Thus, we cannot provide effective recommendations for land managers, until these methods have been systematically evaluated. However, we suggest some management actions that, according to anecdotal evidence, have been successful in certain areas. Du Plessis (2013) reviewed the management actions used to reduce livestock predation by Black-backed Jackal and suggested that non-lethal, preventative and exclusion techniques are the most conservation-friendly. Lethal management should be avoided, if possible, as Black-backed Jackal populations compensate for increased mortality by producing larger litters at younger ages (Minnie et al. 2016). However, when lethal management is employed, it should be applied selectively to ensure that the territorial structure of Black-backed Jackals remains intact. This may reduce compensatory immigration from neighbouring areas (Minnie 2016). Avoiding these compensatory life-history responses (reproduction and immigration), which may negate population reduction efforts (Minnie et al. 2016), may assist in the effective management of Black-backed Jackal predation on livestock and valued ungulate species. Further, Black-backed Jackals are highly adaptable, and it has been suggested that they habituate to specific methods. Thus, we suggest using a combination of management strategies and rotating between these to reduce the chances of Black-backed Jackals learning to bypass these measures. Finally, it is of the utmost importance that land managers keep accurate records of the population trends and the efficacy of management interventions, which will assist in effective adaptive management.
In the North West Province, game farmers and livestock farmers do often leave out carcasses (similarly to Vulture Restaurants), with the intent to reduce ungulate predation, though this has only anecdotal support, there is some support that it can work, given their diet (see Van der Merwe et al. 2009), as well as dependence on Vulture Restaurants in some areas (Yarnell et al. 2015). Whether this published or not, practitioners are not concerned on this.
The following management programmes, information centres and collaborative initiatives should be of interest to managers, game ranchers and farmers, who seek information and assistance with regards to the management of conflicts between Black-backed Jackals and humans:
- Canis–Caracal Programme (CCP), run by the African Large Predator Research Unit (ALPRU), University of the Free State (UFS): aims at finding solutions to reduce the widespread impact of predation on the livestock industry (national). Contact details: Prof. H.O. de Waal, Department of Animal, Wildlife and Grassland Sciences and African Large Predator Research Unit (ALPRU), PO Box 339, Internal Box 70, University of the Free State, Bloemfontein, 9300, South Africa. Email: dewaalho@ufs.ac.za.
- Predation Management Information Centre (PMIC): collating and analysing reliable information on predation and predation management methods, which will be made available continuously to a management information system (MIS). A team of dedicated staff members handles calls and enquiries. Experts in the team are available to provide advice to farmers. The centre is also responsible for the management of information and resources. Aim: to generate information that can be used to reduce the widespread impact of predation on the livestock industry. A collaborative initiative between the UFS and the Predation Management Forum (PMF). Contact details: Email: PredationMC@ufs.ac.za. Telephone: 051 401 2210.
- Scientific Assessment on Livestock Predation in South Africa (PredSA): a collaborative initiative between the Centre for African Conservation Ecology, Nelson Mandela University, Department of Environmental Affairs, Department of Agriculture, Forestry and Fisheries, Red Meat Research Development Planning Committee, National Wool Growers Association, Cape Wools and Mohair Trust. Contact details: Prof. Graham Kerley, Centre for African Conservation Ecology (ACE), PO Box 77000, Nelson Mandela Metropolitan University, Port Elizabeth, 6031, South Africa. Email:graham.kerley@nmmu.ac.za. The final assessment can be downloaded here: https://predsa.mandela.ac.za/predsa/media/Store/documents/PREDSA-eBook-2018.pdf
Research priorities: Black-backed Jackals have been cited as the dominant predators of livestock in the assessment region (e.g. van Niekerk 2010; du Plessis 2013; Badenhorst 2014; Minnie et al. 2016; Kerley et al. 2017; Drouilly et al. 2021) and cause large financial losses to the livestock production industry (van Niekerk 2010; Turpie & Babatopie 2018). Given this dominant role in livestock predation, it is expected that adequate research would have been conducted. However, this is not the case, as most research was spatially and temporally isolated with a limited scope (du Plessis et al. 2015). Many research groups have addressed some of these priority knowledge gaps since the last assessment. Below we highlight the thematic areas where research still needs to be prioritised and we suggest some applied research questions that need to be addressed to reduce human–carnivore conflict.
Ecology: A prerequisite for adaptive management is a sound ecological understanding of the focal species. Without this information it is difficult to predict the efficacy of management interventions and the effects thereof on the behaviour and ecology of target animals. Most importantly, there is a severe lack of accurate population estimates for Black-backed Jackals throughout the assessment region. Thus, a more systematic and regular monitoring programme is required to understand local fluctuations in population densities and to provide important baseline information.
