Xeric four-striped Grass Rat
Rhabdomys pumilio

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Rhabdomys pumilio – (Sparrman, 1784)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Rhabdomys – pumilio
Common Names: Xeric four-striped Grass Rat (English)
Synonyms: Mus pumilio Sparrman, 1784
This is the new taxonomic concept for R. pumilo. Du Toit et al. (2012) provided molecular evidence based on a mitochondrial marker (CO1) for the presence of three distinct taxa within the arid lineage R. pumilio which, comprises R. pumilio sensu stricto that is more closely related to the R. dilectus group than to the other two taxa of the arid lineage R. bechuanae and R. intermedius. (Du Toit et al. 2012).
Taxonomic Note: Very little is known on the genetic structure of the species. A long-term extensive study of Rhabdomys pumilio populations in Geogap Nature Reserve provided extensive knowledge on its habitat requirements, diet, behavioural ecology and population genetics. R. pumilio coastal b, described as a subclade of R. pumilio (du Toit et al. 2012), was reported as two populations in Fort Beaufort and Birha coastal region. Further studies are necessary to clarify the status and the distribution of this subclade.
Red List Status: LC – Least Concern
Assessment Information
Assessors: Ganem, G.1 Schradin, C.2,3 & da Silva, J.M.4
Reviewer: Pillay, N.3
Contributor: Patel, T.5
Institutions: 1Montpellier University,2Institut pluridisciplinaire Hubert Curien,3University of the Witwatersrand, 4South African National Biodiversity Institute,5Endangered Wildlife Trust
Previous Assessors and Reviewers: Du Toit, N., Pillay, N., Ganem, G. & Relton, C.
Previous Contributors: Child, M.F., Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Mondajem, A., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
Rhabdomys spp. are widespread, versatile and common species, coexisting with humans in disturbed, semi-urban and agricultural landscapes. R. pumilio is restricted to the low-altitude coastal region within the Fynbos and Succulent Karoo Biomes from Oranjmound in Namibia, to Richtersveld in the west down to Port Elizabeth in the Eastern Cape Province. Drought periods and climate change can lead to the local extinction of small subpopulations. It appears to require no direct conservation interventions at present. Locally (in Geogap Nature Reserve), its behavioural ecology and population genetics has benefited from in-depth studies. Research should focus on delimiting the geographical extent of the species, the status of R. pumilio clade b, and research on the species ecology to other populations.
