Suni
Nesotragus moschatus

2025 Red list status
Endangered
Regional Population Trend
Declining
Change compared
to 2016
No Change
Overview
Nesotragus moschatus – Von Dueben, 1846
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Nesotragus – moschatus
Common Names: Suni (English), Soenie (Afrikaans), Nhlengane (Swati, Tsonga, Zulu)
Synonyms: Neotragus moschatus (Von Dueben, 1846)
Taxonomic Note:
There are two subspecies of the Suni within southern Africa (Meester et al. 1986); Neotragus moschatus livingstonianus (Kirk 1865) from northeastern Zimbabwe northward, and Neotragus moschatus zuluensis (Thomas 1898) from northern KwaZulu-Natal Province (KZN) South Africa, southern Mozambique and south-eastern Zimbabwe. Although individuals from different regions of Africa are phenotypically indistinguishable, cytogenetic differences between N. m. akeleyi (from Kenya) and N. m. zuluensis indicate that they should be recognised as distinct management units (Kingswood et al. 1998). Globally, five subspecies have been described, but the boundaries between forms are not clearly delineated (IUCN SSC Antelope Specialist Group 2016).
Red List Status: EN – Endangered, A2b; B2ab(ii,iii,v); C2a(i) (IUCN version 3.1)
Assessment Information
Assessors: Streicher, J.P.1,2,3, Ramesh, T.4 & da Silva, J.M.1
Reviewer: Downs, C.T.3
Institutions: 1South African National Biodiversity Institute, 2BirdLife South Africa, 3University of KwaZulu-Natal, 4Sálim Ali Centre for Ornithology and Natural History, Coimbatore, India
Previous Assessors: Hunnicutt, A., Matthews, W. & Somers, M.J.
Previous Reviewers: Hanekom, C. & Child, M.F.
Previous Contributors: Roxburgh, L., Kyle, S., Page-Nicholson, S. & Mulqueeny, C.
Assessment Rationale
The Suni is a woodland and thicket dwelling (especially sand forest) species on the edge of its range within the assessment region, occurring within northern KZN. Although anecdotal reports suggest Suni presence in northeastern Kruger National Park, no recent records confirm this. The current extent of occurrence (EOO) based on confirmed records is estimated to be 6,807 km2 and the area of occupancy (AOO) is estimated to be 476 km2. This is likely to be an overestimate as not all forest patches, especially in the matrix between protected areas, will be occupied because of poaching pressure and human disturbance. Natural habitat in KZN was lost at an estimated rate of 1.2% per annum between 1994 and 2011. Similarly, it is suspected that there is an ongoing loss of mature individuals from illegal poaching for bushmeat and sport hunting with dogs, especially around protected area edges and in private or communal lands. Between 2000 and 2013, there was a 5.6% and 1.1% rate of urban and rural expansion in KZN, respectively, which is considered as a proxy for the latter threats. A significant threat to this species is increasing Nyala (Tragelaphus angasii), African Elephant (Loxodonta africana), and other sympatric browser density that denude the shrub layer necessary for Suni persistence.
Population size is difficult to determine because of the Suni’s shy nature and patchy occurrence, but is estimated to range between 440–4,890 individuals (220–2,445 mature individuals using a 50% mature population structure). The largest subpopulation (based on forest fragments) is estimated to range between 62–750 mature individuals. There has been a genuine decline in the largest historical subpopulation, Tembe Elephant Park: faecal pellet-based abundance in 1984 was 3.58 middens / 100 m but only 0.54 middens / 100 m in 2013, which corresponds to a > 50% population decline in the formerly largest subpopulation in the assessment region over more than three generations (14 years). Thus, Suni are listed as Endangered A2b, based on an estimated decline in the protected stronghold of the species; B2ab(ii,iii,v) considering its restricted AOO and ongoing loss of suitable habitat, loss of mature individuals from poaching and loss of habitat quality from increased browser densities; and C2a(i) using the lower end of population and subpopulation size ranges. However, further subpopulation surveys are necessary to corroborate this listing by quantifying overall population size and trends more accurately. Key interventions for this species include: better management of African Elephant and Nyala populations which involves de-stocking Nyala on private land; management of predator numbers; preservation of understory native vegetation for maintaining the structure and function of forests; reintroduction or supplementation from captive-stock into the suitable habitat patches; protected area expansion, either formally or through biodiversity stewardship schemes; involvement of local communities in suni conservation in sensitive edge forested habitat; and increased training of protected area managers and subsequent prosecution of protected area transgressors.
