Thick-tailed Bushbaby
Otolemur crassicaudatus

2025 Red list status
Near Threatened
Regional Population Trend
Stable
change compared
to 2016
Uplisted
Overview
Otolemur crassicaudatus – (É. Geoffroy Saint-Hilaire, 1812)
ANIMALIA – CHORDATA – MAMMALIA – PRIMATES – GALAGIDAE – Otolemur – crassicaudatus
Common Names: Thick-tailed Bushbaby, Thick-tailed Greater Galago, Greater Bushbaby, Greater Galago, Large-eared Greater Galago (English), Bosnagaap (Afrikaans), Impukunyoni (Ndebele), Chimhavira (Shona), Xideweta (Xitsonga), Sinkwe (Zulu)
Synonyms: Galago crassicaudatus É. Geoffroy Saint-Hilaire, 1812
Otolemur argentatus was distinguished by Kingdon (1997) and O. monteiri by Grubb et al. (2003) as separate species. No evidence has been provided, however, that either is more than a subspecific variant of O. crassicaudatus (Groves 2001). Both taxa are on the large side of the body size range, and have a silvery-grey pelage.
Taxonomic Note:
The genus Otolemur (the eared-lemur) was proposed by Coquerel (1859) for the Small-eared Greater Galago (O. garnetti) found in eastern Kenya, Tanzania and the offshore East African islands. The generic rubric was lost for much of the 20th century, but later resurrected (Olson 1981; Nash et al. 1989; Groves 2001) and is now accepted terminology. Otolemur differs from other galago genera on the basis of its large size (adult body weight 800-1500g), relatively flat basicranium, low-crowned molars and reduced third upper molars (M3). Both species have a cornified region of skin proximal to the plantar pads of the hind foot that is rubbed noisily against the substrate during periods of agitation or insecurity. The distal margins of the nails are convex in young animals, but wear quickly to form a semi-lunar, concave margin in adults. Grubb et al. (2003) separated out the form O. monteiri as a distinct species. However, there is a wide area of overlap in the distribution of forms attributable to O. monteiri and O. crassicaudatus in Tanzania (Groves 2001). For the current assessment, O. monteiri is regarded as a subspecies of O. crassicaudatus based on information regarding its skull morphology (Masters and Lubinsky 1988) and karyotype (Masters 1986). Greater galago subspecies show minor differences in skull size but not in proportions, while skulls of the two recognised Otolemur species can readily be distinguished on the basis of skull proportions. The subspecies O. c. crassicaudatus (Groves 2001), occurs in Mpumalanga, Limpopo and KwaZulu-Natal provinces in South African and Mozambique south of the Limpopo River. Across this region the pelage is variable, ranging from dark grey to chocolate brown, and various admixtures of the two. O. crassicaudatus subspecies have been defined mainly on pelage coloration, which may be adapted to the foliage in which the animals rest during the day, rather than for specific-mate recognition. Hence, the subspecies that occupy dry forests in miombo woodlands (O. c. monteiri, O. c. argentatus) are pale in colour, while those from the dune forests of KwaZulu-Natal and the montane forests of Limpopo Province (O. c. crassicaudatus) are darker. Nevertheless, recent research on a population living within Afromontane Mistbelt forests of the Lajuma Research Centre in the Soutpansberg mountains have documented both pale and darker coat variants within the same area as well as both rufous and pale colouration of tail and extremities for both coat variants. Genomic data and phylogenetic analyses are clearly needed to clarify species and sub-species distinctions for Otolemur crassicaudatus.
| Red List Status |
|
NT- Near Threatened B2ab(iii) (IUCN version 3.1) |
Assessment Information
Assessors: Cuozzo, F.P2., Sauther, M.L3., Linden, B4. & da Silva, J.5
Reviewer: Scheun, J.6
Institutions: 1 Formerly University of Fort Hare South Africa, 2 Mammal Research Institute, University of Pretoria, South Africa, 3Department of Anthropology, University of Colorado, USA, 4Aberystwyth University, UK, University of Venda, South Africa, 5South African National Biodiversity Institute, 6Department of Nature Conservation, Tshwane University of Technology.
Previous Assessors: Génin, F.1 † Masters, J.1 †
Previous Reviewer: Child, M.F.
