Yellow-spotted Rock Hyrax
Heterohyrax brucei

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Heterohyrax brucei – (Gray, 1868)
ANIMALIA – CHORDATA – MAMMALIA – HYRACOIDEA – PROCAVIIDAE – Heterohyrax – brucei
Common Names: Bush Hyrax, Hoggar Hyrax, Yellow-spotted Hyrax, Yellow-spotted Rock Hyrax (English), Geelkoldassie, Geelkosdas (Afrikaans), Imbila (Ndebele), Tshwanyê (Setswana), Busch Schliefer (German), Daman de steppe (French)
Synonyms: Hyrax brucei Gray, 1868; Heterohyrax antineae (Heim de Balsac & Begouen, 1932); Heterohyrax chapini (Hatt, 1933)
Taxonomic Note:
Two subspecies have been described in southern Africa (Meester et al. 1986): H. b. ruddi, with a range that extends from western Mozambique to eastern Botswana and H. b. granti, in the Limpopo Province of South Africa, which may be endemic to South Africa (Wroughton 1910). It is the only species that belongs to the genus Heterohyrax but taxonomy is currently considered uncertain, and distinct differences may exist between the two subspecies. Overall, 25 subspecies have been described, and while the validity of many is in doubt, some may represent distinct species (Barry & Shoshani 2000; Barry & Hoeck 2013). There is an endemic subspecies, H. b. granti, which is suspected to be a separate species that will in future be in need of independent assessment. Furthermore, molecular research (based on mitochondrial DNA and nuclear DNA sequences) reveals two distinct genetic lineages (P. Bloomer unpubl. data): one restricted to the Limpopo valley and the other from the Soutpansberg south and east to the Mpumalanga escarpment. Confirming the taxonomy is crucial as the status of some of the range-restricted forms may be threatened.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Smith, C.,1 & da Silva, J.2
Reviewer: Patel, T.1
Institutions: 1Endangered Wildlife Trust, 2South African National Biodiversity Institute
Previous Assessors: Bloomer, P., Kruger, J. & Maguranyanga, M.
Previous Reviewer: Child, M.F.
Previous Contributor: Relton, C.
Assessment Rationale
Listed as Least Concern in view of its wide distribution within the assessment region, its occurrence in a number of protected areas (including Kruger National Park and Mapungubwe National Park), and because there are no major threats expected to cause population decline. Additionally, its distribution is connected with other range states through largely intact habitat. Its preferred habitat (rocky outcrops) is unlikely to be extensively transformed. Pending taxonomic resolution, this species (currently listed as the subspecies H. b. granti) may require reassessment if it is shown to be endemic to South Africa.
Regional population effects: There are habitat linkages with Botswana, Mozambique and Zimbabwe, and thus dispersal is probable (Barry & Shoshani 2000), and this species occurs in both the Greater Limpopo Transfrontier Park and Greater Mapungubwe Transfrontier Park. They are suspected to have adequate dispersal abilities (Hoeck 1982; Skinner & Chimimba 2005), and thus rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Smith C & da Silva JM. 2025. A conservation assessment of Heterohyrax brucei. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
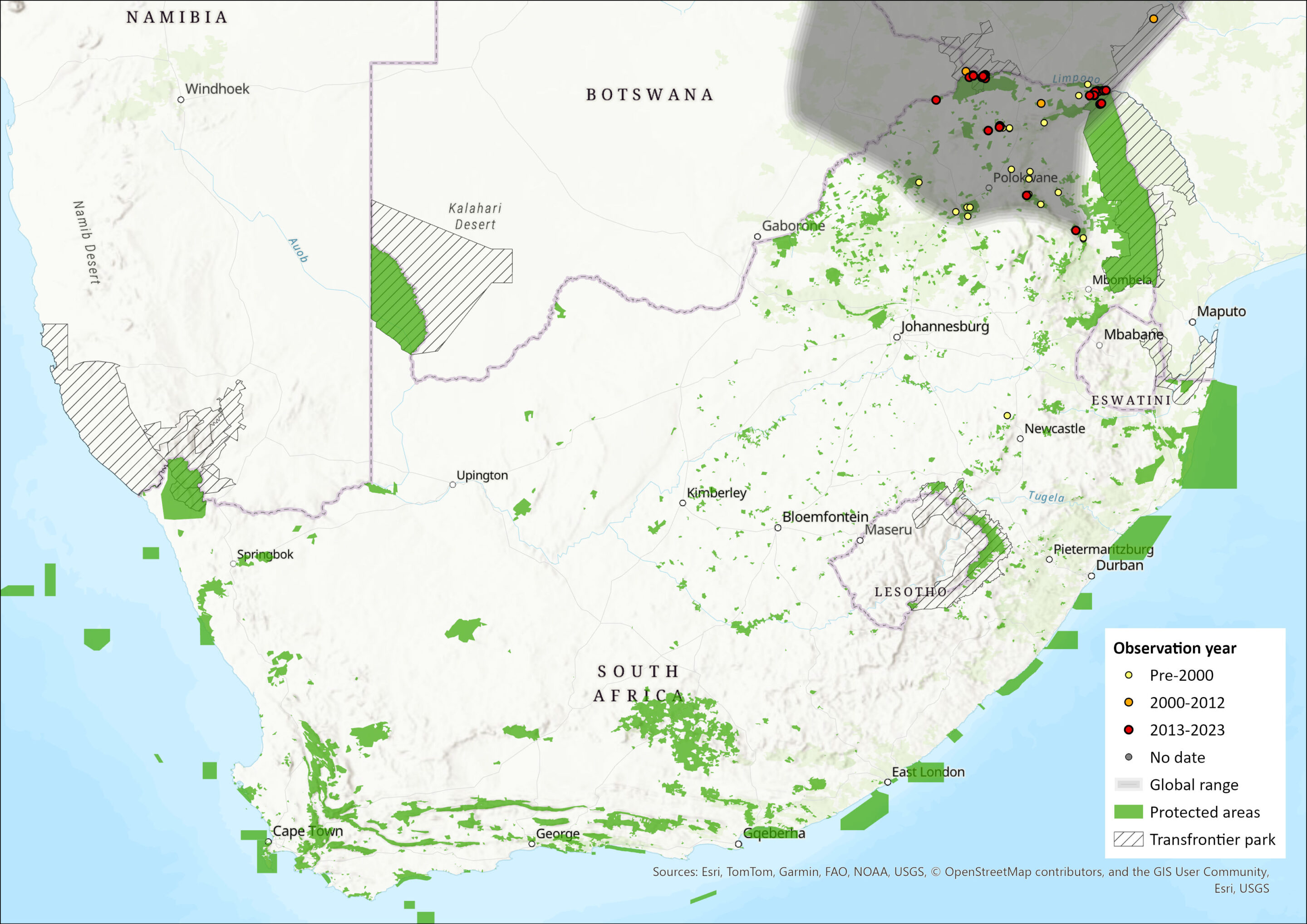