Regional population effects: Although there are potential dispersal corridors between northern KZN and southern Mozambique through the Lubombo Transfrontier Conservation Area, especially through Tembe Elephant Park and Ndumo Game Reserve to the Maputo Special Reserve, high poaching pressure in the matrix between protected areas is suspected to repress the establishment of dispersers, especially around the Kosi Bay area. Furthermore, anecdotal evidence suggests that poaching pressure may be so severe in southern Mozambique that dispersal rates into South Africa may decrease. Until research has demonstrated otherwise, the regional criteria were not applied as no significant rescue effects are suspected.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Streicher JP, Ramesh T & da Silva JM. 2025. A conservation assessment of Nesotragus moschatus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Globally, Suni occur along the eastern part of the African continent in thickets and coastal regions from Kenya to KZN, with their southerly limit being around Lake St. Lucia (IUCN SSC Antelope Specialist Group 2016). They are associated with evergreen vegetation consisting of closed-canopy woodland with shrub cover and thickets in coastal regions and associated hinterlands, stretching from Kenya to KZN, where their southerly limit is around Lake St. Lucia in northeastern KZN (c. 28°S) (Skinner & Chimimba 2005).
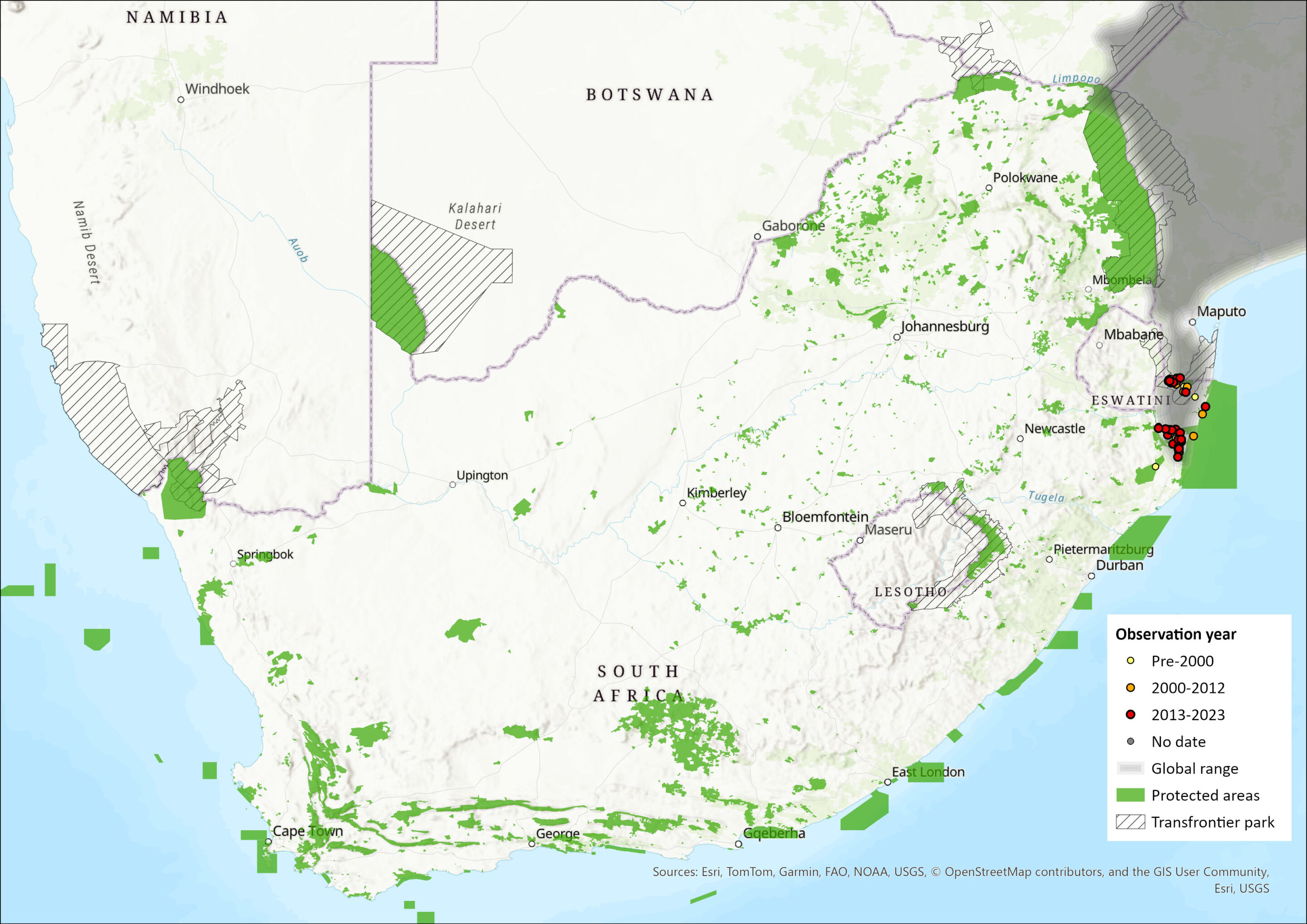
Within the assessment region, which is the southern edge of its global range, Suni occur in woodlands, thickets and forests, especially Sand Forests in Maputaland (van Eeden 2006; Belton et al. 2008; Ramesh et al. 2016), in the northeastern parts of KZN. Sand Forests and thicket habitats have been lost to agricultural, urban and rural sprawl, and thus, these habitat types are highly fragmented, which makes the Suni population subject to edge effects and isolation. Suni occur in protected areas, such as Tembe Elephant Park and Ndumo Game Reserve, and in some coastal areas, such as sections of the iSimangaliso World Heritage Site, including False Bay Park and Mkhuze Game Reserve, as well as on private lands. Although recent records in Kosi Bay are confirmed (c. 10 individuals), poaching pressure