White-tailed Rat
Mystromys albicaudatus

2025 Red list status
Vulnerable
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Mystromys albicaudatus – (A. Smith, 1834)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – NESOMYIDAE – Mystromys – albicaudatus
Common Names: White-tailed Rat, African White-tailed Rat, White-tailed Mouse, Southern African Hamster (English), Witstertmuis (Afrikaans)
Synonyms: Otomys albicaudatus A. Smith, 1834
Taxonomic Note: Originally described in the genus Otomys, molecular studies have confirmed that Mystromys belongs in the Nesomyidae family and represents a separate ancient lineage (the Mystromyinae) divergent from the Cricetomyinae, Dendromurinae, Petromyscinae and Nesomyinae (Jansa et al. 1999). This genus is monotypic and only one species is presently recognised, so this species is a phylogenetic rarity.
Red List Status: VU – Vulnerable
Assessment Information
Assessors: Wilson, B.,1 Patel, T.,2 Ross, N.3 & da Silva, J.M.4
Reviewer: Gazzard, A.5
Institutions:1Freelance Conservation Biologist,2Endangered Wildlife Trust,3Stellenbosch University,4South African National Biodiversity Institute,5IUCN SSC Small Mammal Specialist Group
Previous Assessors: Avenant, N., Wilson, B., Power, J., Palmer, G. & Child, M.F.
Previous Reviewer: Child, M.F.
Previous Contributors: Roxburgh, L., Avery, M., Baxter, R., MacFadyen, D., Monadjem, A. & Taylor, P.
Assessment Rationale
The White-tailed Rat is a Highveld grassland specialist (whilst also marginally occurring in Succulent Karoo and Fynbos biomes; Dean 1978) and is endemic to South Africa and Lesotho. Although it occurs widely across the assessment region, it has an extremely patchy and severely fragmented area of occupancy due to its preference for microhabitats within vegetation types and transitory habitats created after fire. Out of a total extent of occurrence of 737,337 km2, we calculated an area of occupancy (AOO) of 1,364 km2 (based on a grid size of 2 by 2 km), using all known occurrence records for this species. It is one of the rarest species in the small mammal community, as demonstrated by consistently low trapping records. There is an inferred continuing decline from grassland habitat loss (due to expansion in crop agriculture, urban and industrial development, and climate change) and grassland habitat degradation, primarily from suppression of natural fire regimens. This may represent an emerging threat to this species if fire is increasingly suppressed