Vlei Rat (Grassland type)
Otomys auratus

2025 Red list status
Near threatened
Regional Population
Trend
Declining
Change compared
to 2016
No Change
Overview
Otomys auratus – Wroughton, 1906
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Otomys – auratus
Common Names: Vlei Rat, Southern African Vlei Rat (English), Leboli (Sotho, Southern), Vleirot (Afrikaans)
Synonyms: No Synonyms
Otomys irroratus has been split into two species (Otomys irroratus and Otomys auratus) based on genetic evidence and niche modelling by Engelbrecht et al. 2011.
Taxonomic Note: South Africa
This species was until recently included in Otomys irroratus and the two cannot be distinguished on morphological grounds; however, O. auratus is closely associated with the Grassland Biome in South Africa and the Eastern Highlands of Zimbabwe, whilst O. irroratus is closely associated with the Fynbos and Thicket biomes of the Western Cape and Eastern Cape provinces of South Africa (Monadjem et al. 2015). Otomys auratus was shown to significantly differ from O. irroratus on molecular, chromosomal and ecological grounds (Taylor et al. 2009; Engelbrecht et al. 2011).
Red List Status: NT – Near Threatened
Assessment Information
Assessors: Taylor, P.1 & da Silva, J.M.2
Reviewer: Oosthuizen, M.3
Institutions:1University of the Free State,2South African National Biodiversity Institute,3University of Pretoria
Previous Assessors and Reviewers: Taylor, P., Baxter, R. & Child, M.F.
Previous Contributors: Roxburgh, L., Avenant, N., Avery, M., MacFadyen, D., Monadjem, A., Palmer, G. & Wilson, B.
Assessment Rationale
This near-endemic grassland species is becoming increasingly threatened by grassland contraction and wetland loss, with niche modelling showing that it will undergo a 47–61% reduction in suitable habitat between 1975 and 2050 from climate change (6–8% per decade). Already one major subpopulation, located in Soutpansberg Mountains, Limpopo Province, has been completely replaced at a key contact zone by the savannah species O. angoniensis over the past 90 years due to thicket habitats replacing grasslands. Ongoing grassland and wetland loss, primarily from agricultural and settlement expansion, and grassland degradation due to over-grazing, may synergise with climate change to accelerate area of occupancy (AOO) reduction. Since this species is localised to montane or plateau grassland patches, it is more sensitive to habitat loss as it probably cannot utilise degraded habitats. Nationally, there was a 32.8% decline in natural wetlands from 1990–2013/14, which is a combination of both genuine wetland loss through anthropogenic activities and the generally drier conditions currently that in 1990. Specifically, most distribution records (36%) are located in the Mesic Highveld Grassland Bioregion where there has been a 19% loss of moist grasslands since 1990 (Skowno et al. 2021). In KwaZulu-Natal specifically, there was a loss of 7.6% of the natural habitat between 2005 and 2011 (1.3% per annum), which, when extrapolated, equates to a 13% loss of habitat over a ten year time period. Thus, multiple lines of evidence corroborate past, continuing and future habitat loss. Under a precautionary purview, we continue to list this species as Near Threatened A4c as a 20–30% decline in area of occupancy (AOO)—with a similar decline in population size—over ten years is possible due to the synergising effects of climate change and land use change (Taylor et al. 2016). We encourage more long-term surveys, such as that in the Soutpansberg, to identify other subpopulations that have been lost to landscape and climate changes.