continues to cause a decline in the area (S. Kyle pers. comm. 2015). There are reports that Suni occur in northeastern parts of Kruger National Park, Mpumalanga Province, and Pongola Nature Reserve, KZN (Skinner & Chimimba 2005), but no recent records confirm this. Suni are also likely to occur in Eswatini, such as on the east-facing slopes of the Lubombo Mountains or in Milwane Wildlife Sanctuary, although their occurrence is not confirmed (Monadjem 1998). There are suspected to be very few Suni left in southern Mozambique because of high poaching pressure (S. Kyle pers. comm. 2015), so immigration into South Africa is increasingly unlikely. There have, however, been sightings of Suni in Maputo Special Reserve in recent years, although these remain infrequent (C. Hanekom pers. comm. 2015).
The estimated extent of occurrence (EOO), including northeastern Kruger National Park, of Suni is 10,778 km2. If the EOO is calculated based on confirmed records only (i.e., excluding northern Kruger National Park), it is 6,807 km2. The AOO calculated using the IUCN method of occurrence within a 2 by 2 km grid yields an AOO of 224 km2, using all occurrence records, or an AOO of 180 km2 using occurrence records from 2000 to 2024. Using all forest patches as a proxy for AOO yields 476 km2 (using the 2018 National Vegetation Map, SANBI). Of 1930 forest patches within the EOO, the average size is 0.25 ± 1.6 km2, with a maximum size of 48 km2 (Northern Coastal Forest). The largest protected area in which Suni occurs is Tembe Elephant Park (c. 300 km2), of which an estimated 65% (195 km2) comprises suitable thicket woodland habitat (W. Matthews & C. Hanekom unpubl. data).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Suni (Nesotragus moschatus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Eswatini | Presence Uncertain | Native | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
According to Matthews (2001), once adversely affected, Sand Forest exhibits little to no capacity for recovery. Given that climate change negatively impacts Sand Forest distribution (Gaugris et al. 2008) and given Suni’s predilection for Sand Forests (Ramesh et al. 2016), this may impact the species range distribution and its populations negatively in the future.
