Woodland Dormouse
Graphiurus murinus

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Graphiurus murinus – (Desmarest, 1822)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – GLIRIDAE – Graphiurus – murinus
Common Names: Woodland Dormouse, Forest African Dormouse (English), Boswaaierstertmuis (Afrikaans), Mokomane, Nthuê, Nthufê, Nthuhê, Motsêkêtsêkê, Tsikôtsikô (Tswana)
Synonyms: alticola, cineraceus, cinerascens, collaris, erythrobronchus, griseus, isolatus, johnstoni, lalandianus, raptor, saturates, selindensis, soleatus, vulcanicus, zuluensis. See full list in Holden (2013)
Taxonomic Note: Taylor et al. (1994) reported that specimens from Mkhuze in KwaZulu-Natal, Simunye in Swaziland and Waterpoort in Limpopo differed morphologically (reduced upper molar, inflated bullae) from other specimens of Graphiurus murinus and from the Stone Dormouse (G. platyops). Similarly, recent evidence reported differences in the morphology of two populations of G. murinus in the Eastern Cape (Kryštufek et al. 2004). Although karyotypes were the same for both populations, they differed from previously recorded karyotypes for this genus in Africa (Kryštufek et al. 2004). Additionally, a high degree of chromosomal diversity has been recorded in this species (Dippenaar et al. 1983; Holden 1993). Taxonomic research and DNA analysis is therefore necessary to carry out a systematic revision of this species complex (Skinner & Chimimba 2005; Holden 2013).
Red List Status: LC – Least Concern
Assessment Information
Assessors: Smith, C.1 & da Silva, J.M.2
Reviewer: Patel, T.1
Institutions: 1Endangered Wildlife Trust,2South African National Biodiversity Institute
Previous Assessors & Reviewers: Madikiza, Z., Lamani, S., Baxter, R., Kryštufek, B., MacFadyen, D. & Do Linh San, E.
Previous Contributors: Relton, C., Avenant, N., Avery, M., Monadjem, A., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
The Woodland Dormouse is listed as Least Concern in view of its wide distribution within the assessment region and presumed large population. It is present in many protected areas and can co-occur with human habitation. Although the species is thought to occur in naturally low numbers, it is not suspected to be declining as there are no major threats. As this rodent is arboreal, it is difficult to trap on the ground and thus may be more abundant than expected. For future surveys, it is recommended that traps are set in trees. This species may have to be reassessed as the species complex is disentangled through ongoing molecular work.
