Zorilla
Ictonyx striatus

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Ictonyx striatus – (Perry, 1810)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – MUSTELIDAE – Ictonyx – striatus
Common Names: Zorilla, African Skunk, Cape Polecat, Striped Polecat (English), Stinkmuishond (Afrikaans), iQaqa (Ndebele, Xhosa, Zulu), Nakêdi (Sotho, Tswana), Ehidembo (Shona), Licaca (Swati), Thuri (Venda), Inganakazana (Zulu)
Synonyms: Bradypus striatus Perry, 1810
Taxonomic Note:
The type locality of this species is the Cape of Good Hope (Hollister 1915). Although Meester et al. (1986) and Coetzee (1977) listed at least ten subspecies, it is likely that many of these will be discredited following further taxonomic research (Skinner & Chimimba 2005). The Zorilla is easily distinguished from the closely related African Striped Weasel (Poecilogale albinucha) by its larger size, longer hair, and the three characteristic white facial markings (Rowe-Rowe 1997)
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Power, R.J.1, da Silva, J.M2, Hayder, F.3 & Do Linh San, E.4
Reviewer: Nicholson, S.K.5
Institutions: 1North West Provincial Government, 2South African National Biodiversity Institute,3University of Fort Hare,4Sol Plaatje University, 5Endangered Wildlife Trust
Previous Assessors and Reviewers: Rowe-Rowe, D., Stuart, C., Stuart, M. & Do Linh San, E.
Previous Contributors: Madikiza, Z., Relton, C. & Nicholson, S.
Assessment Rationale
The Zorilla is listed as Least Concern as it is common across its wide distribution range, is present in numerous protected areas, and faces no major threats. Threats, such as road mortalities and domestic dogs, may cause local subpopulation declines, but this is not currently suspected to impact on overall population trends. Education and awareness campaigns should be used to raise the public profile of this species. Citizen science monitoring can contribute to refining our knowledge of its distribution and population trends, especially so as to avoid confusion with the rarer African Striped Weasel.
Regional population effects: Dispersal is expected between regions, as the Zorilla’s range is continuous across Africa south of the Sahara, and this species is not limited by fences.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Power RJ, da Silva JM, Hayder F & Do Linh San E. 2025. A conservation assessment of Ictonyx striatus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The Zorilla has a wide distribution, ranging south of the Sahara from Mauritania and Senegal in the west, to Sudan, southeastern Egypt, Ethiopia and Djibouti in the east, and south to South Africa (Stuart & Stuart 2013). It occurs from sea level to more than 4,000 m asl on Mt Kenya (Young & Evans 1993).