Population information
Globally, the Suni population is estimated at c. 365,000 animals (IUCN SSC Antelope Specialist Group 2016). Within the assessment region, it occurs in fragmented and low-density subpopulations. While Suni occurs in higher densities throughout the rest of its African range, such as 13–17 individuals/2 in Zanzibar Island (Tanzania) and Lengwe National Park (Malawi) (IUCN SSC Antelope Specialist Group 2016), densities in South Africa are typically lower. For example, 0.9 individuals/2 in Mkhuze Game Reserve (East 1999). Very few individuals are suspected to occur between Kosi Bay and Lake St. Lucia (S. Kyle pers. comm. 2015).
Estimating the population size of Suni is difficult because of their secretive and shy nature. During 2013/2014, 641 individuals were estimated to occur in four Ezemvelo-KZN Wildlife (EKZNW) nature reserves (I.unpubl. data.): 393 in Mkhuze Game Reserve, 80 in Makhasa Nature Reserve, 158 in False Bay Park, and 10 in the Coastal Forest Reserve. Hunnicutt et al. (unpubl. data) estimate total population numbers at < 3,000 individuals based on changes in faecal pellet counts and available habitat. Considering social groups consist of two adults and one or two juveniles, we assume a 50% mature population structure, which yields c. 1,500 mature individuals. Similarly, by extrapolating a density range of 0.9–10 individuals/2 across the AOO (489 km2), the Suni population size is estimated to be 440–4,890 individuals (220–2,445 mature individuals). If we use forest patches as a proxy for localities suitable for Suni, the largest fragment is 137 km2, which yields a range of 62–685 mature individuals. Historically, the largest protected site was Tembe Elephant Park in KZN, which was estimated at 3,000 individuals in the late 1980s (Lawson 1986). However, recent research indicates a significant decline, with numbers likely to be fewer than 1,500 individuals (750 mature individuals) (A. Hunnicutt unpubl. data), and could be even more severe a decline based on anecdotal encounter rates (W. Matthews pers. obs. 2015, Ramesh et al. 2016). Faecal pellet-based abundance in 1984 was 3.58 middens/m in Tembe Elephant Park (Lawson 1986), but was only 0.54 middens/m in 2013 (A. Hunnicutt unpubl. data). Generation length has been calculated as 4.8 (Pacifici et al. 2013), which yields a 14-year three-generation period (1999–2013). Thus, there has been an estimated > 50% population decline in Tembe Elephant Park over more than three generations. Similarly, game counts at Mkhuze Game Reserve show an estimated decline from 550 Suni in 2008 to 393 in 2014 (A. Hunnicutt unpubl. data). Current numbers at Phinda Private Game Reserve are low as it was found to have an abundance rating of 0.22 middens/m (A. unpubl. data). More positively, numbers in False Bay Park have been stable over three generations (1999: 126 animals; 2014: 159 animals; C. Mulqueeny unpubl. data). In Ndumo Game Reserve, numbers may have started to increase in recent years with vegetation recovery following the reduction in abundance of game species such as Nyala (C. Hanekom unpubl. data).
The decline in Tembe Elephant Park is likely a result of multiple factors: increasing African Elephant density (habitat alteration leading to a decline in the density of shrubs), increasing Nyala density (habitat alteration and resource competition), and predation from high Lion (Panthera leo) density and introduced Wild Dogs (Lycaon pictus), although whether Wild Dogs are a threat is debatable (C. Hanekom unpubl. data). However, in Mkhuze Game Reserve, with possibly the second largest Suni subpopulation in a protected area, a recent reintroduction of Wild Dogs and Lion has added markedly to the predation pressure on Suni. These pressures are not unique to Tembe and Mkhuze and may exist in other reserves with Suni. More research is urgently needed to assess population trends in other protected areas.
Due to their small home range size and low dispersal rates, Suni are subject to considerable fragmentation with limited gene flow between patches. Habitat in the region is becoming increasingly fragmented with anthropogenic changing land use (e.g., residential and industrial development) which disconnects forest patches (Jewitt et al. 2015).
