
Temminck’s Hairy Bat
Myotis tricolor

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Myotis tricolor – (Temminck, 1832)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – VESPERTILIONIDAE – Myotis – tricolor
Common Names: Temminck’s Hairy Bat, Cape Hairy Bat, Cape Myotis, Temminck’s Mouse-eared Bat, Three-coloured bat (English), Temminck se langhaarvlermuis, Temminck-langhaarvlermuis, Kaapse Langhaarvlermuis (Afrikaans)
Synonyms: Eptesicus loveni Granvik 1924
Taxonomic Note: Originally described in 1832 under the name V[espertilio] tricolor Temminck, in Smuts, Enumer. Mamm. Capensium, 106. The type locality is listed as South Africa: Cape province: Cape Town [33 56 S, 18 28 E]. The colloquial name is after C.J. Temminck, who was the author of Monographies de Mammalogie (1827) (Taylor 2005; ACR 2024).
Red List Status: LC – Least Concern (IUCN version 3.1)
Assessment Information
Assessors: Richards, L.R.1, Howard, A.2 & da Silva, J.M.3
Reviewer: Moir, M.4
Institutions: 1Durban Natural Science Museum, 2University of the Free State, 3South African National Biodiversity Institute, 4Stellenbosch University
Previous Assessors and Reviewers: Monadjem, A., Jacobs, D., Cohen, L., MacEwan, K., Richards, L.R., Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Nicholson, S.K., Raimondo, D. & Child, M.F.
Assessment Rationale
Listed as Least Concern in view of its wide distribution (extent of occurrence in the assessment region is 932,732 km2), its occurrence in multiple protected areas across its range, its known large population (colonies up to 1500 individuals; Taylor 1998) and because there are no major identified threats that could be causing widespread population decline. However, its dependence on large caves as roosting sites makes colonies vulnerable to human disturbance and its migratory behaviour may make it vulnerable to wind farm operation. Such threats should be monitored for their impacts on subpopulations and the population overall.
Regional population effects: The range of this species is continuous across the borders of the assessment region into Zimbabwe and Mozambique through its occurrence in transfrontier conservation areas. The species undergoes local migrations between winter hibernacula and summer maternity roosts (Monadjem et al. 2020). Therefore, rescue effects from nearest neighbour colonies are possible, particularly where habitats are continuous across geopolitical borders.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Richards LR, Howard A & da Silva JM. 2025. A conservation assessment of Myotis tricolor. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species has been patchily recorded in sub-Saharan Africa from Ethiopia to South Africa. In West Africa, it has been reported from the northwestern uplands of Liberia (Koopman et al. 1995; ACR 2024), while in central Africa it is known only from a few records in the Democratic Republic of the Congo and Rwanda (Hayman et al. 1966; Baeten et al. 1984). It is much more widely recorded in East Africa, ranging from Ethiopia in the north, through Uganda, Kenya, Tanzania, Malawi, Zambia, Mozambique and Zimbabwe through to southern South Africa (ACR 2024). Within the assessment region, it is widespread across eastern regions of South Africa, including Lesotho and Eswatini. It occurs from Cape Town east along the coast to the Eastern Cape (Herselman & Norton 1985), then north through Lesotho and the Free State (Watson 1990) to northern South Africa and east to KwaZulu-Natal (Monadjem et al. 2020). It is restricted to areas with suitable caves for roosting, which may explain its absence from flat and featureless terrain (although it has been recorded from the Mpumalanga Highveld, de Jong et al. 2024), and its general association with mountainous areas (Monadjem et al. 2020). The estimated extent of occurrence in the assessment region is 932,732 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 18 m asl (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 3,869 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