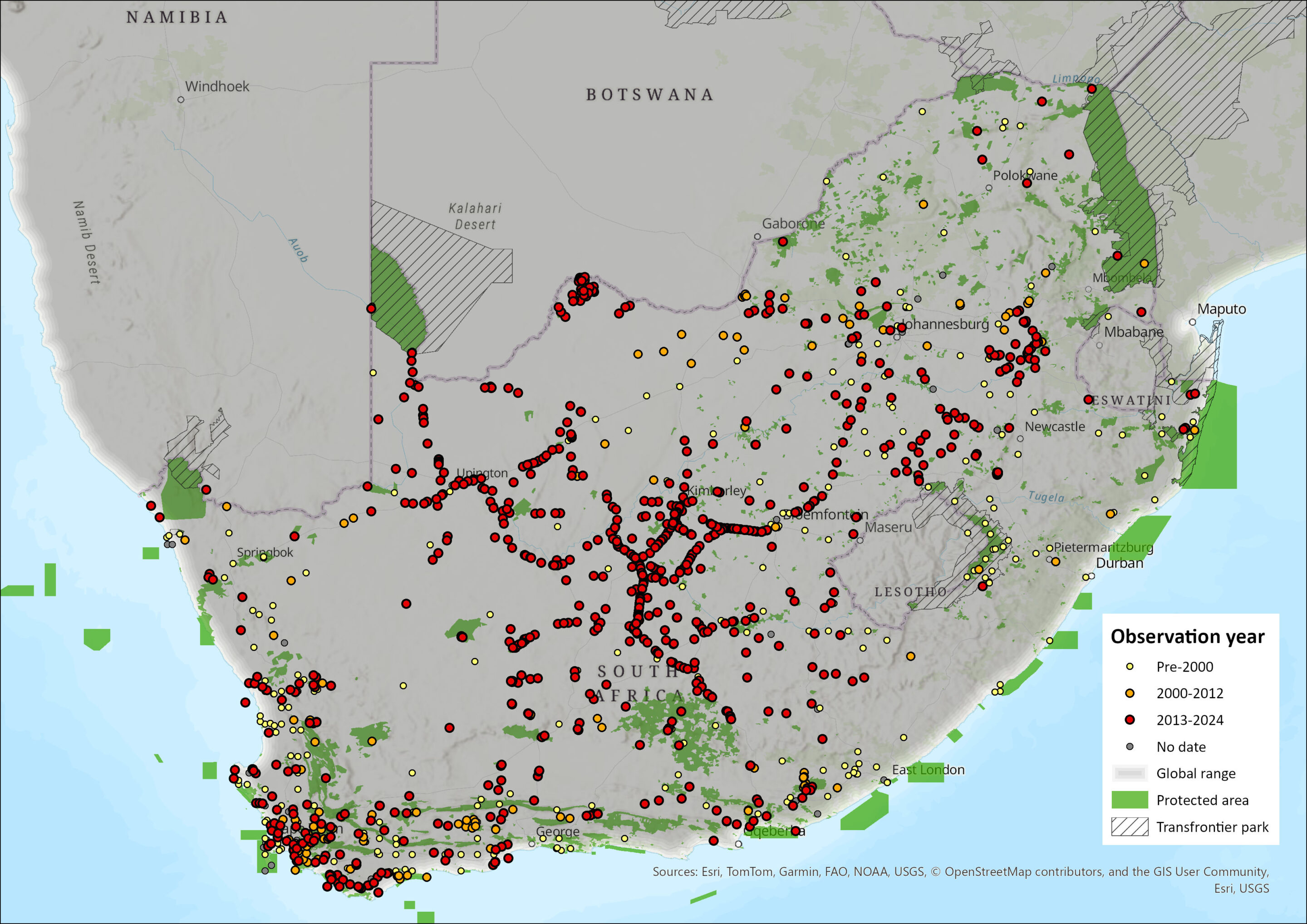
This species is widely distributed throughout southern Africa and, within the assessment region, is present in all South African provinces. Zorillas show a wide habitat tolerance, often ranging into agricultural areas, such as pastoral areas (Williams et al. 2018; Da Rosa 2019), wheatlands of southwest South Africa (Stuart 1981), and exotic plantations of KwaZulu-Natal (Rowe-Rowe 1992; Stuart & Stuart 2013), and rangelands throughout the country (Blaum et al. 2008; Williams et al. 2018; Schurch et al. 2021; Woodgate et al. 2023). Of late, Zorilla has been confirmed in the Northern Cape’s Tswalu Kalahari Reserve (Smith et al. 2023), Eastern Cape’s Mountain Zebra National Park (Smith et al. 2023), Western Cape’s farms in the Nuweveldberg near Beaufort West (Schurch et al. 2021), KwaZulu-Natal’s farmlands adjacent Phinda Resource Reserve (Da Rosa 2019). The species appears more important in the arid central, and southwest of the country, where provinces such as the Eastern Cape, Western Cape, Northern Cape, Free State and Northwest are important (Figure 1). Since the last assessment, there seem fewer occurrences in KwaZulu-Natal and Limpopo of the species. The linearity in occurrence records across much of the central part of the country are likely roadkill records (Figure 1). In the North West Province they occur across most of the province (Power et al. 2019) but are more prevalent in the arid west. They have been reported from the eastern Highveld of Mpumalanga (Loock & Swanepoel 2019) but seem rare in the Lowveld of that province (Figure 1).
Zorilla occur in Lesotho (Lynch & Watson 1990).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Zorilla (Ictonyx striatus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Djibouti | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
It is important to consider a species’ ecological and biological traits to determine the likely mechanisms of climate change impact and quantify these using expert knowledge (Foden & Young 2016). An increase in shrub cover as predicted by many climate change models (Foden and Young 2016), would negatively impact the Zorilla.
However, countering this, general regional drying conditions might enhance range expansion (Rowe-Rowe & Taylor 1996), which is another climate change prediction, and that of expanded areas of aridity (see Foden and Young 2016). However, if excessively dry, it may impact on all prey populations, such as rodents (Milton et al. 2022).
