
Wahlberg’s Epauletted Fruit Bat
Epomophorus wahlbergi

2025 Red list status
Least Concern
Regional Population Trend
Declining
Change compared
to 2016
No Change
Overview
Epomophorus wahlbergi – (Sundevall, 1846)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – PTEROPODIDAE – Epomophorus – wahlbergi
Common Names: Wahlberg’s Epauletted Fruit Bat (English), Wahlberg se Vrugtevlermuis (Afrikaans)
Synonyms: unicolor (Gray 1870), neumanni Matschie 1899, stuhlmanni Matschie 1899, Matschie 1899
Taxonomic Note:
No subspecies are recognised (Monadjem et al. 2020). Adult Epomophorus wahlbergi can be distinguished from the sympatric E. crypturus by the presence of just one post-dental palatal ridge (two in E. crypturus) and broader muzzle (Taylor & Monadjem 2008).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Richardson, E.1, Lötter, C.2, & da Silva, J.M.3
Reviewers: Moir, M.4
Institutions: 1Richardson & Peplow Environmental, 2Inkululeko Wildlife Services (Pty) Ltd, 3South African National Biodiversity Institute, 4Stellenbosch University
Previous Assessors and Reviewers: Schoeman, C., White, W., Monadjem, A., Richards, L.R., Cohen, L., Jacobs, D., MacEwan, K., Sethusa, T. & Taylor, P.
Previous Contributors: Roxburgh, L., Raimondo, D., Page-Nicholson, S. & Child, M.F.
Assessment Rationale
Though threatened by loss of habitat, increasing fatalities due to wind turbines, and increasing frequency and duration of droughts in parts of its range, the species is listed as Least Concern in view of its wide distribution (estimated extent of occurrence is 765,224 km2), presumed large population, its occurrence in many protected areas and modified habitats, and because it is unlikely to be declining fast enough to qualify for listing in a more threatened category. However, with increasing loss of habitat, and with the increasing number of wind farms across the eastern part of the assessment region where this species occurs, fatalities from wind turbines may become a severe threat and must, therefore, be carefully monitored and mitigated.
