Verreaux’s Mouse
Myomyscus verreauxii

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Myomyscus verreauxii – (Smith, 1834)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Myomyscus – verreauxii
Common Names: Verreaux’s Mouse (English), Verreaux se muis (Afrikaans), Verreaux’s White-footed Rat (English)
Synonyms: Myomys verreauxii (Smith, 1834)
Taxonomic Note: The genus Myomyscus is currently represented by three African species, which can be distinguished from the similar Mastomys species based on their longer tail relative to head and body length, whiter ventral colour, and number of nipples (Monadjem et al. 2015), as well as molecular data (Lecompte et al. 2005). Musser and Carleton (2003) suggest that the Myomyscus should be restricted to the type species (M. verreauxii), and a new genus should be described for the remaining taxa (Monadjem et al. 2015). However, further analyses are required before this hypothesis can be confirmed. Verreaux’s Mouse can be easily distinguished from the other Myomyscus species based on its distribution. No subspecies have been described.
Red List Status: LC – Least Concern
Assessment Information
Assessors: Smith, C.1 & da Silva, J.M.2
Reviewer: Taylor, P.3
Institutions: 1Endangered Wildlife Trust,2South African National Biodiversity Institute,3University of the Free State
Previous Assessors & Reviewers: Relton, C., Avery, M. & Palmer, G.
Previous Contributors: Child, M.F., Avenant, N., Baxter, R., MacFadyen, D., Monadjem, A., Taylor, P. & Wilson, B.
Assessment Rationale
This endemic species remains listed as Least Concern in view of its wide (albeit fragmented) distribution within the Western and Eastern Cape, and because its habitat is largely inaccessible and unlikely to be extensively transformed. The estimated extent of occurrence is 150,917 km2. There are no major threats expected to cause range-wide population decline. However, although around 76% of the Western Cape is still considered natural or near-natural, information from the Western Cape Nature Conservation Board indicates that there is a continuing decline in natural habitat from agricultural expansion, especially on lower- to mid-slope areas (below 1,000 m) from planted pastures and rooibos, wine and fruit cultivation, which may impact the species in the future as climate change makes higher elevation habitats more suitable for agriculture. Additionally, there may be localised losses of habitat quality due to the spread of invasive alien species, inappropriate fire regimes, and edge effects associated with agricultural and residential land-uses (for example, use of pesticides and predation from domestic pets). Although the species remains widespread and regularly encountered, proactive mitigation measures, including protected area expansion and habitat restoration, should be continued to counteract habitat loss. Protected area expansion to connect fragmented subpopulations, especially in the low-lying fynbos areas, would especially benefit this endemic species.
Red List Index
Red List Index: No change
Recommended Citation: Smith C & da Silva JM. 2025. A conservation assessment of Myomyscus verreauxii. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
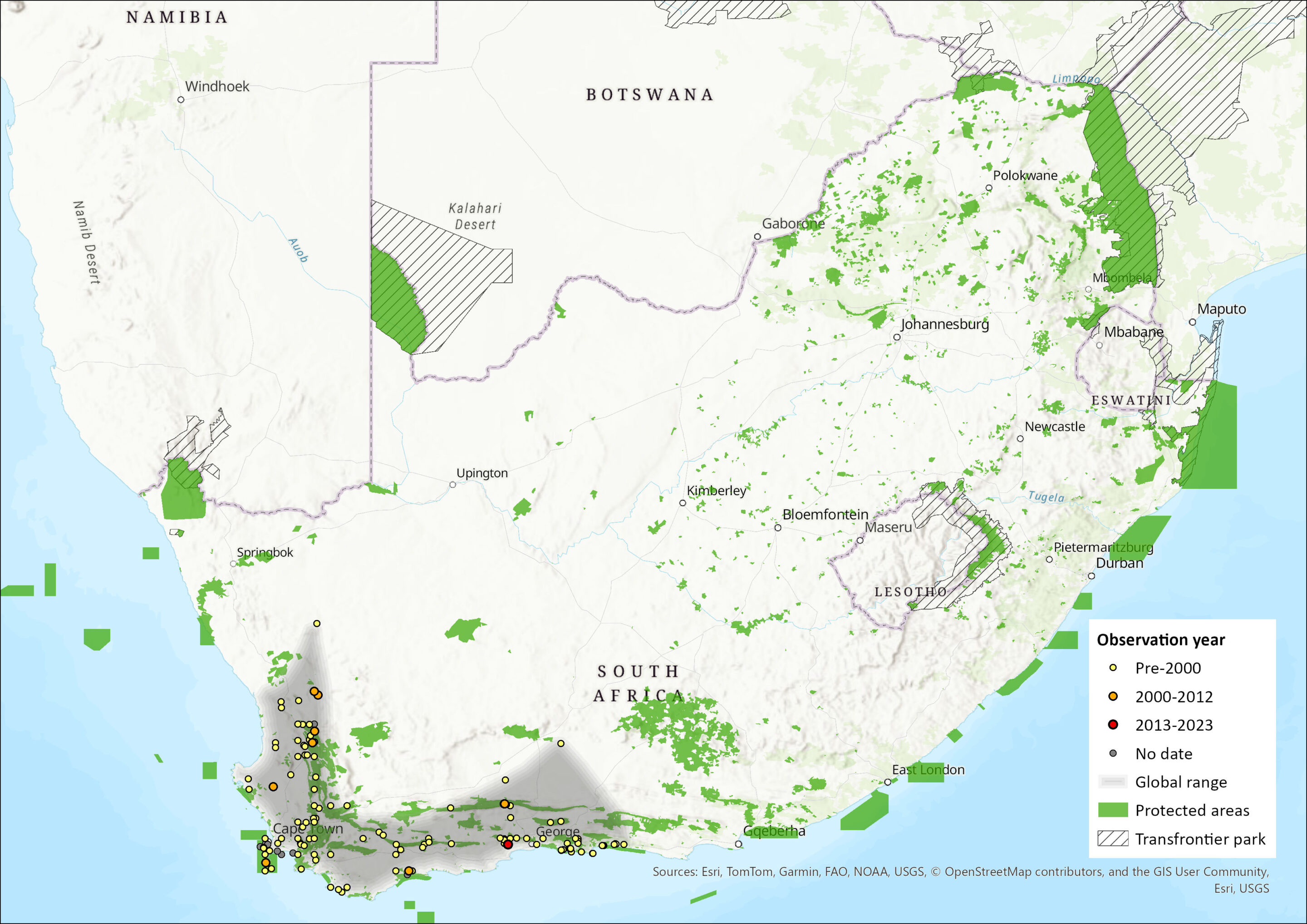