Map
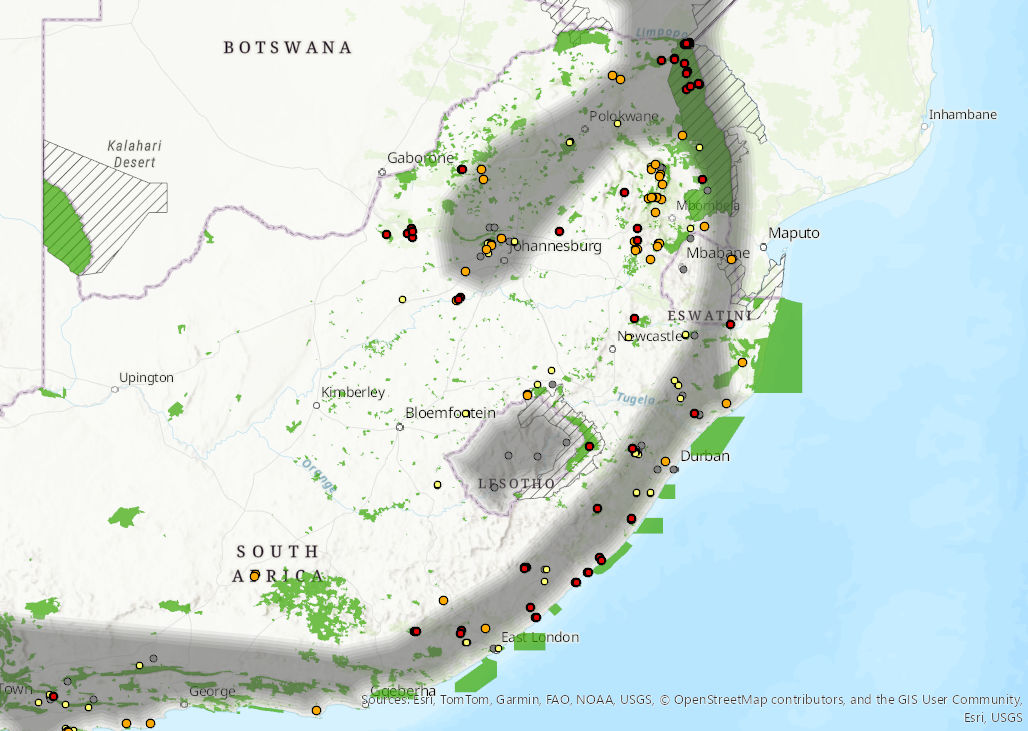 Figure 1. Distribution records for Temminck’s Hairy Bat (Myotis tricolor) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Temminck’s Hairy Bat (Myotis tricolor) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Liberia | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Sao Tome | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
No formal study of the direct effects of climate change on this species has been conducted. Most studies of the effects of climate change on bats are based on predictive species distribution modelling; thus, there is a lack of empirical studies measuring behavioural, physiological, phenological or genetic responses to extreme and seasonal climatic changes, especially in the Global South (Festa et al. 2023; Pio et al. 2014).
Globally, there have been documented declines in bat populations, species richness and distributions in relation to water availability, with increasing global aridity, which may become a growing concern as heat waves and maximum temperatures are expected to increase over much of South Africa (Adams & Hayes 2021; Mbokodo et al. 2020).
Population Information
Appears to be uncommon or rare throughout most of its range besides the assessment region. Here it has been recorded from many localities of the eastern regions, comprising colonies of hundreds up to 1,500 animals (Taylor 1998, 2000, Monadjem et al. 2020).
Current population trend: Stable
Continuing decline in mature individuals? Not suspected
Extreme fluctuations in the number of subpopulations: Cannot be determined
Continuing decline in number of subpopulations: Not suspected
All individuals in one subpopulation: Presumed so.
Number of mature individuals in largest subpopulation: Unknown, however, based on known colonies figure is likely >5,000, if not more (see below).
Number of Subpopulations: Although further studies are required, it appears to be one metapopulation (see Moire et al. 2020).
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Unlikely as there are many known colonies within the assessment region.
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Unlikely.
Probability of extinction in the wild within 100 years: Cannot be determined with available data.
Population Genetics
A phylogenetic study on the genus Myotis revealed two significantly distinct evolutionary lineages of Myotis tricolor – one of which is within South Africa (Patterson et al. 2019). Similarly, a population genetic study found population structuring with strong haplotypic segregation between Mpumalanga and northern KwaZulu-Natal, and between southern KwaZulu-Natal and Eastern Cape (Moir et al. 2020). However, given that these studies employed only mitochondrial genes and limited samples, further research employing an integrative taxonomic approach is highly recommended.
Assuming the species exists as a single metapopulation within the assessment region, connected to colonies in adjacent countries, and considering several colonies exist and some colonies are comprised of up to 1,500 individuals, it is highly likely that at least 5,000 individuals exist, translating to an effective population size of at least 500, meeting the threshold for a healthy, and stable population. A population genetic study is needed to verify this and to inform whether additional sub-structuring exists within the assessment region.
