
Swinny’s Horseshoe Bat
Rhinolophus swinnyi

2025 Red list status
Vulnerable
Regional Population Trend
Declining
Change compared
to 2016
No Change
Overview
Rhinolophus swinnyi – Gough, 1908
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – RHINOLOPHIDAE – Rhinolophus – swinnyi
Common Names: Swinny’s Horseshoe Bat (English), Swinny se Saalneusvlermuis (Afrikaans)
Synonyms: No Synonyms
Taxonomic Note:
Named after H. H. Swinny who collected the original specimens from the Ngqueleni district, Eastern Cape Province (Skinner & Chimimba 2005). Based on morphological similarities, it was suggested that Rhinolophus swinnyi might be a subspecies of R. denti (Csorba et al. 2003). However, based on morphology, molecular sequencing and echolocation, it has been shown that not only is this not the case, but northeastern populations are a separate species, R. rhodesiae, and is limited to the southeastern portion of the assessment region (Taylor et al. 2018, Demos et al. 2019; Taylor et al. 2024). Species delimitations are not fully resolved in the genus Rhinolophus and some publications show discrepancies in phylogenetic relationships. The primary cause of phylogenetic discrepancies is likely the type of marker(s) used. Specifically, the use of mitochondrial DNA has been shown to reflect introgression between lineages (Puechmaille et al. 2011; Benda et al. 2024). Nuclear introns have provided useful reconstructions of phylogenetic relationships, specifically if used in sets and in combination with additional independent markers (Dool et al. 2016). Phenotypic traits such as baculum morphology and acoustic data should be part of a comprehensive approach to resolve species/lineage delimitations in this genus.
Red List Status: VU – Vulnerable C2a(i) (IUCN version 3.1)
Assessment Information
Assessors: Taylor, P.1, Richards, L.R.2, Balona, J.3, Howard, A.1 & da Silva, J.M.4
Reviewers: Moir, M.5 & Mutumi, G.6
Institutions: 1University of the Free State, 2Durban Natural Science Museum, 3Gauteng and Northern Regions Bat Interest Group, 4South African National Biodiversity Institute, 5Stellenbosch University, 6Weblyfe Biological Consulting; University of California.
Previous Assessors and Reviewers: Jacobs, D., Schoeman, C., Cohen, L., MacEwan, K., Monadjem, A., Richards, L.R., Sethusa, T. & Taylor, P.
Previous Contributors: Roxburgh, L., Raimondo, D. & Child, M.F.