This species is endemic to southwestern South Africa, largely restricted to the Fynbos Biome in the Western Cape Province (Mugo et al. 1995), and partially extending into the Northern and Eastern Cape provinces (Avery and Avery 2011). Their range may extend from the Olifants River in the west to the Knysna district and Plettenberg Bay in the east. They occur in a variety of habitats, including grasslands, fynbos and forests (Monadjem et al. 2010). The estimated extent of occurrence using all records is 150,917 km². Further field surveys are necessary to confirm current occupancy within its range.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Verreaux’s Mouse (Myomyscus verreauxii) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
To quantify the potential threat of climate change on the species habitat, species distribution modelling under future climate projections were performed. From a conservation perspective, the models show a consistent reduction in habitat for Myomyscus verreauxii ranging between 12%-22% by 2040 (Cowan 2025).
Population
Although this species has a naturally fragmented distribution and is uncommon, it is regularly recorded and can be locally abundant (Happold 2013). For example, it comprised 50% of small mammals trapped in the forested valleys of the Cederberg, Western Cape (Rautenbach and Nel 1980). Low trapping efficiency for the Nama-Karoo. One individual recorded on the Grassland (Nadine 2020). No population estimates are currently available.
Population Information
Current population trend: Stable
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: Yes, naturally fragmented by mountain ranges.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While a molecular assessment of the family Murinae has been conducted, there have been no population genetic studies undertaken to date. And it is considered widespread, because the species has a naturally fragmented distribution it might exist as of multiple genetically isolated subpopulations. It is not anticipated that any extinctions of potential subpopulations have taken place. Consequently, the Convention on Biological Diversity’s Global Biodiversity Framework’s complementary genetic indicator – proportion of populations maintained (PM) – is estimated at 1.0 (all populations remaining).
Given the lack of information on the species’ population structure and size (or density), it is not possible to estimate effective population sizes (Ne) for the possible subpopulations.
It is highly recommended that a population genetic study be undertaken to better understand the population genetic structure and health of the species and to quantify the estimated indicator values.
Habitats and ecology
This nocturnal species is located within both lowland and montane fynbos vegetation. They have been recorded as fairly abundant in riverine forests, living in scrub on grassy slopes and the edges of forests (Rautenbach and Nel 1980). They are also found on rocky slopes, particularly cliffs (Frazer 2021). For example, in the Cederberg region, they are found in forested valleys. In the Knysna area, this species occurs in damp grasslands and vleis, seeking shelter under fallen trees (De Graaff 1981). Happold (2013) describes them as inhabiting meadow banks near the coast, and near fallen trees or in grassy vleis in forests. It is unknown whether disturbed or modified regions form viable habitats for this species, but it has not been recorded from pine plantations (Armstrong et al. 1996). It breeds in October-February (Frazer 2021).