Yellow-spotted Rock Hyraxes are endemic to the continent and extensively distributed across southern and East Africa (Skinner & Chimimba 2005; Barry & Hoeck 2013). Their range extends from the Limpopo Province of South Africa northwards to Sudan, Ethiopia and Somalia (Barry & Hoeck 2013). Historical gene flow may have occurred between Mapungubwe and Matopos (Zimbabwe) (P. Bloomer unpubl. data), which may represent the range of H. b. ruddi. Northern Namibia needs to be investigated in terms of species identity, where individuals may belong to either Procavia or Heterohyrax. Limpopo and Mpumalanga provinces mark the southern edge of its distribution. Across this range, they are limited to rocky habitats (Skinner & Chimimba 2005).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 3800
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Yellow-spotted Rock Hyrax (Heterohyrax brucei) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified
Biogeographic Realms
Biogeographic Realm: Afrotropical, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | Resident |
| Botswana | Extant | Native | – | Resident |
| Burundi | Extant | Native | – | Resident |
| Congo, The Democratic Republic of the | Extant | Native | – | Resident |
| Djibouti | Extant | Native | – | Resident |
| Eritrea | Extant | Native | – | Resident |
| Ethiopia | Extant | Native | – | Resident |
| Kenya | Extant | Native | – | Resident |
| Malawi | Extant | Native | – | Resident |
| Mozambique | Extant | Native | – | Resident |
| Namibia | Presence Uncertain | Native | – | Resident |
| Somalia | Extant | Native | – | Resident |
| South Africa | Extant | Native | – | Resident |
| South Sudan | Extant | Native | – | Resident |
| Sudan | Extant | Native | – | Resident |
| Tanzania, United Republic of | Extant | Native | – | Resident |
| Uganda | Extant | Native | – | Resident |
| Zambia | Extant | Native | – | Resident |
| Zimbabwe | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
No recent research or literature has focused on how climate change might affect this species in the future; however, populations have been seen to fluctuate, due to reduced fecundity, based on rainfall patterns in the past (Barry & Mundy 1998). Increased aridity and prolonged drought conditions threaten to limit the availability of forage resources (Bartholomew & Rainy 1971). More recent research is needed on this.
Population
They are widespread and common throughout East Africa, with population densities declining and distributions becoming more localized towards southern Africa (Barry & Hoeck 2013). Densities in the Matobo National Park in Zimbabwe ranged from 0.5–1.1 individual / ha from 1992–1996 (Barry & Mundy 1998). Similar densities are expected within South African protected areas. Rainfall and, to a lesser extent, predation appear to be the primary factors affecting population abundance (Barry & Mundy 1998). Population fluctuations have been shown to be annual (largely corresponding to rainfall) but not cyclical (Barry et al. 2015). Fossil remains indicate there were once hyraxes the sizes of oxen, which may explain its gestation period of seven or eight months, unusually long for an animal of its size.
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| No | – | – |