Regional population effects: Unless conditions are favourable, this species may occur in low numbers across the central, eastern and northern parts of the assessment area. Where it occurs in Mozambique, Zimbabwe, Botswana and Namibia, immigration is expected where continuous woodland is found or where patchy woodlands are connected by riverine forests or similar wooded corridors.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citations: Smith C & da Silva JM. 2025. A conservation assessment of Graphiurus murinus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
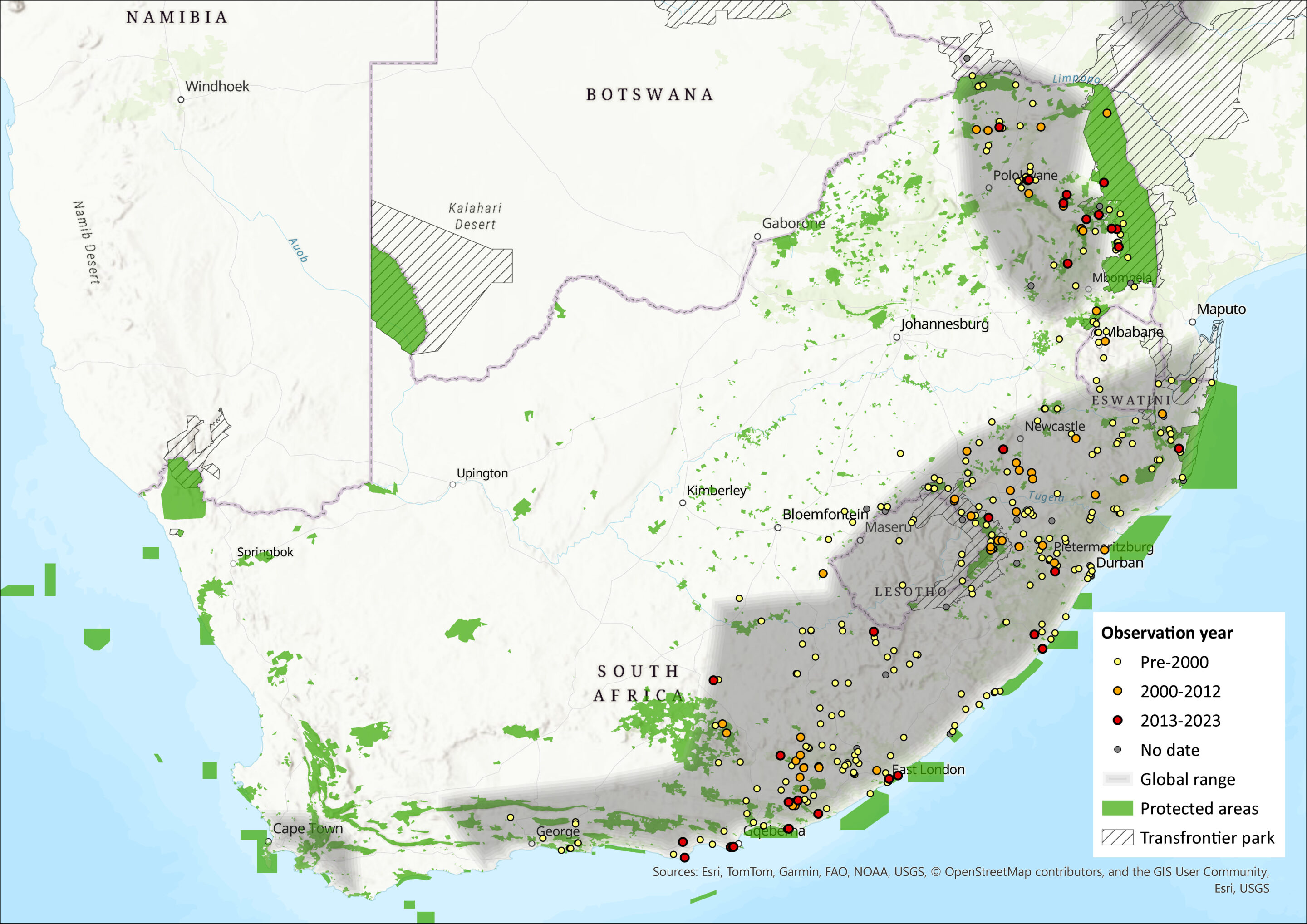
The Woodland Dormouse has a scattered distribution that stretches from Ethiopia to South Africa (reaching as far west as the Western Cape) and Lesotho. Within the assessment region, it ranges across Limpopo, North West, Gauteng, Mpumalanga, KwaZulu-Natal, and Eastern Cape provinces and marginally into the Western and Northern Cape. It also occurs in Eswatini (Monadjem 1998) and Lesotho (Lynch 1994).
Within the North West Province, only one individual was captured post-2000, which was taken in Terminalia sericea woodland near Mositha in the Mafikeng Bushveld vegetation type (Power 2014). However, numerous dead specimens were found throughout the province as Woodland Dormice often nest in electrical switchboxes and sometimes die when in torpor.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Woodland Dormouse (Graphiurus murinus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Botswana | Possibly Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Presence Uncertain | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
This species occurs across Limpopo, North West, Gauteng, Mpumalanga, KwaZulu-Natal, and Eastern Cape provinces, and marginally into the Western and Northern Cape. South Africa. It also occurs in Eswatini and Lesotho. There is a slight possibility that it might be affected by the 1.5-4°C increase in temperature predicted under various climate change scenarios (Engelbrecht et al. 2024). This species is arboreal and might be able to avoid some temperature extremes by seeking refuge nests in trees. Rainfall is predicted to decrease by 10% and increase by 10% across its range.