Assessment Rationale
This is a woodland and forest habitat species occurring widely but sparsely within the eastern parts of the assessment region. It qualifies as Vulnerable C2a(i) as the regional population is suspected to consist of fewer than 10,000 mature individuals, with no subpopulation having more than 1,000 mature individuals. Since this species has low wing loading, we assume that subpopulations are fragmented with limited dispersal between localities. The population is declining due to loss of habitat as a result of the re-mining of existing adits, and loss and degradation of woodland habitat.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Taylor P, Richards LR, Balona J, Howard A & da Silva JM. 2025. A conservation assessment of Rhinolophus swinnyi. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Within the assessment region, this species has been found only in KwaZulu-Natal and Eastern Cape provinces and is endemic to the assessment region (Monadjem et al. 2020). The extent of occurrence is 129,099 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 81 m asl (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 1,588 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
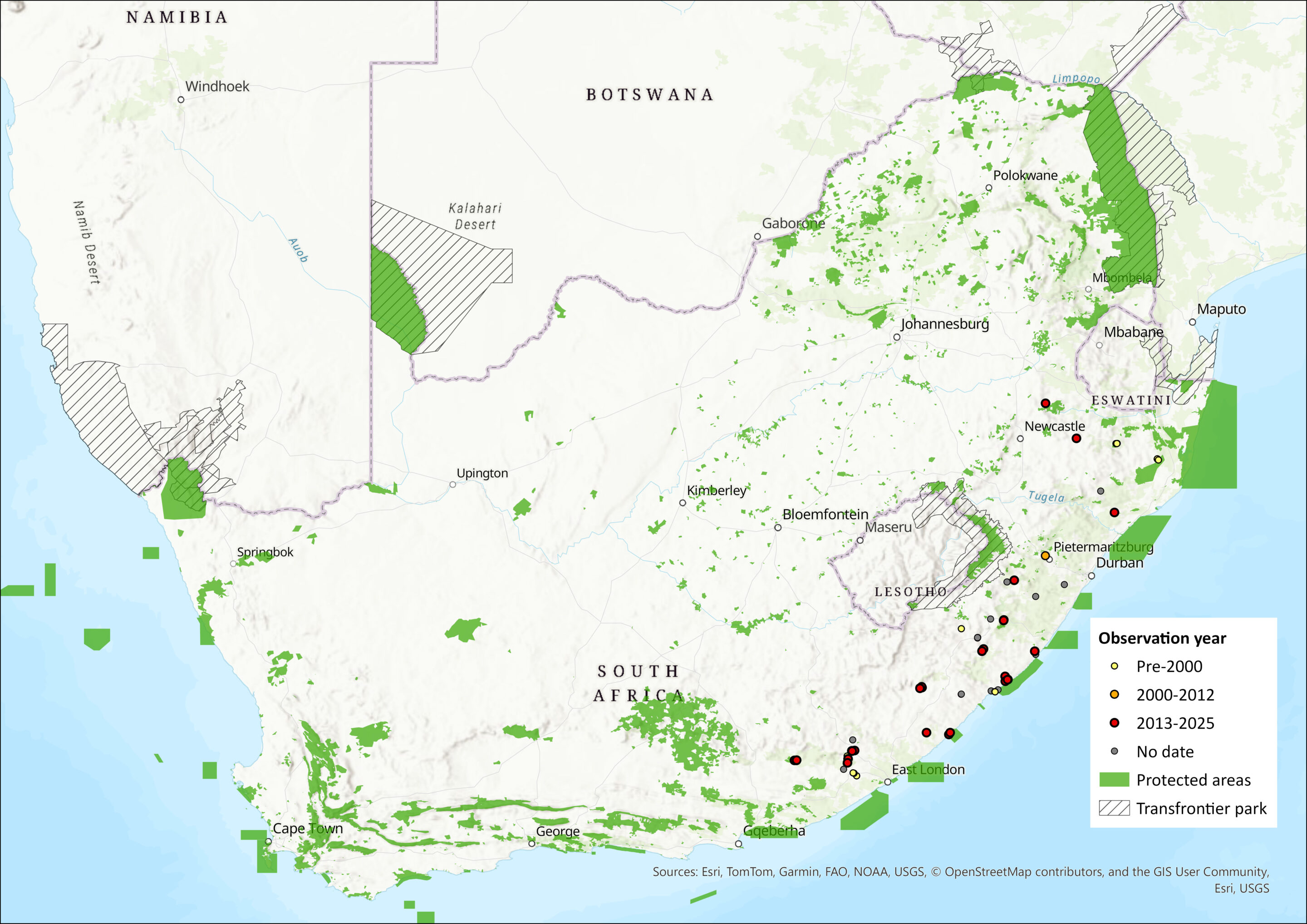 Figure 1. Distribution records for Swinny’s Horseshoe Bat (Rhinolophus swinnyi) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Swinny’s Horseshoe Bat (Rhinolophus swinnyi) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
A study by Taylor et al. (2024) reported minimal range changes for this species under future climate models by suggesting the high-elevation mountains provide a potential refugia for temperate montane species.
Most studies on the effects of climate change on bats are based on predictive species distribution modelling, thus there is a lack of empirical studies measuring behavioural, physiological, phenological or genetic responses to extreme and seasonal climatic changes, especially in the Global South (Festa et al. 2023; Pio et al. 2014). Globally, there have been documented declines in bat populations, species richness and distributions in relation to water availability with increasing global aridity, which may become a growing concern as heat waves and maximum temperatures are expected to increase over much of South Africa (Adams & Hayes 2021; Mbokodo et al. 2020).
