
White-bellied House Bat
Scotophilus leucogaster

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Scotophilus leucogaster – (Cretzschmar, 1826)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – VESPERTILIONIDAE – Scotophilus – leucogaster
Common Names: White-bellied Yellow Bat, Cretzschmar’s Brown Bat, Lesser Yellow House Bat, White-bellied House Bat (English)
Synonyms: Nycticejus leucogaster (Cretzschmar 1826), flavigaster (Heuglin, 1861), murinoflavus (Heuglin, 1861), Scotophilus damarensis O. Thomas, 1906
Taxonomic Note:
South Africa
The genus has undergone extensive taxonomic work, yet it still requires revision. Scotophilus leucogaster occurs in sympatry with S. viridus through part of its’ range. The two species can be discerned from one another based on the pale cream belly colouring in S. leucogaster versus the richer yellow belly colouring of S. viridis. Historically the two taxa were treated as conspecifics (Meester et al. 1986), however Robbins et al. (1985) recognised them as separate species based on morphological analyses, a view corroborated by molecular analyses (Jacobs et al. 2006; see Monadjem et al. 2020).
Demos et al. (2018) considers S. leucogaster a species complex and recommends further taxonomic revision. For example, Robbins et al. (1985) considered white-bellied specimens from Namibia (damarensis) to be conspecific with populations of leucogaster from other parts of Africa. Additionally, the bats identified as S. leucogaster by Robbins et al. (1985) show a discontinuous African distribution, mostly absent from between 5°N and 12°S. This evidence suggests that the southern population is phylogenetically distinct, which would render S. damarensis specifically distinct as a southern African endemic (Monadjem et al. 2020).
Red List Status: LC – Least Concern (IUCN version 3.1)
Assessment Information
Assessors: Richardson, E.J.1, Richards, L.R.2 & da Silva, J.M.3
Reviewer: Smith, C.4
Institutions: 1Independent Consultant at Richardson & Peplow Environmental, 2Durban Natural Science Museum, 3South African National Biodiversity Institute, 4Endangered Wildlife Trust
Previous Assessors and Reviewers: Taylor, P., Richards, L.R., White, W. & Child, M.F.
Previous Contributor: Monadjem, A.
Assessment Rationale
This species is sparsely distributed wherever it is found in the assessment region, although it is found in a large, protected area (Kruger National Park) in northeastern South Africa. Further field surveys and vetting of distribution records should be undertaken to improve the accuracy of its range map within the region. Within the assessment region this species has an estimated extent of occurrence of 4,183 km2 and less than 10 locations: this is close to being assessed as Vulnerable B1 but in the absence of any information on population sizes and changes this assessment has to remain Least Concern. Further molecular research is urgently needed to assess whether S. damarensis is a distinct species, in which case a reassessment will be necessary.