David (1978) and Breytenbach (1982) found that Verreaux’s Mouse depends largely on Proteaceae seeds, for example Leucodendron album. Additionally, Rautenbach and Nel (1980) reported that insects may also form a substantial part of their diet.
This species has been recognised as a valuable, and often essential, pollinator for ground Protea species (David 1978).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
Life History
| Generation Length | Justification | Data Quality |
| 1-2 | Inferred (see Pacifici et al. 2013) | good |
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
The lower-lying areas of this species’ range are threatened by habitat loss and fragmentation as a result of urban and agricultural expansion (Pence 2014a), while the inaccessible montane portions of its range are unlikely to be extensively transformed. However, climate change may make higher elevations more suitable for agricultural expansion (see below) and thus represents an emerging threat.
The effects of global climate change on the endemic Fynbos Biome of South Africa, and its interaction with land transformation has been assessed by Midgely et al. (2003), who predict the potential range contraction and elimination of certain Proteaceae species. Climate change was predicted to have more of an impact on Proteaceae species than land transformation; as land transformation was considerably less prevalent in regions of higher altitude (Midgley et al. 2003). Additionally, altered fire regimes due to climate change and the expansion of invasive alien plant species may lead to habitat degradation and fragmentation, thus affecting the availability of shelter and forage resources for this species. The fynbos habitat is impacted in parts by invasive alien plants which, despite control efforts, are expanding (van Wilgen et al. 2012). Replacement of natural vegetation by non-indigenous species may reduce food resources and lead to altered fire regimes, which may become a threat if the fire return interval becomes so short that seeds preferred by the Verreaux’s Mouse cannot be produced. Ground proteas are dependent on this species for pollination (Nel 2018).
Habitat is currently declining, in the lowland fynbos habitats. However, as 76% of the province remains natural or near-natural and the protected area network continues to expand (Pence 2014b), we suspect that habitat loss from agricultural expansion will only impact the species if higher-elevation areas are increasingly converted to crops, which may become likely as climate change makes such areas suitable. For example, climate change is projected to increase the suitability of upslope habitats for viticulture, increasing the footprint of winelands by 14% by 2050 (Hannah et al. 2013). Such trends in transformation of mid- and upper-slopes should be monitored.
Current habitat trend: Declining, in the lowland fynbos habitats. However, as 76% of the province remains natural or near-natural and the protected area network continues to expand (Pence 2014b), we suspect that habitat loss from agricultural expansion will only impact the species if higher-elevation areas are increasingly converted to crops, which may become likely as climate change makes such areas suitable. For example, climate change is projected to increase the suitability of upslope habitats for viticulture, increasing the footprint of winelands by 14% by 2050 (Hannah et al. 2013). Such trends in transformation of mid- and upper-slopes should be monitored.
Conservation
This species is present within a number of protected areas of the assessment region, such as the West Coast National Park, De Hoop Nature Reserve and Vrolijkheid Nature Reserve. Although no specific conservation interventions are necessary, this species would benefit from protected area expansion, thus connecting lowland fynbos areas to patches of inaccessible montane habitat. Progress is being made in protected area expansion in the Western Cape, especially in Critical Biodiversity Areas (CBAs) (Pence 2014b). Stewardship on private lands may be particularly promising. For example, the Biodiversity Stewardship Programme has added over 490 km2 to the Western Cape’s formal protected area network since its launch in 2003, by entering into biodiversity agreements with private landowners (Maree et al. 2015). Such agreements should be enhanced through best practice management techniques for both viticulture and biodiversity, a new field dubbed vinecology, which is actively implemented in South Africa (reviewed in Viers et al. 2013).
Recommendations for land managers and practitioners:
- It is critical that the fight to eradicate invasive alien plant species from our protected areas continues and that every effort be made to maintain the “natural” fire regime within the Fynbos Biome. Landowners should be incentivised to employ the Working for Water Programme (Department of Environmental Affairs) to restore habitats.
Research priorities:
- Rate of future habitat loss in the Western Cape, especially in higher altitude areas due to climate change and viticultural expansion.
- Estimating population size through density estimates and total natural habitat available. This would enable a threshold of habitat loss to be calculated below which the population is expected to be fewer than 10,000 mature individuals.
- Effectiveness of invasive alien plant removal in increasing small mammal occurrence and abundance.
- Effectiveness of implementing vinecology management on small mammals should be monitored and evaluated.
- Research into how this species responds to fire frequency and the extent of dependence on nut-fruited plants and mammal-pollinated plants.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- The consistent clearance of invasive alien plant species across areas of the Cape Floristic Region reduces habitat degradation and water loss for native biodiversity.
- Plant indigenous vegetation in gardens and ensure corridors of natural vegetation remain to allow local movements.
