
Sundevall’s Leaf-nosed Bat
Hipposideros caffer

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Hipposideros caffer – (Sundevall, 1846)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – HIPPOSIDERIDAE – Hipposideros – caffer
Common Names: Sundevall’s Leaf-nosed Bat, Common African Leaf-nosed Bat, Lesser Leaf-nosed bat, Cape Leaf-nosed bat, Sundevall’s Roundleaf Bat, Sundevall’s African Leaf-nosed Bat, South African Lesser Leaf-nosed Bat (English), Sundevall se bladneusvlermuis, Sundevall-bladneusvlermuis, Sundevall se blaarneusvlermuis, Kaapse Blaarneusvlermuis (Afrikaans)
Synonyms: Rhinolophus caffer Sundevall, 1846; Phyllorrhina gracilis (W. C. H. Peters, 1851); Phyllorrhina patellifera (W. C. H. Peters, 1852) [not used as valid]; bicornis (von Heuglin, 1861); Phyllorrhina angolensis (de Seabra, 1898), Hipposideros nanus J. A. Allen in J. A. Allen, Lang, & Chapin, 1917; Hipposideros caffer aurantiacus De Beaux, 1924 (ACR 2024, Mammal Diversity Database 2025)
Taxonomic Note:
This species was historically listed under the family Rhinolophidae. Taxonomic revision is required as Hipposideros caffer is considered a species complex (Vallo et al. 2008; Monadjem et al. 2013; Patterson et al. 2020). Three subspecies have been listed by Simmons (2005), including: H. c. angolensis Seabra, 1898; H. c. nanus J.A. Allen, 1917; and H. c. tephrus Cabrera, 1906. Vallo et al. (2008) recognises several species lineages within the caffer-ruber complex throughout Africa, and assign southern African populations to H. c. caffer following Meester et al. (1986). The H. caffer 4 lineage comprise specimens from Eswatini, Mozambique and the type locality South Africa, thus it is probable that this lineage represents the true species (Patterson et al. 2020). The presence of multiple cryptic species in this caffer-ruber complex is supported by Monadjem et al. (2013), Patterson et al. (2020). Integrative taxonomic studies in combination with enhanced molecular data are required to resolve the taxonomy of this species complex and confirm the status of populations within the assessment region.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Richards, L.R.1 & da Silva, J.M.2
Reviewer: Smith, C.3
Institutions: 1Durban Natural Science Museum, 2South African National Biodiversity Institute, 3Endangered Wildlife Trust
Previous Assessors and Reviewers: Monadjem, A., Jacobs, D., Cohen, L., Richards, L.R., Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Roxburgh, L., Raimondo, D., Nicholson, S. & Relton, C.
Assessment Rationale
Sundevall’s Leaf-nosed Bat is listed as Least Concern as it is widely distributed across the northern and eastern regions of the assessment region, and it is inferred to have a large population. The species has an addition of new occurrence records, and no major threats have been identified that could cause extensive population declines. It can utilise manmade structures as roosting sites (both day and night roosts). As in the case of other chiropteran species, protecting and limiting disturbance to key roost sites will benefit this species.