In KwaZulu-Natal, it is mentioned that in dry years, the species distribution range expands eastwards, and retracts westwards in wet years (Rowe-Rowe & Taylor 1996). Increasing shrub cover in Kalahari rangelands affects carnivore species, where for the Zorilla, they are negatively affected as they prefer open savanna habitats (Blaum et al. 2007). Increasing temperatures in the Western Cape Karoo combined with a severe drought from 2015 to 2021 caused a 36% dieback in perennial shrub cover. The percentage of heuweltjies occupied by Whistling Rats (Parotomys brantsii) declined from 44% to 20% occupation between 2005 and 2021 during unusually hot, dry conditions (Milton et al. 2022). Given Zorillas’s are murid hunters (Smithers 1971; Rowe-Rowe 1978a), it can be expected that this change in prey abundance may impact them too. Zorilla were detected in their study area, and they might be impacted by loss in these prey species (Milton et al. 2022).
Population Information
The Zorilla ranges extensively across the assessment region and Shortridge (1934) described this species as one of the most ubiquitous mammals in southern Africa. Although Zorillas are considered rare in some parts of their range (Skinner & Chimimba 2005), they are fairly common elsewhere and are easily overlooked (Blaum et al. 2008), as they may be difficult to trap, though not impossible (Little & Crowe 2004), at least locally (E. Do Linh San pers. obs. 2005–2011). Estes (1991) stated that they are most common in open rangelands where wild or domestic grazers keep grass short. On Kalahari rangelands, fluctuations in the relative abundance of small carnivores like Zorilla are reported (Blaum et al. 2008), yet they are said to be one of the dominant species (Blaum et al. 2007; 2009).
In Nuweveld mountains of the Western Cape Karoo, with a change of management to wildlife-friendly farming, Zorilla steadily increased in abundance over a four-year period up until 2019 (Schurch et al. 2021). In the North West, in two comparable periods involving extensive driving up to the limit of the travel allowance (2010-2013), and (2021-2023), reported equally on five Zorilla road kills recorded in each period (NW Directorate of Biodiversity Management records, 2020-2023), which suggests a similar rate of killing for them, suggesting a similar population size.
In East Africa, Hendrichs (1972) estimated population density at 0.1–0.2 individual / km². They are widespread and regularly observed only locally within the assessment region. Zorilla were detected in only 1 out of 7 Zululand reserves (Pretorius 2019), suggesting low population sizes there.
Using an average density of 0.1 individual / km², and assuming similar densities hold across most of the assessment region, we estimate that there are well over 10,000 mature individuals in the population.
Population Information
Current population trend: Stable, based on wide habitat tolerance and extent of occurrence.
Continuing decline in mature individuals: Unknown, but probably not.
Number of mature individuals in population: Probably > 10,000.
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: It is not currently possible to determine the extent or number of subpopulations.
Severely fragmented: No. This species has a broad habitat tolerance and can exist in agricultural landscapes.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While a complete mitochondrial genome has been assembled for I. libycus (Gray et al. 2022), and I. striatus has been investigated in a phylogenetic context (Winter et al. 2023), no population genetic studies have been undertaken on the species.
Because they are widely distributed across the assessment region and considered quite abundant, it is expected that they exist as a single metapopulation that extends into neighbouring countries. No subpopulations are thought to have gone extinct in the past and hence, the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF’s) complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (all populations remaining).
While no census (Nc) or effective population size (Ne) data exists for the species, density estimates in some areas suggest that across the assessment region there is well over 10,000 individuals. Even though the 10,000 value is not an actual census count, the quantity can still be used as a proxy for quantifying the GBF’s headline genetic indicator – proportion of populations with an Ne > 500. Based on the assumption that the species is likely to occur as a single metapopulation, the Ne of the species is ~1,000-3,000 individuals, based on a Ne/Nc conversion ratio of 0.1-0.3. Having the 1 population above Ne 500, translates to an indicator score of 1.0 (1/1 population with Ne > 500).