Current population trend: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of subpopulations: Unknown. While Friedmann and Daly (2004) listed two subpopulations (Limpopo valley for H. b. ruddi and the Soutpansberg–Mpumalanga escarpment for H. b. granti), further molecular work is needed to determine population structure.
Severely fragmented: No. Habitat largely connected and dispersal capacity adequate.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
To date, no population genetic studies have been undertaken on this species within or including animals from the assessment region; however, animals in Tanzania have been investigated using microsatellite data (Gerlack & Hoeck 2001). Despite the studied Tanzanian metapopulation having low genetic diversity, no inbreeding was detected, likely owing to female dispersal patterns.
It is assumed the species exists as a metapopulation within the assessment region; however, this would need to be confirmed with molecular evidence. It is possible genetic structure may be detected. Despite the lack of information, it is assumed that all potential (sub)populations are maintained (still existing), equating to a populations maintained (PM) indicator value of 1.0. Given limited information on population size and density, the effective population size (Ne) for the metapopulation cannot be quantified, and hence neither can the Ne 500 indicator.
Habitats and ecology
The Yellow-spotted Rock Hyrax is predominantly diurnal, highly gregarious and exhibits behavioural thermoregulation (Bartholomew & Rainy 1971). Similar to, and often in association with Procavia capensis, the Yellow-spotted Rock Hyrax occupies rocky habitats, including outcrops, cliffs and piles of boulders (Barry & Shoshani 2000; Barry & Hoeck 2013). However, in contrast to P. capensis, outlying or isolated rocky outcrops are less likely to be occupied by this species (Smithers & Wilson 1979; Skinner & Chimimba 2005). Yellow-spotted Rock Hyraxes and Rock Hyraxes often coexist within the same crevices and are seen basking together on rocky outcrops (Skinner & Chimimba 2005), but no interbreeding occurs between the species since both their mating behaviours and the anatomy of their sex organs differ. This heterospecific behaviour is considered beneficial for the avoidance of predators by increasing group size, and consequently improving vigilance, particularly when offspring are present (Barry & Mundy 2002). Births are often associated with peaks in rainfall. They are predominantly browsers but are known to infrequently supplement their diets with grasses (Hoeck 1975). They have a well-developed digestive system and are hind gut fermenters (Freeman 2018). The bulk of their diets consist of leaves, stems, flowers, fruit and bark. They are cautious feeders, stopping on each mouthful to scan the surroundings and make short foraging trips close to their family base. Barry and Hoeck (2013) and Barry and Shoshani (2000) provide comprehensive reviews of its ecology and characteristics. The very prominent distinguishing features are the white eyebrows, grey colour and the yellow hairs surrounding the dorsal spot, although the latter is variable across their broader distribution. They also have a rounded head and blunt nose that is pointed and rodent-like, which helps in identifying this species.