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| Yes | – | Suspected from snaring, especially on the edge of protected areas. There is also habitat transformation within and outside protected areas where Suni occur. |
Current population trend: Declining. Based on scat-based abundance in Tembe Elephant Park and game counts in Mkhuze Game Reserve.
Number of mature individuals in population: 220–2,445
Number of mature individuals in largest subpopulation: 62–750
Number of subpopulations: c. 5
Severely fragmented: Yes. Forest habitat is fragmented and isolated.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
A comprehensive population genomic study has not been conducted on this species, even within the assessment region. However, a study incorporating a mitochondrial marker and five nuclear microsatellite loci, was able to uncover some structure amongst three reserves in South Africa (Swanepoel 2019). Animals from Tembe and Ndumu were found to be genetically similar, and slightly differentiated from those in Phinda Nature Reserve; however, low levels of genetic diversity were observed throughout. While the total number of subpopulations cannot be inferred from this information, their small home ranges and low dispersal abilities likely suggest the potential for additional genetically distinct subpopulations to exist. Considering the total population size for the assessment region is estimated between 440–4,890 (with the higher end likely to be an overestimate based on current estimates of decline; see Population Section), it is unlikely that any property/reserve/subpopulation will be able to sustain sufficient levels of genetic diversity in the long term. This is based on estimated effective population size for each subpopulation to be less than 500 (based on a Ne/Nc conversion ratio of 0.1-0.3 and a maximum Nc of 4890 for the total population).
Habitats and ecology
Within the assessment region, Suni prefer vegetation types with high undergrowth stem density for both predation evasion and access to foodstuffs (Lawson 1986; Belton et al. 2008). In northern KZN, they occur in dry woodland, bushveld and thickets on sand or clay soils (Rowe-Rowe 1994). The highest Suni densities in Sand Forest (van Eeden 2006), but they also occur in sandveld woodland thickets and riparian woodlands. In some areas within their distribution range, they may benefit from the expansion of secondary thicket habitat associated with human activities (IUCN SSC Antelope Specialist Group 2016). Suni showed strong single-habitat preference (Sand Forest) with a habitat occupancy of 60%, followed by Makatini clay thicket (34%), woodland (21%), dry forest thicket (20%) in in the Maputaland Conservation Unit of South Africa (Ramesh et al. 2016). Similar preference for Sand Forest and woodland habitat preference was identified in a repeat study (Streicher et al. 2025).
Suni occur as solitary adults (77%), in pairs (12%) or in families consisting of a male, female and offspring (Lawson 1986). They are shy and, if disturbed, they freeze before jumping away into the nearest thicket (Skinner & Chimimba 2005). Suni have small territories: Lawson (1986) recorded male territories as 0.005–0.011km2, with little overlap, and females as 0.009–0.046km2, almost entirely overlapping within male territories. Males reach maturity at eight months; females at 12 months. Inter-calving interval is approximately seven months and the generation length is four years.
Suni cannot digest cellulose properly, and feed primarily on leaves, with a preference for freshly fallen leaves, and they also eat wild fruits, flowers and tips of shoots (Lawson 1986). They have also been observed to feed on mushrooms (Heinichen 1972).
Ecosystem and cultural services: Suni are a flagship species for northern KZN’s threatened Sand Forests. They are also an important food source for Crowned Eagles (Stephanoaetus coronatus), Martial Eagles (Polemaetus bellicosus) and Southern African Pythons (Python sebae).
“Nhlegane” is the Zulu name for the Suni and means “why is it beautiful?”. According to Northern Maputaland folklore this came from King Shaka who declared the Suni the most beautiful animal in his forest.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist | – | Suitable | – |
| 14.6. Artificial/Terrestrial -> Artificial/Terrestrial – Subtropical/Tropical Heavily Degraded Former Forest | – | Marginal | – |
Life History
| Generation Length | Justification | Data Quality |
| 4.8 | – | – |
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Suni are used on a subsistence level as bushmeat, which may lead to localised declines. Trophy hunting is also seen in parts of its distribution range, including South Africa, but no information is available on the numbers hunted or impacts on the population. More research should be undertaken to determine possible sustainable offtake rates. Small subpopulations exist on private game farms and conservancies, but are likely fragmented in that they seldom experience gene flow with each other or larger protected subpopulations.