Habitats and ecology
Zorillas are highly adaptable and exhibit a wide habitat tolerance, as they are found in open grassland, savannah woodland, thornbush, rocky habitats, agricultural areas, forest, and even desert (usually along drainage lines, provided there is some scrub cover). They are absent from the dense evergreen forests of the Congo basin and West Africa (Stuart & Stuart 2013). The important biomes for the species are the Grasslands, arid western Savannas, Nama Karoo, Succulent Karoo and Fynbos biomes.
It was noted that, during dry years, their distribution extended further eastwards, receding westwards (to drier regions) during times of high rainfall (Rowe-Rowe & Taylor 1996). Zorillas prefer dry, open grassland (Webster et al. 2021), with moderate levels of shrub cover (Blaum et al. 2007), and also utilise rocky outcrops (Webster et al. 2021). The relative abundance of the species was correlated to rodent prey abundance (Da Rosa 2019).
The species has benefitted from agricultural activity and was said to be common in wheatfields of the Western Cape (Stuart 1981; Williams et al. 2018). In north-eastern Limpopo communal lands, Zorilla were relatively more abundant in croplands than natural rangelands (Williams et al. 2018). The species has been known to reside close to human habitation (Stuart 1981).
Zorillas are carnivorous, with insects and murids predominating in their diet. They are also known to occasionally supplement their diet with reptiles, amphibians, birds, spiders, scorpions, centipedes and millipedes (Stuart 1981; Rautenbach 1982; Lynch 1983; Smithers 1983). Competition between this species and the African Striped Weasel is reduced somewhat by the Zorilla’s more diverse diet and adaptable habits (Rowe-Rowe 1978a). They have been observed scavenging (Shaw & Ryan 2015). The Zorilla has been observed to form foraging parties with the Cape Fox Vulpes chama, which appeared to be variably mutually beneficial to each (Panano et al. 2022).
In Maputaland, KwaZulu-Natal, Zorilla were detected less often at cameras where Leopards Panthera pardus were detected (Ramesh et al. 2017). At camera sites where Spotted Hyenas Crocuta crocuta were detected, Zorilla detection probability was higher in the closed open habitat (Ramesh et al. 2017). In the Succulent Karoo of the Northern Cape, Zorilla coexisted with other small carnivores and showed minor some degree of niche partitioning, mostly temporal there, and avoided the African Wild Cat at finer-scales (De Satge et al. 2017). There is no clear temporal separation of small carnivores from more dominant ones, and this should be investigated at finer scales (Smith et al. 2023). Zorilla are often detected in such low frequencies that it is not possible to examine temporal overlap (Smith et al. 2023).
Zorillas are generally solitary, but pairs and family groups are occasionally sighted (Rowe-Rowe 1992). They are entirely nocturnal (Shortridge 1934; Smithers 1971; De Satgé et al. 2017); Ramesh et al. 2017), resting during the day in rock crevices, burrows, under buildings or in hollow logs (Shortridge 1934; Smithers 1971; Stuart 1981).
In the assessment region, Zorillas mate and give birth between August and December (Rowe-Rowe 1978b; Stuart 1981; Rautenbach 1982). However, females who lose their young likely mate again, as demonstrated in captivity (Rowe-Rowe 1978b). Generally, one to three young are born after a gestation period of 36 days. Postnatal physical and behavioural development has been studied thoroughly (Rowe-Rowe 1978b). Rowe-Rowe (1992) estimated longevity in the wild at 4–5 years.
Ecosystem and cultural services: Zorillas may locally have a predatory impact on small rodents (e.g. Rattus spp.) and invertebrates. This is especially true in agricultural areas, where rodents feed on crops and insects the dung of domestic farm animals (Kingdon 1977). In pastures, Zorillas consume beetle larvae that feed on roots and grasses (Kingdon 1977).
In an agroecosystem of north-eastern Limpopo, Zorilla were detected in grazing rangelands and croplands, and given the proportion of rodents in their diet, could be a good candidate for an ecosystem service (Williams et al. 2018).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 8.1. Desert -> Desert – Hot | – | Suitable | – |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Marginal | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
There are reports of Zorillas being kept as pets after anal glands have been surgically removed (Kingdon 1977), as well as their scent being used as perfume (Shortridge 1934). This was reported in the previous assessment (Rowe-Rowe et al. 2016), though this is likely to be rare currently.