Population
This species is thought to generally occur at low densities, although these can vary drastically where the species experiences “boom and bust” cycles, possibly in response to variations in rainfall and food availability. In the Great Fish River Nature Reserve (GFRNR), Eastern Cape, densities have been estimated to vary between 2 and 16 individuals / ha depending on the season and year (Madikiza 2010; Z.J.K. Madikiza pers. obs. 2006–2012) in riverine Combretum forests, where it can be the dominant small mammal. It is, however, unclear whether the presence of wooden nest boxes at the study site might have improved nesting conditions and offspring survival (Madikiza et al. 2010a), hence leading to overall higher densities than usual. Woodland Dormice exhibit low densities in Telperion Nature Reserve (Mpumalanga) in areas where large rocks are present but have been recorded from various accommodation units at the study site (MacFadyen 2014).
Although it has been noted that this species is seldom caught in traps and is more commonly recorded from houses, it is likely because in most studies traps were set on the ground and not on logs or in trees. In GFRNR, Madikiza et al. (2010b) did not make a single capture on the ground, either inside or outside the riverine forest, during a 13-month period. In contrast, trapping success in trees averaged 13.3 captures per 100 trap nights, with a minimum average of 5.1 captures in winter and a maximum average of 19.0 captures in summer. In a subsequent study in the same area, Lamani (2014) only recorded 10% of captures on the ground over the four seasons of the year. In a recent study by Mampa (2023), 26 individuals were caught in Sherman traps in GFRNR.
The population in the southern temperate forest in KwaZulu-Natal was stable throughout a year (Madikiza et al 2019).
Population Information
Continuing decline in mature individuals? (Not specified)
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: (Not specified)
Number of Subpopulations: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Graphiurinae is a monogeneric subfamily, solely including the genus Graphiurus (Hennekam 2021). While this genus has been investigated in a phylogenetic context (Montgelard et al. 2003), no population genetic assessment has been undertaken on the species.
In general, G. murinus is believed to have a wide distribution across the assessment region and can be found in a range of habitats in parts of its range, likely suggesting it exists as a single metapopulation. There is no knowledge of subpopulation extinctions, therefore the Convention on Biological Diversity’s Global Biodiversity Framework’s complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (1/1 populations remaining).
While no population estimates are known, it is presumed that at least 5 000 –10 000 mature individuals exist within the assessment region, and hence the species is thought to exceed the Ne 500 threshold (assuming a 0.1-0.3 conversion ratio of effective population size [Ne] to census size [Nc]). While the presumed size is not an actual census count, the quantity can still be used as a proxy for quantifying the proportion of populations with an Ne > 500 indicator, which is valued at 1.0 (1/1 subpopulation with Ne > 500).
A population genomic study investigating the population genetic structure and diversity within this species is recommended to verify these indicator values.
Habitats and ecology
This species inhabits woodland, savannah, grassland and rocky areas (Skinner & Chimimba 2005). In parts of its range, it is found in either Afromontane forest or riverine forest dominated by Combretum (Qwede 2003; Kryštufek et al. 2004; Madikiza 2010; Photo 1). In Rolfontein Nature Reserve, Northern Cape Province, two specimens were caught in the Acacia karroo community (Jooste & Palmer 1982), as it is dependent on large trees for nesting. It uses holes in trees for its nest, mainly in Acacia and Combretum (Madikiza et al 2019). It can persist in secondary habitats, and in some regions occurs in various types of buildings. For example, it is recorded regularly in buildings at Tswalu Kalahari Reserve (D. MacFadyen pers. obs. 2007) and can reach pest proportions in dwellings in the Soutpansberg. This species has also been recorded using piles of debris deposited by high floods near seasonal rivers (Skinner & Chimimba 2005). The diet has only been studied in GFRNR based on remains found in nest boxes. There, arthropods – mainly beetles and millipedes – were dominant (99% occurrence) in all seasons, followed by molluscs (16%) and the fruits of the Cross-berry (Grewia robusta) and the Buffalo Thorn (Ziziphus mucronata) (14%).