Regional population effects: Further research on its dispersal capacity and connectedness is necessary. However, since it occurs in the Greater Limpopo Transfrontier Park and its habitat is thus connected to Mozambique and Zimbabwe (Monadjem et al. 2020), we assume rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No Change
Red List Index
Red List Index: No Change
Recommended citation: Richardson EJ, Richards LR & da Silva JM. 2025. A conservation assessment of Scotophilus leucogaster. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The White-bellied Yellow Bat is widely distributed north of 5°N but sparsely and discontinuously distributed in the central savannahs of southern Africa, occurring in northern South Africa, southern Mozambique, and southern Zimbabwe, with a separate population in northern Zimbabwe, central Mozambique and southern Zambia, extending west to northern Botswana and Namibia, with an isolated record from central Angola (Monadjem et al. 2020). Habitat models suggest it might be more widespread in southern Africa than currently recorded (Monadjem et al. 2010), but there is also uncertainty about the validity of its range in southern Africa that encompasses Zambia, Botswana, South Africa and Angola. As such, further molecular research is needed to delimit its range more accurately. Within the assessment region, it occurs in Kruger National Park (KNP) in the Limpopo Province. Specimens from Punda Maria (KNP) include large and small animals as well as specimens with both yellow and no yellow in their ventral pelage, which necessitates re-examination of the specimens to determine whether they correspond to either S. leucogaster or S. viridis (Monadjem et al. 2010). The type specimen for the subspecies S. l. damarensis is from northern Namibia (Monadjem et al. 2010, 2020). The estimated extent of occurrence for this species within the assessment region is 4,183 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 6 m asl (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 2,153 (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
Map
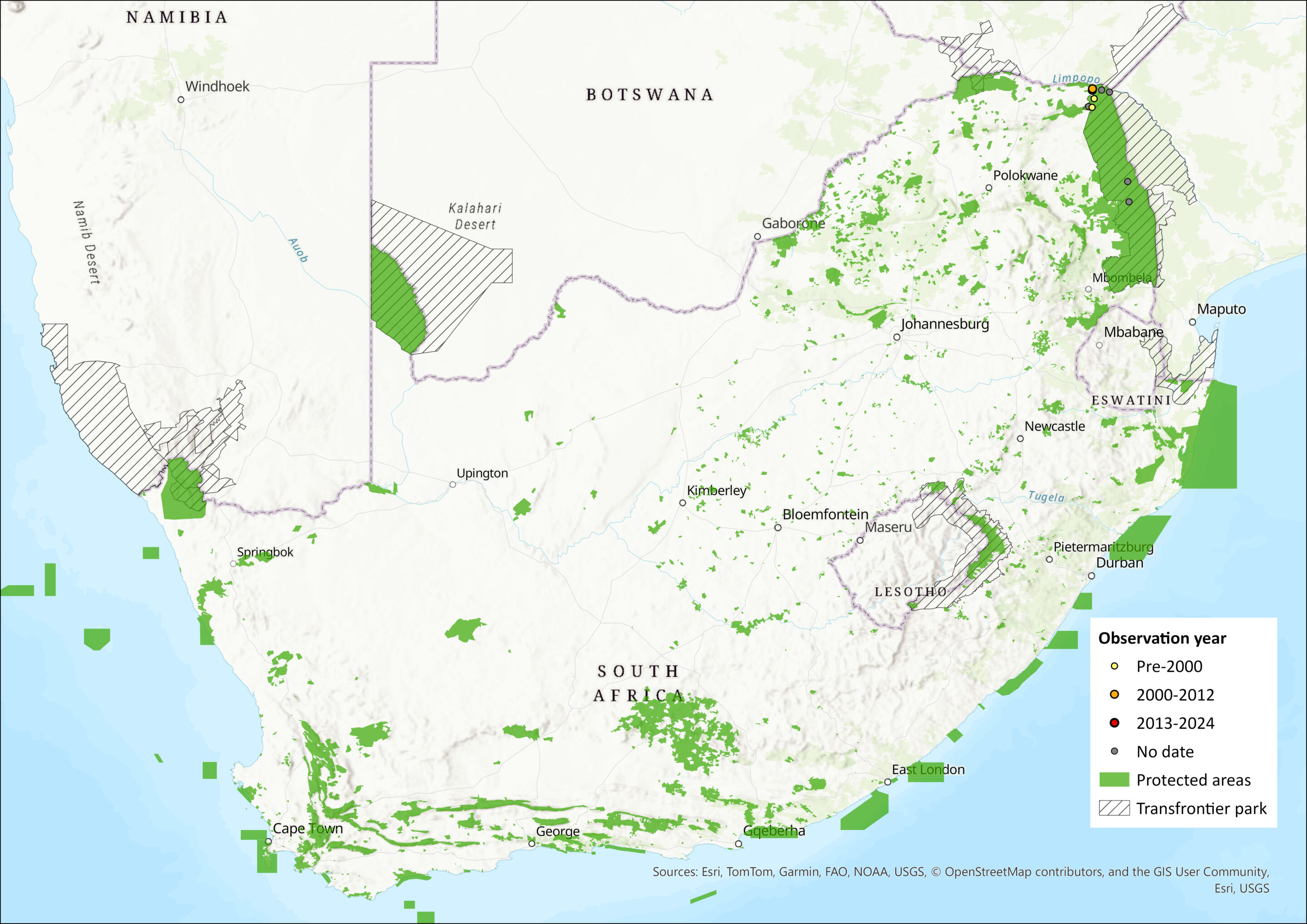 Figure 1. Distribution records for White-bellied Yellow Bat (Scotophilus leucogaster) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for White-bellied Yellow Bat (Scotophilus leucogaster) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: (Not specified)
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | |
| Benin | Extant | Native | ||
| Botswana | Extant | Native | – | – |
| Burkino Faso | Extant | Native | ||
| Cameroon | Extant | Native | ||
| Central African Republic | Extant | Native | ||
| Chad | Extant | Native | ||
| Côte d’Ivoire | Extant | Native | ||
| Ethiopia | Extant | Native | ||
| Gambia | Extant | Native | ||
| Ghana | Extant | Native | ||
| Guinea | Extant | Native | ||
| Guinea-Bissau | Extant | Native | ||
| Kenya | Extant | Native | ||
| Mali | Extant | Native | ||
| Mauritania | Extant | Native | ||
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | ||
| Nigeria | Extant | Native | ||
| Saudi Arabia | Extant | Native | ||
| Senegal | Extant | Native | ||
| Sierra Leone | Extant | Native | ||
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | ||