Red List Index
Red List Index: No change
Recommended Citation: Ganem G, Schradin C & da Silva JM. 2025. A conservation assessment of Rhabdomys pumilio. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
It cannot be stipulated with certainty whether the current distribution differs significantly from the historical distribution, given that the genus taxonomy was revealed roughly 12 years ago. Rhabdomys pumilio appears to be well adapted to the succulent karoo biome. To the best of available knowledge, R. pumilio is parapatric to the other Rhabdomys taxa, while the subclade R. pumilio costal b, thrive further east to R. pumilio distribution, and where it occurs in sympatry with R. dilectus individuals. Unlike first thought, it is not endemic to South Africa where it occurs along a narrow coastal strip within the Fynbos and Succulent Karoo Biomes from Port Elizabeth/Fort Beaufort in the east (Eastern Cape) to the mouth of the Gariep (Orange) River in the Richtersveld in the west (Northern Cape) (du Toit et al. 2012), because a large population of the species was identified in South West Namibia (Ganem et al. 2020) . Further sampling and genotyping along Namibia’s coastal and central area as well as in the Northern Cape Province would be necessary to assess the northern distribution limits of R. pumilio and identify potential contact zones with R. bechuanae.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): 2300
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
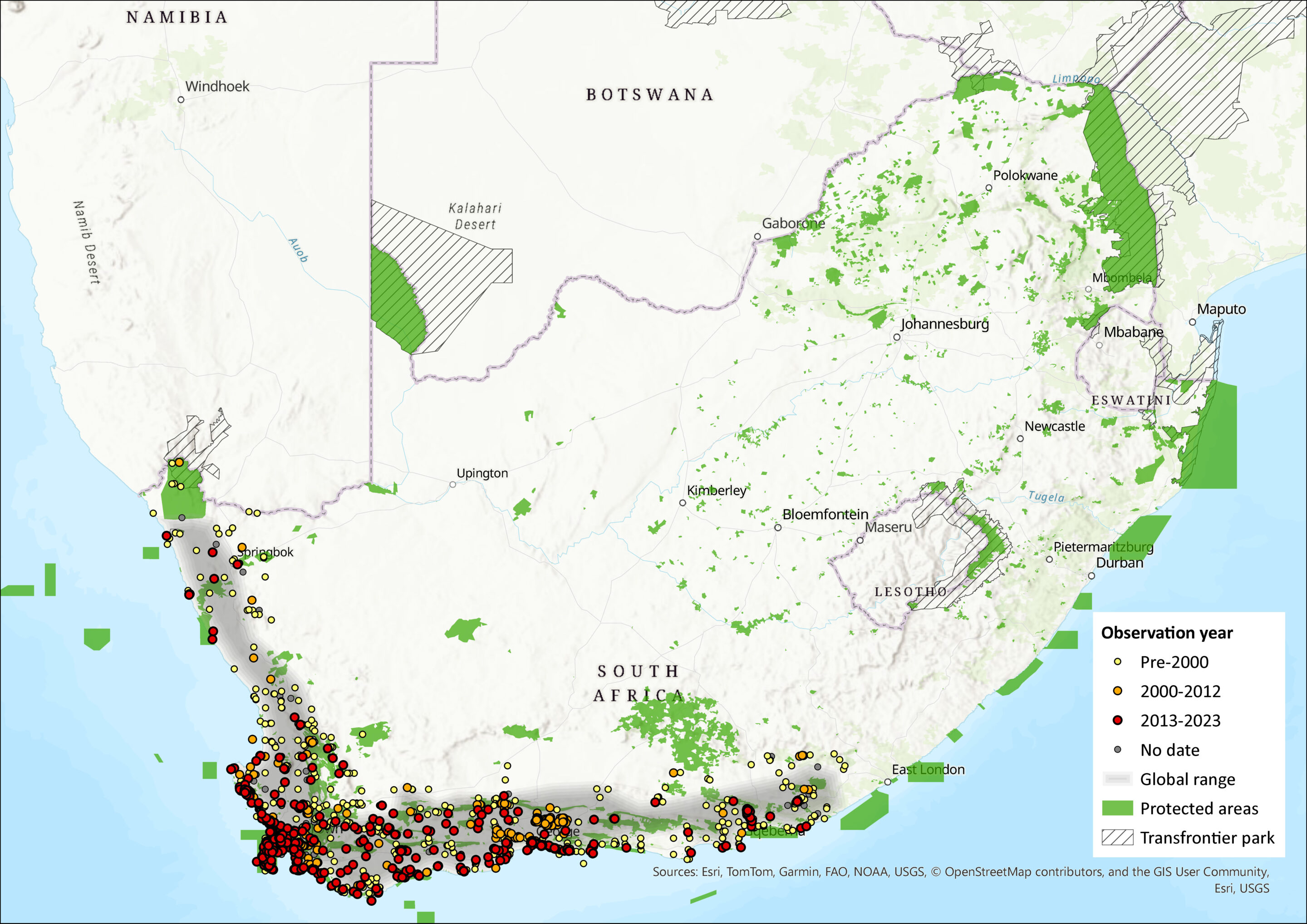
Map
Figure 1. Distribution records for Xeric four-striped Grass Rat (Rhabdomys pumilio) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Namibia -> Oranjmound | Extant | Native | Resident | |
| South Africa | Extant | Native | – | Resident |
| South Africa -> Eastern Cape Province | Extant | Native | – | Resident |
| South Africa -> Northern Cape Province | Extant | Native | – | Resident |
| South Africa -> Western Cape | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Drought periods and climate change can lead to the local extinction of small subpopulations of R. pumilio (Nater et al. 2018).
