Yellow Mongoose
Cynictis penicillata

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Cynictis penicillata – (G.[Baron] Cuvier, 1829)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – HERPESTIDAE – Cynictis – penicillata
Common Names: Yellow Mongoose (English), Geel meerkat, Geelmuishond, Rooimeerkat, Witkwasmuishond (Afrikaans), Mosha, Pipi (Sepedi, Sesotho), Moa, Mofse, Mofê, Ramofse (Tswana), Fuchsmanguste (German), Mangouste fauve, Mangouste jaune (French), Mangusta gialla (Italian)
Synonyms: No Synonyms
Taxonomic Note:
Cynictis penicillata belongs to the clade of solitary mongooses, the subfamily Herpestinae (Veron et al. 2004), and is the sister-taxon of Selous’ Mongoose (Paracynictis selousi) (Flynn et al. 2005). No phylogeographical structure or subspecies delimitation was found by an analysis of molecular variance (van Vuuren & Robinson 1997).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Adams, E.C.1 & da Silva, J.M.2
Reviewer: Do Linh San, E.3
Institutions: 1Endangered Wildlife Trust,2South African National Biodiversity Institute,3Sol Plaatje University, South Africa
Previous Assessors: le Roux, A., Balmforth, Z., Mbatyoti, K.V., Bizani, M. & Avenant, N.
Previous Reviewers: Cavallini, P. & Do Linh San, E.
Previous Contributors: Child, M.F.
Assessment Rationale
Within the assessment region there is no reason to change the status of this species from Least Concern. The Yellow Mongoose is relatively widespread, common and resilient, adaptable to change (whether anthropogenic or not), and is not facing any immediate threats to its distribution or population. Its distribution in Eswatini and Lesotho needs to be assessed. As it is often persecuted for its perceived status as a damage-causing animal, the main interventions for this species are awareness and education campaigns for landowners and the provision of incentives for sustaining a natural prey base.
Regional population effects: There is dispersal between regions and its range is continuous.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: Not change
Red List Index
Red List Index: No change
Recommended citation: Adams EC & da Silva JM. 2025. A conservation assessment of Cynictis penicillata. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