Locally, this species (especially its bones and organs) was used in traditional medicine, although the small-scale use was not expected to have any significant impact on the population (Rowe-Rowe et al. 2016). Use of the Zorilla has been documented in Eastern Cape (Simelane & Kerley 1998), Gauteng (Williams & Whiting 2016) and Western Cape (Nieman 2018; Nieman et al. 2020). In some areas of South Africa, there is an overall clientele consisting of 60 – 80% of South African citizens who seek cures from wild animals (Nieman 2018). In the Western Cape, there has been an increase in medicinal use trade (Nieman 2018). In traditional healer communities in the Western Cape, body parts such as skins of the Zorilla appeared in 23.5% of communities and they were of moderate importance there (Nieman 2018). The skin pieces of the Zorilla have been used to ostensibly protect from being attacked, create conflict and be a headache treatment (Nieman et al. 2018), and could be sold for R600 – R700 (Nieman 2018). In Gauteng, they were used to remedy bad luck (Williams & Whiting 2016).
In the Western Cape, the species had a Cultural Significance Index (CSI) of 1.5, which was low amongst several others that are a lot more important such as the Leopard for instance (Nieman 2018). The quantified use value had a low rating of 0.06, as it was not utilised to a high degree in that part of the Western Cape, and only utilised for medicinal purposes (Nieman 2018).
In the Eastern Cape, 10% of herbalist shops stocked the Zorilla (Simelane & Kerly 1998). In Gauteng, at the Faraday market, mammals were frequently used taxa in traditional medicine, and some species, including the Zorilla, had multiple uses (Williams & Whiting 2016), which was also the case in the Western Cape (Nieman et al. 2020). There may be regional importance of a species when it comes to medicinal uses, which may be a function of its regional abundance.
There has been a decline in interest to hunt Zorilla in South Africa, with large off-takes taking place in the Eastern Cape up until 2012 (SANBI Professional Hunters register, 2010-2019). In the North West, the hunting demand was not high for this species, as one permit was issued in 2014 and they have not been in great demand to hunt (NWPG 2017). Recently, in the province, since 2020, there have been no permit requests to hunt the species (North West Directorate of Biodiversity Management records, 2020-2023). This suggests waning interest in utilising the species.
As rarely seen carnivores, Zorillas are intriguing to see, and there is a desire by purist naturalist to see and view them. This has though not been quantified and the importance of it is unknown.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Bushmeat and traditional medicine. | Yes | Bushmeat and traditional medicine, and the pet trade. |
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | – | – | – | – |
| 3. Medicine – human & veterinary | – | – | – | – |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: (Not specified)
Threats
There are no major threats to the species. Most mortality is attributed to motor vehicles and free-roaming domestic dogs (Kingdon 1977), and in some areas it may be undergoing local declines due to increasing rural human populations and predation by domestic dogs (Stuart & Stuart 2013). Not only are Zorillas killed by free-roaming domestic dogs, but they also compete with them for food in many rural areas (Rowe-Rowe 1997).
Persecution killing
Zorilla are occasionally targeted and killed for preying on poultry (Rowe-Rowe 1992) or gamebird eggs and chicks (Rowe-Rowe 1997), and efforts have been aimed at reducing their abundance (Little & Crowe 2004).
Dogs
Dogs have been observed either disturbing or killing Zorilla in the Western Cape (Nieman et al. 2020). The use of hunting dogs has been one of the biggest threats to Zorilla populations (Rowe-Rowe et al. 2016).
Habitat management
Zorilla may be influenced by subtle changes in habitat, which may be linked to anthropogenic drivers, such as habitat management (Blaum et al. 2009; Schurch et al. 2021). Husbandry of rangeland had an influence on the occurrence of Zorilla, where they were impacted by excessively high stocking rates (Blaum et al. 2009), extended grazing by livestock (Schurch et al. 2021), and general lethal control (Schurch et al. 2021; Woodgate et al. 2023).
Zorilla had higher probabilities of occurrence outside protected areas than inside them, which has been found across a range of ecosystems from the arid Karoo and transitional Fynbos (Woodgate et al. 2023) to the humid coastal plain of Zululand (Da Rosa 2019).
Trophy hunting
Other than the Eastern Cape as the biggest utiliser of the species, provinces such as KwaZulu-Natal, Limpopo, Mpumalanga and North West feature, while surprisingly Northern Cape did not (SANBI Professional Hunters register, 2010-2019), which is interesting as there is likely to be an extensive population there (Figure 1).