Previous Contributors: Ehlers-Smith, Y. & Page-Nicholson, S.
Assessment Rationale
Previously listed as Least Concern, this IUCN rating was based on limited data with no long-term studies of this species, especially at single localities in northern South Africa. All studies prior to the past decade were short term (less than or up to one year) often based solely on behavioural and/or ecology data, and frequently occurring in areas of disturbed habitats, such as farms in Limpopo (formerly Transvaal) Province. Over the past decade, one ongoing long-term study at a single locality (western Soutpansberg, Lajuma Research Centre, Limpopo Province), and broader literature based on ad hoc data provide a much different and more comprehensive picture of this species ecology, behaviour, health, genetics, growth and development, and most important, the varied threats to this species’ continued existence. Documented, quantified threats now include various forms of linear infrastructure (roads, power lines, and electric fences), frequent road kills on primary and secondary roadways, domestic dog kills, and some traditional cultural practices. The species is relatively widespread within the assessment region and in sub-Saharan Africa, and present in several protected areas in South Africa (including Kruger National Park). Northern and northeastern South Africa, however, comprise the southernmost limit of its distribution, within which the species is confined to moist savannah, riverine and Afromontane forest, which are naturally fragmented habitats, and Thick-tailed Bushbaby subpopulations are highly localised. The situation of fragmented habitats has been rapidly exacerbated by human actions, with habitats being both reduced in size, and further fragmented due to roads, fences, power lines, and the loss of intact forests due to increasing agricultural areas. Both the relatively large size of the animals and their habit of sleeping in tree holes during the day make them favoured bushmeat in northern KwaZulu-Natal, and their presence in markets attests to their use in traditional medicine. The effect of this culling on population viability cannot be assessed from available information. The species’ range is unlikely to have expanded as a result of the conversion of land use from livestock to savannah wildlife ranching and is likely to have contracted due to urbanisation and transformation of suitable habitat to agricultural fields. Removal of dead trees from these areas is also likely to limit available day-time resting sites and consequent protection from predators. In addition, at least one locality in Limpopo Province (the Mokopane Biodiversity Centre, a satellite station of the National Zoological Garden, Pretoria Zoo (NZG), has witnessed an extirpation of the Otolemur crassicaudatus population, due to the construction of a dam upriver of the centre, which has led to a rapid deterioration of the riverine forest in the reserve.
With an ever-increasing amount of agricultural, residential, and industrial development within this species’ known range across northern South Africa, the threats documented across the past decade, along with new data on health, migration, reproduction, and survival, this species is best listed as Near Threatened, pending continued, geographically diverse, and longitudinal data collection. Landcover change within the range of the species within the assessment region is estimated to be 3.6% per decade and 5.38 to 6.45% for 3 generations (depending on whether we estimate the generation length as 5 or 6 years). This is higher than the country average of 2.06% per decade (calculated from landcover change data shared by Andrew Skowno, SANBI). The area of occupancy for this species, based on the IUCN method of a 2 x 2km grid is 748 km2. This falls within the <2000 km2 range for Vulnerable. In addition, based on landcover change, we can infer a continuing decline in area, extent and quality of habitat, and a fragmented population due to their habitat preferences, vulnerability to mortality on linear infrastructure and likely limited dispersal abilities. Although it can be debated whether their habitat is severely fragmented or not, they are close to qualifying to Vulnerable B2ab(iii).
Regional population effects: The species’ range is apparently continuous through tropical and subtropical sub-Saharan Africa, but areas of suitable habitat within this range are fragmented and likely to confer a degree of genetic isolation. One example of this fragmentation is seen in Soutpansberg Mountains in northern Limpopo Province, South Africa, which is dissected by the heavily trafficked (including large freight trucks) N1 highway, which presents a major barrier for arboreal animals to traverse. Numerous road kills of Otolemur crassicaudatus have been recorded on this major highway (Cuozzo et al. 2020, Linden unpublished data). Dispersal is suspected to occur along the northern border of South Africa through the Greater Mapungubwe and Greater Limpopo Transfrontier conservation areas between Zimbabwe, the Tuli Block and Mozambique, and into northeastern KwaZulu-Natal, via riparian and dune forest corridors through the Lubombo Transfrontier Conservation Area, however genetic data is needed to confirm this. The South African population comprises the southerly extreme of the species’ distribution, and much of the available habitat is likely to be marginal.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: The main threats have increased, new threats have been identified, new information has become available on population genetics.