Regional population effects: Rescue effect is not possible due to the disjunct distribution of populations within the assessment region and Zimbabwe.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Taylor P & da Silva JM. 2025. A conservation assessment of Otomys auratus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
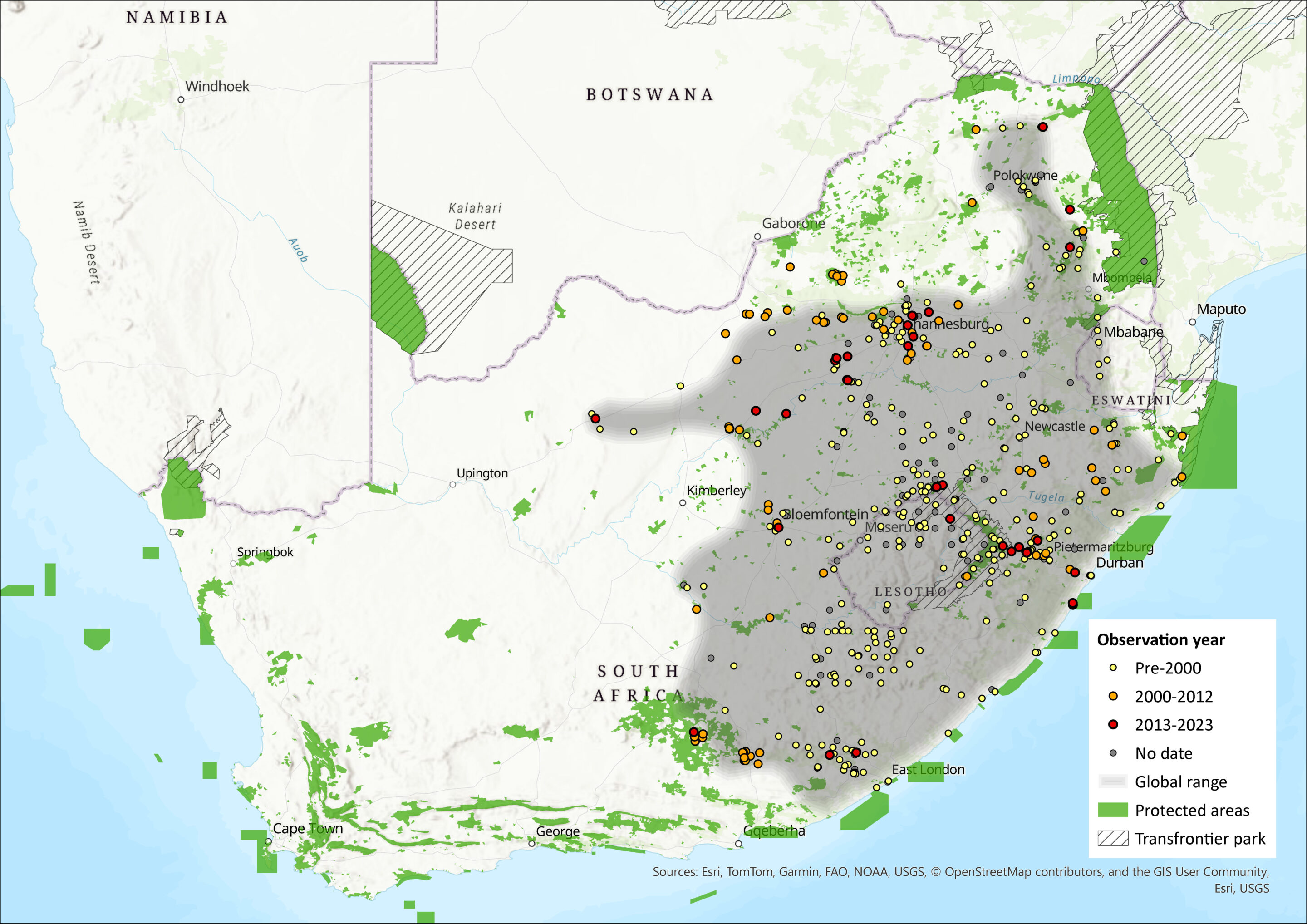
The species is widely distributed throughout the Highveld grasslands and Drakensberg Escarpment of South Africa, Lesotho and Eswatini, with isolated populations in the Soutpansberg Mountains of northern Limpopo and the Eastern Highlands of Zimbabwe (Monadjem et al. 2015). While indiscernible from O. irroratus, Engelbrecht et al. (2011) used mitochondrial data to delineate between the two cryptic species with the only known contact zone between the two species being Alice in the Eastern Cape. As this species has been split from O. irroratus, museum records will need vetting through molecular research to delimit distribution more accurately.
Worryingly, there has been a documented range contraction in the Soutpansberg Mountains of Limpopo Province due to habitat shifts from grassland to thicket. Comparison of occurrence data at a zone of sympatry between 1923 and 2013 revealed that while O. auratus was previously dominant, only O. angoniensis is currently found at the site (Taylor et al. 2016). This could indicate a savannah species displacing a grassland species as grasslands contract and are replaced by thicket woodlands due to climate change. Data from the wider Soutpansberg area support this trend: recent surveys in suitable grasslands habitats at > 1,300 m asl (typically O. auratus habitats) have revealed only O. angoniensis while O. auratus was only sampled at one locality in the eastern Soutpansberg (Taylor et al. 2016).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Vlei Rat (Otomys auratus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Eswatini | Extant | Native | – | Resident |
| Lesotho | Extant | Native | – | Resident |
| Mozambique | Extant | Native | – | Resident |
| South Africa | Extant | Native | – | Resident |
| Zimbabwe | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Climate change models predict a loss of suitable habitat for Otomys auratus, with estimates ranging between 7 and 15% (Cowan 2025). This decline is primarily expected on the northern and western fringes of its distribution range, where environmental shifts are rapidly altering landscapes. Given their strong dependence on wetland ecosystems, Otomys auratus is particularly vulnerable to habitat degradation and loss. Wetlands are increasingly threatened by climate change-driven factors such as altered precipitation patterns, rising temperatures, and human encroachment. As these ecosystems shrink or deteriorate, populations of Otomys auratus may face reduced access to resources, heightened competition, and increased risk of decline. Increases in extreme weather events can also negatively impact on animals, in all climates.