The species is nocturnal and arboreal, and to a lesser extent terrestrial (Lamani 2014; Lombard 2014; Maswoba 2019). At night it forages singly in trees or on rocks – depending on habitat – in search of insects and other food items. In GFRNR, Lamani (2014) found that, in riverine forests, Woodland Dormice select areas with dense canopy cover and a high percentage of arboreal connectivity when active at night, possibly to reduce predation risk and facilitate movements. This species tends to be crepuscular on overcast days on the Highveld (D. MacFadyen pers. obs. 2009) and has been observed during the afternoon at Telperion Nature Reserve (D. MacFadyen pers. obs. 2009). It is inactive at low temperatures in the winter months on the Highveld (MacFadyen 2014). Woodland Dormice are competent thermoregulators (Ellison & Skinner 1991; Webb & Skinner 1996; Whittington-Jones & Brown 1999). In the Eastern Cape, they have been found torpid at different periods of the year (Madikiza 2010), even entering a state of hibernation during the colder months (Mzilikazi et al. 2012). The longest torpor bout that dormice can undergo without arousal is about 8 days, and torpid dormice can drop their body temperature to a minimum of 1.5°C (Mzilikazi et al. 2012).
During the day Woodland Dormice sleep inside branches and trunks (Lamani 2011), and locally in rock crevices. Dead logs and underground sites (burrows) are rarely used (Lamani 2011). In GFRNR, Cape Bushwillows (Combretum caffrum) are the predominant trees used for the resting sites, probably due to their abundance in the forest, and their propensity to rot from the inside and provide natural cavities. Woodland Dormice use several resting sites throughout the year, but site fidelity on consecutive days is very high during winter (Lamani 2011; Z.J.K Madikiza & E. Do Linh San unpubl. data). Specific resting sites (both natural and nest boxes) are used by several different dormice, and simultaneous sharing is frequent, taking place between all combinations of male, female and juvenile dormice, with different patterns observed depending on the season of the year (Madikiza et al. 2010a; Madikiza 2010, 2017).
Woodland dormice are seasonal breeders, starting from October up to February (Qwede 2003; Madikiza 2010), and females can produce up to two litters 6–8 weeks apart (Madikiza 2010). At least in some areas, reproduction seems to be associated with high availability and abundance of insects, fruits and high rainfall (De Graaff 1981). After a gestation of about 24 days (Kingdon 1974), females in natural populations give birth to 3–6 young (Lynch 1989). Using extensive trapping and nest box monitoring data, Madikiza et al. (2011) found high intra- and intersexual home range overlaps, with the home ranges of males twice as large as those of females. This, coupled with asynchronous sexual receptivity in females and a complex network and dynamics of sleeping associations (Madikiza 2010, 2017) suggests that Woodland Dormice have a promiscuous mating system. In addition, some females may engage in communal breeding (Madikiza 2010, 2017).
Ecosystem and cultural services: The Woodland Dormouse is one of the core small mammals in the forest ecosystem, as observed notably in some study sites in the Eastern Cape. It is therefore likely to play a significant ecological role, for example in seed dispersal and/or as a food source for small carnivores (Matolengwe 2010) and aerial predators.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 5.1. Wetlands (inland) -> Wetlands (inland) – Permanent Rivers/Streams/Creeks (includes waterfalls) | – | Suitable | – |
| 14.1. Artificial/Terrestrial -> Artificial/Terrestrial – Arable Land | – | Unknown | – |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Unknown | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Marginal | – |
| 14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas | – | Marginal | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): head-body length of 7.8–11.3 cm and tail length of 5.8–9.4 cm (Skinner and Chimimba 2005)
Size at Birth (in cms): 1.8-4 cm
Gestation Time: 24 days
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: 3-6 per litter
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
The Woodland Dormouse seems to be growing in popularity in the pet trade in Europe and USA. We suspect that a large majority of pet Woodland Dormice originate from captive breeding programmes rather than from the wild. There are also anecdotal records of schoolboys keeping them as pets in Zimbabwe (Skinner & Chimimba 2005).