or controlled with the rise of intensive wildlife breeding. Grasslands are the most threatened biome in the assessment region with at least 33% transformed already. Thus, we list this species as Vulnerable B2ab(iii) under a precautionary purview. Further field surveys are needed to estimate population size and trend, and this species should be reassessed once more data are available. Key interventions include protected area expansion of grassland habitat and the implementation of suitable grazing and fire management systems for nature reserves, livestock and wildlife ranches. Research and monitoring are desperately needed to test the effectiveness of such interventions and to produce baseline information on its ecology and biogeography.
Red List Index
Red List Index: No change
Recommended Citation: Wilson B, Patel T, Ross N & da Silva JM. 2025. A conservation assessment of Mystromys albicaudatus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
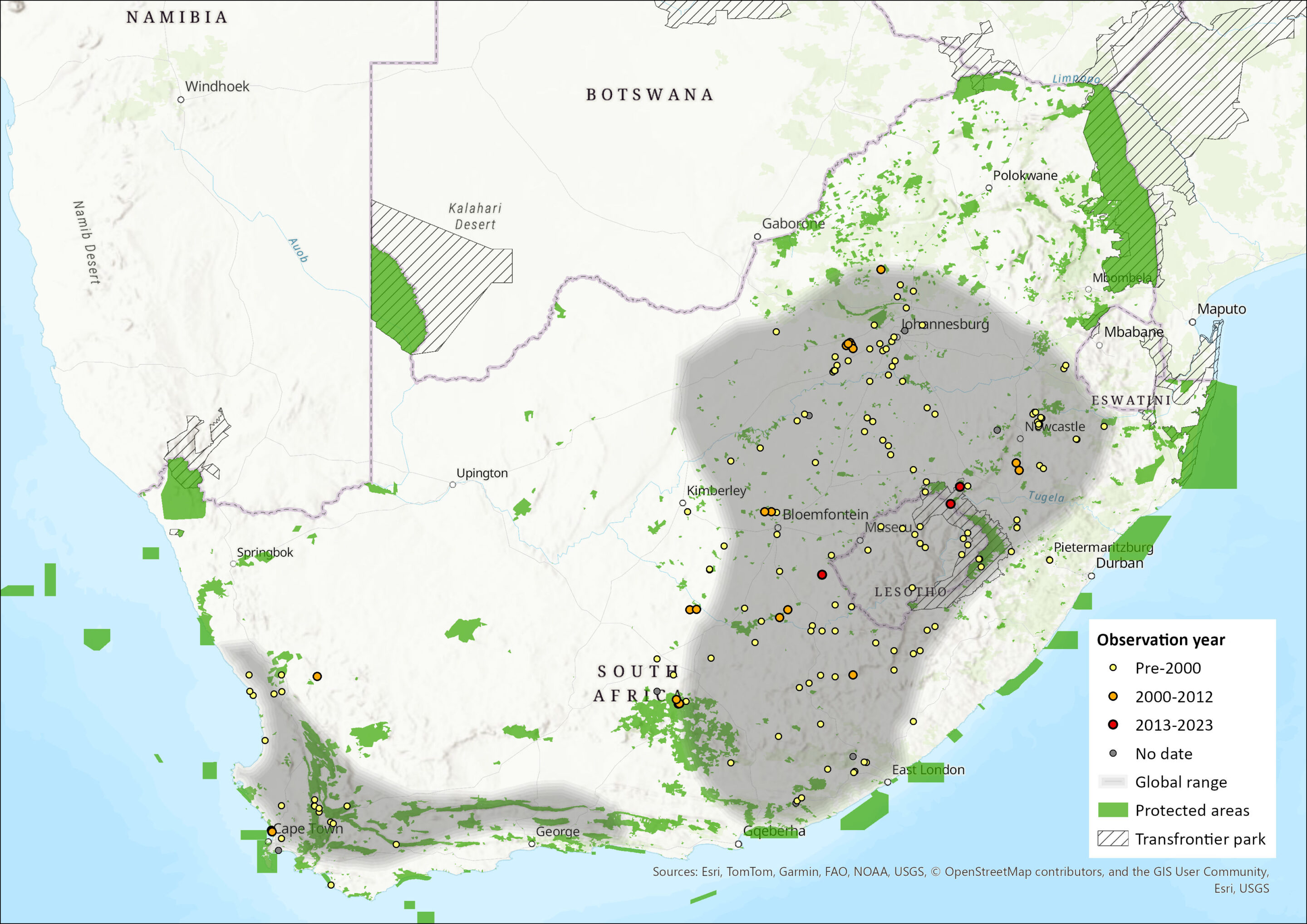
White-tailed Rats are endemic to South Africa and Lesotho, where they inhabit Highveld grasslands primarily, but also Succulent Karoo and Fynbos (Dean 1978). Within South Africa, it occurs in the following provinces: southern Mpumalanga, Free State, the high-lying areas of KwaZulu-Natal (KZN), Eastern Cape, southeastern North-West, and marginally into Northern Cape (Skinner and Chimimba 2005, Avery and Avery 2011). There is a potentially relict subpopulation in the Fynbos of the Western Cape where it probably once occurred extensively throughout the southern grasslands, heathlands and renosterveld of the Cape. There are no records from Eswatini, despite extensive surveys (Monadjem pers. comm. 2015). No recent range expansions or contractions have been documented. However, it is likely that the population is fragmented and will contract in the future as grasslands continue to decline (Mucina and Rutherford 2006, Driver et al. 2012). Additionally, there is a suggestion that, historically, its range had already contracted southwards in the Northern Cape region: owl pellet analysis reveals Pleistocene records from Equus and Wonderwerk Caves, as well as records of the species in 10 other localities in the Kuruman area and surrounds from the Holocene period, while more recent owl pellet records (1930 to present) only reveal localities from the previously-mentioned regions of the province (Avery and Avery 2011), which suggests a long-term range contraction.