The private sector may have generally had a positive effect on this species as it has been reintroduced onto private properties within its natural distribution. However, private landowners often overstock Nyala, which can reduce the shrub cover necessary for this species (Lawson 1986; Coates & Downs 2005; Belton et al. 2008; Lagendijk et al. 2012). With recent increases in Nyala prices, there is potential for ranches to increase their Nyala densities, which may have adverse effects on Suni overall.
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
Yes |
Anecdotal observations of poaching, also compliance incidents recorded by EKZNW staff. |
Yes |
Anecdotal reports of trophy hunting on private lands. |
National Commercial Value: Yes
International Commercial Value: No
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
1. Food – human |
true |
true |
– |
– |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: (Not specified)
Threats
Historically, the greatest threat to Suni was habitat loss from both agricultural expansion, human settlement expansion and large herbivore impacts, which left their preferred habitat fragmented and their subpopulations isolated. Although suitable habitats of northern KZN are currently well-protected, habitat loss continues, especially through human settlement expansion (see below). Currently, the major threats affecting Suni are:
Bushmeat poaching: Anecdotal observations suggest high mortality rates of Suni from snares, even within protected areas. Bushmeat poaching may be increasing, especially along protected area edges where human densities tend to be highest (Wittemyer et al. 2008). Because they use pathways through dense underbrush, they are easily snared, and poaching is suspected to lead to higher mortality than natural predation (Skinner & Chimimba 2005). Lawson (1986) observed that in northern KZN, Suni had disappeared outside protected areas from habitat loss and overhunting. On several farms, particularly in the Mkhuze area, new landowners have opened their land to bushmeat and traditional medicine hunting.
Sport hunting: Illegal hunting with domestic dogs (Canis familiaris) for sport or gambling is similarly suspected of causing high mortality rates (sensu Grey-Ross et al. 2010), especially on private or communal lands in the matrix between protected areas.
Continuing loss of habitat: Current habitat loss, especially from urban and rural expansion (see below), contributes to further fragmentation of the Suni population, lowering gene flow rates and thus reducing the resilience of the population to environmental changes.
High African Elephant and Nyala densities: High or artificially inflated African Elephant and Nyala abundance reduce the shrub cover needed by Suni and consequently have a negative relationship with Suni abundance (Lawson 1986; Belton et al. 2008; Lagendijk et al. 2012). In many areas, the increase in Nyala, particularly on private lands, has been suggested to contribute to local Suni extinctions, which has similarly been shown to impact Bushbuck (Tragelaphus sylvaticus) abundance (Coates & Downs 2005). Similarly, increased Sand Forest disturbance (likely by sympatric browsers), which affects forest structure, negatively affects Suni presence (Belton et al. 2008; Lagendijk et al. 2012; A. Hunnicutt unpubl. data).
Loss of habitat quality from resource extraction: Firewood collection and charcoal production in and near forests are likely to decrease habitat quality for the species by removing shrub cover, and also decreasing the effective AOO.
Current habitat trend: Declining. Habitat for Suni has decreased in quality because of anthropogenic factors, including human habitation, slash and burn agricultural practices, and resource extraction from forests and woodlands (for example, removal of small stems and undergrowth for fuelwood and building materials). This is particularly the case in northern Maputaland. Within protected areas, the transformation of habitat structure and quality is potentially increasing from the high densities of African Elephants and Nyala.
Between 2000 and 2013, there was a 5.6% and 1.1% rate of urban and rural expansion in KZN, respectively (GeoTerraImage 2015), which we are inferred to be increasing rates of habitat degradation. Recent satellite imagery data confirm an ongoing loss of forest habitat within the assessment region between 2000 and 2014 (A. Skowno unpubl. data). As Suni are at their highest densities in Sand Forest (van Eeden 2006), the loss of this forest type will be especially detrimental to the population. Overall, there was a 20.4% loss of natural habitat in KZN from 1994 to 2011, with an average loss of 1.2% per annum (Jewitt et al. 2015). If this rate of loss continues , there will be an estimated 14.4% loss of habitat over the next three generations.