Potential research questions:
- What drives distribution and variation in local densities?
- How do seasonal fluctuations in resource availability influence density and social structure?
- What are the environmental drivers of predation?
- What is the level of dispersal between various land uses and what drives this dispersal (sensu Minnie 2016)?
- To what extent, and under which conditions do Black-backed Jackals display compensatory reproduction (sensu Minnie et al. 2016)?
- Can “problem individuals” (i.e. sections of a population that consume more livestock/ungulates than others) be identified?
- What is the ecological role of the Black-backed Jackal (i.e. impacts on associated predator and prey populations)?
- What are some of the limiting factors on the species distribution (ie coastal humid Zululand, montane Fynbos)?
Predator and livestock management techniques: Relatively little research has focussed on the role of Black-backed Jackals as predators of livestock (Beinart 1998; van Sittert 1998; du Plessis 2013) and ungulates, as well as the various predator and livestock management methods employed to reduce livestock predation (e.g. du Plessis 2013; McManus et al. 2014; Drouilly et al. 2023). This limits our ability to effectively manage livestock and ungulate predation. Most information on these methods is contained in popular literature (du Plessis 2013). Therefore, research needs to focus on accurately assessing the efficacy of various management methods. Preferably this should be conducted within an adaptive management framework, which will assist in developing effective management regimes aimed at reducing human–carnivore conflict. Additionally, quantitative information on the extent of livestock and wildlife predation across the entire assessment region is required to substantiate reported losses and provide scientifically defensible grounds for management decisions.
Potential research questions:
- What are the spatial and temporal patterns of predation on livestock/ungulates?
- Which factors drive variation in local predation patterns?
- What is the viability and efficacy of various management methods? (for farmers’ reported efficacy, see Drouilly et al. 2023)
- Which variables influence the local efficacy of various management methods?
North West Province authorities have produced a document: Proof of concept: Feasibility of jackal restaurants to reduce jackal predation in rangelands of the North West province of South Africa – which outlines the need to simulate apex predators role by replacing these with humans, where carcasses are placed in the field with the intent to possibly reduce predation. This is a proposal open to any researchers to examine, and conduct should they wish to, and any prospective researchers can simply e-mail JPower@nwpg.gov.za.
Economics: Even though Black-backed Jackals are the dominant predators of livestock in the assessment region, surprisingly little information on the extent of this predation is available (see Strauss 2009; van Niekerk 2010; Turpie & Babatopie 2018). Thus, researchers need to focus on estimating the extent of predation, the economic consequences of this, as well as the economic costs of employing various livestock and predator management techniques.
Potential research questions:
- What are the direct and indirect economic costs of predation?
- What are the short- and long-term costs and benefits of various management techniques?
Social: In many cases, the perception of the landowner is more important in driving management decisions than the actual predation impact (e.g. Minnie et al. 2015; Drouilly et al. 2021). However, there is a paucity of information on the perceptions of various stakeholders towards Black-backed Jackals, predation and associated management actions. Understanding these perceptions and their drivers may assist in developing sustainable management strategies. Without this information, the acceptance of any proposed management interventions is unlikely (du Plessis 2013).
Potential research questions:
- Who are the interested and affected parties?
- What drives the perceptions of these stakeholders?
Considering the interrelatedness of these various aspects, it is important that all stakeholders – including ecologists, social scientists, economists, wildlife managers, farmers, ranchers and government officials contribute to the development of a sustainable, adaptive management strategy. Future research should be conducted systematically, as a combined effort, to ensure that the priority knowledge gaps are filled (see du Plessis 2013).
Ongoing research projects and research foci:
- Centre for African Conservation Ecology (ACE), Nelson Mandela University and University of Mpumalanga (UMP): Diet, reproduction, population structure, regional dispersal and the impacts of lethal management on population structure.
- Centre for Wildlife Management, University of Pretoria: Diet and social structure.
- Cheetah Conservation Fund: Livestock guarding dog programme.
- Endangered Wildlife Trust: Human–carnivore conflict.
- Institute for Communities and Wildlife in Africa, University of Cape Town (UCT): Human–carnivore conflict and economics.
- National Museum Bloemfontein and Centre for Environmental Management, University of the Free State: Ecology, socio-economics and effects of management on ecosystem integrity.
- North-West Parks: Social structure, space use and interactions with top-predators.
- UMP and Rhodes University: Diet, human–carnivore conflict and livestock management techniques.