Bibliography
Armstrong AJ, van Hensbergen HJ, Scott DF, Milton SJ. 1996. 1996. Are Pine Plantations “Inhospitable Seas” around Remnant Native Habitat within South-western Cape Forestry Areas? South African Forestry Journal 176: 1-9.
Avery, D.M. and Avery, G. 2011. Micromammals in the Northern Cape Province of South Africa, past and present. African Natural History 7: 9-39.
Breytenbach, G.J. 1982. Small mammal responses to environmental gradients in the Groot Swartberg of the Southern Cape. University of Pretoria.
Cowan, O.S. 2025. Modelling the change in potential species distribution of Verreaux’s Mouse (Myomyscus verreauxii) under predicted future climate change scenarios. Technical Report, Endangered Wildlife Trust.
David, J.H. 1978. A survey of vertebrate fauna on the Rooiberg, Ladysmith, Cape. Unpublished Report. Zoology Department, University of Cape Town.
Fraser, M., 2022. Mammals of the Cape of Good Hope Nature Reserve, Western Cape, South Africa. Biodiversity Observations, 12, pp.15-46.
Hannah, L., Roehrdanz, P.R., Ikegami, M., Shepard, A.V., Shaw, M.R., Tabor, G., Lu, Z., Marquet, P.A. and Hijmans, R.J. 2013. Climate change, wine, and conservation. Proceedings of the National Academy of Sciences of the United States of America 110: 6907-6912.
Happold, D.C.D. 2013. Myomyscus verreauxii Verreaux’s Meadow Mouse (White-footed Mouse). In: Kingdon. J., Happold. D., Butynski. T., Hoffmann. M., Happold. M. and Kalina. J. (eds), Mammals of Africa, pp. 505. Bloomsbury Publishing, London, UK.
IUCN. 2017. The IUCN Red List of Threatened Species. Version 2017-2. Available at: www.iucnredlist.org. (Accessed: 14 September 2017).
Lecompte, E., Denys, C. and Granjon, L. 2005. Confrontation of morphological and molecular data: the Praomys group (Rodentia, Murinae) as a case of adaptive convergences and morphological stasis. Molecular Phylogenetics and Evolution 37: 899–919.
Maree, K.S., Pence, G.Q.K. and Purnell, K. 2015. Western Cape Protected Area Expansion Strategy: 2015–2020. Produced by CapeNature, Cape Town, South Africa, Unpublished report.
Midgley, G.F., Hannah. L., Millar. D,, Thuiller. W. and Booth, A. 2003. Developing regional and species-level assessments of climate change impacts on biodiversity in the Cape Floristic Region. Biological Conservation 112: 87–97.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa – a biogeographic and taxonomic synthesis. De Gruyter, Berlin/Munich/Boston.
Mugo, D.N., Lombard, A.T., Bronner, G.N. and Gelderblom, C.M. 1995. Distribution and protection of endemic or threatened rodents, lagomorphs and macrosceledids in South Africa. Journal of Zoology 30: 115-126.
Musser, G.G. and Carleton, M.D. 1993. Family Muridae. In: D.E. Wilson and D.A. Reeder (eds), Mammal species of the world: A taxonomic and geographic reference, pp. 501-736. Smithsonian Institution Press, Washington, DC, USA.
Musser, G.G. and Carleton, M.D. 2005. Superfamily Muroidea. In: D.E. Wilson and D.A. Reeder (eds), Mammal Species of the World: a geographic and taxonomic reference, pp. 894-1531. The John Hopkins University Press, Baltimore, USA.
Nadine, A.H., 2020. Spatial variation in small mammal communities across the Karoo Shale Gas Development Area of South Africa.
Nel, T.H., Wurz, S. and Henshilwood, C.S., 2018. Small mammals from marine isotope stage 5 at Klasies River, South Africa–reconstructing the local palaeoenvironment. Quaternary International, 471, pp.6-20.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Pence, G.Q.K. 2014. Western Cape Biodiversity Framework 2014 status update: Critical Biodiversity Areas of the Western Cape. CapeNature, Cape Town.
Rautenbach, I.L. and Nel, J.A.J. 1980. Mammal diversity and ecology in the Cedarberg wilderness area, Cape Province. Annals of the Transvaal Museum 32: 101-124.
Richardson, D.M. and van Wilgen, B.W. 2004. Invasive alien plants in South Africa: how well do we understand the ecological impacts? South African Journal of Science 100: 45-52.
Viers, J.H., Williams, J.N., Nicholas, K.A., Barbosa, O., Kotzé, I., Spence, L., Webb, L.B., Merenlender, A. and Reynolds, M. 2013. Vinecology: pairing wine with nature. Conservation Letters 6: 287-299.
de Graaff, G. 1981. The Rodents of Southern Africa: Notes on their Identification, Distribution, Ecology, and Taxonomy. Butterworths, Durban, Pretoria, South Africa.