Population information
Although presently it is a common species in some areas, MaxEnt modelling based on continued grassland contraction from land-use and climate change is projected to lead to a 47–61% population reduction by 2050 (Taylor et al. 2016). This species is K-selected and thus has a relatively long generation length, estimated to be 1.8 years (Pacifici et al. 2013). There has been repeat observed population declines of this species in Rietvlei Dam, Albert Falls, Chelmsford and Vryheid Hill.
Population Information
Current population trend: Declining
Continuing decline in mature individuals: No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: Yes. Dispersal is dependent on continuous areas of riparian grassy vegetation around wetlands, dams and riparian corridors. Such patches may become too isolated for effective dispersal (Taylor et al. 2016).
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
While molecular evidence supported Otomys auratus as being differentiated from O. irroratus despite indistinguishable morphologies (Taylor et al. 2009; Engelbrecht et al. 2011), no population genetic studies have been conducted on this species. However distinct molecular subclades have been described occurring west and east of the Drakensberg (Engelbrecht et al. 2011).
Habitats and ecology
This species is associated with mesic grasslands and wetlands within alpine, montane and sub-montane regions (Monadjem et al. 2015), typically occurring in dense vegetation in close proximity to water (for example, Wandrag et al. 2002, Watson 2006). In the Drakensberg Range, O. angoniensis occurs on the lower slopes in savanna habitats, O. auratus and O. laminatus occur at mid and higher elevations in grasslands and O. sloggetti at the highest elevations in alpine heath habitats (Monadjem et al. 2015). Where O. auratus and O. angoniensis co-occur at the same site, the former is associated with sedges and grasses adapted to densely vegetated wetlands with wet soils, while the latter is associated with plant species that typically grow in the drier margins of wetlands (Davis 1973). Vlei rats are exclusively herbivorous, with a diet mainly comprised of grasses. Cranial size of both O. auratus and O. angoniensis decreased significantly over the past 100 years possibly in response to climate change (Nengovhela et al. 2016).
Ecosystem and cultural services: Vlei rats are important food for a number of mammalian predators, as well as raptors such as marsh owls and barn owls (Skinner and Chimimba 2005, Monadjem et al. 2015). For example, Vlei rats are favoured food by the Serval (Leptailurus serval) (Bowland 1990), so their range expansion could be interrelated (Power 2014).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 4.7. Grassland -> Grassland – Subtropical/Tropical High Altitude | – | Suitable | – |
| 5.4. Wetlands (inland) -> Wetlands (inland) – Bogs, Marshes, Swamps, Fens, Peatlands | – | Suitable | – |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Suitable | – |
Life History
| Generation Length | Justification | Data Quality |
| 1-2 | see Pacifici et al. 2013 | medium |
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: At least 4 years in captivity
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not traded or utilised in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are three main threats that may cause significant population decline in the near future:
- Grassland and wetland habitat loss from agricultural expansion, human settlement sprawl and mining. In Mpumalanga, for example, only 51% of the grasslands are still natural and not previously ploughed and 40% of the grassland vegetation types are listed as threatened (Lötter 2015). Water abstraction or filling in of wetlands from human settlement and industrial expansion also leads to habitat loss. Similarly, suppression of natural ecosystem processes, such as fire, can also lead to habitat degradation through bush encroachment or loss of plant diversity through alien invasive species, and is suspected to be increasing with human settlement expansion. Overall, 45% of our remaining wetland area exists in a heavily modified condition, due primarily to onsite modification from crop cultivation, coal mining, urban development, dam construction, and overgrazing (and thus erosion) and off-site modifications from disruptions to flow regime and deterioration of water quality (Driver et al. 2012).
- Overgrazing the vegetation around wetlands reduces ground cover and thus leads to decreased small mammal diversity and abundance (Bowland and Perrin 1989, 1993). The expansion of wildlife ranching will have to be monitored in this regard, as game overstocking may also affect wetland condition.