- University of Fort Hare and ACE: Diet and interactions with top-predators.
- University of the Free State and African Large Predator Research Unit: Human–carnivore conflict and diet.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Record and report predation on livestock and ungulate species to the provincial wildlife authorities and to the Predation Management Information Centre (PMIC; email: PredationMC@ufs.ac.za).
- Individuals actively managing this species should report all the dead animals (trapped, shot or poisoned), as well as confirmed predation events, including photographs and GPS coordinates, to the national Predation Management Information Centre (PMIC; email: PredationMC@ufs.ac.za).
- Livestock farmers can assist by encouraging scientific research on their properties.
- Use holistic predator and livestock management methods. Additionally, employ these methods in an adaptive management framework to ensure the collation of information.
- Livestock farmers can actively monitor and record the effectiveness of management methods within an adaptive management framework. Through liaison with the scientific community, results can be accurately recorded and analysed to determine best-management practices.
- Citizen scientists that have been involved in radio-collaring studies of the species are encouraged to get their results published, as it is unethical not to
Bibliography
Ansell, W. F. H. 1960. Mammals of Northern Rhodesia. The Government Printer, Lusaka, Northern Rhodesia.
Atickem A, Stenseth NC, Drouilly M, Bock S, Roos C, Zinner D. 2017. Deep divergence among mitochondrial lineages in African jackals. Zoologica Scripta: 1–8 (early online).
Avenant NL, Steenkamp E, de Waal HO. 2009. Reviewing a case study on the effects of different management options to reduce predation on small livestock in the Karoo. Southern African Wildlife Management Association Symposium. Cape Town, South Africa.
Avenant NL. 2004. Conserving Mountain Biodiversity in Southern Lesotho: Mammal report. United Nations Development Plan, Lesotho.
Avenant, N.L. and du Plessis, J.J. 2008. Sustainable small stock farming and ecosystem conservation in southern Africa: a role for small mammals? Mammalia 72: 258-263.
Badenhorst CG. 2014. The economic cost of large stock predation in the North West Province of South Africa. M.Sc. Thesis. University of the Free State.
Bagniewska JM, Kamler JF. 2014. Do black-backed jackals affect numbers of smaller carnivores and prey? African Journal of Ecologyvvvv 52: 546–567.
Beinart W. 1998. The night of the jackal: sheep, pastures and predators in the Cape. Past & Present 158: 172–206.
Bellan SE, Cizauskas CA, Miyen J, Ebersohn K, Kusters M, Prager KC, van Vuuren M, Sabeta C, Getz WM. 2012. Black-backed jackal exposure to rabies virus, Canine Distemper Virus, and Bacillus anthracis in Etosha National Park, Namibia. Journal of Wildlife Diseases 48: 371–381.
Bergman DL, de Waal HO, Avenant NL, Bodenchuk MJ, Marlow MC, Nolte DL. 2013. The need to address black-backed jackal and caracal predation in South Africa. In: Armstrong JB, Gallagher GR. (ed.), Wildlife Damage Management Conference., pp. 86–94. Lincoln, USA.
Bernard RTF & Stuart CT. 1992. Correlates of diet and reproduction in the black-backed jackal. South African Journal of Science 88: 292–294.
Bingham J, Purchase GK. 2002. Reproduction in the jackals Canis adustus Sundevall, 1846, and Canis mesomelas Schreber, 1778 (Carnivora: Canidae), in Zimbabwe. African Zoology 37(21–26).
Blaum, N., Tietjien, B., and Rossmanith, E. 2009. Impact of livestock husbandry on small- and medium-sized carnivores in Kalahari Savannah Rangelands. Journal of Wildlife Management 73: 60-67.
Bothma J du P. 1971. Notes on movement by the black-backed jackal and the aardwolf in the western Transvaal. Zoology Africana 6: 205–207.Bothma JDP. 2012. Literature review of the ecology and control of black-backed jackal and caracal in South Africa. Cape Nature.
Brassine MC. 2011. The diet and ecological role of black-backed jackals, Canis mesomelas, in two conservation areas in the Eastern Cape Province, South Africa. M.Sc. Thesis. Rhodes University.
Brassine, M.C. and Parker, D.M. 2012. Does the presence of large predators affect the diet of a mesopredator? African Journal of Ecology 50: 243–246.
Carruthers, J. & Nattrass, N. 2018. History of Predator-Stock Conflict in South Africa. In: Livestock predation and its management in South Africa: a scientific assessment (Eds Kerley, G.I.H., Wilson, S.L. & Balfour, D.). Centre for African Conservation Ecology, Nelson Mandela University, Port Elizabeth, 30-52.