Habitats and ecology
It occupies widely varied habitats, including montane forests, rainforests, coastal forests, savannah woodlands (including mopane and miombo), arid thickets, mixed grassland and fynbos. It has a close association with mountainous terrain which may be due to its cave-roosting requirements (Monadjem et al. 2020). Within the assessment region, it occurs mainly in woodland savannah habitats but also dry grassland savannahs and montane grasslands in the Drakensberg Mountains (Lynch 1994; Taylor 1998). Most records in the region are from areas of > 500 mm of annual rainfall (Rautenbach 1982), which indicates that its distribution is probably limited by rainfall and the availability of humid caves and mines (Salata 2012). It may tolerate disturbed habitats to a degree. For example, it was recorded along the polluted Umbilo River in the Durban region in 2008 (Naidoo et al. 2011). However, this may have been influenced by the presence of nearby Paradise Valley Nature Reserve (Naidoo et al. 2011). It has been mist-netted in open spaces close to trees and close to the surface of running water (for example, Sirami et al. 2013; Howard et al. 2022; de Jong et al. 2024).
Roosts by day in moist caves and mine shafts hanging freely from ceilings or clinging to walls. Mostly found in larger caves usually containing pools of water where disturbance is minimal (Roberts 1951; Herselman & Norton 1985), but this is not always the case. In the North-West Province, it was recorded in old mine tunnels in the Vredefort Dome area (Power 2014). Throughout the assessment region, this species migrates hundreds of kilometres between warmer summer maternity caves (such as De Hoop Gauno Cave in the Western Cape) and colder winter hibernation caves (Monadjem et al. 2020). It is a clutter-edge forager with a diet consisting of Coleoptera, Hemiptera, Diptera, Neuroptera and Hymenoptera (Monadjem et al. 2020); aquatic insects can comprise a substantial portion of its diet (Moyo and Jacobs 2020; G. de Jong and L.R. Richards, unpublished data). The intermediate wing morphology of M. tricolor allows the flexible foraging guilds of an aerial forager, gleaner (takes insects off a substrate) and trawler (takes prey from the water surface) according to Jordaan & Jacobs (2009).
Ecosystem and cultural services: This insectivorous species has been recorded from macadamia orchards (Taylor et al. 2013; Weier et al. 2021) and is likely to assist with insect pest control. Worldwide, insectivorous bats contribute to controlling insect populations that damage crops (Boyles et al. 2011; Kunz et al. 2011). Ensuring a healthy population of insectivorous bats can thus decrease the need for pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Unknown | – |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Unknown | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 4.4. Grassland -> Grassland – Temperate | – | Suitable | – |
| 7.1. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Caves | – | Suitable | Yes |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: Wingate (1986) reported most females attain sexual maturity at 4-5 months of age; the remainder of the observed population attained maturity a year later.
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: Mean forearm length 5.04 ±0.18 cm; Mean total length = 10.60 ± 0.56 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Mean forearm length 4.96 ±0.15 cm; Mean total length = 10.40 ± 0.62 cm (Monadjem et al. 2020)
Longevity: Unknown
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: Estimated to be ca. 63 days
Reproductive Periodicity: Exhibit seasonal monoestry, with births occurring in the summer (November-December) (Barnard 1982).
Average Annual Fecundity or Litter Size: One pup per year
Natural Mortality: Natural predators include snakes, owls and other nocturnal raptors
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Migratory; undergo local migrations between winter hibernacula and summer maternity roosts (Monadjem et al. 2020).
Congregatory: Yes; some cave-roosting colonies can harbour 1,500 individuals (Taylor 1998, 2000).
Systems
System: Terrestrial
General Use and Trade Information
A recent study by Tarango et al. (2025) listed this species as imported into the U.S.A. Online e-commerce platforms should be monitored for illegal trade in bat taxidermy and specimens from the assessment region.
Local Livelihood: None reported
National Commercial Value: May provide essential ecosystem services in the form of pest insect control in macadamia orchards.
International Commercial Value: May provide essential ecosystem services in the form of pest insect control in macadamia orchards; an important export product.
End Use: N/A
Is there harvest from captive/cultivated sources of this species? None reported from the assessment region.