This species is widespread across the assessment region but patchily distributed. It has an EOO of 737,337 km2. However, we suspect this is a huge overestimate as they it often occurs in just one of the sampled sites in a region and often is not found in sites where tit is expected to occur (for example, Vermeulen 2005; O’Farrell et al. 2008; Avenant 2011; Kok et al. 2012; N.L. Avenant, G. Palmer, B. Wilson unpubl. data). For example, although Rondeberg Private Nature Reserve shares similar habitat types with the Blaauwberg Conservation Area (BCA) in the Western Cape Province and was identified as a site where the species should occur, they have only been recorded at BCA (Vermeulen 2005). More worryingly, a province-wide survey in the North-West did not reveal a single record for the species, where the last museum record was from Boskop Dam Nature Reserve in 1990 (Power 2014). This is most likely due to the heavy transformation of the grassland biome in the province (Driver et al. 2012). Similarly, although an owl pellet indicating the presence of the species in Tygerberg Nature Reserve, Western Cape, was found in 1992, a recent survey did not find any individuals (Vermeulen 2005). Furthermore, they are suspected to exist in transitory habitats created after fire, having been found equally in burnt and unburnt patches in the Western Cape Province and in low to mid-successional habitats following fire in the Free State Province (Kuyler 2000, Kaiser 2006, Specker 2006, Avenant and Cavallini 2007, Avenant 2011, Avenant and Schulze 2012, Morwe 2013). Although they are predicted to occur widely in Gauteng Province, they have not been found in the Cradle of Humankind region, possibly because the area does not burn. Thus, this species likely occurs in microhabitats (see Habitats and Ecology) that are too fine scale to be approximated by vegetation types and that occur in a shifting mosaic of patches following fire.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for White-tailed Rat (Mystromys albicaudatus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Lesotho | Extant | Native | – | Resident |
| South Africa | Extant | Native | – | Resident |
| South Africa -> Eastern Cape Province | Extant | Native | – | Resident |
| South Africa -> Free State | Extant | Native | – | Resident |
| South Africa -> Gauteng | Extant | Native | – | Resident |
| South Africa -> KwaZulu-Natal | Extant | Native | – | Resident |
| South Africa -> Mpumalanga | Extant | Native | – | Resident |
| South Africa -> North-West Province | Extant | Native | – | Resident |
| South Africa -> Northern Cape Province | Extant | Native | – | Resident |
| South Africa -> Western Cape | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
The species inhabits specific, fragmented habitats, which puts it at risk due to climate change. The small population of this endemic rodent is fragmented as it seems to occur in specific, fire effected, areas within its grassland, karoo and fynbos ranges. These preferred microhabitats lead to M. albicaudatus having an estimated AOO of 1,364 km2 and as such they are likely to be affected by climate change to a large degree. The predicted effects of climate change are increases in drought that may affect the abundance of the micro habitats of this species, potentially causing even more fragmentation, fluctuations and overall decline in population (Lawal et al. 2019). It is already implied that habitat loss and degradation indicate a decline on the overall population of M. albicaudatus.
Population information
Dean (1978) regards the White-tailed Rat as a relict species. Although conservationists have been concerned with it for over forty years, it still persists at low densities. While no empirical population estimates or trends are available, they are consistently one of the rarest species encountered and infrequent capture rates during surveys indicate that population size is low. Friedmann and Daly (2004) reported 5–10 specimens in 15–20,000 trap nights / year in the Free State Province (but only a small percentage of those trap nights took place in habitats where one expected to find the species). Similarly, with over 7,500 small mammal records collected over the current Northern Cape (concentrating in the old demarcated provincial boundaries) and current North-West (the western areas which used to be Northern Cape) provinces, only two records (from Benfontein Farm, near Kimberley) have been archived in the McGregor Museum (0.03% reporting rate, B. Wilson unpubl. data). One other record, 25km south of De Aar was trapped and released in February 2022 (B. Wilson pers comm. 2025). The most recent museum record from North-West Province was from Boskop Dam Nature Reserve (de Beer 1990), and a trapping effort of 240 trap nights in 2013 did not detect the species (Power 2014). Recently, in the Blaauwberg Conservation Area, Western Cape, two studies had a trap success of 1.9% and 1.3% respectively (900 and 800 trap nights corresponding to 17 and 10 individuals respectively (Vermeulen 2005, Specker 2006). The species may have a low sighting record as it is naturally rare and uses a burrow system.