Red List Index
Red List Index: Uplisted
Recommended citation: Cuozzo FP, Sauther ML, Linden B & da Silva JM. 2025. A conservation assessment of Otolemur crassicaudatus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
At the species level, O. crassicaudatus has a “Zambezian” distribution, from Angola in the west to Kenya and Tanzania in the east, and KwaZulu-Natal Province, South Africa, in the south. Its preferred habitat is a natural mosaic of coastal or riverine forest-savannah transition. In dry regions, Thick-tailed Bushbabies follow riverine forest, creating a discontinuous distribution. On the east coast it is found in coastal forest and thicket, and the population was probably continuous in the recent past but is less so today because of incursions from urbanisation and agriculture (sugar cane and forestry plantations).
The only subspecies to occur within the assessment region is O. c. crassicaudatus. Its range extends north from the vicinity of Mokopane and Makhado into northern Limpopo Province, including across the Soutpansberg mountains and eastwards through Mpumalanga and Eswatini into northern KwaZulu-Natal and southern Mozambique, occurring marginally on the Eastern Cape coast (Skinner & Chimimba 2005). In western Eswatini, it has adapted to wattle forests and is found around Mbabane (Skinner & Chimimba 2005). Otolemur c. crassicaudatus grades into and is replaced by O. c. kirkii in Zimbabwe, the Botswanan Tuli Block, southern Malawi (Chikwawa region) and central Mozambique. The pelage of this subspecies is paler than that of O. c. crassicaudatus, and the tail and extremities have a rufous brown tint. Further north, in Angola, Zambia, Malawi and northern Mozambique (predominantly in Brachystegia woodland), and into southern Democratic Republic of the Congo and southern Tanzania (Tabora), O. c. kirkii is replaced by O. c. monteiri, a silvery-grey subspecies with brown ear pinnae and extremities. Otolemur c. monteiri has also been reported from Rwanda, northern Tanzania and Kenya, where dark brown to black ears and extremities have led to it being given the name O. c. argentatus by some researchers.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): 1800
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realms
Biogeographic Realm: Afrotropical
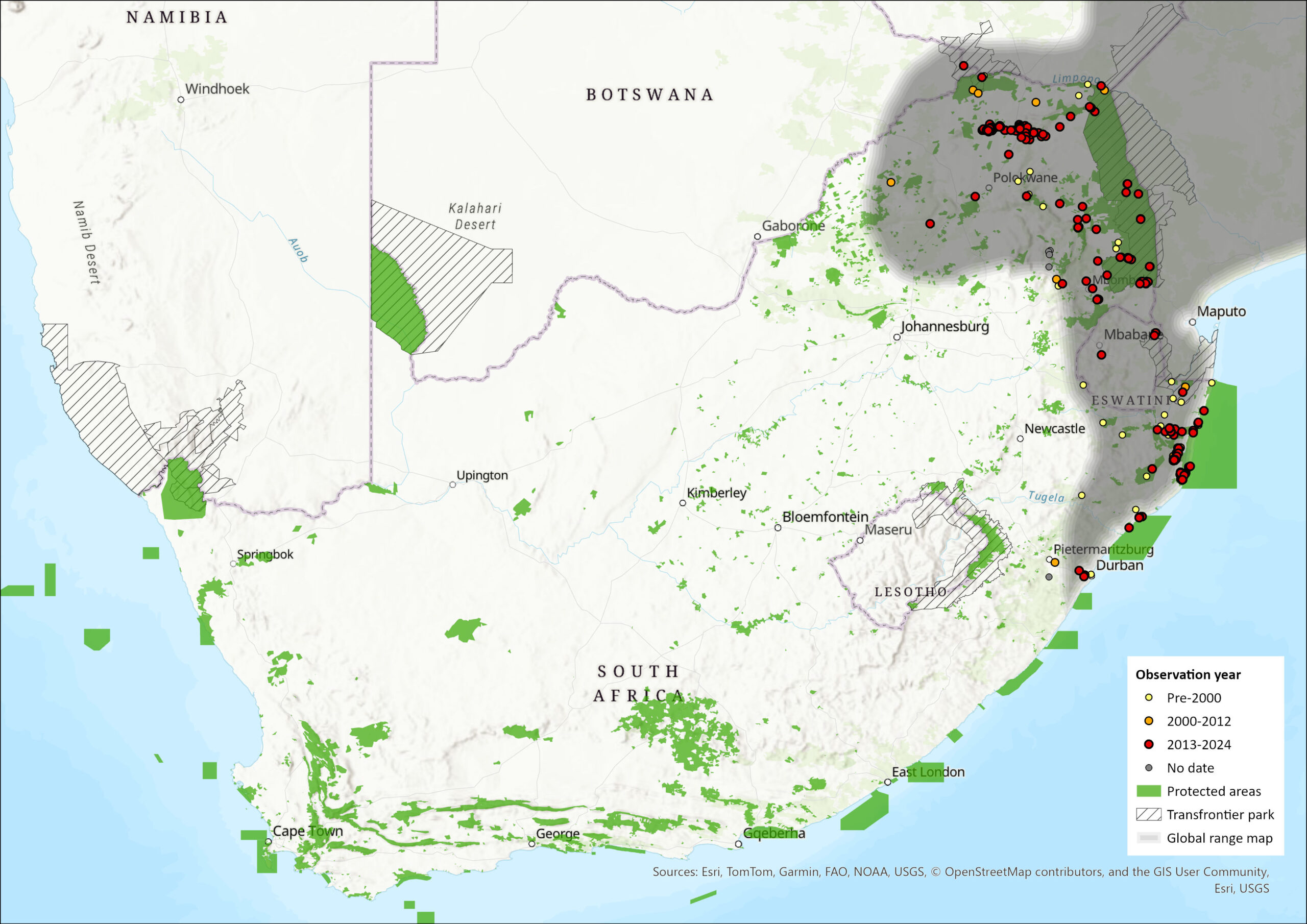
Map
Figure 1. Distribution records for Thick-tailed Bushbaby (Otolemur crassicaudatus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Primates in general are highly susceptible to anthropogenic pressures, with over 60% of primate species now threatened with extinction (Estrada et al. 2017). An important component includes a species physiological response to environmental change to assess primate resilience. A recent study of the effect of seasonality in wild O. c. crassicaudatus fecal glucocorticoid