Conservation
Important protected Suni subpopulations in KZN occur in Tembe Elephant Park (largest subpopulation and a source for immigrants and/or dispersal), Mkhuze Game Reserve, Phinda Private Game Reserve , Ndumo Game Reserve, and False Bay Park, St. Lucia. They may marginally occur in Kruger National Park, but no recent records are available. The most important interventions are as follows:
Protected area expansion: To conserve and consolidate remaining Sand Forest patches and other suitable habitats into a connected metapopulation. For example, in Maputaland, a new transfrontier landscape is being proposed that will, amongst other species, presumably benefit Suni by extending the existing Lubombo Transfrontier Conservation Area to the south and east of Tembe Elephant Park and the west of Ndumo Game Reserve (Smith et al. 2008).
Biodiversity stewardship schemes: To conserve suitable Sand Forest patches and other suitable habitats outside protected areas and prevent ongoing habitat loss or disturbance to such sites. Similarly, stewards and private landowners should be incentivised to manage their African Elephant and Nyala to support healthy Suni subpopulations.
Training: Protected area managers and governmental agencies need further training and capacity development to monitor and enforce regulations on illegal development, timber harvesting and dog hunting. This will minimise mortality and disturbance to Suni.
Reintroduction: Reintroduction and augmentation of existing subpopulations from captive-bred stock could help create demographically and ecologically self-sustaining subpopulations and increase the overall AOO (this needs further investigation). Suni breed well in captivity, and captive-bred individuals can be used as a source population. For example, a Suni breeding project (currently not active) was initiated in Punda Maria, Kruger National Park, in 1989, to gain information on the species in captivity (Pretorius et al. 1996). However, reintroduction methods and attempts need further research and documentation, as a previous attempt was unsuccessful: in 1995, 39 captive-bred individuals were reintroduced into northeastern Kruger National Park, but no individuals could be found by 1998 (East 1999). Another translocation to Kruger National Park from Tembe Elephant Park yielded high mortality for the captured individuals, thus safer capture methods should be employed for future translocations. Furthermore, research showed that the subspecies N. m. zuluensis should be managed as a separate conservation unit (Kingswood et al. 1998), and thus the introduction of extra-limital subspecies to augment existing subpopulations within the assessment region is discouraged. Suni have small home ranges, which may make them suitable for reintroduction into private conservancies.
Recommendations for land managers and practitioners:
- Captive breeding projects can be used to supplement existing subpopulations or reintroduce subpopulations to areas within the natural range. However, captive subpopulations should not mix alien subspecies with N. m. zuluensis. Evidence from captive-bred individuals indicates higher perinatal mortality in subspecies hybrids (Kingswood et al. 1998). Recording the success or failure of any reintroduction event is crucial to create an evidence base.
- Stocking levels of Nyala and African Elephant may adversely affect Suni, thus both species should be kept at stocking densities. Similarly, monitoring the effects of African Elephants on habitats inhabited by Suni is required.
- Systematic Suni subpopulation monitoring is required to estimate population size and trends, as well as current AOO patterns better.
Research priorities:
An abundance index based on faecal pellet counts was initiated. The project plans to collect abundance data annually at Tembe Elephant Park and Phinda Private Game Reserve to compile an index for Suni. This project could increase its impact and effectiveness by sampling from more reserves where Suni are thought to be present. Further research priorities include:
- Surveys of Suni subpopulations to generate accurate subpopulation size estimates.
- Studies investigating the rate of dispersal of individuals between South Africa and Mozambique.
- Studies generating information on ranching and commercial utilisation of this species, as well as the potential for reintroduction into small private reserves or conservancies.
- Studies to further understand Suni diet and relationship with sympatric foragers, as well as the risk of predation by introduced carnivores.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Reduce Nyala on private properties to keep stocking rates low.
