Yellow Golden Mole
Calcochloris obtusirostris

2025 Red list status
Near threatened
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Calcochloris obtusirostris – (Peters, 1851)
ANIMALIA – CHORDATA – MAMMALIA – AFROSORICIDA – CHRYSOCHLORIDAE – Calcochloris – obtusirostris
Common Names: Yellow Golden Mole (English), Geel Gouemol (Afrikaans), Gelber Goldmull (German)
Synonyms: Chrysochloris obtusirostris Peters, 1851
Taxonomic Note:
This genus has been variably assigned to Chrysochloris (Broom 1907), Amblysomus (Simonetta 1968, Petter 1981) or Calcochloris (Meester 1974, Bronner 1995a). The name Calcochloris predates Chrysotricha (Broom 1907) commonly used for this genus until 1953 (Ellerman 1953, Meester 1974) and also Huetia (Forcart 1942). Bronner (1995a) placed Calcochloris obtusirostris in the subgenus Calcochloris, and Calcochloris leucorhinus in the subgenus Huetia (Forcart 1942), which previously included Calcochloris tytonis [incertae sedis]. Huetia has been elevated to full generic status based on phylogenetic analyses of combined morphological and molecular characters (Asher et al. 2010), leaving Calcochloris to include only C. obtusirostris and C. tytonis [incertae sedis].
Three subspecies are recognised, C. o. obtusirostris, C. o chrysillus, and C. o. limpopoensis, all of which were once considered valid species; subspecies are discriminated by subtle morphological differences, but intergrade in size, making distinguishing among them difficult (Bronner 2013).
Red List Status: Near Threatened NT, B1ab(iii) (IUCN version 3.1)
Assessment Information
Assessor: Patel, T.1 & da Silva, J.2
Reviewer: Mynhardt, S.1
Institutions: 1Endangered Wildlife Trust, 2South African National Biodiversity Institute
Previous Assessor: Maree, S.
Previous Reviewers: Bronner, G. & Taylor, A.
Previous Contributors: Bennett, N.C., Child, M.F. & Relton, C.
Assessment Rationale
This is an edge of range species within the assessment region, occurring in two isolated subpopulations in northern KwaZulu-Natal and north-eastern Limpopo, which correspond to the two subspecies C. o. obtusirostris and C. o chrysillus respectively. The total combined (for both subspecies) extent of occurrence is estimated to be 11,970 km2 and area of occupancy is 48 km2, which would qualify the species as Vulnerable B1 ab (iii) due to local habitat degradation from overgrazing and fuelwood harvesting. Assessing each subspecies separately would qualify only C. o. obtusirostris as Vulnerable B2ab(iii), as there is presumably no habitat degradation in the area where C. o chrysillus occurs. However, we do not assess at subspecies level as both subspecies occur in transfrontier conservation areas (Great Limpopo Transfrontier Park and Lubombo Transfrontier Conservation and Resource Area for C. o. obtusirostris and C. o chrysillus respectively) that connect the assessment region to the bulk of the global population in Mozambique and Zimbabwe. As such, habitat loss is not a major threat within the assessment region. Globally, the species is widespread, tolerates mild habitat alteration and is not heavily impacted by human activities, so the presumably large global population is unlikely to be in decline. Thus, we employ the regional criterion and list the species as Near Threatened B1ab(iii) because the global population is unlikely to decline, and the assessment region subspecies are connected to the global population.