Their patchy distribution may explain the lack of trapping success, but once a patch is found they are trapped easily if present. No home range or dispersal rate data are available, but records indicate a clumped distribution within suitable habitats. The two individuals trapped on Benfontein Farm, just east of Kimberley, were trapped in the same area, although several years apart, despite consecutive annual surveys at similar trapping effort (B. Wilson, unpub. data). Within the Free State Province, colonies did not seem to be in close proximity as, within a homogenous area-trap plot of 0.027 km2 (100 traps spaced 5 m apart with a border area of 25 m around the traps), a maximum of one colony was present (N.L. Avenant unpubl. data). Furthermore, a colony is found in approximately one in every 20 area-trap plots, which means colony density can be preliminarily estimated at one colony / 0.55 km2 or 1.8 colonies / km2 (N.L. Avenant unpubl. data). We can assume a minimum of two mature adults / colony (colonies are small, consisting of < 10 individuals). If we use maximum density as 1 colony / 0.027 km2 and minimum density as one colony / 1.05 km2 (assumed to be the positive interval between the maximum and medium density estimates), a range of population sizes can be estimated. Colony numbers and corresponding mature population size ranges over orders of magnitude, depending on the AOO estimate. The most likely density estimates (medium and minimum) range from 3,499 colonies (6,997 mature individuals) to 22,131 colonies (44,262 mature individuals). Given that colonies (breeding pairs) are suspected to be isolated and may effectively represent a single breeding unit, colony number is probably a more accurate measure of mature population size (sensu IUCN Standards and Petitions Subcommittee 2014).
Defining subpopulations for this species is difficult as we lack basic information about its ecology and distribution. However, we suspect that using vegetation types as proxies for subpopulation distribution may be an adequate approximation since the species may rely on fire to create suitable habitat patches and fire frequency will depend on the fuel load and climate of the vegetation type or bioregion. Subpopulations are suspected to be severely fragmented due to the fragmented nature of the grassland biome (Neke and du Plessis 2004), as well as the prevention of fire as an ecological process or cessation of burning as a management tool by landowners in many areas (A. Taylor unpubl. data).
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| Yes | Inferred | – |
| Extreme fluctuations in the number of subpopulations | Justification |
| No | – |
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: No
Number of mature individuals in largest subpopulation: 712-1521
Number of Subpopulations: 2
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
No population genetic study has been undertaken on this species, nor has it been investigated in a phylogenetic context. However, it is believed that the Fynbos and Grassland animals likely represent genetically differentiated groups (possibly evolutionary significant units [EUSs] or even separate species) due to the large distributional gap within the species. Within each vegetational group, additional genetic subpopulations are suspected due to the fragmented nature of the biomes, especially the grassland biome (Neke and du Plessis 2004), as well as the prevention of fire as an ecological process or cessation of burning as a management tool by landowners in many areas (A. Taylor unpubl. data). It is also possible that some subpopulations have gone extinct, but the number or specific subpopulations are unknown.
To gain a better understanding of the genetic structure and diversity within this species, a fine scale molecular investigation is needed.
Due to the uncertainty in number of subpopulations and their sizes, the Convention on Biological Diversity’s population genetic indicators cannot be quantified.
Habitats and ecology
The colloquial and scientific names of this species refer to its pure white tail. Typically, somewhat hamster-like in appearance, this stocky, grey-coloured rat has a very short tail (about 35–40% of its head and body length) covered in dense short white hairs from which it derives its name. It is terrestrial and nocturnal and has several unusual physiological features. Their stomachs are adapted, via a complex microfauna and ruminant-like digestive system, to digest a wide range of plant foods that contain chemical defences and are thus not eaten by other species (Perrin and Maddock 1983, Downs and Perrin 1995), but there are no available data on preferred plant species from wild populations. It has a high metabolic rate well adapted to the Highveld winter (Monadjem et al. 2015), during which it reduces its activity and foraging times. However, there is no evidence suggesting that White-tailed Rats enter torpor (Perrin 2013). There remains no information about social and reproductive behaviour in the wild, and most data come from captive individuals which breed easily. Litter sizes vary between one and six and it is suspected that breeding takes place year-round. They construct high-sided, cup-shaped nests which “look like Elizabethan neck ruffs” (Skinner and Chimimba 2005: 191), which is probably why the generic name is derived from the Greek mystron (meaning “spoon”) and mys (meaning “mouse”).