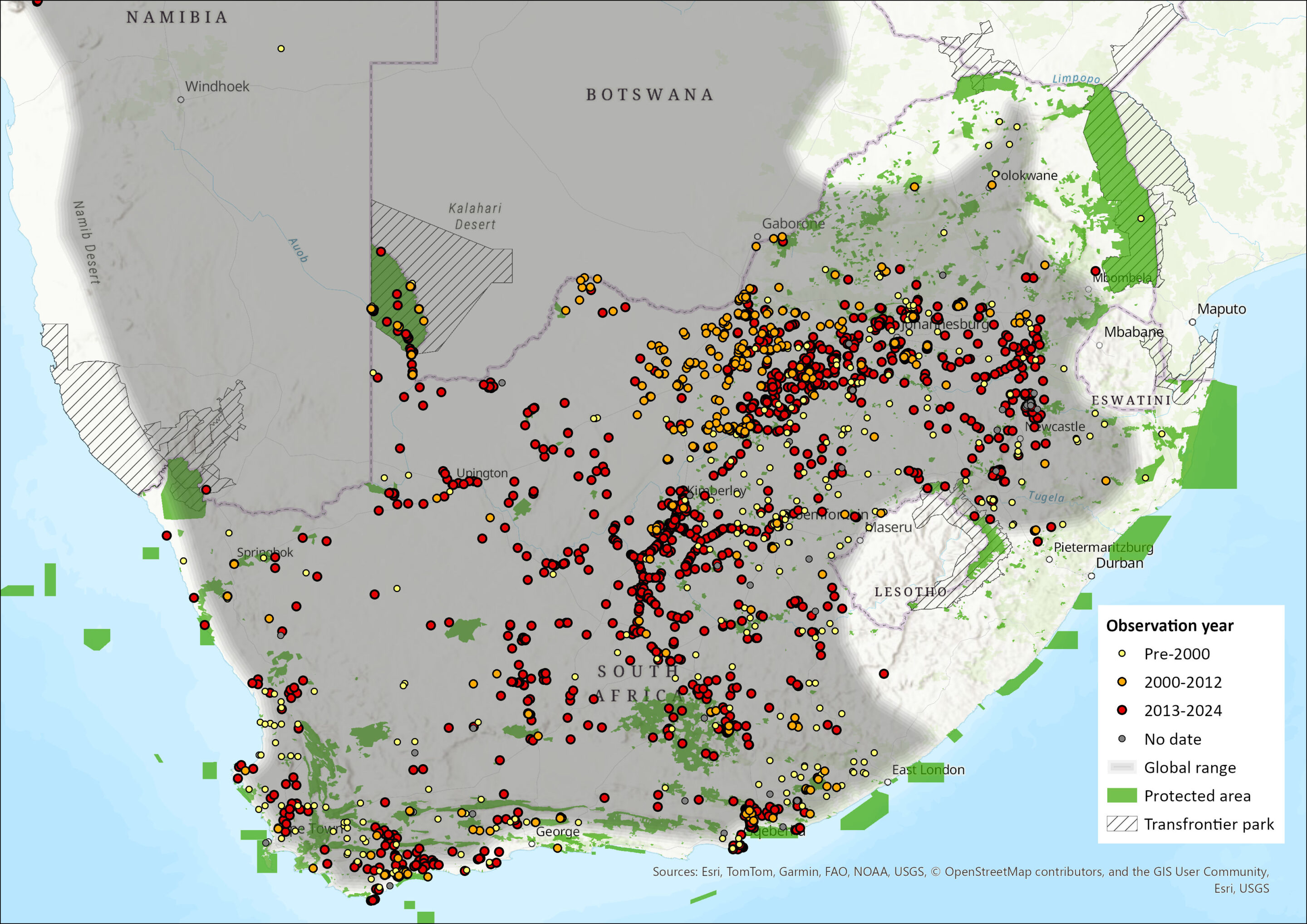
The Yellow Mongoose is largely confined to southern Africa. Within this range, it occupies the drier western parts, being widespread in Namibia, Botswana – as far north as the top of Chief’s Island in the Okavango Delta (K. Collins pers. comm. 2016) – and much of central and western South Africa (Lynch 1989). It is however absent from the Namib Desert and the Caprivi Strip in Namibia, the Lesotho Highlands (Lynch 1994, Avenant 2004, ERM 2010), and the easterly regions of southern Africa (Taylor 2013). The status in Zimbabwe is unclear: the species appears generally absent, except for an unconfirmed sighting in Hwange National Park suggesting that it is still present in the Zambezi basin (Purchase et al. 2007).
Within the assessment region, Yellow Mongooses occur everywhere, except in the south coast forest, the east coast (from East London northwards), the Lesotho Highlands, and the lowveld and bushveld areas of Limpopo Province (Figure 1). Several recent studies referred to below (see Population and Habitats and Ecology) confirm the existence of healthy populations of Yellow Mongooses at various locations in South Africa. For example, in the Free State Province, they have been recorded from both protected areas, including Sandveld Nature Reserve (Avenant and Watson 2002), Soetdoring Nature Reserve (Kuyler 2000), Tussen-die-Riviere Nature Reserve (Watson 2006), Golden Gate National Park (Avenant 1997), and in modified or disturbed areas (Avenant 2008, 2011a, 2011b), including farmlands in the Western Cape (Vidya et al. 2009). There have also been numerous recent sightings and rabies cases (with Yellow Mongooses as the vector) reported in the eastern Free State (Bishop et al. 2010). Yellow Mongooses are common in the Northern Cape Province (Bishop et al. 2010), including the rangelands (Blaum et al. 2007, 2008). In the North West Province, a recent survey revealed that they were common in the arid south and west regions, especially the southern grasslands, but few were recorded in the Kalahari habitats of the Molopo region (Power 2014). Additionally, a small carnivore survey confirmed their presence in Namibia (Kauffman et al. 2007).