Population Information
No abundance information is available for this species and, although they can be locally and temporally abundant, their densities are known to fluctuate High annual survival rates in the Succulent Karoo resulted in significantly higher population densities compared to drier grassland regions (Schradin and Pillay 2005). In the Succulent Karoo, average population densities of 36.6 ± 21.2 mice / ha (N = 5 groups) were reported in September, increasing to a peak of 171.1 ± 40.9 mice / ha (N = 7 groups) in December, and then declining through the rest of summer and into winter (Schradin and Pillay 2005). In other parts of South Africa, densities of d 30–300 individuals / ha were recorded in the Cape Flats (David and Jarvis 1985).
Population Information
Continuing decline in mature individuals? Unknown
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: Unknown
All individuals in one subpopulation: No
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Phylogenetic analyses have provided great insights into the distribution and genetic diversification of species within the Rhabdomys pumilio complex (du Toit et al. 2012; Ganem et al. 2020). A coastal and interior lineage are apparent based on mitochondrial CO1 sequence data (du Toit et al. 2012; Ganem et al. 2020). Whether there exists more fine scale population genetic structure remains unknown. A more comprehensive fine scale molecular investigation incorporating nuclear markers is highly encouraged.
Based on the available information, it is possible to quantify one of the two population genetic indicators in the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF). The complementary genetic indicator – proportion of populations maintained (PM), would receive a value of 1.0, considering at least two subpopulations (lineages) are known and exist (2/2 populations remaining).
Due to the lack of effective population size or abundance data, and considering population densities can fluctuate considerably, the GBF’s headline indicator – proportion of species with an effective population size (Ne) greater than 500 – cannot be estimated.
Habitats and ecology
Rhabdomys pumilio occurs in the Fynbos and Succulent Karoo Biomes. Rhabdomys is diurnal and crepuscular omnivores with a diet of seeds, fruits, green plant material, Acacia pods, Protea flower bracts, Pinus bark and arthropods (Fuller and Perrin 2001, Skinner and Chimimba 2005, Monadjem et al. 2015). They are primarily granivorous, but depending on the season, are known to be opportunistically omnivorous, occasionally consuming insects (Perrin and Curtis 1980). R. pumilio shows intra-specific variation in social organisation. From solitary living when population density is very low at the onset of the breeding season, to large groups of up to 30 adults of both sexes with communal breeding and helpers at the nest (Schradin and Pillay 2004, 2010). Intra-specific variation in social organisation is an adaptation to variation in population density both over time (Schradin et al. 2010, 2019) and geographically (Schradin et al. 2020). The main driver is reproductive competition, with females becoming solitary to avoid infanticide by other females, which is only possible when population density is low and free territories are available (Schradin et al. 2010; Schoepf et al. 2012). Outside the breeding season, when no reproductive competition occurs, R. pumilio is always group living (Schradin et al. 2010, 2019). The breeding season commences in August (Schradin and Pillay 2005) and terminates in December in the Succulent Karoo. The start of the breeding season is dependent on a combination of food availability, temperature and rainfall. Litter size is affected by female age and body mass, and females produce an average of two (and maximum of five) litters per season (David and Jarvis 1985). Litter size averaged at 4.9 (range, 2–9) on the Cape Flats (David and Jarvis 1985). In the wild, average life expectancy is documented as 1.5 months (David and Jarvis 1985), but individuals may live for up to 24 months in captivity (Brooks 1974).
Ecosystem and cultural services: Rhabdomys species are particularly important forage species for diurnal raptors, snakes and small mammals, including Caracal (Caracal caracal), Black-backed Jackal (Lupulella mesomelas), African Wildcat (Felis silvestris) and several species of mongoose (De Graaff 1997), as they are one of the few diurnal rodent species. R. pumilio is a pollination agent in the Cape Fynbos.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 14.1. Artificial/Terrestrial -> Artificial/Terrestrial – Arable Land | – | Suitable | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Suitable | – |
| 14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas | – | Suitable | – |
Life History
| Generation Length | Justification | Data Quality |
| 1 | see Pacifici et al. 2013 | medium |
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Rhabdomys is currently being utilised for field and lab-based research and museum collections. It is an important research model to assess social evolution and ecological diversification.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
No major threats have been recognised for this species. R. pumilio can become locally extinct during droughts (Schradin et al. 2020) even though modelling indicates a low extinction risk (Nater et al. 2018), though the data underlying this modelling excluded the year 2003 with the severest drought, when a large population of 200 individuals/5 ha declined to only 1 individual (Schradin, pers. comm.).