Harvest Trend Comments: N/A
Threats
Overall, there are no major known threats to this species (ACR 2024). To date, disturbance of roost sites in caves due to tourism, and traditional ceremonies and rituals has been the largest threat to this species (Barnard 2013). It is also threatened by habitat loss around roost sites due to agricultural expansion and poor land-use management practices, as well as alien and invasive plants infestations, which depletes insect biomass (the prey base for this species). In parts of its range, (for example, Mpumalanga), the species is threatened by legal and illegal mining, and re-commissioning of old mines, as well as new threats from proposed wind farms. Wind energy may pose a significant threat as this species has a medium to high risk of mortality from wind turbine blades due to its migratory habits (Sowler et al. 2020). One South African wind farm fatality is documented in the Durban Natural Science Museum specimen records.
Conservation
This species occurs in more than ten protected areas within the assessment region, including Great Limpopo Transfrontier Park, and is a well-conserved species. It must, however, be noted that in parts of its range, large populations often occur in caves and mines outside formally protected areas. As such, continued roost protection is necessary. Additionally, this species would benefit from holistic land management that reduces pesticide use and conserves buffer strips of natural vegetation to sustain insect biomass (Weier et al. 2021). To operational wind farms may pose a threat, especially along migration routes. Mortalities from turbine collisions on wind farms should be reported and mitigated through interventions such as using ultrasound to deter bats and curtailing turbines at low wind speeds (Baerwald et al. 2009; Berthinussen et al. 2010; Arnett et al. 2011).
Recommendations for land managers and practitioners:
- Reduce pesticide use in agricultural landscapes and maintain buffer strips of natural vegetation.
- Data sharing by wind farm managers into a national database is needed to be able to calculate cumulative impacts and thereafter implement collaborative mitigation and management efforts.
- Conservation of riparian habitats, aquatic biodiversity and associated invertebrate prey species.
Research priorities:
- Quantification of severity of local threats. For example, monitoring mortalities linked with windfarm operations and assessing impact on populations.
- Systematic monitoring to measure overall population size, trends and regional migration routes.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
- Minimise disturbance at caves when visiting.
Bibliography
ACR. 2024. African Chiroptera Report. 2024. AfricanBats NPC, Pretoria. i – xv; 1 – 1397.
Arnett, E.B., Huso, M.M., Schirmacher, M.R. and Hayes, J.P. 2011. Altering turbine speed reduces bat mortality at wind-energy facilities. Frontiers in Ecology and the Environment 9: 209–214.
Baerwald, E.F., Edworthy, J., Holder, M. and Barclay, R.M. 2009. A large-scale mitigation experiment to reduce bat fatalities at wind energy facilities. Journal of Wildlife Management 73: 1077–1081.
Baeten, B., Van Cakenberghe, V. and De Vree, F. 1984. An annotated inventory of a collection of bats from Rwanda. Revue de Zoologie Africaine 98(1): 183-196.
Bernard, R.T.F. 1982. Monthly changes in the female reproductive organs and the reproductive cycle of Myotis tricolor (Vespertilionidae: Chiroptera). South African Journal of Zoology, 17(2), pp.79-84.
Bernard, R.T.F. 2013. Myotis tricolor, pp 706-708. In: The Mammals of Africa. Volume IV: Hedgehogs, Shrews and Bats. Eds: M. Happold and D.C.D. Happold. Bloomsbury Publishing, London.
Berthinussen, A., Richardson, O.C. and Altringham, J.D. 2010. Bat Conservation: Global Evidence for the Effects of Interventions. Synopses of Conservation Evidence. Pelagic Publishing, Exeter.
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
de Jong, G.J., Melville, H.I. and Richards, L.R. 2025. The Insectivorous Bat Assemblage in Telperion Nature Reserve, Mpumalanga, South Africa: Species Inventory and Call Library. Acta Chiropterologica, 26(2), 315-327.
Festa, F., Ancillotto, L., Santini, L., Pacifici, M., Rocha, R., Toshkova, N., Amorim, F., Benítez‐López, A., Domer, A., Hamidović, D. and Kramer‐Schadt, S. 2023. Bat responses to climate change: a systematic review. Biological Reviews, 98(1), pp.19-33.
Hayman, R.W., Misonne, X. and Verheyen, W. 1966. The bats of the Congo and of Rwanda and Burundi. Annales Musée Royal de l’Afrique Centrale, Sciences Zoologiques 154: 1-105.
Herselman, J.C. and Norton, P.M. 1985. The distribution and status of bats (Mammalia: Chiroptera) in the Cape Province. Annals of the Cape Provincial Museums 16: 74-126.