Apart from Lesotho, this current distribution is not significantly different from the species’ historical distribution. Yellow Mongooses are expected to occur in natural habitat in the western lowlands of Lesotho, as they are common in the eastern Free State on the northwest side of the Caledon River (Lynch 1975, N.L. Avenant pers. obs. 1995–2016), but there are only six sightings recorded since the early 1980s (Lynch 1994). Similarly, they have not been recorded from Eswatini (Monadjem 1998), although, based on suitable habitat, they may well occur in this country. A range expansion into the south coast of the Western Cape, due to the expansion of crop agriculture, especially wheat lands, in these regions, may have occurred over the past decades (Figure 1, Taylor and Meester 1993).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Yellow Mongoose (Cynictis penicillata) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
No research has focused on how the Yellow Mongoose will be affected by climate change in the future, however research has been conducted on similar sized species such as Suricates (Suricata suricatta) and Cape Ground Squirrels (Xerus inauris) in arid environments (Fuller et al. 2021). It has been noted that smaller animals such as the Yellow Mongoose may be at risk as temperatures continue to increase as a result of the greater surface area to volume ratio, resulting in them increasing their body temperature faster than larger animals (Fuller et al. 2021). This could put particularly the arid populations more at risk of extinction if they fail to regulate their activity patterns accordingly, such as through practicing crepuscular rather than diurnal activity.
Population Information
The Yellow Mongoose population appears to be stable. Population densities vary across the assessment region, probably correlating with habitat productivity and predation pressure, with the highest densities reported for grassland regions of South Africa. For example, Earlé (1981) reported a density of 133–200 individuals / km2 on a site on the Vaal River, which was an island for much of the year and thus probably enabled high population density. A population density of 6–7 individuals / km2 was estimated in the West Coast National Park of South Africa (Cavallini 1993a, Cavallini and Nel 1995). Similar densities were observed in the Great Fish River Nature Reserve (Eastern Cape; 4–10 individuals / km2; E. Do Linh San unpubl. data). Two widely distant and well-studied sites (Kuruman River Reserve near Vanzylsrus, Northern Cape, and farmland along the south coast, near Bredasdorp/Heidelberg, Western Cape) also exhibited thriving subpopulations (Vidya et al. 2009). The density for the farmland was 23–26 individuals / km2 during the period 2001–2003 (Balmforth 2004), whereas that for Kuruman River Reserve was 4–14 individuals / km2 (Le Roux et al. 2008, 2009).
Research has focused on groups within and outside of protected areas, highlighting that this species is not restricted to nature reserves (e.g., Le Roux et al. 2009; Makenbach et al. 2013; see also Lynch 1980, 1983, 1989). It is flexible in terms of social behaviour, diet, and heterospecific interactions (see Habitats and Ecology). Often thriving in farmland, it is not strongly affected by a growing human population size. Its highly adaptable behaviour will likely ensure the survival and growth of the Yellow Mongoose population across southern Africa. Overall, studies reporting on mongoose behaviour or rabies outbreaks continue to strongly suggest that this species is common (e.g., Davis et al. 2007), widely distributed, and adaptable. As such, given their large range and high densities, we infer there are > 10,000 mature individuals within the assessment region and that the population is stable.
Current population trend: Stable
Continuing decline in mature individuals: No. However, local declines are expected in areas of high human density or sprawl.
Number of mature individuals in population: > 10,000
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown, but likely to constitute one large, connected population (van Vuuren & Robinson 1997).
Severely fragmented: No. This species can survive in multiple habitat and land-use types.
Population Information
Continuing decline in mature individuals? (Not specified)
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: Unknown
Number of mature individuals in largest subpopulation: (Not specified)
Number of Subpopulations: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
A population genetic study utilising Rapid Fragment Length Polymorphisms (RLPFs) found no correspondence between mtDNA phylogeography and previous subspecies designations, nor did it find any genetic structure across the assessment region (van Vuuren & Robinson 1997). While RFLPs are generally considered less robust due to their lower resolution, the findings support current thinking related to the dispersal capabilities of the species and them forming a contiguous population. With this information and given the current extent of the species range, it is assumed no previous genetically distinct group has gone extinct. Consequently, the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF’s) complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (all populations remaining).