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no major threats to this species. However, habitat loss and fragmentation from mining and agriculture is likely to cause local subpopulation declines. This species is unlikely to persist in agricultural monocultures with few or no trees. In transformed areas, it is only likely to remain within the rocky, inaccessible habitats, which are unsuitable for agriculture. However, it is adaptable and is known to make use of secondary habitats, including roofs of buildings or thatched huts, switch boxes, water pumps and transformers. Resultantly, it can become a nuisance by causing electrical short circuits (Skinner & Chimimba 2005).
Conservation
Within the assessment region, the Woodland Dormouse is present within many protected areas across its range, including the Kruger National Park (Limpopo and Mpumalanga), Telperion Nature Reserve (Mpumalanga), Ezemvelo Nature Reserve (Gauteng), Tswalu Kalahari Reserve (Northern Cape), Silaka, Hluleka, Dwesa, Mpofu, Fort Fordyce and the Great Fish River nature reserves (Eastern Cape). It is also present in the forests of the Amathole Mountains. Although no specific conservation interventions are necessary, protected area expansion to ensure corridors of suitable woodland for movement would benefit this species.
Recommendations for land managers and practitioners:
- Protect woodland areas.
- Influence agricultural policy to ensure connected patches of woodland are retained.
Research priorities:
- Taxonomic revision is required to disentangle this species complex.
- The boundaries between G. murinus and G. microtis should be clearly defined.
- A better knowledge of the geographic distribution of this and other dormice species is needed. As trap placement is critical, with traps generally having to be set in trees in order to successfully catch this species, it means that Woodland Dormice could well be present in areas where they have not previously been detected.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Plant suitable indigenous trees in gardens and ensure corridors of natural vegetation remain to allow local movements.
Bibliography
Dippenaar NJ, Meester J, Rautenbach IL, Wolhuter DA. 1983. The status of southern African mammal taxonomy. Annales, Musée Royal de l’Afrique Centrale, Sciences Zoologiques 237: 103–107.
Ellison, G.T.H. and Skinner, JD. 1991. Thermoregulation and torpor in African woodland dormice, Graphiurus murinus, following cold acclimation. Zeitschrift für Säugetierkunde 56: 41–47.
Engelbrecht, FA., Steinkopf, J., Padavatan, J. & Midgley, GF. 2024. Projections of future climate change in southern Africa and the potential for regional tipping points. In: von Maltitz, G.P., et al. Sustainability of Southern African Ecosystems under Global Change. Ecological Studies, vol 248. Springer, Cham. https://doi.org/10.1007/978-3-031-10948-5_7
Hennekam, J.J., 2022. Comparative morphology of the dormouse skull and the influence of size and ecology. Journal of Anatomy, 240(5), pp.914-935.
Holden ME. 1993. Family Myoxidae. In: Wilson DE, Reeder DM (ed.), Mammal Species of the World: A Taxonomic and Geographical Reference, pp. 763–770. Smithsonian Institution Press, Washington, DC, USA.
Holden ME. 2013. Graphiurus murinus Forest African Dormouse. In: Happold DCD (ed.), Mammals of Africa. Volume III: Rodents, Hares and Rabbits, pp. 124–126. Bloomsbury Publishing, London, UK.
Jooste JF, Palmer NG. 1982. The distribution and habitat preference of some small mammals in the Rolfontein Nature Reserve. South African Journal of Wildlife Research 12: 26-35.
Kingdon, J. 1974. East African Mammals: An Atlas of Evolution in Africa. Academic Press, London, USA.
Kryštufek B, Haberl W, Baxter RM, Zima J. 2004. Morphology and karyology of two populations of the woodland dormouse Graphiurus murinus in the Eastern Cape, South Africa. Folia Zoologica 53: 339–350.
Lamani S. 2011. Resting site ecology of the woodland dormouse Graphiurus murinus at the Great Fish River Reserve, Eastern Cape. B.Sc. Honours Thesis. University of Fort Hare.