metabolite concentrations within the Afromontane Mistbelt forest of the Lajuma Research Centre in the Soutpansberg found that seasonal variation in key food resource availability affected concentrations; reduction in gum availability was associated with higher concentrations (Long et al. 2021). In addition, females had higher concentrations overall than males, with lactation associated with the highest concentrations. Overall, this indicates reduction in gum availability for this gum specialist could be difficult and females may be especially sensitive to environmental change.
Population information
No population estimates have been made to date. However, the fragmented nature of its preferred habitat and the potential for genetic isolation that this confers indicate in favour of population studies to investigate local levels of genetic diversity. The only study on population genetics for this species at the Lajuma Research Centre in the Soutpansberg documented a single haplotype, indicating selective barriers in and out of this population (Phukuntsi et al. 2020). The same study found male-biased gene flow, but in general dispersal ability is unknown. Given the lack of data we cannot determine whether the population is stable, but the species is present in many protected areas (32 known). A formal survey throughout the assessment region is needed.
Population Information
Current population trend: Assumed stable
Continuing decline in mature individuals? No
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Severely fragmented: Yes. Incursions into natural forest patches (for example, Ngoye) for firewood; increasing destruction of natural forest on KwaZulu-Natal Province north coast by plantations and urban developments.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Due to the fragmented nature of O. c. crassicaudatus within this assessment region as well as the effect of linear infrastructure on mortality (Linden et al. 2022) there is an urgent need for population genetic assessments. To date, only one population genetic study using mitochondrial DNA has been undertaken for this species, specifically at the Lajuma Research Centre located in a high altitude Mistbelt forest in the Soutpansberg mountains. A single haplotype was uncovered, indicating selective barriers in and out of this population (Phukuntsi et al. 2020), and potentially very low levels of genetic diversity. Given that the same study found male-biased gene flow, a different genetic picture might be drawn using more fine-scale nuclear markers. This cannot be accurately inferred given that the dispersal ability of males is unknown. A comprehensive population genetic survey and analysis throughout this species’ distribution using nuclear markers is needed to understand population genetic structuring and diversity within this species.