While precise population sizes (e.g. census [Nc]) or effective population size (Ne) data do not exist, it is assumed the population is stable with over 10,000 individuals in the assessment region. Even though the 10,000 value is not an actual census count, this quantity can still be used as a proxy for quantifying the GBF’s headline genetic indicator – proportion of populations with an Ne > 500. Based on the assumption that the species is likely to occur as a single metapopulation, the Ne of the species is ~1,000-3,000 individuals, based on a Ne/Nc conversion ratio of 0.1-0.3. Having the 1 population above Ne 500, translates to an indicator score of 1.0 (1/1 population with Ne > 500).
It is recommended that a follow-up population genetic study be undertaken using more fine-scale molecular techniques to better examine the genetic structure and diversity (health) within the species, and to more accurately quantify the indicators.
Habitats and ecology
The Yellow Mongoose is widespread and adaptable to many habitats, including human-transformed landscapes. It appears to prefer areas with short grass and/or shrub, with soft to medium-hard sand. Generally, this species is found on “burrow-able” substrate in savannah, shrubland, grassland and arid environments at various altitudes (e.g., Taylor and Meester 1993, Popp et al. 2007). Rocky substrate and heavy clay soils are likely to be avoided due to reliance on boltholes/sleeping burrows with multiple entrances. Within urban environments, Yellow Mongoose tend to preferentially select open grasslands (Cronk & Pillay 2019). Sleeping burrows may be shared with other terrestrial species, including Suricates and Cape Ground Squirrels (e.g., Lynch 1980, Le Roux et al. 2009), and these associations have been hypothesised to mutually facilitate predator detection, at least locally and/or punctually (Do Linh San and Somers 2006, Makenbach et al. 2013). Shrubs are important refuges and are often – but not always – preferred den sites when nursing young and are utilised to protect offspring from avian predators (Rasa et al. 1992, Blaum et al. 2007).
This mongoose species is essentially diurnal, with emergence from, and return to the sleeping burrow correlating with sunrise and sunset times, respectively (Cavallini 1993b, Mbatyoti 2012). Some early evening activity can, however, be recorded, especially on warm and bright nights (E. Do Linh San pers. obs. 2005–2011). During the day, activity is generally negatively affected by adverse climatic conditions, such as very hot or conversely low temperatures, rainy weather or wet soil (Mbatyoti 2012). Urban mongoose were seen to be more crepuscular with activity peaks in the early morning or late afternoon (Cronk & Pillay 2019b). The diet is primarily based on invertebrates (essentially beetles, termites and grasshoppers/locusts/crickets), although mammals, small birds and reptiles may be hunted opportunistically (Avenant and Nel 1992, Taylor and Meester 1993, Nel and Kok 1999, Bizani 2014; Cronk and Pillay 2019a). Arthropods are consumed throughout the year, but particularly so in summer and autumn, whereas mammal consumption tends to peak in winter and spring (Bizani 2014). The diurnal Four-striped Mice (Rhabdomys spp.) were found to be the main rodent prey in the Great Fish River Nature Reserve (Bizani 2014), and this may well be the case in most of the Yellow Mongoose’s range. Within urban environments, anthropogenic food items are also exploited, particularly during the winter months (Cronk & Pillay 2019b).
The Yellow Mongoose is a facultatively social species: den, territory and sometimes foraging excursions may be cooperatively shared with family members in areas of high habitat saturation (Balmforth 2004, Le Roux et al. 2009, Vidya et al. 2009). Variations in social organisation and social behaviour seem to be affected by habitat productivity, with richer habitats allowing for higher population density, larger group sizes and more complex social interactions. In low density populations, females defend a group home range, males have much larger ranges (up to four times bigger) that overlap broadly with each other and with those of males from numerous neighbouring groups, and no regular border patrolling is observed (Cavallini 1993a, Mbatyoti 2012). At slightly higher density, only alpha males are responsible for territory defence and border scent-marking, whilst subordinates only mark near the cores (Le Roux et al. 2008). In medium- to high-density populations both males and females defend a common territory (Balmforth 2004, Earlé 1981). The group territory is used equally by all group members (Balmforth 2004) and territory limits are patrolled regularly and scent-marks and faeces deposited at border latrines (Earlé 1981). Individual home range size varies between 0.05 km2 and 2.4 km2 (Earlé 1981, Cavallini 1993a, Balmforth 2004, Le Roux et al. 2008, Mbatyoti 2012), with smaller ranges expectedly observed in higher-density populations and likely richer habitats. Urban Yellow Mongooses had an average home range size of 0.12 km2 (Cronk & Pillay 2019b).