The habitat requirements and ecology of White-tailed Rats merits further urgent research. They are often associated with calcrete soils within grasslands. They are never found on soft, sandy substrate, rocks, wetlands or riverbanks. Furthermore, records from the Free State Province and Borakalalo Nature Reserve, North-West Province show that they can occur in disturbed areas (heavily grazed, D. MacFadyen pers. obs.) and in sparse grasslands (Kuyler 2000, Kaiser 2006, Avenant and Cavallini 2007, Avenant and Schulze 2012, Morwe 2013); for example, on shallow limestone substrate (Kuyler 2000, Morwe 2013, N.L. Avenant unpubl. data). Similarly, Vermeulen (2005) surveyed the Blaauwberg Conservation Area (BCA), Western Cape Province and found they had a preference for Dune Thicket on sloped clay soils, which disproves Friedmann and Daly’s (2004) assertion that they require sandy soils. In Mpumalanga, they were recorded from areas of dense grass, as well as rocky areas with grass (Rautenbach 1978). In Lesotho and KZN, they are found along dolerite ridges, rocky slopes, basalt koppies (Lynch 1994, Taylor 1998). In the Maclear district of the Eastern Cape Province, it was found in habitats with crests and ridges and trapped on bare patches with sparse vegetation (Armstrong and van Hensbergen 1996a). They may have a preference for the Carletonville Dolomite Grasslands in North-West Province as is suggested by the museum records (Power 2014). On the Bokkeveld Plateau near Nieuwoudtville, Northern Cape, it was trapped in Dolerite Plains vegetation (O’Farrell et al. 2008). They do not occur in transformed habitat (croplands, fallow fields, or old fields). However, as they select microhabitats, such as slopes and ridges, small numbers may survive in non-arable patches within an agricultural matrix.
A controlled and replicated study analysing the effects of fire on the species in the BCA showed that about half the individuals trapped occurred in unburnt versus burnt habitats (60% in unburnt) whereas > 90% of other small mammals were recorded in unburnt areas (Specker 2006). This may indicate that the species is fire-adapted and Specker (2006) suggests that fire may trigger breeding. However, the study also suggests that unburnt areas are utilised in tandem with burnt areas and are perhaps needed as refuges from predation. Thus, a patch mosaic burning regimen, rather than a block-burn, may create the most desirable micro-habitat for this species (but see Parr and Andersen 2006). The species may require phase diversity, as observations suggest it exists in transitory habitat post-fire: they appear to enter grassland habitat about six months after a patch has burned and exit as the vegetation recovers (Kuyler 2000, Kaiser 2006, Avenant and Schulze 2012). Thus, the species follows plant succession closely, making disturbance important.
As they are nocturnal, they are often preyed on by Barn Owls (Tyto alba) as documented in studies analysing owl pellets (Avenant 2005, Avery et al. 2005). They live in burrows or crevices (de Graaf 1981, Armstrong and van Hensbergen 1996b), and can also swim (Hickman and Machiné 1986).
Ecosystem and cultural services: White-tailed Rats are burrowers. They have been recorded as a forage species for owls (Dean 1978; Avery et al. 2002, 2003, 2005; Avery and Avery 2011). Previously they were considered susceptible to plague which occurs sporadically in free-living populations, but the National Institute for Communicable Diseases (2005) did not list them as plague-carrier.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | Yes |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | Yes |
Life History
Generation Length: 2.1 years (Pacifici et al. 2013)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: 2,5 years
Average Reproductive Age: (Not specified)
Maximum Size (in cms): 240mm
Size at Birth (in cms): 6,5g
Gestation Time:37 days
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: Breeds year-round with litters of 2-5 young
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
They are used extensively in laboratory research (Hall 3rd et al. 1967). They may occasionally be utilised as bushmeat but are not a target species.
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
Yes |
– |
– |
– |
National Commercial Value: No
International Commercial Value: No
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
1. Food – human |
true |
– |
– |
– |
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Habitat loss of grasslands, both from agricultural and industrial or urban expansion (accounting for 97% of the transformed land in North-West Province, NW READ 2014), as well as in the future from climate change (through bush encroachment, van Jaarsveld and Chown 2001), is the greatest threat to this species. This species is also threatened by afforestation of Highveld grasslands (Armstrong and van Hensbergen 1996b); for example, Mpumalanga has already lost at least 9% of its natural vegetation to forestry plantations (Lötter et al. 2014). Mining is also a major ongoing cause of habitat loss, where 25% of the surface of Mpumalanga is under mining application (Lötter et al. 2014), most of which is coal mining.
It appears as if this species has adapted to a mosaic of disturbances, such as that brought about through patch burns (through natural vegetation fires) and/or overgrazing (perhaps from when herds were migratory across the Highveld). Even in formally protected areas, most of these disturbances are managed, thereby prohibiting natural fluctuations. Suppression of fire is thus suspected to be the next most severe threat, as the microhabitats that the species requires are not created or recycled. Although Coetzee and Monadjem (2008) list grazing pressure as contributing to habitat loss, data from Lesotho indicate that the species occurs in overgrazed areas (Avenant 2011, N.L. Avenant unpubl. data).