Coe, M.J. and Skinner, J.D. 1993. Connections, disjunctions and endemism in the eastern and southern African mammal faunas. Transactions of the Royal Society of South Africa, 48(2), pp.233-255.
Collinson WJ, Parker DM, Bernard RTF, Reilly BK, Davies-Mostert HT. 2015. An inventory of vertebrate roadkill in the Greater Mapungubwe Transfrontier Conservation Area, South Africa. African Journal of Wildlife Research 45: 301–311.
Conradie B. 2012. Are hunting clubs the solution to small stock depredation? The case of Ceres, 1979 and 1980. Agrekon 51: 96–113.
da Rosa, B.R.P. 2019. Optimizing Small Mammal Relative Abundance Measures Using Non-Invasive Sampling and Assessment of its Contribution to Occupancy Modelling of Small Carnivores in Dry Woodland Savannah of South Africa (Master’s thesis, Universidade de Lisboa (Portugal)).
Deacon F. 2010. Aspekte rakende die ruimtelike ekologie van die rooijakkals (Canis mesomelas) as probleemdier in die Suid-Vrystaat. M.Sc. Thesis. University of the Free State.
Do Linh San E, Malongwe NB, Fike B, Somers MJ, Walters M. 2009. Autumn diet of black-backed jackals (Canis mesomelas) in the thicket biome of South Africa. Wildlife Biology in Practice 5: 96–103.
Dinets, V. (2015). The Canis tangle: a systematics overview and taxonomic recommendations. Vavilov Journal of Genetics and Breeding, 19(3), 286-291.
Drouilly, M., Clark, A., & O’Riain, M. J. 2018a. Multi-species occupancy modelling of mammal and ground bird communities in rangeland in the Karoo: A case for dryland systems globally. Biological Conservation, 224, 16-25.
Drouilly, M., Nattrass, N., & O’riain, M. J. 2018b. Dietary niche relationships among predators on farmland and a protected area. The Journal of Wildlife Management, 82(3), 507-518.
Drouilly, M., & O’Riain, M. J. 2019a. Wildlife winners and losers of extensive small-livestock farming: a case study in the South African Karoo. Biodiversity and Conservation, 28, 1493-1511.
Drouilly, M., Nattrass, N., & O’riain, M. J. 2019b. Global positioning system location clusters vs. scats: comparing dietary estimates to determine mesopredator diet in a conflict framework. Journal of Zoology, 310(2), 83-94.
Drouilly, M., Tafani, M., Nattrass, N., & O’Riain, J. 2019c. Spatial, temporal and attitudinal dimensions of conflict between predators and small-livestock farmers in the Central Karoo. African Journal of Range & Forage Science, 35(3-4), 245-255.
Drouilly, M., Kelly, C., Cristescu, B., Teichman, K. J., & O’riain, M. J. 2020. Investigating the hidden costs of livestock guarding dogs: a case study in Namaqualand, South Africa. Journal of Vertebrate Biology, 69(3), 20033-1.
Drouilly, M., Nattrass, N., & O’Riain, M. J. 2021. Beauty or beast? Farmers’ dualistic views and the influence of aesthetic appreciation on tolerance towards black-backed jackal and caracal. PLoS One, 16(3), e0248977.
Drouilly, M., Nattrass, N., & O’Riain, M. J. 2023. Small-livestock farmers’ perceived effectiveness of predation control methods and the correlates of reported illegal poison use in the South African Karoo. Ambio, 52(10), 1635-1649.
du Plessis JJ. 2013. Towards the development of a sustainable management strategy for Canis mesomelas and Caracal caracal on rangeland. Ph.D. Thesis. University of the Free State.
du Plessis SS. 1972. Ecology of blesbok with special reference to productivity. Wildlife Monographs 30: 3–70.
du Plessis JJ, Avenant NL, de Waal HO. 2015. Quality and quantity of the scientific information available on black-backed jackals and caracals: contributing to human–predator conflict management? African Journal of Wildlife Research 45: 138–157.
Estes, R.D. 1991. The Behavior Guide to African Mammals: including Hoofed Mammals, Carnivores and Primates. University of California Press, Berkeley and Los Angeles, California, USA.
Fairall N. 1968. The reproductive seasons of some mammals in the Kruger National Park. Zoologica Africana 3: 189-210.
Favaretto, B. S., Tambling, C. J., & Kerley, G. I. 2024. The black-backed jackal as a seed disperser. Journal of Arid Environments, 220, 105099.