Yellow Mongooses reproductive season generally runs from August to February, with a first peak of oestrus in August and a second in October or December (Rasa et al. 1992). Gestation period is 60–62 days and litter size at emergence from the burrow varies between one and three young, on average two (Rasa et al. 1992). A study carried out in a medium-density population revealed that Yellow Mongooses may exhibit alloparental behaviour (cooperative breeding) – including guarding, feeding and grooming others’ young – and communal vigilance (Balmforth 2004). Alarm vocalizations are used almost exclusively when individuals are in a group rather than solitary (Le Roux et al. 2008).
The Yellow Mongoose is the primary host for the viverrid strain of rabies endemic to the interior plateau of South Africa, where its population densities are highest (Ngoepe et al. 2024). The role of this species as a maintenance host is promoted by its diurnal and colonial burrow-living habits (Taylor 2013).
Ecosystem and cultural services: Similar to other herpestids, the Yellow Mongoose is a prolific snake mobber; yet, the species’ snake fighting abilities have not translated into common folklore or other stories. The current image of the Yellow Mongoose is that it is a pest that preys on lambs (reportedly eating the lamb alive, starting at the nose), is a dangerous carrier of rabies (Bishop et al. 2010), and an egg thief. It is generally a rather bold species, commonly seen in rest camps in nature reserves, and on farmland. Therefore, it does not have the (untapped) potential of making laypeople aware of the plight of the mongoose family in general. The species’ inquisitiveness, social flexibility, and potential as a controller of termites could possibly enhance its public profile. More broadly, its ability as a pest control agent in farmland areas – as insect prey almost certainly includes some pest species – should be quantitatively investigated.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 60-62 days
Reproductive Periodicity: August to February
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
To our knowledge this species is not utilised in any way.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no major threats to the species. Minor threats include incidental/accidental poisoning, or occasional deliberate hunting/poisoning (Taylor and Meester 1993). Hunting and shooting of Yellow Mongooses have been reported on farmland in the Western Cape (between 2001 and 2003) and on some farms in the Free State (Lynch 1980, 1983), where the species is temporarily regarded as a pest around the maize fields, largely because these small carnivores dig up the pits when they are planted (N.L. Avenant pers. obs. 1995–2016). However, persecution does not appear to impact the population, which occurs at higher density on farmland than in natural areas in the Western Cape Province (Balmforth 2004, Vidya et al. 2009). It may also be persecuted as a vector for rabies, especially in the Free State Province (Bishop et al. 2010). Lynch (1980) mentioned that Yellow Mongooses are also persecuted in some areas in the Free State due to the general belief that they kill new-born sheep. However, this is a low level threat and appears to have little, if any, impact on the population, which has remained at high density (A. le Roux pers. obs. 2012–2016). Another minor, localised threat may be the impact of road mortalities in some areas (W. Collinson unpubl. data; Z. Balmforth pers. obs. 2001–2003). Finally, in the Lesotho Lowlands, high human population density and “associated” dogs are expected to have led and be responsible for maintenance of the current extremely low densities of the species (Lynch 1994, N.L. Avenant pers. obs. 2000–2014). Climate change could impact the species, particularly the populations in arid environments.