- Climate change is projected to reduce area of occupancy significantly by reducing temperate grasslands and wetland habitats. Climate modelling shows that a 47–61% loss in AOO is expected between 1975 and 2050 depending on the dispersal ability of the species (Taylor et al. 2016). While the model predicts a range shift along the Indian Ocean Coastal Belt, where it occurs narrowly at present, this area is highly developed (Driver et al. 2012) and thus may not provide suitable alternative habitat. Coastal habitat transformation in KwaZulu-Natal is due primarily to sugarcane monocultures, forestry plantations and residential developments. Thus, the future distribution of the species may be severely fragmented, resulting in local extinctions (Taylor et al. 2016). More recently, climate change models predict a loss of suitable habitat for Otomys auratus, with estimates ranging between 7 and 15% (Cowan 2025).
Land-use change and climate change may synergise to cause non-linear and accelerating population decline. For example, trapping data suggest that O. angoniensis has already replaced O. auratus throughout most of the Soutpansberg Mountain range except in the Entabeni area (Taylor et al. 2016). More research is needed to validate these trends in other regions.
Habitat trend: Declining. Wetlands are the most threatened ecosystem in South Africa (Driver et al. 2012). The South African National Land-Cover change report found a 32.8% decline in natural wetlands nationally from 1990 – 2013/14, which is a combination of both genuine wetland loss through anthropogenic activities and the generally drier conditions currently that in 1990 (GeoTerraImage 2015). Habitat loss due to land transformation in the surrounding matrix further isolates wetlands from one another and exacerbates the degradation of individual wetlands. For example, sugarcane and forestry plantations are often planted right up to wetlands edges, not respecting the appropriate buffer. In KwaZulu-Natal alone, there an average loss of natural habitat of 1.2% per annum between in 1994 and 2011 from agriculture, plantations, built environments and settlements, mines and dams (Jewitt et al. 2015). Although no specific rates of habitat loss are available, 61% of Mpumalanga’s land surface between 2000 and 2014 have come under pressure from prospecting applications (Lötter 2015). Compounding habitat loss from land-use change is climate change. Projected range shifts under the A2 emission scenario of the Intergovernmental Panel on Climate Change showed increases (O. angoniensis) and decreases (O. auratus) that closely mirrored those expected for the savanna and grassland biomes respectively (Taylor et al. 2016).
Conservation
They occur in many protected areas, such as Kruger National Park in Limpopo/Mpumlanga, Pilansberg National Park in the North West (Power 2014), Rietvlei Nature Reserve in Gauteng (Taylor et al. 2016), Tussen-die-Riviere and Seekoeivlei Nature Reserves in the Free State (Ferreira and Avenant 2003), and Asante Sana Nature Reserve and Mountain Zebra National Park in the Eastern Cape (Kok et al. 2012). Mitigating habitat loss outside of protected areas is urgently. The following interventions should be implemented:
- Using previously cultivated areas for development instead of remaining natural areas: In Mpumalanga, for example, old lands or previously ploughed areas now left fallow make up 8.9% of the grassland biome (Lötter 2015), and these areas should be prioritised for further development. Similarly, in KwaZulu-Natal, abandoned agricultural fields on marginal lands offer an opportunity for further development instead of transforming virgin land and at least 4% of the landscape is available for this (Jewitt et al. 2015).
- Wetland conservation and restoration: land managers should maintain a vegetation buffer to reduce impacts of land-use practices (Driver et al. 2012). For example, the frequency of O. auratus and O. angoniensis has not changed between 1972 and 2013 as the wetland vlei areas have not changed significantly in structure (Taylor et al. 2016). Restoration will also create corridors between suitable habitat patches to allow for dispersal in responses to climate change.
- Holistic management of ranchlands: including de-stocking, rotational grazing and buffering wetland vegetation, are encouraged.
Recommendations for land managers and practitioners:
- Land managers should decrease stocking rates to conserve vegetation around wetlands.
- Long-term, systematic monitoring is needed to establish subpopulation trends and threat levels.
- Prioritise old fields for development in systematic conservation planning.
Research priorities:
- Long-term, systematic monitoring is needed to establish subpopulation trends and threat levels.
- Fine-scale studies on habitat loss and inferred impact on the species across the range of the species.
- Effects of overgrazing on the density and viability of this species.
- Effects of habitat connectivity on dispersal rates.
- Further vetting of museum records to delimit distribution more accurately.