Regional population effects: Its habitat is connected to the rest of its southern African range where it faces the same challenges, although rescue effects may be possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Richardson E, Lötter C & da Silva JM. 2025. A conservation assessment of Epomophorus wahlbergi. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Wahlberg’s Epauletted Fruit Bat is found in Central Africa, East Africa and southern Africa where it is widespread and abundant in the eastern regions (Monadjem et al. 2020). It has been recorded from Cameroon, Equatorial Guinea, Gabon, Congo and Angola in the west, through the southern Democratic Republic of the Congo and Rwanda. The species is also distributed in East Africa from Uganda, Kenya, southern Somalia, Tanzania (including Zanzibar), Zambia, Malawi and Mozambique into Zimbabwe (ACR 2024). Within the assessment region, the species is distributed in the southern and eastern areas of South Africa, where it has been recorded from the Western Cape through Eswatini (Skinner & Chimimba 2005; Monadjem et al. 2020). The type specimen is from near Durban, KwaZulu-Natal (Monadjem et al. 2010). Its estimated extent of occurrence is 765,224 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 2000
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
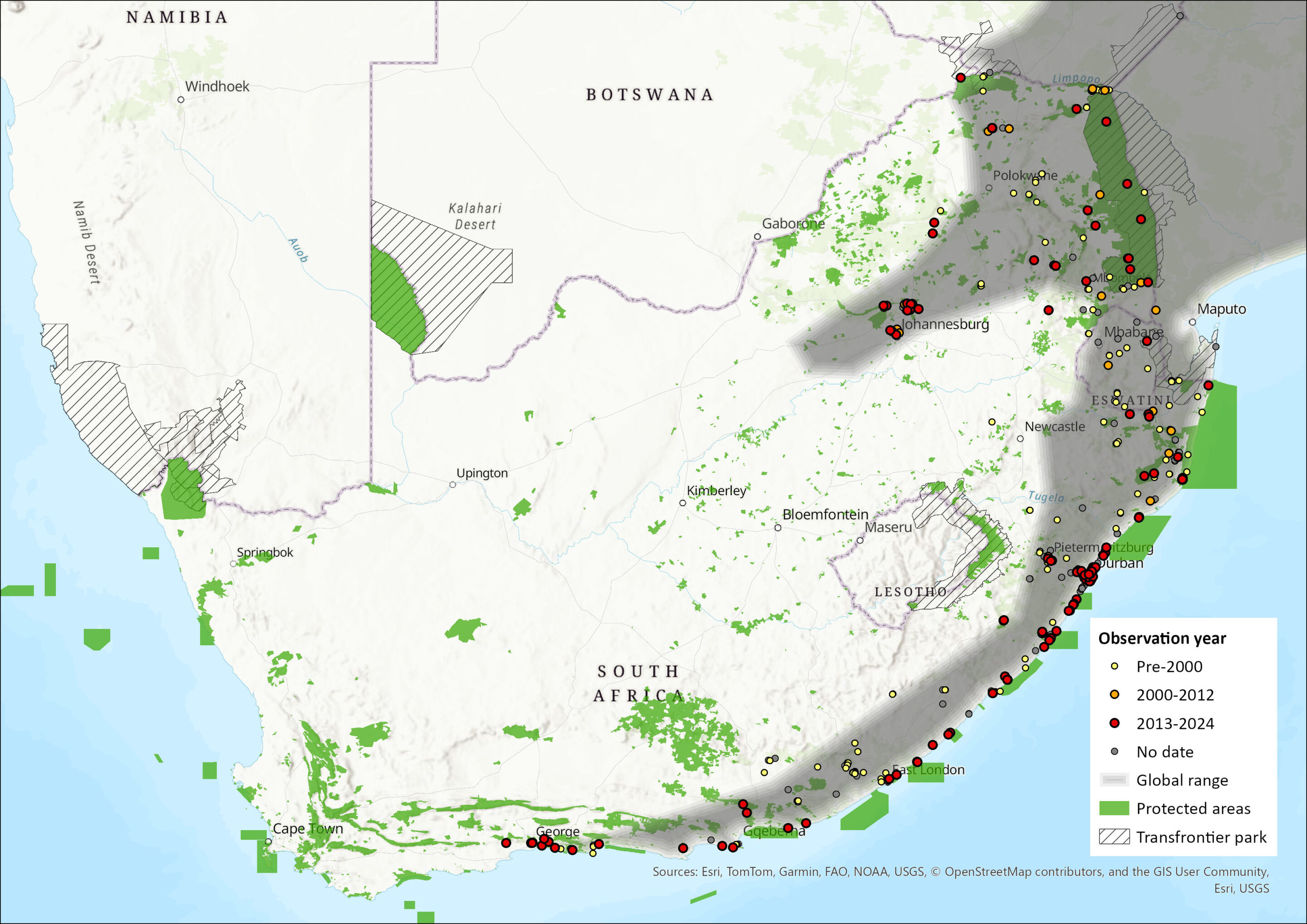 Figure 1. Distribution records for Wahlberg’s Epauletted Fruit Bat (Epomophorus wahlbergi) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Wahlberg’s Epauletted Fruit Bat (Epomophorus wahlbergi) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Presence Uncertain | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Equatorial Guinea | Presence Uncertain | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Arumoogum et al. (2019) used ensemble niche modelling to investigate the relative influence of abiotic and biotic variables on the potential current and future distributions of three fruit bat species in southern Africa. Abiotic variables associated with productivity (synergistic effects of temperature and rainfall) were found to be the primary determinants of habitat suitability for Wahlberg’s Epauletted Fruit Bat. As such, this species may become increasingly threatened with a possible increase in the frequency and duration of droughts within its distribution range.
All of the Pteropodids are susceptible to extreme weather conditions, especially heat waves (see, for example Matthew et al. 2022). Increased deaths associated with the increasing frequency and severity of extreme weather events may seriously impact species numbers.