| Togo | Extant | Native | ||
| Uganda | Extant | Native | ||
| Yemen | Extant | Native | ||
| Zambia | Extant | Native | – | |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
Nothing is known about the potential impact of climate change on this species, but globally, there have been documented declines in bat populations, species richness and distributions in relation to water availability with increasing global aridity which may become a growing concern as heat waves and maximum temperatures are expected to increase over much of South Africa (Adams & Hayes 2021; Mbokodo et al. 2020). Bats may also be more susceptible to extreme weather events than to climate shift (see, for example, Matthew et al. 2020).
Kock (1969) reported of lethargy and clumsy flights in bats from Sudan when temperatures feel below 34°C, suggesting that individuals may be physiologically adapted to the high temperatures of the region. Conversely, bats of the closely related S. dinganii have been recorded leaving their roof roosts and hanging exposed on the exterior walls of houses during extremely hot days (Bats KZN data); it is possible that this species also may be affected by heat waves and increasing maximum temperatures.
Barclay (1985) reports of dietary shifts linked to changing season in Zimbabwe individuals; Coleoptera and Hemiptera comprise important prey items in the wet season, whereas the diet comprised mostly Lepidoptera in the dry season. Climate change and the predicted increase in precipitation and temperature seasonality is likely to affect the insect biomass, and consequently the distribution and diet of bat species.
Population Information
Little information is available on the population abundance or size of this species across its range but is presumably uncommon. This species is not well represented in museums, with only 46 records examined in Monadjem et al. (2020), which is due to its more restricted distribution compared with S. dinganii.
Current population trend: Stable
Continuing decline in mature individuals: No
Extreme fluctuations in the number of subpopulations: Cannot be determined/unknown
Continuing decline in number of subpopulations: Unknown
All individuals in one subpopulation: Likely
Number of mature individuals in largest subpopulation: Not known
Number of Subpopulations: Has not been determined
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Cannot be determined based on available data
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Cannot be determined based on available data
Probability of extinction in the wild within 100 years: Cannot be determined based on available data
Population Genetics
No population genetic studies have been conducted on this species; however, given its highly restricted distribution, it is presumed to exist as a single population, connected to colonies in adjacent countries (e.g., Zimbabwe, Mozambique).
Due to the limited knowledge of this species population size, its effective population size cannot be estimated. Population genetic studies would be beneficial to better inform the genetic health of this species within the assessment region.