Population information
This species is relatively widely and sparsely distributed across the eastern regions of South Africa and is known to roost singly or in small groups of up to five individuals (Monadjem et. al. 2020). The overall population size within the assessment region is likely to be less than 10,000. Individuals of this species were captured in Ngeli State Forest, Gomo State Forest, Baziya State Forest, Kologha State Forest, Fort Fordyce Nature Reserve, Mbotyi State Forest and Dwesa Nature Reserve. This species was acoustically detected in Xumeni State Forest, Umtamvuna Nature Reserve, and Silaka Nature Reserve (Moir et al. 2020a).
Current population trend: Declining
Continuing decline in mature individuals: Yes, inferred.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: 1,000
Number of Subpopulations: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Phylogenetic studies have been conducted involving this species (e.g., Moir et al. 2020b, Taylor et al. 2024), with little to no genetic structure detected across the sample localities based on two mitochondrial markers (Moir et al. 2020b). Based on this information, it would indicate the species exists as a single subpopulation within the region, at least historically. However, there is evidence of differentiation of KwaZulu-Natal (Eshowe to Kokstad) and coastal localities (Dwesa and Mbotyi) from other central localities, suggestive of reduced gene flow between these areas (Moir et al. 2020b). The species is considered to have limited dispersal capability, and given that the species is forest-dependent, it is possible that further population structure has gone undetected. A more comprehensive study incorporating nuclear markers is highly recommended to investigate this. Such a study would also prove valuable for obtaining an estimate of the effective population sizes (Ne) for each genetically distinct group.
Currently, the only approximate measure has been estimated by Moir et al. (2020). Estimates of Ne dating from ~70,000 years based on Bayesian Skyline plots indicates that there has been an increase in Ne over time, and while the results in the study approximate current Ne at approximately 600,000 individuals (~CI: 300,000 – 4,000,000), caution must be taken in extrapolating contemporary Ne based on analyses designed to measure historical processes. Broad-based mitochondrial markers, which are generally accepted as most useful for inferring patterns (e.g., phylogenies), are generally insufficient for inferring processes shaping population history due to influences from selection, sex-biased dispersal, mutation rates and sampling (Zink & Barrowclough 2008; Edwards & Bensch 2009; Schmidt & Garroway 2021); and hence, may not accurately quantify contemporary genetic structure and abundance (see Heller et al. 2013). Based on assumptions of the species numbering < 10000 individuals in the assessment region, the best approximation for Ne is <1000 (1,000 – 3,000; based on a Ne/Nc conversion ratio of 0.1-0.3).
Habitats and ecology
This species is found in temperate Afromontane forests (Monadjem et al. 2020). For example, Roberts (1951) recorded it in the Pirie Forest, Eastern Cape Province. The distribution of R. swinnyi is dependent on the availability of suitable shelter in which to roost during the day (Skinner & Chimimba 2005) as well as suitable wooded/forested habitat for foraging (Bronner 1990). It is dependent on caves, mines and similar habitats for roosting. It appears to be sparsely distributed in the assessment area, the species is recorded from the Mesic Highveld Grassland, Lowveld, Sub-Escarpment Grassland, Indian Ocean Coastal Belt, Sub-Escarpment Savanna and Drakensberg Grassland.
R. swinnyi is a clutter forager requiring dense vegetation where it primarily preys on Lepidoptera species (Monadjem et al. 2020). Females have been recorded to give birth to a single young (Monadjem et al. 2020)
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Suitable | – |
| 7.1. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Caves | – | Suitable | – |
| 7.2. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Other Subterranean Habitats | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: Unknown
Average Reproductive Age: Unknown
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: Unknown
Reproductive Periodicity: Unknown
Average Annual Fecundity or Litter Size: One
Natural Mortality: Unknown
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Populations may be locally threatened by the use of timber and firewood, and the general conversion of land to agricultural use (ACR 2015). Re-mining of old mine adits is a significant threat in the assessment region, as it results in the loss of suitable habitat for this species. Implementation of the Mine Closure Strategy of the South African Department of Mineral Resources and Energy (Government Notice 446, 21 May 2021) poses a serious threat to colonies of this species.