Habitats and ecology
Thick-tailed Bushbabies are mostly found in forests, moist savannah woodland, riparian bushland and thickets in higher rainfall regions, but penetrate into drier areas along riparian corridors (Skinner & Chimimba 2005). They can occur in urban gardens and farmland where there is sufficient tree cover for shelter and fruit-bearing species to provide food. Thick-tailed Bushbabies feed on a mixed diet that can include insects and molluscs, although small vertebrates are occasionally taken (Masters et al. 1988). Gum (Vachellia karoo) is especially important and is used year-round (Harcourt 1986), reflecting their characterisation as an intensive exudativore, with both dental and digestive adaptations for gum feeding (Burrows and Nash, 2013). They also include fruit depending on the season (Bearder, 1974).
Like all living galagos, Thick-tailed Bushbabies have an elongated ankle region that allows some leaping, but this species is primarily a quadrupedal climber. Spaces between trees are generally crossed by “bridging” (the animal retains its grip on the original tree with its hind feet and secures a branch on the destination tree with its hands before attempting to cross). Other, more saltatory species, like Galago moholi, leap across open spaces.
Thick-tailed Bushbabies communicate primarily by vocalisations – including the long distance “crying baby” calls from which the animals derive their colloquial name – as well as by olfaction. Otolemur crassicaudatus is one of several galago species that practise “urine-washing”, a stereotyped behaviour in which urine is dribbled onto the palm of one hand and wiped across the sole of the ipsilateral foot. The behaviour is then repeated with the contralateral hand and foot. Urine-washing enables galagos to leave a scent trail behind them as they pursue their nightly activities. Thick-tailed Bushbabies also have a bare patch of skin on the sternum which is rich in scent glands. The major component of the chest/sternal gland secretion is benzyl cyanide, and the complex odour changes with time as the volatile components evaporate, enabling neighbours to track one another’s movements over a period of hours. Thick-tailed Bushbabies sleep alone or in groups of 2–6 and disperse at sunset to forage solitarily or in small groups. Mating generally occurs in June and July, when there is a great increase in the frequency of the loud calls as potential mating partners locate each other, and the established adult males remind rivals of their locations. Thick-tailed Bushbabies restrict their activities to home ranges, the borders of which are patrolled, announced, and scent-marked regularly. When mating occurs, it involves prolonged bouts of copulation (up to 45 minutes); the adult male penis is furnished with epidermal spines that point backwards (Dixson & Van Horn 1977) and provide a “genital lock”. Penile spines occur in several galago species, and the pattern of the spines is species-specific. Female Thick-tailed Bushbabies give birth in early November to 1–3 infants per year, and twinning is common.
Ecosystem and cultural services: The frugivorous habits of Thick-tailed Bushbabies mean that they play a role in seed dispersal for indigenous thicket, woodland and forest plant species. As fruit-eating primates they carry seeds for greater distances and create a more scattered seed dispersal compared to terrestrial seed-dispersers (Entwistle & Dunstone 2000). No detailed study of the effects of galagos on forest regeneration or diversity has been carried out to date. Galagos are also likely to assist in the control of insect numbers.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.9. Forest-> Subtropical/Tropical Moist Montane | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Marginal | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Marginal | – |
Life History
Generation Length: 5
Age at Maturity: Female or unspecified: 2 years (1.5 years from Lajuma)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: 12 years (Lajuma study) (18 in captivity)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 133 days
Reproductive Periodicity: Annual
Average Annual Fecundity or Litter Size: 1.5
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is used locally for the traditional medicine trade and also as bushmeat (Svensson et al. 2016). Although the impacts have not been measured, it is often seen in muthi markets which sell traditional medicines (for example, Whiting et al. 2011).