Conservation
The Yellow Mongoose occurs in numerous protected areas across its range in southern Africa and within the assessment region. No specific conservation interventions are necessary. However, education and awareness campaigns might be used, in conjunction with other affected species, to reduce persecution on farmlands. In areas where the Yellow Mongoose is a recognized, major vector for rabies, for example, in the northern parts of the Eastern Cape, the Free State, southern parts of Gauteng and south western parts of Mpumalanga (Bishop et al. 2010), such campaigns may be less effective. Similarly, holistic management of ranch lands and game farms should be encouraged, particularly habitat or ecosystem management that tends to sustain a natural prey base, such as conserving patches of natural habitat and lowering stocking densities (Bowland and Perrin 1989, Eccard et al. 2000).
Recommendations for land managers and practitioners: Since the species is an important vector of rabies (Zumpt and Hassel 1982, Bishop et al. 2010), its abundance close to settlements should be monitored, and relevant health departments informed should rabies cases appear. No active monitoring is taking place, although some subpopulations have been under recent study for epidemiological and behavioural research. In most of these cases, population dynamics is not the central focus of the research. However, it is typically included in the monitoring of small carnivores (e.g., Blaum et al. 2008), species check lists (e.g., Avenant 1997, Avenant and Watson 2002, Watson 2006) and environmental assessments (e.g., Avenant 2008).
Research priorities:
- Field surveys in Lesotho and Eswatini to confirm Yellow Mongoose presence/absence and distribution in these countries.
- Degree to which this species is still a rabies vector, and in which specific areas within its distribution range.
- Effectiveness of awareness and education campaigns in reducing persecution on farmlands.
- The impact of high Black-backed Jackal (Lupulella mesomelas) densities on Yellow Mongoose abundance and distribution (sensu Kamler et al. 2012).
- Level of reproductive suppression in cooperatively breeding groups and how/whether this varies with density (as other aspects of sociality do). For example, hormone and genetic studies to determine levels of suppression (how many females are breeding per group in different populations) and whether suppression ever takes place pre-gestation (vs via infanticide post-gestation).
- The effects of climate change on this species throughout its range.
- Population health and genetics across its range.
Encouraged citizen actions:
- While this species is not under threat, it would be ideal for visitors to neighbouring countries such as Lesotho and Eswatini to be actively looking out for Yellow Mongoose, and report sightings on virtual museum platforms (e.g., iNaturalist and MammalMAP).
- When in any doubt, take carcasses (using plastic gloves to place the carcasses in a bag) to a veterinarian for rabies testing.
Bibliography
Avenant NL, Nel JAJ. 1992. Comparison of the diet of the yellow mongoose in a coastal and a Karoo area. South African Journal of Wildlife Research 22: 89–93.
Avenant NL, Watson JP. 2002. Mammals recorded in the Sandveld Nature Reserve, Free State province, South Africa. Navorsinge van die Nasionale Museum, Bloemfontein 18: 1-12.
Avenant NL. 1997. Mammals recorded in the QwaQwa National Park (1994-1995). Koedoe 40: 31-40.
Avenant NL. 2004. Conserving Mountain Biodiversity in Southern Lesotho: Mammal report. United Nations Development Plan, Lesotho.
Avenant NL. 2008. CN Code: the mammals and herpetofauna at Beatrix Main and Beatrix No. 4 slime dam complexes. Report to H2ON Environmental Specialists. Bloemfontein.
Avenant NL. 2011b. Small mammal report. . Integrated Report on Biomonitoring at Kolomela Mine, Kumba Iron Ore. Centre for Environmental Management, University of the Free State, Bloemfontein, South Africa.
Avenant, N.L. 2011. The potential utility of rodents and other small mammals as indicators of ecosystem “integrity” of South African grasslands. Wildlife Research 38: 626–639.
Balmforth, Z.E. 2004. The demographics, spatial structure and behaviour of the yellow mongoose, Cynictis penicillata, with emphasis on cooperative breeding. PhD Thesis, University of Sussex.
Bishop GC, Durrheim DN, Kloeck PE, Godlonton JD, Bingham J, Speare R. 2010. Rabies Guide for the Medical, Veterinary and allied Professions. Department of Agriculture, Forestry and Fisheries, Pretoria, South Africa.
Bizani M. 2014. Diet of the yellow mongoose (Cynictis penicillata) in the Albany Thicket Biome of South Africa. M.Sc. Thesis. University of Fort Hare.