Conservation
This species is located in several protected areas across its range. No conservation interventions are deemed necessary for this species. R. pumilio should benefit from the expansion of lowland fynbos protected areas in the Western Cape.
Research priorities:
- Effect of climate change and especially heat and drought on population dynamics.
- Rate of future habitat loss.
- Species geographical distribution, morphometrics, genotyping of populations and taxonomic assessment. This includes vetting of existing museum specimens.
- Species social and general biology.
- Ecology and mechanisms of coexistence with other Rhabdomys species.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas. However, due to their morphological similarities, identification to species level may prove difficult, especially in zones of sympatry.
Bibliography
Brooks, P.M. 1974. The ecology of the four-striped field mouse, Rhabdomys pumilio(Sparrman, 1984), with particular reference to a population on the Van Riebeeck Nature Reserve, Pretoria. D.Sc. Thesis. University of Pretoria.
David J.H.M. and Jarvis J.U.M. 1985. Population fluctuations, reproduction and survival in the striped fieldmouse Rhabdomys pumilio on the Cape Flats, South Africa. Journal of Zoology 207: 251–276.
De Graaff, G. 1997. Striped mouse Rhabdomys pumilio. In: G. Mills and L. Hes (eds), The Complete Book of Southern African Mammals, Struik Publishers, Cape Town, South Africa.
du Toit , N., Jansen van Vuuren , B., Matthee, S. and Matthee, C.A. 2012. Biome specificity of distinct genetic lineages within the four-striped mouse, Rhabdomys pumilio (Rodentia: Muridae) from southern Africa with implications for taxonomy. Molecular Phylogenetics and Evolution 65: 75–86.
Fuller, J.A. and Perrin, M.R. 2001. Habitat assessment of small mammals in the Umvoti Vlei Conservancy, KwaZulu-Natal, South Africa. South African Journal of Wildlife Research 31: 1-12.
Ganem, G., Dufour, C. M. S., Avenant, N. L., Caminade, P., Eiseb, S. J., Tougard, C., & Pillay, N. 2020. An update on the distribution and diversification of Rhabdomys sp. (Muridae, Rodentia). Journal of Vertebrate Biology, 69(2), 1-17. doi: 10.25225/jvb.20013
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of sub-Saharan Africa: a biogeographic and taxonomic synthesis. De Gruyter, Berlin, Germany.
Nater, C. R., Benthem, K. J., Canale, C. I., Schradin, C. & Ozgul, A. 2018. Density feedbacks mediate effects of environmental change on population dynamics of a semidesert rodent. Journal of Animal Ecology, 87, 1534-1546.
Perrin, M.R. and Curtis, B.A. 1980. Comparative morphology of the digestive system of 19 species of Southern African myomorph rodents in relation to diet and evolution. South African Journal of Zoology 15: 22–33.
Schoepf, I. & Schradin, C. 2012. Better off alone! Reproductive competition and ecological constraints determine sociality in the African striped mouse (Rhabdomys pumilio). Journal of Animal Ecology, 81, 649-656.
Schradin, C. & Pillay, N. 2004. The striped mouse (Rhabdomys pumilio) from the succulent karoo of South Africa: A territorial group living solitary forager with communal breeding and helpers at the nest. J Comp Psychol, 118, 37-47.
Schradin, C. and Pillay N. 2005. Demography of the striped mouse (Rhabdomys pumilio) in the succulent karoo. Mammalian Biology 70: 84–92.
Schradin, C., König, B. & Pillay, N. 2010. Reproductive competition favours solitary living while ecological constraints impose group-living in African striped mice. Journal of Animal Ecology, 79, 515-521.
Schradin, C., Pillay, N. & Bertelsmeier, C. 2019. Social flexibility and environmental unpredictability in African striped mice. Behavioral Ecology and Sociobiology, 73, 94.
Schradin, C., Drouard, F., Lemonnier, G., Askew, R., Olivier, C. A. & Pillay, N. 2020. Geographic intra-specific variation in social organization is driven by population density. Behavioral Ecology and Sociobiology, 74, 113.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.