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | – | – | – |
| 3. Medicine – human & veterinary | true | – | – | – |
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
The past decade has seen a dramatic increase in the number of studies of Otolemur crassicaudatus’ ecology, biology, health, anatomy, genetics, growth and development, and conservation threats. This includes a now decade plus comprehensive study of this species at the Lajuma Research Centre, in northern Limpopo Province, South Africa. Although long rated as an IUCN Least Concern species (e.g., Masters and Genin, 2016), in part due to its broad traditional range and frequent sightings in protected areas or and residential gardens, this species is facing a number of new and increasing threats, both human induced, or at least human influenced. Two areas of rapidly increasing threat are varied forms of linear infrastructure (roads, power lines, and electric fences) and newly quantified (though likely not a new threat) data on kills by domestic dogs, in varied contexts, and endemic carnivores in disturbed habitats (Cuozzo et al. 2020, 2021, 2022; Linden et al. 2022). In recent years, a number of deaths have occurred through electrocution (power lines and electric fences) in the Soutpansberg Mountains (Cuozzo et al. 2020; Linden et al. 2022). Additionally, an incident of a thick-tailed bushbaby being chased down and killed by domestic hunting dogs due to superstitious reasons linked to witchcraft was reported in the Soutpanberg area (Tshikudo, 2015). Although this was a single record, it is likely that bushbabies are persecuted based on a lack of understanding of the animal (Linden, 2015; Cuozzo et al. 2022). In addition, changing habitats, especially the reduction in natural forests of several types, may be leading to shifts in predation by endemic carnivores. One example was documented in Limpopo Province, where the first record of a Caracal preying upon a Think-tailed Bushbaby was recorded, occurring in disturbed habitat on a private farm along the R522 corridor (Cuozzo et al. 2021). The species occurs in several protected areas but may be locally threatened in parts of its range through the loss of suitable forest habitat. Within the assessment region, timber plantations and human settlement have fragmented the Thick-tailed Bushbaby’s habitat, which may lead to inbreeding in isolated subpopulations. For example, in the Soutpansberg, Limpopo Province, pine and eucalyptus plantations and residential housing expansion reduced forest cover by 20% over a 16-year period between 1990 and 2006 (Munyati & Kabanda 2009). The animals are also illegally harvested for the pet trade, and traditional medicine, and are also poached for human consumption (for example, in KwaZulu-Natal Province), which may result in local declines.
Conservation
Among the newly identified and increasing threats posing conservation pressure on this species is documentation of death resulting from various forms of linear infrastructure and from domestic dogs. Since 2013 and emanating from the long-term project based at the Lajuma Research Centre, both direct observations and observations through a broad set of citizen scientists have provided the first empirical data on these increasing threats. Deaths of O. crassicaudatus on power lines and roads have been collated from national data bases (Linden et al. 2022) illustrating that many taxa, including this species, are threatened by these forms of infrastructure. Observations stemming from the bushbaby project at the Lajuma Research Centre indicate that road kills occur frequently, on both primary highways such as the N1, and secondary roads including the R522 and R524. These deaths occur in areas of higher human population density, such as within the city limits of Louis Trichardt (Mahkado), and in outlying areas dominated by agriculture. Direct observations of additional linear infrastructure threats include electrocution on power lines and electric fences, common in many areas of the region, and are likely increasing in their use as agricultural areas continue to expand (Cuozzo et al. 2020; Linden 2022)
Recommendations for conservationists, land managers and practitioners:
- Thick-tailed Bushbabies’ requirement for moist riparian woodland and thicket in the interior regions of its range is likely to limit dispersal. Protected area expansion and connection of forest habitats through corridors and transfrontier conservation areas, such as that being developed in Maputaland (Smith et al. 2008), are important interventions for this species. Protection of riparian forest passageways are particularly important. Conservationists should continue to enforce protected area rules and prevent the illegal harvesting of firewood.
- Landowners should continue to form conservancies to protected critical forest habitat.
- Reserve managers should prevent the removal of dead trees for firewood. These old trees comprise an important source of wood boring insects, as well as cavities for shelter.
- Given the necessity of guard dogs on farms and at residences in more urban areas, one simple mitigation measure is to ensure that outdoor food for these dogs be covered or moved during the evening hours when bushbabies are most active, and are known to come to the ground, especially in areas where food is accessible (Cuozzo et al. 2022).