Conservation
In the assessment area, the species is present in the protected areas Fort Fordyce Nature Reserve, Dwesa Nature Reserve, Umtamvuna Nature Reserve, and Silaka Nature Reserve (Moir et al. 2020a).
Key interventions for this species include protection of known roost sites through appropriate management to limit disturbance, and protected area expansion to incorporate further subpopulations.
Recommendations for land managers and practitioners:
- Report discoveries of new roost sites.
- Reduce logging and habitat conversion within the species’ range
Research priorities:
- Further field surveys to discover new roost sites to inform protected area expansion.
- Monitoring of known subpopulations to establish population trends.
Encouraged citizen actions:
- Limit disturbance to roost sites.
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
Bibliography
ACR. 2015. African Chiroptera Report 2015. Pretoria, South Africa.
Adams, R.A. and Hayes, M.A. 2021. The importance of water availability to bats: climate warming and increasing global aridity. 50 years of bat research: foundations and new frontiers, pp.105-120.
Bronner, G.N. 1990. New distribution records for four mammal species, with notes on their taxonomy and ecology. Koedoe 33: 1–7.
Csorba, G.P., Ujhelyi, P. and Thomas, N. 2003. Horseshoe Bats of the World. Alana Books, Shropshire, England.
Edwards, S. and Bensch, S. 2009. Looking forwards or looking backwards in avian phylogeography? A comment on. Molecular ecology, 18(14).
Festa, F., Ancillotto, L., Santini, L., Pacifici, M., Rocha, R., Toshkova, N., Amorim, F., Benítez‐López, A., Domer, A., Hamidović, D. and Kramer‐Schadt, S. 2023. Bat responses to climate change: a systematic review. Biological Reviews, 98(1), pp.19-33.
Mbokodo, I., Bopape, M.J., Chikoore, H., Engelbrecht, F. and Nethengwe, N. 2020. Heatwaves in the future warmer climate of South Africa. Atmosphere, 11(7), p.712.
Moir, M., Richards, L.R.R., Rambau, R.V. and Cherry, M.I. 2020a. Bats of Eastern Cape and southern KwaZulu-Natal forests, South Africa: diversity, call library and range extensions. Acta Chiropterologica, 22(2):365-381
Moir, M., Richards, L.R.R., Cherry, M.I. and Rambau, R.V. 2020b. Demographic responses of forest-utilizing bats to past climate change in South Africa. Biological Journal of the Linnean Society, 130(4): 850-868.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis, 2nd Ed. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Pio, D.V., Engler, R., Linder, H.P., Monadjem, A., Cotterill, F.P., Taylor, P.J., Schoeman, M.C., Price, B.W., Villet, M.H., Eick, G. and Salamin, N. 2014. Climate change effects on animal and plant phylogenetic diversity in southern Africa. Global Change Biology, 20(5), pp.1538-1549.
Roberts, A. 1951.The Mammals of South Africa. The Trustees of the Mammals of South Africa. Central News Agency, Johannesburg, South Africa.
Schmidt, C. and Garroway, C.J. 2021. The conservation utility of mitochondrial genetic diversity in macrogenetic research. Conservation Genetics, 22(3), pp.323-327.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Stofferg, S. Submitted. Molecular phylogenetics and the evolution of high-frequency echolocation in horsheshoe bats (Genus Rhinolophus). University of Cape Town.
Taylor, P.J. 2000. Bats of Southern Africa: Guide to Biology, Identification, and Conservation. University of Natal Press, Pietermaritzburg, South Africa.
Taylor, P.J., MacDonald, A., Goodman, S.M., Kearney, T., Cotterill, F.P.D., Stoffberg S., Mondajem, A., Schoeman, M.C., Guyton, J., Naskrecki, P. and Ricahrds L.R. 2018. Integrative taxonomy resolves three new cryptic species of small southern African horseshoe bats (Rhinolophus). Zoological Journal of the Linnean Society, 184: 1249-1276.
Zink, R.M. and Barrowclough, G.F. 2008. Mitochondrial DNA under siege in avian phylogeography. Molecular ecology, 17(9), pp.2107-2121.