Encouraged citizen actions:
- Report Vlei Rat sightings on MammalMAP: The feeding signs of this species are easy to detect by short, chopped lengths of grass and green moist faecal pellets (Skinner and Chimimba 2005).
Bibliography
Bowland, A.E. and Perrin, M.R. 1989. The effect of overgrazing on the small mammals in Umfolozi Game Reserve. Zeitschrift für Säugetierkunde 54: 251–260.
Bowland, J.M. 1990. Diet, home range and movement patterns of serval on farmland in Natal. Department of Zoology and Entomology, University of Natal.
Bowland, J.M. and Perrin, M.E. 1993. Wetlands as reservoirs of small-mammal populations in the Natal Drakensberg. South African Journal of Wildlife Research 23: 39–43.
Cowan, OS. 2025. Modelling the change in potential species distribution of Vlei Rat (Otomys auratus) under predicted future climate change scenarios. Technical Report, Endangered Wildlife Trust.
Davis, R.M. 1973. The ecology and life history of the vlei rat, Otomys irroratus (Brants, 1827), on the van Riebeeck Nature Reserve, Pretoria. Ph.D Thesis. University of Pretoria.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Engelbrecht, A., Taylor, P.J., Daniels, S.R. and Rambau, R.V. 2011. Cryptic speciation in the southern African vlei rat Otomys irroratus complex: evidence derived from mitochondrial cyt b and niche modelling. Biological Journal of the Linnean Society 104: 192–206.
Ferreira, S.M. and Avenant, N.L. 2003. Modelling the effects of trap-spacing on small mammal community descriptors in grasslands at Tussen-die-Riviere Nature Reserve, Free State Province, South Africa. Navorsinge van die Nasionale Museum, Bloemfontein 19: 21-30.
GeoTerraImage. 2015. 1990-2013/14 South African National Land-Cover Change. DEA/CARDNO SCPF002: Implementation of Land-Use Maps for South Africa. Project Specific Data Report.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Kok, A.D., Parker, D.M. and Barker, N.P. 2012. Life on high: the diversity of small mammals at high altitude in South Africa. Biodiversity and Conservation 21: 2823–2843.
Lötter, M.C. 2015. Technical Report for the Mpumalanga Biodiversity Sector Plan – MBSP. Mpumalanga Tourism & Parks Agency, Mbombela.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa – a biogeographic and taxonomic synthesis. De Gruyter, Berlin/Munich/Boston.
Nengovhela, A., Baxter, R.M. and Taylor, P.J. 2016. Temporal changes in cranial size in South African vlei rats (Otomys): evidence for the “third universal response to warming.”. Zoology 50: 233-239.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Pence, G.Q.K. 2012. Contribution of C.A.P.E. Business and Biodiversity Initiatives to conservation of critical biodiversity, landscape connectivity and ecological support areas: Post-baseline assessment (2010). A Green Choice Alliance project report. Conservation South Africa, Kirstenbosch, South Africa.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Skowno AL, Jewitt D, Slingsby JA. 2021. Rates and patterns of habitat loss across South Africa’s vegetation biomes. S Afr J Sci. 2021;117(1/2), Art. #8182. https://doi.org/10.17159/sajs.2021/8182
Taylor, P.J., Maree, S., van Sandwyk, J., Baxter, R. and Rambau, R.V. 2009. When is a species not a species? Uncoupled phenotypic, karyotypic and genotypic divergence in two species of South African laminate-toothed rats (Murinae: Otomyini). Journal of Zoology 277: 317–332.
Taylor, P.J., Nengovhela, A., Linden, J. and Baxter, R.M. 2016. Past, present, and future distribution of Afromontane rodents (Muridae: Otomys) reflect climate-change predicted biome changes. Mammalia 80: 359–375.
Wandrag, G.F., Watson, J.P. and Collins, N.B. 2002. Rodent and insectivore species diversity of Seekoeivlei Provincial Nature Reserve, Free State province, South Africa. South African Journal of Wildlife Research 32: 137–143.
Watson, J.P. 2006. Check list of the mammals of Tussen-die-Riviere Provincial Nature Reserve, Free State Province, South Africa. Koedoe 49: 111–117.
Taylor, P. J., Kearney, K., Dalton, D., Mataruse, G., Kelly, C. M. R. and Barker, N. P. 2020. Biomes, geology and past climate drive speciation of laminate-toothed rats on South African mountains (Murinae: Otomys). Zoological Journal of the Linnean Society 189: 1046–1066, https://doi.org/10.1093/zoolinnean/zlz134.