Population Information
Wahlberg’s Epauletted Fruit Bat is relatively common within its range, and is well represented in museums, with over 280 specimens examined in Monadjem et al. (2020). The population trend is difficult to determine, as roosting areas fluctuate depending on availability of seasonal resources and the presence of threats. The overall population within the assessment region may be declining slowly due to i) significant levels of deforestation within KwaZulu-Natal and the Eastern Cape (www.globalforestwatch.org); ii) mortalities caused by wind turbines (MacEwan 2016; Aronson 2022; Inkululeko Wildlife Services unpubl. data); and iii) extended periods of drought in recent years, such as in the Eastern Cape and KwaZulu-Natal (Bat Interest Group KwaZulu-Natal, unpubl. data). Numbers should be systematically monitored to establish population trends.
Current population trend: Fluctuates, but overall, declining slowly.
Continuing decline in mature individuals: Yes
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Extreme fluctuations in the number of subpopulations: Roosting colonies fluctuate in size depending on the availability of resources (e.g., fruiting trees) and the presence of threats (e.g., drought or predators).
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of subpopulations: Unknown, but >50 colonies
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
No population genetic study has been conducted on this species; however, the distribution records suggest the species likely exists as a single metapopulation within the assessment region. Despite the lack of actually census counts or measure of effective population size, it is presumed that there are more than 5,000 individuals within the assessment region.
From this information we can quantify the Convention of Biological Diversity’s two population genetic indicators. As a metapopulation that is still in existence, the populations maintained indicator would receive a value if 1.0 (1/1 populations remain). And with at least 5000 individuals in this metapopulation, and applying a conversion ratio of 0.1-0.3, the effective population size for this metapopulation is estimated between 500- 1500. Correspondingly, the Ne 500 indicator would receive a value of 1.0 (1/1 populations with Ne > 500).
It is highly recommended population genetic studies be undertaken to verify the genetic structure and diversity within this species.
Habitats and ecology
This species is associated with forest and forest-edge habitats, including riparian and mangrove forests with fruiting trees, in peri-urban areas with extensive wooded gardens (Monadjem et al. 2020), as well as woodland savannahs (including miombo, mopane, Acacia-Commiphora deciduous woodlands and thickets). Epomophorus crypturus seems to prefer drier habitats and its capture rate increases in northern regions whereas that of E. wahlbergi increases in the south (Monadjem et al. 2020). Roosting sites are often under the canopy of trees, in thick foliage (Monadjem et al. 2020), with animals roosting singly or in small groups, sometimes present in considerable numbers. The species is somewhat adaptable to habitat modification, with subpopulations recorded from well wooded urban and suburban areas, where it is associated with fruit-bearing trees (especially fruit trees). It occasionally roosts in man-made structures (ACR 2024). This species continually changes its roost site, which appears to be an anti-predation strategy. It has been recorded to travel several kilometres each night to reach fruiting trees (Fenton et al. 1985, Rollinson et al. 2013). In the assessment region, the species is recorded from Eastern Kalahari Bushveld, Upper Karoo, Dry Highveld Grassland, Central Bushveld, Mopane Bioregion, Lowveld, Indian Ocean Coastal Belt, Sub-Escarpment Savanna, Eastern Fynbos-Renosterveld, Rainshadow Valley Karoo, Sub-Escarpment Grassland, Albany Thicket and Drakensberg Grassland. It feeds on fruit (including both indigenous and cultivated species), nectar, pollen and flowers (Monadjem et al. 2020). Fig trees appear to be favoured food resources; for example, in Kruger National Park, it specialises on the fruits of Ficus sycomorus (Fenton et al. 1985; Bonaccorso et al. 2014), where mean foraging range and core use area changes significantly with season, year, and fig abundance (Bonaccorso et al. 2014). Where this species is sympatric with E. crypturus, differences in mating vocalisations may be used by both species to avoid cross-mating (Adams & Snode 2015).