Conservation
Due to its patchy distribution, this species often does not occur in protected areas that one would expect to find them (for example, De Graaff 1974, Vermeulen 2005). Further surveys should document the full list of protected areas in which it occurs. Currently (post-2000 records), there are confirmed records for Tussen-die-Riviere Nature Reserve (NR) (Ferreira and Avenant 2003), Maria Moroka NR, Soetdoring NR, Erfenisdam NR and De Brug Army Training Camp in the Free State Province; Blaauwberg Conservation Area (BCA) in the Western Cape Province; and Abe Bailey Nature Reserve in Gauteng Province and Boskop Dam Nature Reserve in the North-West.
Conservation of grasslands through protected area expansion and biodiversity stewardship schemes is suspected to be the most important intervention for this species. For example, the Maloti-Drakensberg Transfrontier Conservation and Development Area, established in 2001, accounts for the majority of the c. 2% of the protected grassland within the assessment region (Carbutt et al. 2011) and has higher levels of species richness than neighbouring communally grazed areas (Everson and Morris 2006).
Similarly, biodiversity stewardship schemes are suspected to be conserving grassland patches that would benefit this species. For example, a study in the Little Karoo region (overlapping with White-tailed Rat distribution) found that private conservation areas conserved almost three times the amount of land as formally protected areas for both Succulent Karoo and Renosterveld vegetation types (Gallo et al. 2009), both habitats where the species occurs (albeit marginally).
Grassland condition must also be conserved through correct grazing and fire management. Anecdotal observations suggest that overgrazing is not detrimental to White-tailed Rats (N.L. Avenant unpubl. data). Interestingly, research suggests that continuous cattle grazing systems may have the least negative effect on grasslands (as opposed to continuous sheep grazing or high stocking levels and rotational grazing systems, O’Connor et al. 2010), because continuous grazing dissipates impact across the landscape whereas rotational grazing concentrates impacts on small areas while also leading to fragmentation of the landscape through fencing and infrastructure. Thus, depending on stocking level, game and livestock can be the best land use for grasslands (as opposed to intensive dairy farming, crop agriculture, or rural settlements, O’Connor and Kuyler 2009), which supports the occurrence of this species on such land uses, and should be promoted to limit the sprawl of alternative, damaging land uses.
As this species may be connected to post-fire patches, the pristine condition of the grassland may not be as limiting as the lack of ecosystem functioning and thus phase diversity. Landscapes that burn occasionally are suspected to have greater conservation value. Thus, mosaic burning, within the natural fire frequency of the landscape and within the goals of grazing management for the relevant ungulate species, should be used by managers to promote the persistence of White-tailed Rats, and the effects should be monitored. Further field research is needed to produce evidence for the conservation effectiveness of the proposed interventions for this species specifically (see Research Priorities).
Recommendations for land managers and practitioners:
- Incorporate monitoring the species as a possible indicator of disturbed grassland patches (Avenant 2011), as its presence may indicate that the ecosystem is functioning and that patch heterogeneity exists in the landscape.
- Managers must use fire as a management tool.
- Managers should not over-stock ranch lands and should be encouraged to switch to cattle or wildlife.
- Survey work, including the collection of owl pellets, is needed to determine its occurrence in different land-management areas.
Research priorities:
- Basic ecological research on habitat requirements (and ability to exist on modified landscapes), movement/dispersal capacity between patches, and biogeography.
- Research on the effects of different management systems on presence and abundance. This should be compared to a baseline subpopulation in suitable protected habitat (control).
- Research to test the effectiveness of utilising best management practices as identified or hypothesised above.
- Research to determine colony size, reproduction, dispersal/movement between patches.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP). As this species is easily identified from its white tail and white-grey fur, photographs and coordinates of this species, especially outside of protected areas (for example, livestock and wildlife ranches), will help us to determine its habitat preferences and ecology more accurately.
- Collect and submit owl pellets to relevant museums.
- If a game farm or conservancy landowner, use a mosaic fire management as a tool to create microhabitats for this species. Its occurrence should be monitored.
Bibliography
Armstrong, A.J. and van Hensbergen, H.J. 1996. Impacts of afforestation with pines on assemblages of native biota in South Africa. South African Forestry Journal 175: 35-42.
Armstrong, A.J. and van Hensbergen, H.J. 1996. Small mammals in afforestable montane grasslands of the northern Eastern Cape Province, South Africa. South African Journal of Wildlife Research 26: 11–18.