Habitats and ecology
This species has been recorded from both dry and moist savanna habitats. During the day, they roost in a variety of shelters, including holes in trees (Fenton 1983), such as Mopane (Colophospermum mopane) (Monadjem et al. 2020), gaps under bark, and roofs of houses. Roost sites may be changed regularly (Fenton et al. 1985). This species has a strong association with cathedral mopane woodland, especially in the Limpopo, Sebungwe and Zambezi basins of Zimbabwe, where it is often the most abundant microbat (Fenton 1985). At Sengwa Wildlife Research Station, Zimbabwe, the diet of Scotophilus leucogaster comprised mainly Hemiptera and Coleoptera, with Hymenoptera, Homoptera, Orthoptera, Lepidoptera and Diptera present in small numbers (Barclay 1985). Individuals foraged primarily over floodplains for an average of less than one hour at dusk and spent the rest of the night in small tree-cavity roosts (Barclay 1985).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
Life History
Generation Length: Unknown
Age at Maturity: Female or unspecified: Unknown
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Female: Mean total length = 12.30 ±0.43 cm; Mean forearm length 5.03 ±0.09 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: mean total length 12.0 ±0.35 cm; Mean forearm length 4.92 ±0.52 cm (Monadjem et al. 2020)
Longevity: (Not specified)
Average Reproductive Age: Unknown
Maximum Size (in cms): Forearm length Females = 5.15 cm; Males = 5.19 cm (Monadjem et al. 2020)
Size at Birth (in cms): Unknown
Gestation Time: Unknown
Reproductive Periodicity: Reports from southern Africa indicate the species exhibits seasonal monoestry (Barclay 1985)
Average Annual Fecundity or Litter Size: Two pups per litter
Natural Mortality: Preyed upon by Barn owls and Bat hawks (Van Cakenberghe & Happold 2013))
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Unknown
Congregatory: roost in small colonies elsewhere in Africa
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
Local Livelihood: None reported
National Commercial Value: May provide ecosystem services in the form of pest insect control.
International Commercial Value: May provide ecosystem services in the form of pest insect control.
End Use: None reported
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: N/A
Threats
Except for climate change and extreme weather events, there appears to be no major threats to this species as a whole. Similarly, no threats to the South African population have been identified.
Conservation
This species occurs in Kruger National Park in the north of the assessment region. No direct conservation interventions are necessary at present.
Research priorities:
- Taxonomic resolution through integrative molecular and morphometric research.
- Field surveys delimiting geographical distribution and habitat preferences.
- Quantification of threats potentially facing this species.
Encouraged citizen actions:
- Deposit any dead specimens to the Durban Natural Science Museum or Ditsong Museum of Natural History.
- List sightings on citizen science platforms (e.g. iNaturalist)
Bibliography
Adams, R.A. and Hayes, M.A. 2021. The importance of water availability to bats: climate warming and increasing global aridity. 50 years of bat research: foundations and new frontiers, pp.105-120.
Barclay, R.M. 1985. Foraging behavior of the African insectivorous bat, Scotophilus leucogaster. Biotropica 17: 65–70.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Fenton, M.B., Brigham, R.M., Mills. A.M. and Rautenbach, I.L. 1985. The roosting and foraging areas of Epomophorus wahlbergi (Pteropodidae) and Scotophilus viridis (Vespertilionidae) in Kruger National Park, South Africa. Journal of Mammalogy 66: 461–468.
Fenton, M.B. 1985. The feeding behaviour of insectivorous bats: echolocation, foraging strategies, and resource partitioning. Transvaal Museum Bulletin 21: 5–19.
Fenton, M.B. 1983. Roosts used by the African bat, Scotophilus leucogaster (Chiroptera: Vespertilionidae). Biotropica 15: 129–132.
Jacobs, D.S., Eick, G.N., Schoeman, M.C. and Matthee, C.A. 2006. Cryptic species in an insectivorous bat, Scotophilus dinganii. Journal of Mammalogy 87: 161–170.
Kock, D. 1969. Die Fledermaus-Fauna des Sudan (Mammalia, Chiroptera) (pp. 1-238).
Matthew, M. et al. 2022. Estimating flying-fox mortality associated with abandonments of pups and extreme heat events during the austral summer of 2019–20. Pacific Conservation Biology 28, 124-139
Mbokodo, I., Bopape, M.J., Chikoore, H., Engelbrecht, F. and Nethengwe, N. 2020. Heatwaves in the future warmer climate of South Africa. Atmosphere, 11(7), p.712.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum, Pretoria, South Africa.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2010. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Robbins, C. B., De Vree, F. and Van Cakenberghe, V. 1985. A systematic revision of the African bat genus Scotophilus (Vespertilionidae). Annales Musue Royal de l’Afrique Centrale, Sciences Zoologiques 246: 53-84.
Van Cakenberghe, V. & Happold, M. 2013. Scotophilus leucogaster. White-bellied House-bat. pp. 676–678. In: Happold, M. & Happold, D. (Eds.), Mammals of Africa. Volume IV. Hedgehogs, shrews and bats. Bloomsbury, London, New Delhi, New York and Sydney.