Ecosystem and cultural services: As this species is frugivorous, it plays a crucial role in pollination and seed dispersal of many plant species and thus is important for plant community persistence and habitat regeneration within its range (Monadjem et al. 2020; Kunz et al. 2011). This species of bat has been recorded to pollinate Baobabs (Adansonia digitata), an economically important tree in the African savannah (Monadjem et al. 2020). Several studies (Adams and Snode 2013; Bonaccorso et al. 2014; Aziz et al. 2021; Mphethe et al. 2023) have shown that Epauletted fruit bats prefer wild fruits (to cultivated fruit), particularly, figs. Epomophorus bats are indeed key seed dispersers for fig trees (Ficus spp.), which represent a keystone resource for numerous frugivores and other animals, and which are economically important trees in the African savannah.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | Yes |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | Yes |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | Yes |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Marginal | – |
| 3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist | – | Marginal | – |
| 4.4. Grassland -> Grassland – Temperate | – | Marginal | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Marginal | – |
| 14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas | – | Marginal | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified) 12 months
Age at Maturity: Male: (Not specified) 18 months
Size at Maturity (in cms): Female: Forearm mean = 8.12 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Forearm mean = 8.51 cm (Monadjem et al. 2020)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: Births occur mainly in November but occasionally in July
Average Annual Fecundity or Litter Size: One, very rarely two offspring (Monadjem et al. 2020)
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Telemetry work in the Kruger National Park showed that individuals of this species may travel more than 13 km a night between roosting and feeding sites, but most flight activity was within 400 m of a Ficus sycomorus tree with ripe fruits, and home ranges averaged approximately 16 ha (Bonaccorso et al. 2014). In Luvhondo Nature Reserve in the Soutpansberg, Limpopo Province, a tagged female spent most of its time foraging very close to its roost, concentrating its activity over natural habitats with ripe figs, and did not visit litchi orchards during the litchi harvest season (Mphethe et al. 2023). In an urban environment of Pietermaritzburg, KwaZulu-Natal, the mean maximum distance travelled at night by individuals of this species was 1.45 km in winter and 0.88 km in spring, and female and male home ranges were 18-61 ha and 13-54 ha, respectively (Rollinson et al. 2013). If the nightly movements and home ranges of this species are larger during winter when food may be scarce, this may explain why fatalities of this species occur more often during winter at wind farms on the South African south Cape coast (Inkululeko Wildlife Services unpubl. data).
Congregatory: This species roosts singly or in groups of up to 65 animals, depending on the availability of suitable and safe roosts.
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence that this species is traded or utilised within the assessment region.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are several threats that could be causing localised colony declines and a gradual overall population decline. A decline in the population of E. wahlbergi in KwaZulu-Natal has been observed, with the cause suspected to be loss of coastal forest as well as extensive drought (Bat Interest Group KwaZulu-Natal unpubl. data). The species is also threatened by loss of habitat in the Eastern Cape due to deforestation for subsistence fuel wood use. From 2001 to 2023, the Eastern Cape lost 193 kha of tree cover, equivalent to a 12% decrease in tree cover since 2000. This included a loss of 6.58 kha or 7.6% of humid primary forest between 2002 and 2023 (www.globalforestwatch.org). From 2001 to 2023, KwaZulu-Natal lost 624 kha of tree cover, equivalent to a 28% decrease in tree cover since 2000. It is uncertain how much of this comprised primary forest (www.globalforestwatch.org). It is also suspected that the Lagos Bat Virus may have affected the KwaZulu-Natal population as a number of bats caught from Durban and the South Coast tested seropositive for the virus.
Moreover, wind farms in the eastern portion of the assessment region may pose a serious threat for the species. Bats that fly near to turbine blades generally collide directly with a blade, but may experience barotrauma, which is tissue damage caused by rapid changes in air pressure near the blades) (Baerwald et al. 2008; Cryan & Barclay 2009; Rydell et al. 2010). Growing numbers of fatalities of Epomophorus species from wind turbines have been recorded since 2015 and are likely to continue to escalate (MacEwan 2016; Aronson 2022; Inkululeko Wildlife Services unpubl. data). The impact of these threats on the overall population in the assessment region is currently unknown, and the species needs to be monitored, especially in relation to mortalities from wind turbines. Roost disturbance and removal of large trees used as roost sites are also plausible threats in some parts of its range.