Fedriani JM, Fuller TK, Sauvajot RM. 2001. Does availability of anthropogenic food enhance densities of omnivorous mammals? An example with coyotes in Southern California. Ecography 24: 325–331.
Ferguson JWH, Nel JAJ, de Wet MJ. 1983. Social organization and movement patterns of black-backed jackals Canis mesomelas in South Africa. Journal of Zoology 199v: 487–502.
Ferreira NA. 1988. Sekere aspekte van die ekologie en die beheer van die rooikat (Felis caracal) in die Oranje-Vrystaat. Unpublished report. Orange Free State Provincial Administration,irectorate Environmental and Nature Conservation, Bloemfontein, South Africa.
Fourie RM, Tambling CJ, Gaylard A, Kerley GIH. 2015. Short-term foraging responses of a generalist predator to management-driven resource pulses. African Journal of Ecology 53: 521–530.
Fuller, T. K., Biknevicius, A. R., Kat, P. W., Van Valkenburgh, B. and Wayne, R. K. 1989. The ecology of three sympatric jackal species in the Rift Valley of Kenya. African Journal of Ecology 27: 313-323.
Geraads, D. 2011. A revision of the fossil Canidae (Mammalia) of north‐western Africa. Palaeontology, 54(2), 429-446.
Gese, E. and Bekoff, M. 2004. Coyote Canis latrans Say, 1823. In: Sillero-Zubiri, C., Hoffmann, M. & Macdonald, D.W. (ed.), Canids: Foxes, Wolves, Jackals and Dogs. Status Survey and Conservation Action Plan, pp. 81-87. IUCN, Gland and Cambridge.
Gunter Q. 2008. A critical evaluation of historical data on two damage causing predators, Canis mesomelas and Caracal caracal. M.Sc. Thesis. University of the Free State.
Hayward MW, Porter L, Lanszki J, Kamler JF, Beck JM, Kerley GIH, Macdonald DW, Montgomery RA, Parker DM, Scott DM, O’Brien J, Yarnell RW. 2017. Factors affecting prey preferences of jackals (Canidae). Mammalian Biology 85: 70–82.
Hiscocks K, Perrin MR. 1988. Home range and movements of black-backed jackals at Cape Cross Seal Reserve, Namibia. South African Journal of Wildlife Research 18: 97–100.
Hoffmann M. 2014. Canis mesomelas. The IUCN Red List of Threatened Species 2014.
Humphries BD, Ramesh T, Hill TR, Downs CT. 2016. Habitat use and home range of black-backed jackals (Canis mesomelas) on farmlands in the Midlands of KwaZulu-Natal, South Africa. African Zoology 51: 37–45.
James RS, Scott DM, Yarnell RW, Overall ADJ. 2017. Food availability and population structure: how do clumped and abundant sources of carrion affect the genetic diversity of the black-backed jackal? Journal of Zoology 301: 184–192.
Jenner N, Groombridge J, Funk SM. 2011. Commuting, territoriality and variation in group and territory size in a black-backed jackal population reliant on a clumped, abundant food resource in Namibia. Journal of Zoology 284: 231–238.
Kamler JF, Foght JL, Collins K. 2010. Single black-backed jackal (Canis mesomelas) kills adult impala (Aepyceros melampus). African Journal of Ecology 48: 847–848.
Kamler JF, Klare U, Macdonald DW. 2012a. Seasonal diet and prey selection of black-backed jackals on a small-livestock farm in South Africa. African Journal of Ecology 50: 2099–2307.
Kamler, J. F., Stenkewitz, U., Klare, U. Jacobsen, N. F., and Macdonald, D. W. 2012b. Resource partitioning among cape foxes, bat-eared foxes, and black-backed jackals in South Africa. Journal of Wildlife Management 76: 1241-1253.
Kamler, J. F., Stenkewitz, U., and Macdonald, D. W. 2013. Lethal and sublethal effects of black-backed jackals on cape foxes and bat-eared foxes. Journal of Mammalogy 94(295-306).
Kamler, J. F., Stenkewitz, U., Gharajehdaghipour, T., & Macdonald, D. W. 2019. Social organization, home ranges, and extraterritorial forays of black‐backed jackals. The Journal of Wildlife Management, 83(8), 1800-1808.
Kamler, J., Loveridge, A., O’Donnell, H., & Macdonald, D. 2020. Effects of humans and large carnivores on the survival of black‐backed jackals. African Journal of Ecology, 58(3).