Roadkill
There are several roadkill records from Northern Cape (Bullock et al. 2011) and North West (Power 2014). From January to September in 2007, 12 road mortality surveys were carried out along the R360 main road in the Northern Cape for a total distance of 2796 km (Bullock et al. 2011). On these transects, two Zorilla roadkills were discovered (Bullock et al. 2011). In the North West, all Zorilla roadkill records were on tarred surface roads and were in either in the Kalahari or Highveld (NWPG 2024). A local level road kill transect of ˜40 km was driven 39 time from May 202o to February 2021, circuiting the Mafikeng Game Reserve, and it revealed one Zorilla roadkill record, though this was the period during the Covid-19 lockdown (NWPG 2021). Later on, another larger circuit was developed to connect the towns of Lichtenburg and Ottoshoop, between November 2021 and February 2024, which was driven 71 times, and no Zorilla roadkills were recorded (NWPG 2022; 2023, 2024). In one case an adult and juvenile were recorded in the same roadkill event (NWPG 2022).
There may be regional patterns in roadkill mortality which may reflect local abundance.
Roadkills of adult female Zorilla and juvenile in December 2021 on R52 between Rustenburg and Koster (John Power)
Body parts use and trade
In the Western Cape, Zorilla appeared in 23.5% of communities which traded with animal parts (Nieman 2018). Zorilla body parts were increasingly more important in South Africa (Nieman et al. 2018).
Conservation
Zorillas are present in numerous protected areas within the assessment region, and it seems the medium-sized reserves may be more important in conserving the species.
Zorilla have been confirmed to still occur in the Tswalu Kalahari Reserve and Mountain Zebra National Park (Smith et al. 2023). In the North West Province, they have been recorded from both the Molopo Nature Reserve and !Khamab Kalahari Reserves (NWPG 2016; 2017). In the Verloren Vallei Nature Reserve, between 2015 and 2019, Zorilla were found at low frequency by camera traps (Loock & Swanepoel 2019).
Although they occur in the Kgalagadi Transfrontier Park, they do seem sparsely distributed there, and seem more common on farmlands to the south (Blaum et al. 2008; 2009). They are likely very rare in Kruger National Park and given local rarity on protected areas (Da Rosa 2019; Woodgate et al. 2023), farmlands and pastoral areas may also be important to their conservation. However, since they are not formally declared protected areas, the best we have are smaller to medium-sized reserves for them.
Currently, no conservation interventions are required. However, educational campaigns aimed at distinguishing between this species and the African Striped Weasel may help citizen scientists to improve monitoring capacity for these two species (Rowe-Rowe et al. 2016; Nieman et al. 2021).
Recommendations for land managers and practitioners:
- There are no management or conservation plans for this species, and it does not require any at this stage.
Research priorities:
- Quantification of the impact of roads, and especially mortality from motor vehicles, on this species.
- The use of citizen science in generating accurate population trends.
- More information is required on the species basic biology (e.g. reproduction) and ecology (e.g. home range size, habitat use).
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas. Please see Taxonomic notes for differentiating between the Zorilla and the African Striped Weasel.
- Roadkill sightings with clear photographs and accurate GPS coordinates can be submitted to the Endangered Wildlife Trust (email: roads@ewt.org.za; or download the EWT Road Watch app to submit data directly).
Bibliography
Blaum N, Engeman RM, Wasiolka B, Rossmanith E. 2008. Indexing small mammalian carnivores in the southern Kalahari, South Africa. Wildlife Research. 35(1):72-9.
Blaum N, Rossmanith E, Popp A, Jeltsch F. 2007. Shrub encroachment affects mammalian carnivore abundance and species richness in semiarid rangelands. Acta Oecologica. (1):86-92.
Blaum N, Tietjen B, Rossmanith E. 2009. Impact of livestock husbandry on small‐and medium‐sized carnivores in Kalahari savannah rangelands. The Journal of Wildlife Management. (1):60-7.
Bullock KL, Malan G, Pretorius MD. 2011. Mammal and bird road mortalities on the Upington to Twee Rivieren main road in the southern Kalahari, South Africa. African Zoology 46(1):60-71.