Conservation
The species is present in many protected areas, including Kruger National Park. Identification of key roost sites and subsequent protected area expansion, and habitat rehabilitation in areas suffering from habitat loss, would benefit this species. Additionally, the impact of wind farming should be monitored to determine its impact on this species. An effective and standardised method for quantifying the activity of fruit bats at proposed and operational wind farm sites is needed. Thermal binoculars may prove useful in this regard. To mitigate mortalities from turbine collisions, development of wind farms must avoid encroachment into the prescribed buffers around important habitat (e.g., forest, riverine woodland, large fruiting trees) and confirmed and potential roosts of this species (for buffer recommendations see MacEwan et al. 2020 or later). Turbine-fatalities of this species must be reduced below fatality threshold values (as calculated according to MacEwan et al. 2018 or later) with interventions such as curtailing turbines at low wind speeds during times of the year with known fruit bat fatalities (Baerwald et al. 2009; Berthinussen et al. 2010; Arnett et al. 2011). At wind farms near the south-eastern South African coastline, fatalities of this species have occurred most often (but not exclusively) during winter (specifically June and July; Inkululeko Wildlife Services unpubl. data). Therefore, curtailment of turbines to mitigate fatalities of Epomophorus species at wind farms in this part of South Africa may be most (albeit not 100%) effective during winter. As high bat fatalities at wind farms cannot be easily offset (Aronson et al. 2018; Mark Botha pers. comm.), avoidance and minimisation of bat fatalities is critical. Improved conservation (protection, rehabilitation, and expansion) of forest, riverine, and savannah habitats would be beneficial for this species.
Recommendations for land managers and practitioners:
- Development of wind farms must strictly avoid encroachment into the prescribed buffers around roosts and other important habitat for fruit bats, especially since current options for mitigating fatalities at wind farms are more limited for fruit bats compared to insectivorous bats.
- Turbine-fatalities of Epomophorus species must be reduced with interventions such as curtailing turbines at low wind speeds during times of the year with known fatalities.
- Wind farm carcasses of Epomophorus fruit bats should be identified to species level based on the number of post-dental palatal ridges (one in E. wahlbergi; two in E. crypturus) and muzzle width (or DNA sequencing, if feasible).
- Data sharing by wind farm managers into a national database is needed to be able to calculate cumulative impacts and thereafter implement collaborative mitigation and management efforts.
- Farmers should be educated on the ecological benefits of having fruit bats around their orchards, and the importance of preserving natural habitat on their farms.
- Protect large fruit bearing trees.
Research priorities:
- A meta-analysis of all recorded E. wahlbergi fatalities at wind farms in South Africa to date, to assess the cumulative impact of the industry on this species, and to identify any spatial, temporal, or turbine-specific trends, which may assist in devising effective fatality mitigation for future implementation.
- Identification of key roost sites and systematic monitoring to determine subpopulation trends.
- Research to assess the efficacy of different mitigation methods to reduce fruit bat
- mortality at wind farms. For example, it is not known whether turbine blade painting (to mitigate avian
- fatalities) is beneficial or detrimental for fruit bats.
- Wind farm carcasses of this species could be used to obtain greater insight into the biogeography, genetics, reproductive biology, ecology (e.g., diet), and ecosystem services of this species in eastern southern Africa.
Encouraged citizen actions:
- Limit disturbance to roost sites.
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution. This species is easily confused with E. crypturus but can be distinguished by the presence of only one post-dental palatal ridge (two in E. crypturus) (Taylor & Monadjem 2008).
Bibliography
ACR. 2015. African Chiroptera Report 2015. Pretoria, South Africa.
Adams, R. A., Snode, E. R. 2013. Unique insights into dispersion distances among calling males of Wahlberg’s Epauletted fruit bat in Kruger National Park, South Africa. The Open Ecology Journal 6: 54–60.
Adams RA, Snode ER. 2015. Differences in the male mating calls of co-occurring epauletted fruit bat species (Chiroptera, Pteropodidae, Epomophorus wahlbergi and Epomophorus crypturus) in Kruger National Park, South Africa. Zoological Studies 54: 15.
Arnett EB, Huso MM, Schirmacher MR, Hayes JP. 2011. Altering turbine speed reduces bat mortality at wind-energy facilities. Frontiers in Ecology and the Environment 9: 209–214.