Ecosystem and cultural services: It is a primary prey species for a range of predators, including the Verreaux’s Eagle (Aquila verreauxii), Martial Eagle (Polemaetus bellicosus) and other raptors, as well as terrestrial species such as Leopard (Panthera pardus), Black Mamba (Dendroaspis polylepis) and African Rock Python (Python sebae) (Turner & Watson 1965; Hoeck 1982). Adult hyrax species comprise more than 84% of the prey biomass for a diverse range of predators in Matobo National Park, Zimbabwe (Barry & Mundy 1998).
An association with H. brucei has been found to benefit the survival success of young P. capensis, as H. brucei often browse in trees and consequently act as sentinels, warning both species of impending danger (Barry & Mundy 2002).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | No |
| 6. Rocky areas (e.g. inland cliffs, mountain peaks) | – | Suitable | No |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: 16 months (Freeman 20218)
Age at Maturity: Male: 28 months (Freeman 20218)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 6-8 months (Freeman 20218)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: 3 individuals (Freeman 2018)
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Similar to P. capensis, it is hunted for meat and skins opportunistically. Their skins are used for blankets (karosses) in South Africa (Barry & Hoeck 2013). However, this is not considered to negatively influence population stability.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Bushmeat use. | Yes | Local trade in skins. |
National Commercial Value: No
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | – | – | – |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: (Not specified)
Threats
There are no significant threats to this species. However, the expansion of human populations adjacent to protected areas (sensu Wittemyer et al. 2008) may lead to local decline due to hunting for meat and skins. Similarly, mining activities in the Limpopo valley may threaten local subpopulations through direct habitat encroachment or indirect disturbance. Previous observations have found that Rock Hyrax populations have been influenced by drought conditions by reducing fecundity (Barry & Mundy 1998). Yellow-spotted Rock Hyraxes respond behaviourally to heat stress, by seeking shelter in caves and crevices (Turner & Watson 1965) and, when necessary, are able to meet water requirements from browse material alone (Skinner & Chimimba 2005). Additionally, the spread of disease is rapid among gregarious hyrax species and H. brucei is characterised as susceptible to viral pneumonia and tuberculosis (Sale 1969). For example, in the Serengeti an outbreak of mange eliminated an entire colony (Hoeck 1982).
The subspecies H. b. ruddi is not considered to be under any great threat throughout the region, and it is likely that populations are interlinked between those in Botswana and Zimbabwe (Barry & Shoshani 2000). In contrast, H. b. granti, which is endemic to South Africa, may be more threatened by such activities.
Current habitat trend: Stable. They occupy rocky outcrops that are largely inaccessible and not under threat of extensive transformation and the savannah is not threatened within the assessment region (Driver et al. 2012). However, ongoing rural and urban settlement expansion may be causing local habitat loss and increasing the risk of bushmeat harvesting. In Limpopo Province, between 2000 and 2013, rural and urban settlements expanded by 8.8% and 14.9% respectively (GeoTerraImage 2015).
Conservation
Yellow-spotted Rock Hyraxes occur locally in a number of protected areas, including Kruger National Park and Mapungubwe National Park, and their associated transfrontier areas. At present, no specific conservation interventions have been identified. However, the expansion of protected areas and the implementation of biodiversity stewardship policies to safeguard primary habitats could significantly benefit Yellow-spotted Rock Hyraxes, and in particular the subspecies H. b. granti.