Regional population effects: Dispersal is possible. The Lubombo Transfrontier Conservation Area (South Africa, Mozambique and Eswatini) between the Lebombo Mountains and the Indian Ocean now connects (as of 2011) the Maputo Elephant Reserve through the Futi Corridor and Lubombo Conservancy to Tembe Elephant Park and protects large areas of suitable habitat of this subspecies. Similarly, the Great Limpopo Transfrontier Park connects the northern subpopulations of this species. Thus, although the dispersal capability of this species is poor and limited by soil type and high energetic costs associated with digging; dispersal is possible through transfrontier conservation areas across the northern KwaZulu-Natal – Eswatini – Mozambique border and the north-eastern Limpopo – Zimbabwe – Mozambique border.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Patel T & da Silva JM. 2025. A conservation assessment of Calcochloris obtusirostris. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
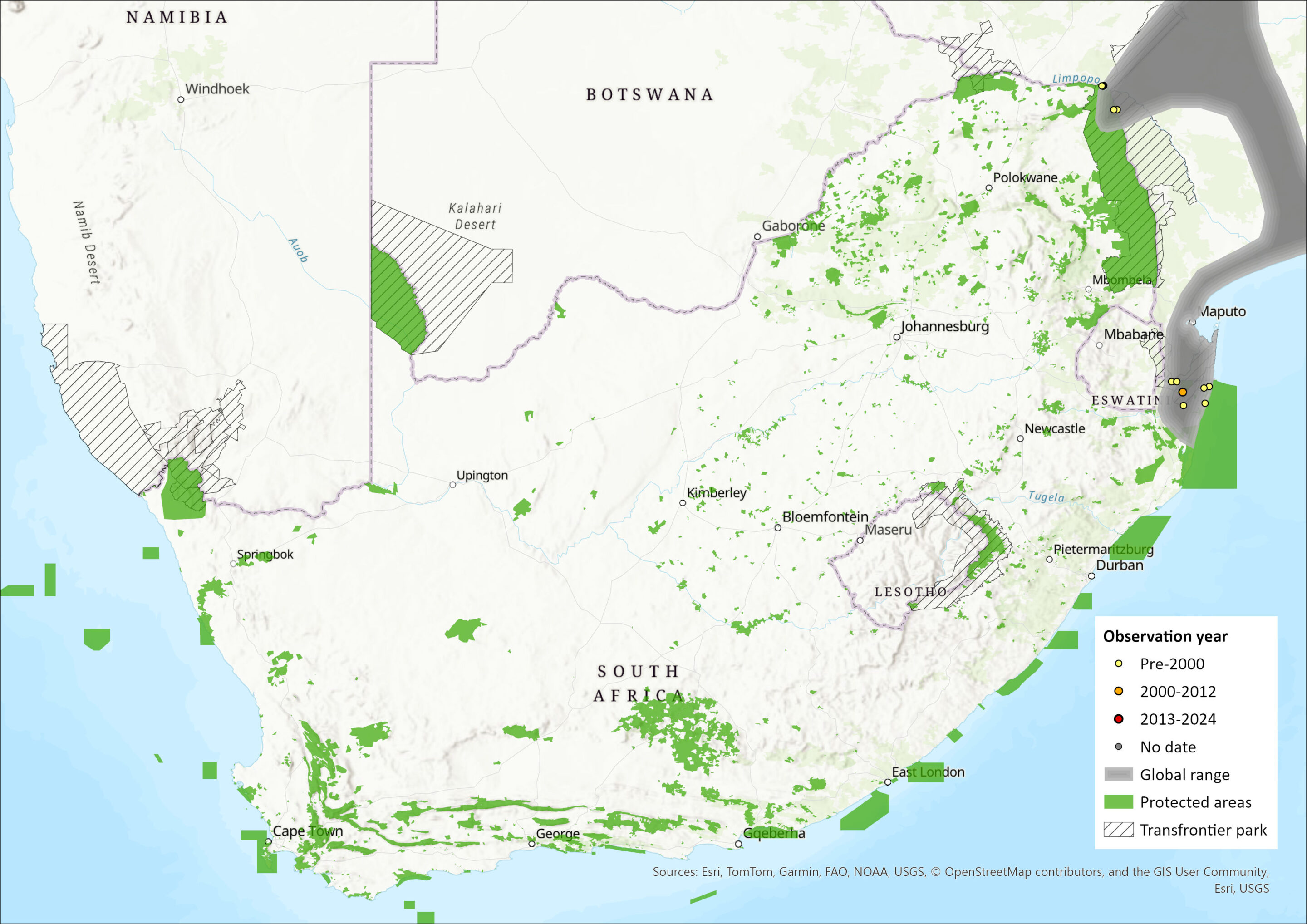
The Yellow Golden Mole is largely restricted to the Mozambique sand plain. It ranges from Inhambane district in Mozambique southwards to northern KwaZulu-Natal, South Africa, and inland to the eastern slopes of the Lebombo Mountains and the south-eastern Lowveld of Zimbabwe.