- Awareness campaigns to educate local people about the harmless nature of these animals should be undertaken
- In urban and residential areas, people should be educated about the effects of directly feeding bush babies and the attraction of bird feeders in gardens.
- In terms of power lines, a simple and cost-effective way to prevent electrocution on power lines is to use basic plastic or rubber covering to insulate live electric lines and to cut back tree branches from the power lines to discourage climbing or jumping onto them, which has been done successfully at the Lajuma Research Centre.
- One way to mitigate road kills is the construction of so-called canopy bridges, which have been used with success in pilot studies in Limpopo Province, as well as in other areas of the world (Linden et al. 2020).
- Little is known about the degree of isolation between, or level of inbreeding within, fragmented populations, and no survey has been undertaken as to the likely size of the South African population. A phylogeographic assessment of the degree of isolation of local subpopulations, and the potential for gene flow among them is urgently needed. A measure of whether fragmentation is increasing, or affecting population sizes and heterozygosity will enable more robust projections of population impacts.
- Investigating the efficacy of educating local communities to decrease poaching rates
Research priorities:
- Population genetic study across the species distribution using fine-scale nuclear markers to understand the genetic structure, connectivity and diversity within this species and its populations.
- Investigating the efficacy of educating local communities to decrease poaching rates
Encouraged citizen actions:
- With respect to habitat preservation, landowners can create conservancies to protect natural forest habitat in KwaZulu-Natal, Limpopo and Mpumalanga. Albizia trees are much preferred by Otolemur crassicaudatus. Any person residing within the assessment region can assist the local wildlife by planting trees and shrubs indigenous to their area, rather than exotic species.
- Sightings outside private lands or protected areas can be reported on virtual museum platforms (for example, iNaturalist and MammalMAP).
Bibliography
Bearder, S.K. 1974. Aspects of the Ecology and Behavior of the Thick-tailed Bushbaby, Galago crassicaudatus. (Unpublished doctoral thesis). University of the Witwatersrand.
Burrows, A.M., Nash,L.T. 2010. Searching for dental signals of exudativory in Galagos. In A.M. Burrows, & L.T. Nash (Eds), The Evolution of Exudativory in Primates. Developments in Primatology: Progress and Prospects (211-233). Springer. https://doi.org/10.1007/978-1-4419-6661-2_12
Cuozzo, F.P., A. Halajian, M.L. Sauther, B. Linden, J. Linden, J. Millette, A.S.W. Tordiffe & D. Romanello. 2020. Human induced threats to a nonhuman primate of “Least Concern” (Otolemur crassicaudatus, Primates: Galagidae) in northern South Africa. American Journal of Physical Anthropology 171(S69): 63.
Cuozzo, F.P., A. Halajian, M.L. Sauther, K.M. Rampedi & J.B. Millette. 2021. First report of the thick-tailed bushbaby (Otolemur crassicaudatus) being preyed upon by an endemic carnivore (Caracal caracal) in South Africa. African Zoology 56(3): 231–235.
Cuozzo, F.P., J. Linden, M.L. Sauther, A. Halajian & B. Linden. 2022. Domestic Dogs Present a Human-Induced Threat to Thick-tailed Bushbabies (Otolemur crassicaudatus) in Northern South Africa. African Primates 16: 59-64.
Estrada, A., Garber, P.A., Rylands, A.B., Roos, C., Fernandez-Duque, E., Di Fiore, A., Nekaris, .KA., Nijman, V., Heymann, E.W., Lambert, J.E. et al. 2017. Impending extinction crisis of the world’s primates: why primates matter. Science Advances 3: 1–17. 10.1126/sciadv.1600946.
Coquerel C. 1859. Notes de Mammalogie. 1. Genre nouveau de Lémurien. Revue et Magasin de Zoologie 2, Part XI: 457-460; 2. Renseignements sur quelques Lémuriens de Madagascar. Revue et Magasin de Zoologie 2 Part XI:4461–464.
Dixson AF, van Horn RN. 1977. Comparative studies of morphology and reproduction in two subspecies of the greater bushbaby, Galago crassicaudatus crassicaudatus and G. c. argentatus. Journal of Zoology 183: 517–526.