Bibliography
Belton LE, Dalerum F, van Rensburg BJ. 2008. Factors associated with suni distribution in Tembe Elephant Park, South Africa: implications for management and conservation of a small antelope. African Journal of Ecology 46: 631-636.
Coates GD, Downs CT. 2005. Survey of the status and management of sympatric bushbuck and nyala in KwaZulu-Natal, South Africa. South African Journal of Wildlife Research 35: 179.
East, R. (compiler). 1999. African Antelope Database 1998. IUCN, Gland, Switzerland and Cambridge, UK.
Gaugris, J. Y., Van Rooyen, M. W., and Bothma, J. d. P. 2008. Growth rate of selected woody species in northern Maputaland, KwaZulu–Natal, South Africa. South African Journal of Botany, 74(1), 85-92. https://doi.org/https://doi.org/10.1016/j.sajb.2007.09.001
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Grey-Ross R, Downs CT, Kirkman K. 2010. An assessment of illegal hunting on farmland in KwaZulu-Natal, South Africa: implications for oribi (Ourebia ourebi) conservation. South African Journal of Wildlife Research 40: 43-52.
Heinichen IG. 1972. Preliminary notes on the suni (Neotragus moschatus) and the red duiker (Cephalophus natalensis). Zoologica Africana 7: 157-165.
IUCN SSC Antelope Specialist Group. 2016. Nesotragus moschatus. The IUCN Red List of Threatened Species 2016: e.T14604A50191073. Available at: http://dx.doi.org/10.2305/IUCN.UK.2016-1.RLTS.T14604A50191073.en.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Kingswood SC, Kumamoto AT, Charter SJ, Jones ML. 1998. Cryptic chromosomal variation in suni Neotragus moschatus (Artiodactyla, Bovidae). Animal Conservation 1: 95-100.
Lagendijk G, Page BR, Slotow R. 2012. Short-term effects of single species browsing release by different-sized herbivores on Sand Forest vegetation community, South Africa. Biotropica 44: 63-72.
Lawson D. 1986. The ecology and conservation of Suni in Natal. Ph.D Thesis. University of KwaZulu-Natal, Pietermaritzburg, South Africa.
Matthews, W. S., van Wyk, A. E., van Rooyen, N., and Botha, G. A. 2001. Vegetation of the Tembe Elephant Park, Maputaland, South Africa. South African Journal of Botany, 67(4), 573-594. https://doi.org/https://doi.org/10.1016/S0254-6299(15)31188-1
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum , Pretoria, South Africa.
Monadjem, A. 1998. Mammals of Swaziland. The Conservation Trust of Swaziland and Big Game Parks.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Pretorius Q, Pretorius BP, Dannhauser CS. 1996. The reproductive behaviour of the suni Neotragus moschatus zuluensis in captivity. Koedoe 39: 123-126.
Ramesh, T., Kalle, R., Rosenlund, H., and Downs, C. T. 2016. Native habitat and protected area size matters: Preserving mammalian assemblages in the Maputaland Conservation Unit of South Africa. Forest Ecology and Management, 360, 20-29. https://doi.org/https://doi.org/10.1016/j.foreco.2015.10.005
Rowe-Rowe, D. T. 1994. The ungulates of Natal. Natal Parks Board, Pietermaritzburg, South Africa.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Streicher, J.P., Ramesh, T. & Downs, C.T. 2025. A long-term reinvestigation using camera traps reveals the resilience of mammalian communities in protected areas of the Maputaland Conservation Unit, South Africa. Landscape Ecology 40: 141.
Swanepoel, M. 2019. MSc Thesis. Conservation genetics of the suni antelope Nesotragus moschatus von Deuben, 1846. University of Pretoria, Pretoria.
Wittemyer, G., Elsen, P., Bean, W.T., Burton, A.C.O. and Brashares, J.S. 2008. Accelerated human population growth at protected area edges. Science 321: 123-126.
van Eeden DG. 2006. Aspects of the ecology and conservation status of selected wildlife in and around Tembe Elephant Park, KwaZulu-Natal, South Africa. M.Sc. Thesis. University of Pretoria, Pretoria, South Africa.