Howard, A., Monadjem, A., MacFadyen, D. and Chimimba, C.T. 2022. Testing the efficacy of bat monitoring methods for identification and species surveys in KwaZulu-Natal province, South Africa. African Zoology, 57(4), pp.180-194.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: implications for biodiversity. South African Journal of Science 111: 1–9.
Jordaan, S. L. and D. S. Jacobs. 2009. Foraging behaviour of Myotis tricolor (Chiroptera: Vespertilionidae) in relation to reproductive state. 50th Anniversary Conference of the Zoological Society of Southern Africa. 21 – 25 July 2009 – Natalia, Illovo Beach, KwaZulu-Natal: 35. URL: http://academic.sun.ac.za/botzoo/zssa/ZSSA_2009_Abstract_Booklet.pdf
Koopman, K.F., Kofrin, C.P. and Chapman, A. 1995. The bats of Liberia: Systematics, ecology and distribution. American Museum Novitates 3148: 1-24.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum, Bloemfontein 10: 177–241.
McDonald, J.T., Rautenbach, I.L. and Nel, J.A.J. 1990. Roosting requirements and behaviour of five bat species at De Hoop Guano Cave, southern Cape Province of South Africa. South African Journal of Wildlife Research 20: 157–161.
Moir, M., Richards, L.R.R., Cherry, M.I. and Rambau, R.V. 2020b. Demographic responses of forest-utilizing bats to past climate change in South Africa. Biological Journal of the Linnean Society, 130(4): 850-868.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis, Second Edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Moyo, S. and Jacobs, D.S. 2020. Faecal analyses and alimentary tracers reveal the foraging ecology of two sympatric bats. PLoS One, 15(1), p.e0227743.
Naidoo, S., Mackey, R.L. and Schoeman, C.M. 2011. Foraging ecology of insectivorous bats (Chiroptera) at a polluted and an unpolluted river in an urban landscape. Durban Museum Novitates 34: 21–28.
Pence, G.Q.K. 2014. Western Cape Biodiversity Framework 2014 status update: Critical Biodiversity Areas of the Western Cape. In: Unpublished CapeNature project report (ed.). CapeNature, Cape Town, South Africa.
Pio, D.V., Engler, R., Linder, H.P., Monadjem, A., Cotterill, F.P., Taylor, P.J., Schoeman, M.C., Price, B.W., Villet, M.H., Eick, G. and Salamin, N. 2014. Climate change effects on animal and plant phylogenetic diversity in southern Africa. Global Change Biology, 20(5), pp.1538-1549.
Power, R.J. 2014. The Distribution and Status of Mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng, South Africa.
Rautenbach, I.L. 1982. Mammals of the Transvaal.Mammals of the Transvaal. No. 1, Ecoplan Monograph.
Roberts, A. 1951. The Mammals of South Africa. The Trustees of the Mammals of South Africa. Central News Agency, Johannesburg, South Africa.
Salata, H.A.B. 2012. Environmental factors influencing the distribution of bats (Chiroptera) in South Africa. Phd Thesis. University of Cape Town
Sirami C., Jacobs D.S. and Cumming G.S. 2013. Artificial wetlands and surrounding habitats provide important foraging habitat for bats in agricultural landscapes in the Western Cape, South Africa. Biological Conservation 164: 30–38.
Sowler, S., MacEwan, K., Aronson, J. and Lötter, C. 2020. South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities. South African Bat Assessment Association.
Tarango, M., Kolby, J.E., Goodman, O.L., Anderson, C.J., Tinsman, J., Kirkey, J., Liew, A., Jones, M., Firth, C. and Reaser, J.K. 2025. Going batty: US bat imports raise concerns for species conservation and human health. One Health, 20, p.100999.
Taylor, P.J. 2000. Bats of Southern Africa: Guide to Biology, Identification, and Conservation. University of Natal Press, Pietermaritzburg, South Africa.
Taylor, P.J. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.
Watson, J.P. 1990. Westward range extension of Temminck’s hairy bat in South Africa and Lesotho. South African Journal of Wildlife Research 20: 119–121.
Weier, S.M., Linden, V.M., Hammer, A., Grass, I., Tscharntke, T. and Taylor, P.J. 2021. Bat guilds respond differently to habitat loss and fragmentation at different scales in macadamia orchards in South Africa. Agriculture, Ecosystems & Environment, 320, p.107588.
Wingate, L .1983. The populations status of five species of Microchiroptera in Natal. MSc thesis, University of Natal, Durban. South Africa.