Aronson, J. 2022. Current state of knowledge of wind energy impacts on bats in South Africa. Acta Chiropterologica 24: 221-238.
Arumoogum, N., Schoeman, M.C., Ramdhani, S. 2019. The relative influence of abiotic and biotic factors on suitable habitat of Old World fruit bats under current and future climate scenarios. Mammalian Biology 98: 188-200.
Baerwald EF, D’Amours GH, Klug BJ, Barclay RM. 2008. Barotrauma is a significant cause of bat fatalities at wind turbines. Current Biology 18: 695–696.
Baerwald EF, Edworthy J, Holder M, Barclay RM. 2009. A large-scale mitigation experiment to reduce bat fatalities at wind energy facilities. Journal of Wildlife Management 73: 1077–1081.
Berthinussen, A., Richardson, O.C. and Altringham, J.D. 2010. Bat Conservation: Global Evidence for the Effects of Interventions. Synopses of Conservation Evidence. Pelagic Publishing, Exeter.
Bonaccorso, F.J., Winkelmann, J.R., Todd, C.M. and Miles, A.C. 2014. Foraging Movements of Epauletted Fruit Bats (Pteropodidae) in Relation to the Distribution of Sycamore Figs (Moraceae) in Kruger National Park, South Africa. Acta Chiropterologica 16(1): 41-52.
Cryan PM, Barclay RM. 2009. Causes of bat fatalities at wind turbines: hypotheses and predictions. Journal of Mammalogy 90: 1330–1340.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Fenton, M. B., Brigham, R. M., Mills, A. M. and Rautenbach, I. L. 1985. The roosting and foraging areas of Epomophorus wahlbergi (Pteropodidae) and Scotophilus viridis (Vespertilionidae) in Kruger National Park, South Africa. J. Mammal 66: 461–468.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
MacEwan KL. 2016. Fruit bats and wind turbine fatalities in South Africa. African Bat Conservation News 42: 3–5.
MacEwan, K., Aronson, J., Richardson, K., Taylor, P., Coverdale, B., Jacobs, D., Leeuwner, L., Marais, W. and Richards, L. 2018. South African Bat Fatality Threshold Guidelines. Edition 2. South African Bat Assessment Association, South Africa.
MacEwan, K., Sowler, S., Aronson, J. and Lötter, C. 2020. South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities. Edition 5. South African Bat Assessment Association, South Africa.
Mo, M., Roache, M., Davies, J., Hopper, J., Pitty, H., Foster, N., Guy, S., Parry-Jones, K., Francis, G., Koosmen, A. and Colefax, L. 2021. Estimating flying-fox mortality associated with abandonments of pups and extreme heat events during the austral summer of 2019–20. Pacific Conservation Biology, 28(2), pp.124-139.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.
Mphethe, V., Weier, S., Westphal, C., Linden, B., Swanepoel, L., Parker, D. and Taylor, P. 2023. Epauletted fruit bats prefer native plants and contribute to seed dispersal in a South African agricultural landscape. African Journal of Ecology 61: 399-410.
Munyati, C. and Kabanda, T.A. 2009. Using multitemporal Landsat TM imagery to establish land use pressure induced trends in forest and woodland cover in sections of the Soutpansberg Mountains of Venda region, Limpopo Province, South Africa. Regional Environmental Change 9(1): 41–56.
Rollinson, D., Coleman, J.C., and Downs, C.T. 2013. Seasonal differences in foraging dynamics, habitat use and home range size of Wahlberg’s epauletted fruit bat in an urban environment. African Zoology 48: 340-350.
Rydell J, Bach L, Dubourg-Savage M-J, Green M, Rodrigues L, Hedenström A. 2010. Bat mortality at wind turbines in northwestern Europe. Acta Chiropterologica 12: 261–274.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor, P.J. and Monadjem, A. 2008. Maxillary shape as a diagnostic tool for identifying fruit bats, Epomophorus crypturus and E. wahlbergi (Mammalia: Chiroptera) from museum specimens and in the field. South African Journal of Wildlife Research 38(1): 22-27.