Blaum N, Engeman RM, Wasiolka B, Rossmanith E. 2008. Indexing small mammalian carnivores in the southern Kalahari, South Africa. Wildlife Research 35: 72–79.
Blaum N, Rossmanith E, Fleissner G, Jeltsch F. 2007. The conflicting importance of shrubby landscape structures for the reproductive success of the yellow mongoose (Cynictis penicillata). Journal of Mammalogy 88: 194–200.
Bowland, A.E. and Perrin, M.R. 1989. The effect of overgrazing on the small mammals in Umfolozi Game Reserve. Zeitschrift für Säugetierkunde 54: 251–260.
Cavallini P. 1993b. Activity of the yellow mongoose Cynictis penicillata in a coastal area. Zeitschrift für Säugetierkunde 58: 281–285.
Cavallini, P. 1993a. Spatial organization of the yellow mongoose Cynictis penicillata in a coastal area. Ethology Ecology and Evolution 5: 501-509.
Cavallini, P. and Nel, J.A.J. 1995. Comparative behaviour and ecology of two sympatric mongoose species (Cynictis penicillata and Galerella pulverulenta). South African Journal of Zoology 30: 46-49.
Cronk, N.E. and Pillay, N. 2019a. Dietary overlap of two sympatric African mongoose species in an urban environment. Mammalia, 83(5), 428-438.
Cronk, N.E. and Pillay, N. 2019b. Flexible use of urban resources by the yellow mongoose Cynictis penicillata. Animals, 9(7), 447.
Davis PL, Rambaut A, Bourhy H, Holmes EC. 2007. The evolutionary dynamics of canid and mongoose rabies virus in Southern Africa. Archives of virology 152: 1251–1258.
Do Linh San E, Somers MJ. 2006. Mongooses on the move: an apparent case of interspecific cooperative vigilance between carnivores. South African Journal of Wildlife Research 36: 201–203.
ERM. 2010. Critical Habitat Assessment, Lesotho Highlands. ERM Southern Africa, Cape Town, South Africa.
Earlé, R.A. 1981. Aspects of the social and feeding behaviour of the yellow mongoose Cynictis penicillata (G. Cuvier). Mammalia 45: 143-152.
Eccard JA, Walther RB, Milton SJ. 2000. How livestock grazing affects vegetation structures and small mammal distribution in the semi-arid Karoo. Journal of Arid Environments 46: 103–106.
Flynn J, Finarelli J, Zehr S, Hsu J, Nedbal M. 2005. Molecular phylogeny of the Carnivora (Mammalia): assessing the impact of increased sampling on resolving enigmatic relationships. Systematic Biology 54.
Fuller, A., Mitchell, D, Maloney, S.K., Hetem, R.S., Fonsêca, V.F., Meyer, L.C., Van de Ven T.M. and Snelling, E.P. 2021. How dryland mammals will respond to climate change: the effects of body size, heat load and a lack of food and water. Journal of Experimental Biology, 224(Suppl_1), jeb238113.
Kamler, J. F., Stenkewitz, U., Klare, U. Jacobsen, N. F., and Macdonald, D. W. 2012b. Resource partitioning among cape foxes, bat-eared foxes, and black-backed jackals in South Africa. Journal of Wildlife Management 76: 1241-1253.
Kauffman MJ, Sanjayan M, Lowenstein J, Nelson A, Jeo RM, Crooks KR. 2007. Remote camera-trap methods and analyses reveal impacts of rangeland management on Namibian carnivore communities. Oryx 41: 70–78.
Kuyler, P. 2000. Veld condition assessment and small mammal community structure in the management of Soetdoring Nature Reserve, Free State, South Africa. Masters Thesis. niversity of the Free State.
Le Roux A, Cherry MI, Gygax L, Manser MB. 2009. Vigilance behaviour and fitness consequences: comparing a solitary foraging and an obligate group-foraging mammal. Behavioral Ecology and Sociobiology 63: 1097–1107.