Kaunda SKK, Skinner JD. 2003. Black-backed jackal diet at Mokolodi Nature Reserve, Botswana. African Journal of Ecology 41: 39–46.
Kerley GIH, Behrens KG, Carruthers J, Diemont M, du Plessis J, Minnie L, Richardson PRK, Somers MJ, Tambling CJ, Turpie J, van Niekerk HN, Balfour D. 2017. Livestock predation in South Africa: the need for and value of a scientific assessment. South African Journal of Science 113: 1–3.
Kingdon, J. 1977. East African Mammals: An Atlas of Evolution in Africa. Volume IIIA (Carnivores). Academic Press, London, UK.
Klare, U., Kamler, J.F., Stenkewitz, U. and Macdonald, D.W. 2010. Diet, prey selection, and predation impact of black-backed jackals in South Africa. Journal of Wildlife Management 74: 1030-1042.
Kok OB & Nel JAJ. 2004. Convergence and divergence in prey of sympatric canids and felids: opportunism or phylogenetic constraint? Biological Journal of the Linnean Society 83: 527–538.
Lamprecht, J. 1978. On diet, foraging behaviour and interspecific food competition of jackals in the Serengeti National Park, East Africa. Zeitschrift für Säugetierkunde 43: 210-223.
Leijenaar S-L, Cilliers D, Whitehouse-Tedd K. 2015. Reduction in livestock losses following placement of livestock guarding dogs and the impact of herd species and dog sex. Journal of Agriculture and Biodiversity Research 4: 9–15.
Loveridge, A. & Macdonald, D. W. 2001. Seasonality in spatial organisation and dispersal of sympatric jackals: implications for rabies management. Journal of Zoology 253: 101-111.
Loveridge, A. J. 1999. Behavioural-ecology and rabies transmission in sympatric Southern African jackals. Wildlife Conservation Research Unit, University of Oxford.
Loveridge, A. J. and Macdonald, D. W. 2002. Habitat ecology of two sympatric species of jackals in Zimbabwe. Journal of Mammalogy 83: 599-607.
Loveridge, A. J. and Macdonald, D. W. 2003. Niche separation in sympatric jackals (Canis mesomelas and Canis adustus). Journal of Zoology (London) 259: 143-153.
Loveridge, A. J. and Nel, J. A. J. 2004. Black-backed Jackal Canis mesomelas Schreber, 1775. In: Sillero-Zubiri, C., Hoffmann, M. and Macdonald, D.W. (eds), Canids: Foxes, Wolves, Jackals and Dogs. Status Survey and Conservation Action Plan, pp. 161-166. IUCN, Gland.
Loveridge, A.J. and Nel, J.A.J. 2013. Canis mesomelas Black-backed Jackal. In: Kingdon, J. & Hoffmann, M. (ed.), Mammals of Africa. Volume 5. Carnivores, Pangolins, Equids and Rhinoceroses, pp. 39-44. Bloomsbury, London.
Lynch, C.D. 1994. The mammals of Lesotho: order Carnivora: family Canidae. Navorsinge van die Nasionale Museum: Researches of the National Museum, 10(4), pp.199-209.
Macdonald DW. 1979. The Flexible Social System of the Golden Jackal, Canis aureus. Behavioral Ecology and Sociobiology 5: 17–38.
Marker LL, Dickman AJ, Macdonald DW. 2005. Perceived effectiveness of livestock-guarding dogs placed on Namibian farms. Rangeland Ecology & Management 58: 329–336.
McKenzie AA. 1990. Co-operative hunting in the black-backed jackal, Canis mesomelas, Schreber. Ph.D. Thesis. University of Pretoria.
McKenzie MM. 1993. Biology of the black-backed jackal Canis mesomelas with reference to rabies. Onderstepoort Journal of Veterinary Research 60: 367–371.
McManus JS, Dickman AJ, Gaynor D, Smuts BH, Macdonald DW. 2014. Dead or alive? Comparing costs and benefits of lethal and non-lethal human–wildlife conflict mitigation on livestock farms. Oryx 49: 1–9.
McManus JS, Dickman AJ, Gaynor D, Smuts BH, Macdonald DW. 2014. Dead or alive? Comparing costs and benefits of lethal and non-lethal human–wildlife conflict mitigation on livestock farms. Oryx 49: 1–9.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum, Pretoria, South Africa.
Minnie L, Boshoff AF, Kerley GIH. 2015. Vegetation type influences livestock predation by leopards: implications for conservation in agro-ecosystems. African Journal of Wildlife Research 45: 204–214.