Avenant, N. and Schulze, E. 2012. Rodent succession in post-fire grassland, Erfenis Dam Nature Reserve, Free State Province, South Africa. 13th Rodens et Spatium Conference – Abstracts: 183.
Avenant, N.L. 2011. The potential utility of rodents and other small mammals as indicators of ecosystem “integrity” of South African grasslands. Wildlife Research 38: 626–639.
Avenant, N.L. and Cavallini, P. 2007. Correlating rodent community structure with ecological integrity, Tussen-die-Riviere Nature Reserve, Free State province, South Africa. Integrative Zoology 2: 212–219.
Avenant, N.L. 2005. Barn owl pellets: a useful tool for monitoring small mammal communities. Belgian Journal of Zoology 135: 39–43.
Avery, D.M. and Avery, G. 2011. Micromammals in the Northern Cape Province of South Africa, past and present. African Natural History 7: 9-39.
Avery, D.M., Avery, G. and Colahan, B.D. 2003. Micromammals and barn owls in the Free State, South Africa: Prey distribution and predator behaviour. Navorsinge van die Nasionale Museum Bloemfontein 19: 2-18.
Avery, D.M., Avery, G. and Palmer, N.G. 2005. Micromammalian distribution and abundance in the Western Cape Province, South Africa, as evidenced by Barn owls Tyto alba (Scopoli). Journal of Natural History 39: 2047–2071.
Avery, D.M., Avery, G. and Roberts, A. 2002. A contribution from barn owl pellets to known micromammalian distributions in KwaZulu-Natal, South Africa. African Zoology 37: 131-140.
Carbutt, C., Tau, M., Stephens, A. and Escott, B. 2011. The conservation status of temperate grasslands in southern Africa. Grassroots 11: 17–23.
Coetzee, N. and Monadjem, A. 2008. Mystromys albicaudatus. The IUCN Red List of Threatened Species 2008: e.T14262A4428195. DOI: 10.2305/IUCN.UK.2008.RLTS.T14262A4428195.en.
De Graaff, G. 1974. Notes on the occurrence of rodents in South African National Parks. Koedoe 17: 173–183.
De Graaff, G. 1981. The Rodents of Southern Africa. Butterworths, Durban, South Africa.
Dean, W.R.J. 1978. Conservation of the white-tailed rat in South Africa. Biological Conservation 13: 133–140.
Downs, C.T. and Perrin, M.R. 1995. The thermal biology of the white-tailed rat Mystromys albicaudatus, a cricetine relic in southern temperate African grassland. Comparative Biochemistry and Physiology 110A: 65–69.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Everson, T.M. and Morris, CD. 2006. Conservation of biodiversity in the Maloti-Drakensberg Range. In: E.M. Spehn, Land Use Change and Mountain Biodiversity, pp. 285-291. CRC Press, Boca Raton, USA.
Ferreira, S.M. and Avenant, N.L. 2003. Modelling the effects of trap-spacing on small mammal community descriptors in grasslands at Tussen-die-Riviere Nature Reserve, Free State Province, South Africa. Navorsinge van die Nasionale Museum, Bloemfontein 19: 21-30.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. Conservation Breeding Specialist Group (SSC/IUCN) and Endangered Wildlife Trust, Parkview, South Africa.
Gallo, J.A., Pasquini, L., Reyers, B. and Cowling, R.M. 2009. The role of private conservation areas in biodiversity representation and target achievement within the Little Karoo region, South Africa. Biological Conservation 142: 446–454.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Hall 3rd, A., Persing, R.L., White, D.C. and Ricketts Jr, R.T. 1967. Mystromys albicaudatus (the African white-tailed rat) as a laboratory species. Laboratory Animal Care 17: 180.
Hickman, G.C. and Machiné, C. 1986. Swimming behaviour in six species of African rodents (Cricetidae, Muridae). Acta Theriologica 31: 449–466.
IUCN Standards and Petitions Subcommittee. 2014. Guidelines for Using the IUCN Red List Categories and Criteria. Version 11. Available at: http://jr.iucnredlist.org/documents/RedListGuidelines.pdf.
IUCN. 2019. The IUCN Red List of Threatened Species. Version 2019-1. Available at: www.iucnredlist.org. (Accessed: 21 March 2019).
Jansa, S.A., Goodman, S.M. and Tucker, P.K. 1999. Molecular phylogeny and biogeography of the native rodents of Madagascar (Muridae: Nesomyinae): a test of the single-origin hypothesis. Cladistics 15: 253–270.
Kaiser, W. 2006. The characteristics of insect and small mammal communities as a reflection of the ecological value of grasslands. M.Sc. Thesis. University of the Free State.