Da Rosa BRP. 2019. Optimizing Small Mammal Relative Abundance Measures Using Non-Invasive Sampling and Assessment of its Contribution to Occupancy Modelling of Small Carnivores in Dry Woodland Savannah of South Africa. Master’s thesis, University of Lisbon, Lisbon.
De Satgé J, Teichman K, Cristescu B. 2017. Competition and coexistence in a small carnivore guild. Oecologia 184:873-84.
Foden WB, Young BE. 2016. IUCN SSC Guidelines for Assessing Species’ Vulnerability to Climate Change. Version 1.0. Occasional Paper of the IUCN Species Survival Commission No. 59. Cambridge, UK and Gland, Switzerland: IUCN Species Survival Commission. x+114pp.
Gray A, Brito JC, Edwards CW, Figueiró HV, Koepfli KP. 2022. First complete mitochondrial genome of the Saharan striped polecat (Ictonyx libycus). Mitochondrial DNA Part B. 7(11):1957-60.
Hendrichs, H. 1972. Beobachtungen und Untersuchungen zur Ökologie und Ethologie, insbesondere zur sozialen Organisation ostafrikanischer Säugetiere. Zeitschrift für Tierpsychologie 30: 146-189.
Hollister N. 1915. The genera and subgenera of raccoons and their allies. United States National Museum: 143–150.
Kingdon, J. 1977. East African Mammals: An Atlas of Evolution in Africa. Volume IIIA (Carnivores). Academic Press, London, UK.
Little RM, Crowe TM. 2004. Effects of a predator control experiment on Grey-winged Francolin (Scleroptila africanus) populations. Ostrich-Journal of African Ornithology. 75(4):285-7.
Lynch, C.D. 1983. The mammals of the Orange Free State, South Africa. Navorsinge van die Nasionale Museum Bloemfontein 18: 1-218.
Lynch CD, Watson JP. 1990. The mammals of Sehlabathebe National Park, Lesotho: species accounts. Navorsinge van die Nasionale Museum: Researches of the National Museum. 6(12):530-43.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum , Pretoria, South Africa.
Milton SJ, Short S, Dean WR. 2022. Decline in whistling rat (Parotomys brantsii) density: Possible response to climate change in the Karoo, South Africa. African Journal of Ecology. 60(4):969-979.
Nieman WA. 2018. Culture, Conflict, and Cuisine: A Quantitative Assessment of Terrestrial Vertebrate Off-Take at the HumanWildlife Interface. Msc thesis, University of Stellenbosch, Stellenbosch.
Nieman, W.A., Leslie, A.J. and Wilkinson, A., 2018. The Use of Animals and Animal-Derived Constituents in African Traditional Medicine and Other Cultural Applications: Townships in the Western Cape Province. Culture, Conflict and Cuisine: A Quantitative Assessment of Terrestrial Vertebrate Off-Take at the Human-Wildlife Interface, p.215.
Nieman WA, Schultz BC, Wilkinson A, Leslie AJ. 2021. Stakeholders’ perceptions of mammal occurrence and abundance on agricultural properties bordering the Boland Mountain Complex, South Africa. African Zoology 56(2): 104-116.
NWPG (North West Provincial Government) 2016. Report examining the status of small carnivores and the detriment of hunting them in the Molopo Bushveld of the Dr Ruth Segomotsi Mompati District of the North West. Department of Rural, Environment and Agricultural Development, North West Provincial Government, Mahikeng.
NWPG (North West Provincial Government) 2017. The status of free ranging carnivores in Bokone Bophirima and impacts of hunting them. Department of Rural, Environment and Agricultural Development, North West Provincial Government, Mahikeng.
NWPG (North West Provincial Government) 2021 The impact of roadkill on mammal populations in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
NWPG (North West Provincial Government) 2022 The impact of roadkill on mammal populations in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
NWPG (North West Provincial Government) 2023 The impact of roadkill on mammal populations in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
NWPG (North West Provincial Government) 2024 The impact of roadkill on mammal populations in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Power RJ, Van Straaten A, Schaller R, Mooke M, Boshoff T, Nel HP. 2019. An inventory of mammals of the North West Province, South Africa. Annals of the Ditsong Museum of Natural History 8: 6-29.
Pretorius, M. 2019. Mesocarnivores in Protected Areas: ecological and anthropogenic determinants of habitat use in northern Kwa-Zulu Natal, South Africa. Msc thesis, University of Cape Town, Cape Town.