Lamani S. 2014. Diet and microhabitat use of the woodland dormouse Graphiurus murinus at the Great Fish River Reserve, Eastern Cape, South Africa. M.Sc. Thesis. University of Fort Hare.
Lombard M. 2014. Activity patterns of the woodland dormouse Graphiurus murinus at the Great Fish River Reserve (Eastern Cape, South Africa): a pilot study. B.Sc. Honours Thesis. University of Fort Hare.
Lynch C.D. 1989. The mammals of the north-eastern Cape Province. Navorsinge van die Nasionale Museum Bloemfontein 25: 1-116.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
MacFadyen DN. 2014. The dynamics of small mammal populations in Rocky Highveld Grassland, Telperion, South Africa. Ph.D. Thesis. University of Pretoria, Pretoria, South Africa.
Madikiza ZJK, Bertolino S, Baxter RM, Do Linh San E. 2010. Nest box use by woodland (Graphiurus murinus): the influence of life cycle and nest box placement. European Journal of Wildlife Research 56: 735–743.
Madikiza ZJK, Bertolino S, Baxter RM, Do Linh San E. 2010. Seasonal, sexual and age-related variations in the live-trapping success of woodland dormice (Graphiurus murinus). Zoological Studies 49: 797–805.
Madikiza ZJK, Bertolino S, Do Linh San E. 2011. Female in space, or female in space and time? First data on the socio-spatial organization and mating system of the woodland dormouse (Graphiurus murinus). Journal of Ethology 29: 375–380.
Madikiza ZJK. 2010. Population biology and aspects of the socio-spatial organization of the Woodland dormouse Graphiurus murinus (Desmarest, 1822) in the Great Fish River Reserve, South Africa. M.Sc. Thesis. University of Fort Hare.
Madikiza ZJK. 2017. Sociality in the African Woodland dormouse. Ph.D. Thesis. University of the Witwatersrand.
Madikiza, Z.J., Bertolino, S. and San, E.D.L., 2019. Population biology of the woodland dormouse Graphiurus murinus in a riverine Combretum forest, South Africa. African Zoology, 54(2), pp.105-113.
Maswoba, S., 2019. Behavioural responses of male woodland dormice (Graphiurus murinus) towards communicatory cues of same-sex conspecifics (Doctoral dissertation).
Mampa, N., 2023. Personality profiles of the Namaqua rock mouse (Micaelamys namaquensis), the Mozambique thicket rat (Grammomys cometes) and the woodland dormouse (Graphiurus murinus).
Matolengwe T. 2010. Diet of the small-spotted genet, Genetta genetta (Carnivora: Viverridae), in the Great Fish River Reserve (South Africa). M.Sc. Thesis. University of Fort Hare.
Monadjem A. 1998. The mammals of Swaziland. Conservation Trust of Swaziland and Big Games Parks, Mbabane, Swaziland.
Mzilikazi N, Madikiza Z, Oelkrug R, Baxter RM. 2012. Hibernation in free-ranging African woodland dormice, Graphiurus murinus. In: Ruf T, Bieber C, Arnold W, Millesi E. (ed.), Living in a Seasonal World, Thermoregulatory and Metabolic Adaptations., pp. 41–50. Springer, Berlin, Germany.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Qwede K. 2003. Observations on two populations of the woodland dormouse, Graphiurus murinus, in the Eastern Cape, South Africa. B.Sc. Honours Thesis. University of Fort Hare.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor, P.J., Richardson, E.J., Meester, J. and Wingate, L. 1994. New distribution records for six small mammal species in Natal, with notes on their taxonomy and ecology. Durban Museum Novitates 19: 59-66.
Webb PI, Skinner JD. 1996. Summer torpor in African woodland dormice Graphiurus murinus (Myoxidae: Graphiurinae). Journal of Comparative Physiology, Series B 166: 325–330.
Whittington-Jones CA, Brown CR. 1999. Thermoregulatory capabilities of the woodland dormouse, Graphiurus murinus. South African Journal of Zoology 34: 34–38.