Minnie L, Gaylard A, Kerley GI. 2016. Compensatory life-history responses of a mesopredator may undermine carnivore management efforts. Journal of Applied Ecology 53: 379-387.
Minnie, L., Zalewski A., Zalewska H. & Kerley, G.I.H. 2018. Spatial variation in anthropogenic mortality induces a source-sink system in a hunted mesopredator. Oecologia, 186, 1–13.
Minnie L. 2009. Socio-economic and ecological correlates of leopard-stock farmer interactions in the Baviaanskloof Mega-Reserve, Eastern Cape. M.Sc. Thesis. Nelson Mandela Metropolitan University.
Minnie L. 2016. Effects of lethal management on black-backed jackal population structure and source–sink dynamics. Ph.D. Thesis. Nelson Mandela Metropolitan University.
Moehlman PD. 1979. Jackal helpers and pup survival. Nature 277: 382–383.
Moehlman, P.D. 1983. Socioecology of silverbacked and golden jackals (Canis mesomelas and Canis aureus). Recent advances in the study of mammalian behavior, pp. 423-453. American Society of Mammologists, Pittsburgh Pennsylvania, USA.
Moehlman, PD. 1987. Social organization in jackals. American Scientist 75: 366–375.
Morwe, J.B. 2013. Determining the direct impact of black-backed jackal (Canis mesomelas) on the springbok (Antidorcas marsupialis) population at Maria Moroka Nature Reserve, Free State, South Africa. B.Sc. Honours Thesis. University of the Free State.
Murison MK. 2014. The roles of black-backed jackals and caracals in issues of human–wildlife conflict in the Eastern Cape, South Africa. M.Sc. Thesis. Rhodes University.
Nattrass, N., & Conradie, B. 2015. Jackal narratives: predator control and contested ecologies in the Karoo, South Africa. Journal of Southern African Studies, 41(4), 753-771.
Nattrass, N., Conradie, B., Stephens, J., & Drouilly, M. 2020a. Culling recolonizing mesopredators increases livestock losses: Evidence from the South African Karoo. Ambio, 49, 1222-1231.
Nattrass, N., Drouilly, M., & O’Riain, M. J. 2020b. Learning from science and history about black‐backed jackals Canis mesomelas and their conflict with sheep farmers in South Africa. Mammal Review, 50(1), 101-111.
Nel JAJ, Loutit RJ, Braby R, Somers MJ. 2013. Resource dispersion, territory size and group size of black-backed jackals on a desert coast. Acta Theriologica 58: 189–197.
Ngoepe, E., Chirima, J.G., Mohale, D., Mogano, K., Suzuki, T., Makita, K. and Sabeta, C.T. 2022. Rabies outbreak in black-backed jackals (Canis mesomelas), South Africa, 2016. Epidemiology & Infection, 150, p.e137.
Nieman, W. A., Leslie, A. J., & Wilkinson, A. 2018. The Use of Animals and Animal-Derived Constituents in African Traditional Medicine and Other Cultural Applications: Townships in the Western Cape Province. In Culture, Conflict and Cuisine: A Quantitative Assessment of Terrestrial Vertebrate Off-Take at the Human-Wildlife Interface, MSc thesis, Stellenbosch University, South Africa.
Oro D, Genovart M, Tavecchia G, Fowler MS, Martínez-Abraín A. 2013. Ecological and evolutionary implications of food subsidies from humans. Ecology Letters 16: 1501–1514.
Pirie TJ, Thomas RL, Fellowes MDE. 2017. Increasing game prices may alter farmers’ behaviours towards leopards (Panthera pardus) and other carnivores in South Africa. PeerJ 5(e3369).
Pohl, C.F. 2015. The diet of caracal (Caracal caracal) in the Southern Free State. Department of Zoology and Entomology, University of the Free State.
Potgieter GC, Kerley GIH, Marker LL. 2016. More bark than bite? The role of livestock guarding dogs in predator control on Namibian farmlands. Oryx 50: 514–522.
Potgieter GC, Marker LL, Avenant NL, Kerley GIH. 2013. Why Namibian farmers are satisfied with the performance of their livestock guarding dogs. Human Dimensions of Wildlife 18: 403–415.
Pretorius, M. 2019. Mesocarnivores in Protected Areas: ecological and anthropogenic determinants of habitat use in northern Kwa-Zulu Natal, South Africa.
Pringle JA & Pringle VL. 1979. Observations on the lynx Felis caracal in the Bedford district. African Zoology 14: 1–4.
Rowe-Rowe DT. 1978. The small carnivores of Natal. Lammergeyer 25: 1–48.