Recommendations for land managers and practitioners: This species may benefit from the establishment of buffer zones around primary habitats.
Research priorities: A taxonomic and genetic study would be valuable to resolve the taxonomy of the potential species complex. Additionally, survey studies are necessary to investigate the population density and range distribution of this species. Particular attention should be paid to H. b. granti as this subspecies is endemic to South Africa. Methods to estimate the area of occupancy of this species would also improve the accuracy of this assessment.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Avoid feeding or keeping as pets.
Bibliography
Barry RE, Chiweshe N, Mundy PJ. 2015. Fluctuations in bush and rock hyrax (Hyracoidea: Procaviidae) abundances over a 13-year period in the Matopos, Zimbabwe. South African Journal of Wildlife Research 45: 17-27.
Barry RE, Mundy PJ. 2002. Seasonal variation in the degree of heterospecific association of two syntopic hyraxes (Heterohyrax brucei and Procavia capensis) exhibiting synchronous parturition. Behavioral Ecology and Sociobiology 52: 177-181.
Barry, R.E. and Hoeck, H.E. 2013. Heterohyrax brucei Bush Hyrax. In: J.S. Kingdon, D.C.D. Happold, Hoffmann, T.M. Butynski, M. Happold and J. Kalina (eds), Mammals of Africa. Vol. I: Introductory Chapters and Afrotheria, pp. 161-165. Bloomsbury Publishing, London.
Barry, R.E. and Mundy, P.J. 1998. Population dynamics of two species of hyraxes in the Matobo National Park, Zimbabwe. African Journal of Ecology 36: 221-233.
Barry, R.E. and Shoshani, J. 2000. Heterohyrax brucei. Mammalian Species 645: 1–7.
Bartholomew GA, Rainy M. 1971. Regulation of body temperature in the rock hyrax, Heterohyrax brucei. Journal of Mammalogy 51: 81-95.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Freeman, M.S. 2018. Hyracoidea. In: Vonk, J., Shackelford, T. (eds) Encyclopedia of Animal Cognition and Behavior. Springer, Cham. https://doi.org/10.1007/978-3-319-47829-6_1133-1
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. Conservation Breeding Specialist Group (SSC/IUCN) and Endangered Wildlife Trust, Parkview, South Africa.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Gerlach, G. & Hoeck. 2001. Islands on the plains: metapopulation dynamics and female biased dispersal in hyraxes (Hyracoidea) in the Serengeti National Park. Molecular Ecology 10(9): 2307-2317.
Hoeck HN. 1975. Differential feeding behaviour of the sympatric hyrax Procavia johnstoni and Heterohyrax brucei. Oecologia 22: 15-47.
Hoeck HN. 1982. Population dynamics, dispersal and genetic isolation in two species of hyrax (Heterohyrax brucei and Procavia johnstoni) on habitat islands in the Serengeti). Zeitschrift für Tierpsychologie 59: 177-210.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum , Pretoria, South Africa.
Sale JB. 1969. Breeding season and litter size in Hyracoidea. Journal of Reproduction and Fertility, Supplement 6: 249-263.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smithers, R.H.N. and Wilson, V.J. 1979. Check List and Atlas of the Mammals of Zimbabwe Rhodesia. Trustees of the National Museums and Monuments of Rhodesia, Salisbury, Rhodesia.
Turner MIM, Watson RM. 1965. An introductory study on the ecology of hyrax (Dendrohyrax brucei and Procavia johnstoni) in the Serengeti National Park. African Journal of Ecology 3: 49-60.
Wittemyer, G., Elsen, P., Bean, W.T., Burton, A.C.O. and Brashares, J.S. 2008. Accelerated human population growth at protected area edges. Science 321: 123-126.
Wroughton RC. 1910. New African mammals of the genera Cricetomys and Procavia. Journal of Natural History 5: 106-110.