Le Roux A, Cherry MI, Gygax L, Manser MB. 2009. Vigilance behaviour and fitness consequences: comparing a solitary foraging and an obligate group-foraging mammal. Behavioral Ecology and Sociobiology 63: 1097–1107.
Le Roux A, Cherry MI, Manser MB. 2008. The audience effect in a facultatively social mammal, the yellow mongoose, Cynictis penicillata. Animal Behaviour 75: 943–949.
Lynch C.D. 1989. The mammals of the north-eastern Cape Province. Navorsinge van die Nasionale Museum Bloemfontein 25: 1-116.
Lynch CD. 1980. Ecology of the suricate, Suricata suricatta and yellow mongoose, Cynictis penicillata with special reference to their reproduction. Navorsinge van die Nasionale Museum, Bloemfontein 14: 1–145.
Lynch, C. D. 1975. The distribution of mammals in the Orange Free State, South Africa. Navorsinge van die Nasionale Museum, Bloemfontein 3: 109-139.
Lynch, C.D. 1983. The mammals of the Orange Free State, South Africa. Navorsinge van die Nasionale Museum Bloemfontein 18: 1-218.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
Makenbach SA, Waterman JM, Roth JD. 2013. Predator detection and dilution as benefits of associations between yellow mongooses and Cape ground squirrels. Behavioral Ecology and Sociobiology 67: 1187–1194.
Mbatyoti OA. 2012. Spatio-temporal ecology of the yellow mongoose Cynictis penicillata in the Great Fish River Reserve (Eastern Cape, South Africa). M.Sc. Thesis. University of Fort Hare.
Monadjem A. 1998. The mammals of Swaziland. Conservation Trust of Swaziland and Big Games Parks, Mbabane, Swaziland.
Nel JAJ, Kok OB. 1999. Diet and foraging group size in the yellow mongoose: a comparison with the suricate and the bat-eared fox. Ethology Ecology & Evolution 11: 25–34.
Ngoepe, C.E., Shumba, W., and Sabeta, C. 2024. Evidence for a host switching in the maintenance of canid rabies variant in two wild carnivore species in the Northern Cape Province, South Africa. Journal of the South African Veterinary Association, 95(1), 16-25.
Popp A, Schwager M, Blaum N, Jeltsch F. 2007. Simulating the impacts of vegetation structure on the occurrence of a small mammalian carnivore in semi-arid savanna rangelands. Ecological Modelling 209: 136–148.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Purchase, G.K., Mateke, C. and Purchase, D. 2007. A review of the status and distribution of carnivores, and levels of human–carnivore conflict, in the protected areas and surrounds of the Zambezi Basin. The Zambezi Society, Bulawayo.
Rasa OAE, Wenhold BA, Howard P, Marais A, Pallett J. 1992. Reproduction in the yellow mongoose revisited. South African Journal of Zoology 27: 192–195.
Taylor PJ, Meester J. 1993. Cynictis penicillata. Mammalian Species 432: 1–7.
Taylor, P.J. 2013. Cynictis penicillata Yellow Mongoose. In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. V. Carnivores, Pangolins, Equids and Rhinoceroses, pp. 334-339. Bloomsbury, London, UK.
Veron, G., Colyn, M., Dunham, A., Taylor, P. and Gaubert, P. 2004. Molecular systematics and origin of sociality in mongooses (Herpestidae, Carnivora). Molecular Phylogenetics and Evolution 30: 582-598.
Vidya TNC, Balmforth Z, Le Roux A, Cherry MI. 2009. Genetic structure, relatedness and helping behaviour in the yellow mongoose in a farmland and a natural habitat. Journal of Zoology 278: 57–64.
Watson JP. 2006. Check list of the mammals of Tussen-die-Riviere Provincial Nature Reserve, Free State Province, South Africa. Koedoe 49: 111-117.
Zumpt IF, Hassel RH. 1982. The yellow mongoose as a rabies vector on the central plateau of South Africa. South African Journal of Science 78: 417–418.
van Vuuren BJ, Robinson TJ. 1997. Genetic population structure in the yellow mongoose, Cynictis penicillata. Molecular Ecology 6: 1147–1153.