Rowe-Rowe DT. 1984. Black-backed jackal population structure in the Natal Drakensberg. Lammergeyer 32: 1–7.
Rowe-Rowe, D. T. 1982. Home range and movements of black-backed jackals in an African montane region. South African Journal of Wildlife Research 12: 79-84.
Rowe-Rowe, D.T. 1992. The Carnivores of Natal. Natal Parks Board, Pietermaritzburg, South Africa.
Schepers A. 2016. The economic impact of predation in the wildlife ranching industry in Limpopo, South Africa. M.Sc. Thesis. University of the Free State.
Skead DM. 1973. Incidence of calling in the black-backed jackal. Journal of the Southern African Wildlife Management Association 3: 28–29.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Slater, K., De Jager, D., van Wyk, A. M., Dalton, D. L., Kropff, A. S., & Du Preez, I. 2022. Population genetics of a lethally managed medium‐sized predator. Journal of Zoology, 318(1), 47-62.
Smithers, R.H.N. 1971. The mammals of Botswana. National Museums of Rhodesia, Museum Memoir 4: 1-340.
Soulsbury CD, Iossa G, Baker PJ, Cole NC, Funk SM, Harris S. 2007. The impact of sarcoptic mange Sarcoptes scabiei on the British fox Vulpes vulpes population. Mammal Review 37: 278–296.
Stadler H. 2006. Historical perspectives on the development of problem animal management in the Cape Province. In: Daly B, Davies-Mostert W, Evans S, Friedmann Y, King N, Snow T & Stadler H, (ed.), Workshop on Holistic Management of Human–Wildlife Conflict in the Agricultural Sector 94 of South Africa, pp. 11–16. Johannesburg, South Africa.
Stewart D. 2004. The Zebra’s Stripes and other African Animal Tales. Struik Publishers, Cape Town, South Africa.
Strauss AJ. 2009. The impact of predation on a sheep enterprise in the Free State Province. M.Sc. Thesis. University of the Free State.
Swanepoel LH, Swanepoel CM, Brown PR, Eiseb SJ, Goodman SM, Keith M, Kirsten F, Leirs H, Mahlaba TaAM, Makundi RH, Malebane P, von Maltitz EF, Massawe AW, Monadjem A, Mulungu LS, Singleton GR, Taylor PJ, Soarimalala V, Belmain SR. 2017. A systematic review of rodent pest research in Afro–Malagasy small-holder farming systems: Are we asking the right questions? PLoS One 12(e0174554).
Turpie, J.K. & Babatopie, A. 2018. The socio-economic impacts of livestock predation and its prevention In South Africa. In: Livestock predation and its management in South Africa: a scientific assessment (Eds Kerley, G.I.H., Wilson, S.L. & Balfour, D.). Centre for African Conservation Ecology, Nelson Mandela University, Port Elizabeth, 53-81.
Van de Ven, T.M., Tambling, C.J. and Kerley, G.I. 2013. Seasonal diet of black-backed jackal in the Eastern Karoo, South Africa. Journal of Arid Environments, 99, pp.23-27.
Van der Merwe, I., Tambling, C.J., Thorn, M., Scott, D.M., Yarnell, R.W., Green, M., Cameron, E.Z. and Bateman, P.W. 2009. An assessment of diet overlap of two mesocarnivores in the North West Province, South Africa. African Zoology, 44(2), pp.288-291.
Viljoen, S., O’Riain, M. J., Penzhorn, B. L., Drouilly, M., Vorster, I., & Bishop, J. M. 2021. Black-backed jackals (Canis mesomelas) from semi-arid rangelands in South Africa harbour Hepatozoon canis and a Theileria species but apparently not Babesia rossi. Veterinary Parasitology: Regional Studies and Reports, 24, 100559.
Viranta, S., Atickem, A., Werdelin, L., & Stenseth, N. C. 2017. Rediscovering a forgotten canid species. BMC Zoology, 2, 1-9.
de Waal HO. 2009. Recent advances in co-ordinated predator management in South Africa. Merino SA Focus 2009(44–46).
Wittenberg, H. 2014. The boer and the jackal: satire and resistance in Khoi orature. Critical arts, 28(4), 593-609.
Wyman, J. 1967. The jackals of the Serengeti. Animals 10: 79-83.
Yarnell, R.W., Phipps, W.L., Dell, S., MacTavish, L.M. and Scott, D.M. 2015. Evidence that vulture restaurants increase the local abundance of mammalian carnivores in S outh A frica. African Journal of Ecology, 53(3), pp.287-294.