Kok, A.D., Parker, D.M. and Barker, N.P. 2012. Life on high: the diversity of small mammals at high altitude in South Africa. Biodiversity and Conservation 21: 2823–2843.
Kuyler, P. 2000. Veld condition assessment and small mammal community structure in the management of Soetdoring Nature Reserve, Free State, South Africa. Masters Thesis. niversity of the Free State.
Lawal, S., Lennard, C., Hewitson, B. 2019 Response of southern African vegetation to climate change at 1.5 and 2.0° global warming above the pre-industrial level. Journal of Climate Services 16: 1-16
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
Lötter, M.C., Cadman, M.J. and Lechmere-Oertel, R.G. 2014. Mpumalanga Biodiversity Sector Plan Handbook. MTPA. Nelspruit.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. . De Gruyter, Berlin, Germany.
Morwe, J.B. 2013. Determining the direct impact of black-backed jackal (Canis mesomelas) on the springbok (Antidorcas marsupialis) population at Maria Moroka Nature Reserve, Free State, South Africa. B.Sc. Honours Thesis. University of the Free State.
Mucina, L. and Rutherford, M.C. 2006. The Vegetation of South Africa, Lesotho and Swaziland. Strelitzia 19. South African National Biodiversity Institute, Pretoria, South Africa.
NW READ. 2014. North West Environment Outlook Report 2013. North West Provincial Government, Mahikeng, South Africa.
National Institute for Communicable Diseases. 2005. Plague Control Guidelines for South Africa. Department ofHeath, South Africa.
Neke, K.S. and du Plessis, M.A. 2004. The threat of transformation: quantifying the vulnerability of grasslands in South Africa. Conservation Biology 18: 466–477.
O’Connor, T.G. and Kuyler, P. 2009. Impact of land use on the biodiversity integrity of the moist sub-biome of the grassland biome, South Africa. Journal of Environmental Management 90: 384–395.
O’Connor, T.G., Kuyler, P., Kirkman, K.P. and Corcoran, B. 2010. Which grazing management practices are most appropriate for maintaining biodiversity in South African grassland? African Journal of Range & Forage Science 27: 67–76.
O’Farrell, P.J., Donaldson, J.S., Hoffman, M.T. and Mader, A.D. 2008. Small mammal diversity and density on the Bokkeveld escarpment, South Africa-implications for conservation and livestock predation: short communications. African Zoology 43: 117–124.
Pacifici M, Santini L, Di Marco M, Baisero D, Francucci L, Grottolo Marasini G, Visconti P, Rondinini C (2013) Database on generation length of mammals. 5427 data records. Online at http://doi.org/10.5061/dryad.gd0m3, version 1.0 (last updated on 2013-08-27), Resource ID: 10.5061/dryad.gd0m3, Data Paper ID: doi: 10.3897/natureconservation.5.5734
Parr, C.L. and Andersen, A.N. 2006. Patch mosaic burning for biodiversity conservation: a critique of the pyrodiversity paradigm. Conservation Biology 20: 1610–1619.
Perrin, M. 2013. Mystromys albicaudata African White-tailed Rat. In: D.C.D. Happold (ed.), Mammals of Africa vol. III: Rodents, Hares and Rabbits., pp. 201–203. Bloomsbury Publishing, London, UK.
Perrin, M.R. and Maddock, A.H. 1983. Preliminary investigations of the digestive processes of the white-tailed rat Mystromys albicaudatus (Smith 1834). South African Journal of Zoology 18: 128–133.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Rautenbach, I.L. 1978. Ecological distribution of the mammals of the Transvaal (Vertebrata: Mammalia). Annals of the Transvaal Museum 31: 131-156.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Specker, S. 2006. Determining the effects of fire on Mystromys albicaudatus within the Blaauwberg Conservation Area. Cape Peninsula University of Technology, Cape Town, South Africa.
Taylor, P. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.
Vermeulen, I. 2005. Determining the abundance, habitat preference and distribution of Mystromys albicaudatus within the Blaauwberg Conservation Area. Cape Peninsula University of Technology, Cape Town, South Africa.
de Beer, T. 1990. Kleinsoogdieropname: Boskop dam natuurreservaat. Internal report. Transvaal Provincial Administration, Pretoria, South Africa.
van Jaarsveld, A.S. and Chown, S.L. 2001. Climate change and its impacts in South Africa. Trends in Ecology & Evolution 16: 13–14.