Calcochloris o. obtusirostris occurs in the Inhambane and Gaza districts of Mozambique, extends westwards along the Changane and Save river systems to marginally intrude into south-eastern Zimbabwe and the Nyadu Sandveld in northern Kruger National Park (Limpopo Province, South Africa). C. o. limpopoensis occurs beyond the assessment region, in the Masiene district in Mozambique southwards to the southern Mozambiquan coastal plains near Maputo where it is replaced by C. o. chrysillus. C. o. chrysillus occurs from Maputo southwards to Maputaland (north of St. Lucia) and in the Ingwavuma and Ubombo districts in northern KwaZulu-Natal. Parts of the geographic ranges of C. o. obtusirostris and C. o. chrysillus are within protected areas, but this is not the case for C. o. limpopoensis (See Threats).
The extent of occurrence for the species overall was calculated at 11,970 km², by clipping the minimum convex polygon of the South African records to the assessment region, which includes the area of the Lubombo Transfrontier Conservation Area in which the species occurs. The area of occupancy was calculated as 48 km², using all 2 by 2 km grid cells in which the species is known to occur, both historically and currently, within the assessment region; and 20 km² and 28 km² for C. o. obtusirostris and C. o. chrysillus respectively.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Figure 1. Distribution records for Yellow Golden Mole (Calcochloris obtusirostris) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Eswatini | Possibly Extant | Native | – | – |
| Mozambique | Extant | Native | – | Resident |
| South Africa | Extant | Native | – | Resident |
| Zimbabwe | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Although the potential impacts of climate change on this species have not been directly assessed, climate change is expected to impact most golden mole species. Since these subterranean animals are restricted to soft soils for burrowing, and have very low vagility, their ability to migrate or shift their distribution ranges in response to climate change is very limited. Climate change is likely to impact golden moles through changes in vegetation type, soil moisture and prey availability.
Population information
This species is locally common in suitable sandy habitats on the coastal plains of Mozambique and northern KwaZulu-Natal (South Africa).
Continuing decline in mature individuals? (Not specified)
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: No
Number of mature individuals in largest subpopulation: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Two of the three recognised subspecies of C. obtusirostris are found within the assessment region and correspond to two geographically isolated subpopulations that differ in habitat and morphology. These differences are expected to translate into genetic differences; however, this has not been confirmed through molecular techniques.
While the species is considered locally common, estimates of population size or densities are unknown, thereby preventing estimates of effective population size for the two subpopulations.
Habitats and ecology
This species is strictly fossorial, restricted to light sandy soils, sandy alluvium and coastal sand dunes in southern African subtropical forest and savannah woodland transitions and mosaics, particularly close to rivers and waterways. C. o. chrysillus occurs in coastal forest and savannah (woodland) transitions and mosaics from southern coastal Mozambique to northern KwaZulu-Natal. C. o. limpopoensis occurs in the miombo savannah (woodland) of eastern Mozambique. C. o. obtusirostris inhabits Acacia and Mopane savannah (thornveld woodland) in south-eastern Mozambique, westwards along the Changane and Save river systems into the Lowveld grasslands of south-eastern Zimbabwe, and the Nyadu Sandveld in northern Kruger National Park, Limpopo Province. The species lives in close proximity to human settlements and thrives in rural and urban gardens. It also occurs in cultivated and pastoral land and commercial forestry plantations, but the latter is considered suboptimal habitat for the species.
Predominantly insectivorous, they are highly sensitive to vibrations produced by live insect prey, which include tenebreonid larvae, termites, grasshoppers, flies, moths and also small lizards. Shallow subsurface foraging tunnels are linked to nest chambers amongst roots of trees. Yellow Golden Moles occupy deep nesting chambers at the base of trees up to 20 cm below the soil surface and create subsurface runs to foraging sites which have been recorded at distances of up to 50 m away from nesting chambers (Roberts 1936). Breeding is suggested to be limited to the wet summer season (Taylor 1998, Skinner and Chimimba 2005).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | Resident | Suitable | Yes |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | Resident | Suitable | Yes |
| 2.1. Savanna -> Savanna – Dry | Resident | Suitable | Yes |
| 2.2. Savanna -> Savanna – Moist | Resident | Suitable | Yes |
| 14.1. Artificial/Terrestrial -> Artificial/Terrestrial – Arable Land | Resident | Suitable | No |
| 14.2. Artificial/Terrestrial -> Artificial/Terrestrial – Pastureland | Resident | Suitable | No |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | Resident | Marginal | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | Resident | Suitable | No |
| 14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas | Resident | Marginal | – |
| 14.6. Artificial/Terrestrial -> Artificial/Terrestrial – Subtropical/Tropical Heavily Degraded Former Forest | Resident | Marginal | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: 9-11
Size at Maturity (in cms): Male: 9-11
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: Seasonally polyoestrous
Average Annual Fecundity or Litter Size: Litter size 2
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species in not known to be utilised or traded in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no known major threats to this species. Minor threats may arise from rural settlement and small-holder ranching expansion, which includes housing and associated roads infrastructure, which may increase rates of habitat degradation through overgrazing and logging. Agriculture and commercial forestry operations no doubt contribute to degradation, fragmentation and loss of its natural soil habitat. However, these are localised threats.