Ramesh T, Kalle R, Downs CT. 2017. Staying safe from top predators: patterns of co-occurrence and inter-predator interactions. Behavioral Ecology and Sociobiology. 71:1-4.
Rautenbach, I.L. 1982. The mammals of the Transvaal. Ecoplan Monograph 1:1-211.
Rowe-Rowe DT, Taylor PJ. 1996. Distribution patterns of terrestrial mammals in KwaZulu-Natal. South African Journal of Zoology 31: 131–144.
Rowe-Rowe DT. 1978a. Comparative prey capture and food studies of South African mustelines (Carnivores: Mustelidae). Mammalia 42: 175–196.
Rowe-Rowe DT. 1978b. Reproduction and post-natal development of South African mustelines (Carnivora: Mustelidae). Zoologica Africana 13: 103–114.
Rowe-Rowe DT. 1997. Striped polecat Ictonyx striatus. In: Mills G, Hes L. (ed.), The Complete Book of Southern African Mammals, pp. 204. Struik, Cape Town, South Africa.
Rowe-Rowe, D.T. 1992. The Carnivores of Natal. Natal Parks Board, Pietermaritzburg, South Africa.
Schurch MP, McManus J, Goets S, Pardo LE, Gaynor D, Samuels I, Cupido C, Couldridge V, Smuts B. 2021. Wildlife-friendly livestock management promotes mammalian biodiversity recovery on a semi-arid Karoo farm in South Africa. Frontiers in Conservation Science 2:6. https://doi.org/10.3389/fcosc.2021.652415
Shaw JM, Ryan PG. 2015. Winter scavenging rates under power lines in the Karoo, South Africa. African Journal of Wildlife Research. 45(1):122-6.
Shortridge, G.C. 1934. The mammals of South West Africa: a biological account of the forms occurring in that region. Heinemann, London, UK.
Simelane TS, Kerley GI. 1998. Conservation implications of the use of vertebrates by Xhosa traditional healers in South Africa. South African Journal of Wildlife Research (4):121-6.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smith K, Venter JA, Peel M, Keith M, Somers MJ. 2023. Temporal partitioning and the potential for avoidance behaviour within South African carnivore communities. Ecology and Evolution. 13(8):e10380.
Smithers, R.H.N. 1971. The mammals of Botswana. National Museums of Rhodesia, Museum Memoir 4: 1-340.
Smithers, R.H.N. 1983. The Mammals of the Southern African Subregion. University of Pretoria, Pretoria, South Africa.
Stuart, C.T. 1981. Notes on the mammalian carnivores of the Cape Province, South Africa. Bontebok 1: 1-58.
Stuart, C. and Stuart, T. 2013. Ictonyx striatus Zorilla. In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. Volume V: Carnivores, Pangolins, Equids and Rhinoceroses, pp. 93-97. Bloomsbury Publishing, London, UK.
Webster AB, Pretorius ME, Somers MJ. 2021. The determinants of mesocarnivore activity patterns in highveld grassland and riparian habitats. African Journal of Wildlife Research. 51(1):178-92.
Williams ST, Maree N, Taylor P, Belmain SR, Keith M, Swanepoel LH. 2018. Predation by small mammalian carnivores in rural agro-ecosystems: an undervalued ecosystem service?. Ecosystem Services 30: 362-371.
Williams VL, Whiting MJ. 2016. A picture of health? Animal use and the Faraday traditional medicine market, South Africa. Journal of Ethnopharmacology. 179:265-73.
Winter, S., Fennessy, J., Janke, A. and Nilsson, M.A. 2023. Northern olingo (Bassaricyon gabbi), zorilla (Ictonyx striatus), and honey badger (Mellivora capensis) mitochondrial genomes and a phylogeny of Musteloidea. Frontiers in Ecology and Evolution, 10, p.1089641.
Woodgate Z, Drouilly M, Distiller G, O’Riain MJ. 2023. The effect of multi-use landscapes on mammal assemblages and its implication for conservation. Land.12(3):599. https://doi.org/10.3390/land12030599
Young, T.P. and Evans, M.R. 1993. Alpine vertebrates of Mount Kenya, with particular notes on the rock hyrax. Journal of the East Africa Natural History Society and National Museum 82(202): 55-79.