Entwistle A, Dunstone N. 2000. Priorities for the conservation of mammalian diversity: has the panda had its day? Cambridge University Press, Cambridge, UK.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Groves, C.P. 2001. Primate Taxonomy. Smithsonian Institution Press, Washington, DC, USA.
Grubb, P., Butynski, T.M., Oates, J.F., Bearder, S.K., Disotell, T.R., Groves, C.P. and Struhsaker, T.T. 2003. Assessment of the diversity of African primates. International Journal of Primatology 24: 1301–1357.
Jewitt D, Goodman PS, Erasmus BFN, O’Connor TG, Witkowski ETF. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: implications for biodiversity. South African Journal of Science 111: 1–9.
Harcourt, C. 1986. Seasonal variation in the diet of South African Galagos. International Journal of Primatology 7: 491-506. https://doi.org/10.1007/BF02693660
Kingdon, J. 1997. The Kingdon Field Guide to African Mammals (first edition}. Academic Press, San Diego, California, USA.
Linden, B. 2015. It is a greater bushbaby, not a tokoloshe. Limpopo Mirror, May 15 https://www.zoutnet.co.za/articles/letters/30900/2015-05-15/it-is-a-greater-bushbaby-not-a-tokoloshe
Linden B., Foord S., Horta-Lacueva Q.J.B., Taylor P.J. 2020. Bridging the gap: how to design canopy bridges for arboreal guenons to mitigate road collisions. Biological Conservation, 246, 108560.
Linden, B., Cuozzo, F.P., Sauther ,M.L., Collinson Jonker, W. 2022. Impact of linear infrastructure on South Africa’s primate fauna: the need for mitigation. Folia Primatologica, 93:235-253. https://doi.org/10.1163/14219980-20211112
Long, C., Tordiffe ,A., Sauther, M., Cuozzo, F., Millette, J., Ganswindt, A., Scheun J. 2021. Seasonal drivers of faecal glucocorticoid metabolite concentrations in an African strepsirrhine primate, the thick-tailed greater galago (Otolemur crassicaudatus), Conservation Physiology: 9coab081, https://doi.org/10.1093/conphys/coab081
Masters JC, Lumsden WHR, Young DA. 1988. Reproductive and dietary parameters in wild greater galago populations. International Journal of Primatology 9: 573–592.
Munyati C, Kabanda TA. 2009. Using multitemporal Landsat TM imagery to establish land use pressure induced trends in forest and woodland cover in sections of the Soutpansberg Mountains of Venda region, Limpopo Province, South Africa. Regional Environmental Change 9: 41–56.
Nash, L.T., Bearder, S.K. and Olson, T.R. 1989. Synopsis of Galago species characteristics. International Journal of Primatology 10: 57–80.
Olson TR. 1981. Systematics and zoogeography of the greater galagos. American Journal of Physical Anthropology 54: 259–259.
Phukuntsi M.A., Du Plessis M., Dalton D.L., Jansen R, Cuozzo F.P., Sauther M..L, Kotze A. 2020. Population genetic structure of the thick-tailed bushbaby (Otolemur crassicaudatus) from the Soutpansberg Mountain range, Northern South Africa, based on four mitochondrial DNA regions, Mitochondrial DNA Part A, 31:1-10, DOI: 10.1080/24701394.2019.1694015
Skinner J.D. and Chimimba C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Smith RJ, et al. 2008. Designing a transfrontier conservation landscape for the Maputaland centre of endemism using biodiversity, economic and threat data. Biological Conservation 141: 2127–2138.
Svensson, M.S., Ingram, D.J., Nekaris, A.I., Nijman, V. 2016. Trade and ethnozoological use of African lorisiforms in the last 20 years. Hyxtrix, the Italian Journal of Mammology 26:153-161. https://doi.org/10.4404/hystrix-26.2-11492
Tshikudo, E. 2015. I killed the tokoloshe. Limpopo Mirror May 8. https://www.zoutnet.co.za/articles/news/30741/2015-05-08/i-killed-the-tokoloshe
Whiting MJ, Williams VL, Hibbitts TJ. 2011. Animals traded for traditional medicine at the Faraday market in South Africa: species diversity and conservation implications. Journal of Zoology 284: 84–96.