Conservation
Its cryptic and adaptable nature allows the Yellow Golden Mole to coexist successfully with humans. C. o. chrysillus is adequately protected in KwaZulu-Natal at Ndumu Game Reserve, Greater St. Lucia Wetland Park, Tembe Elephant Park, Kosi Bay Nature Reserve, Lake Sibaya Nature Reserve, and the Maputaland Coastal Forest Reserve. In Mozambique, this subspecies occurs in the Maputo Elephant Reserve. The Transfrontier Conservation Area (South Africa, Mozambique and Eswatini) between the Lebombo Mountains and the Indian Ocean now connects the Maputo Elephant Reserve through the Futi Corridor and Lubombo Conservancy to Tembe Elephant Park and protects large areas of suitable habitat of this subspecies. C. o. obtusirostris has been recorded from the Nyadu Sandveld in north-eastern corner of Kruger National Park in South Africa, Gonarezhou National Park in south-eastern Zimbabwe and northern part of the Limpopo National Park in Mozambique. These protected areas are part of the Great Limpopo Transfrontier Park. No specific conservation interventions are currently necessary.
Recommendations for land managers and practitioners:
- Monitoring of existing subpopulations, particularly in KwaZulu-Natal, to assess population trends.
- Field surveys to discover additional subpopulations and to establish the distributional limits of subspecies.
- Incentivise landowners and communities to de-stock to reduce overgrazing impacts.
Research priorities:
- Subpopulation trends within and outside protected areas.
- Evidence for rescue effect from transfrontier parks.
- Field studies to determine the life history traits and ecological tolerances of the species.
- Field surveys to discover additional subpopulations and to establish the distributional limits of subspecies.
- Studies assessing the severity of threats outside of protected areas.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (e.g., iNaturalist and MammalMAP), especially outside protected areas.
- Deposit any dead specimens found in a state or provincial museum, together with information on the date and site where found.
- Create native vegetation gardens.
Bibliography
Asher, R.J., Maree, S., Bronner, G., Bennett, N.C., Bloomer, P., Czechowski, P., Meyer, M. and Hofreiter, M. 2010. A phylogenetic estimate for golden moles (Mammalia, Afrotheria, Chrysochloridae). BMC Evolutionary Biology 10: 69 (doi:10.1186/1471-2148-10-69).
Bronner GN. 1995. Systematic revision of the golden mole genera Amblysomus, Chlorotalpa and Calcochloris (Insectivora: Chrysochloromorpha; Chrysochloridae. Ph.D Thesis. University of KwaZulu-Natal, Durban, South Africa.
Bronner, G.N. 2013. Calcochloris obtusirostris. In: J. Kingdon, D. Happold, T. Butynski, M. Hoffmann, M. Happold and J. Kalina (eds), Mammals of Africa, Volume I: Introductory Chapters and Afrotheria, pp. 235-236. Bloomsbury Publishing, London.
Broom, R. 1907. A contribution to the knowledge of the Cape golden moles. Transactions of the South African Philosophical Society 18: 283–311.
Ellerman, J.R., Morrison-Scott, T.C.S. and Hayman, R.W. 1953. Southern African Mammals 1758-1951: A reclassification. Trustees of the British Museum (Nat. Hist.), London, UK.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Meester, J. 1974. Family Chrysochloridae. Smithsonian Institution Press, Washington, DC, USA.
Petter, F. 1981. Remarques sur la systématique des Chrysochloridés. Mammalia 45: 49–53.
Roberts A. 1936. Report on a survey of the higher vertebrates of North-Eastern Zululand. Annals of the Transvaal Museum 18: 163-251.
Simonetta, A.M. 1968. A new golden mole from Somalia with an appendix on the taxonomy of the family Chrysochloridae (Mammalia, Insectivora). Monitore zoologici Italiano, Supplement 2: 27–55.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor, P. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.