Regional population effects: The distribution records for this species are continuous between the northeastern section of the assessment region and Mozambique and Zimbabwe. Dispersal into and out of the assessment region is suspected to be ongoing.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Richards LR & da Silva JM. 2025. A conservation assessment of Hipposideros caffer. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Sundevall’s Leaf-nosed Bat has an extensive distribution across sub-Saharan Africa and the south-western Arabian Peninsula (ACR 2024, Mammal Diversity Database 2025). In Africa, there is an isolated population in Morocco, and their distribution extends from Senegal eastwards to Sudan and Ethiopia, southwards into Kenya and Tanzania, but are absent from the central dense forest areas. Their distribution continues southwards along the eastern and western regions of southern Africa, but this is restricted to the nominate H. c. caffer lineage (Vallo et al. 2008; Patterson et al. 2020). Disjunct populations have been recorded in northern and southern Namibia and Botswana, but they are widespread across Zimbabwe and Mozambique. Within the assessment region, the species occurs in KwaZulu-Natal, Limpopo, Mpumalanga, and the northern areas of the Eastern Cape Province of South Africa, as well as in Eswatini (Skinner & Chimimba 2005; Monadjem et al. 2020). The type specimen is from Durban, South Africa (BM 1848.6.2.16, Syntype) (Monadjem et al. 2020). The species may be underrepresented in acoustic surveys as their high frequency echolocation calls rapidly attenuate (Monadjem et al. 2017). The species has an EOO of 403,688 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 3 m asl (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 3,869 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: (Not specified)
Map
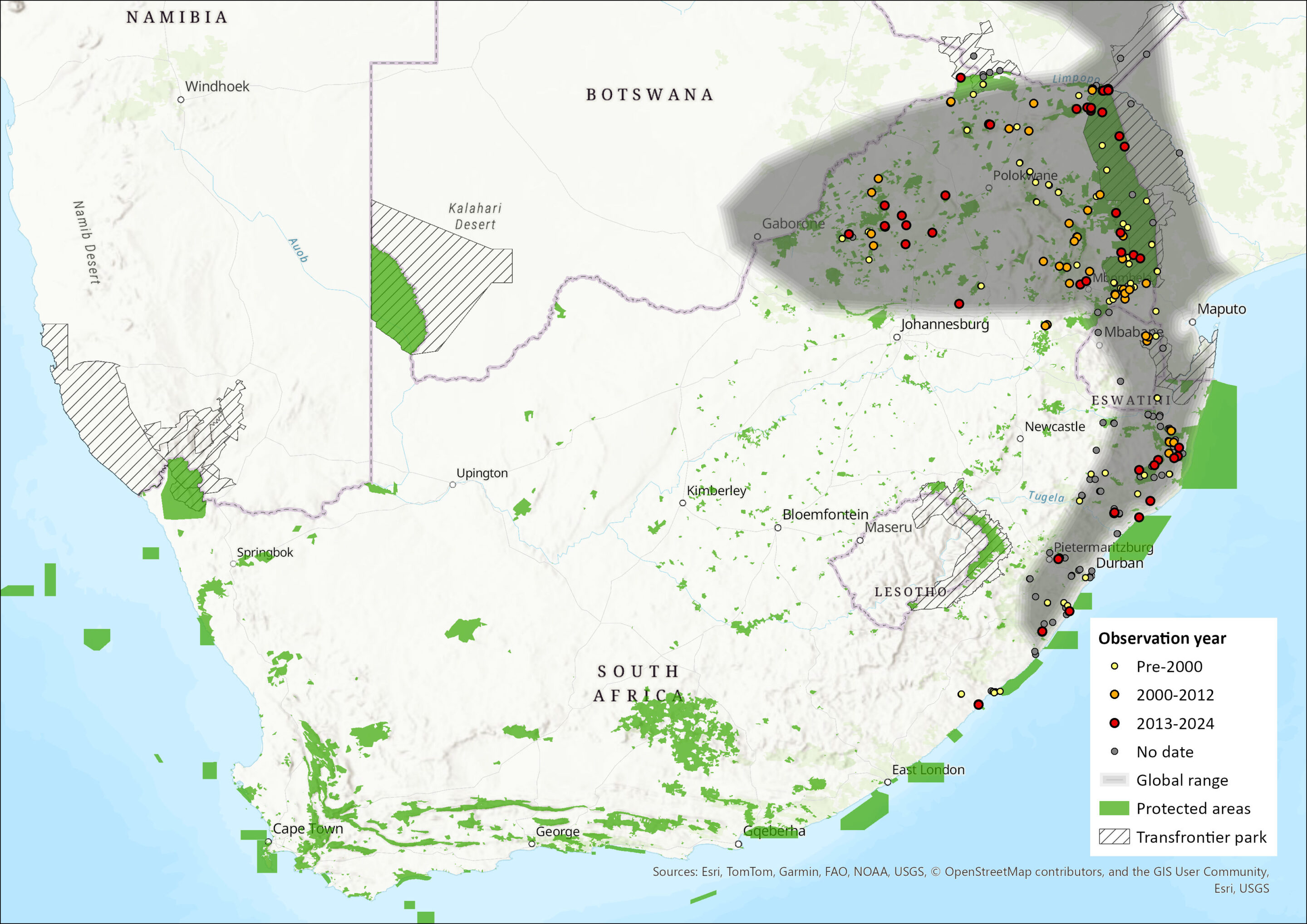 Figure 1. Distribution records for Sundevall’s Leaf-nosed Bat (Hipposideros caffer) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Sundevall’s Leaf-nosed Bat (Hipposideros caffer) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Algeria | Presence Uncertain | Native | – | – |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Liberia | Presence | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Morocco | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Saudi Arabia | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Yemen | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
No formal studies have been conducted on the potential impacts of climate change on the species. Despite this limitation, it is presumed that increasing temperatures, coupled with erratic rainfall patterns and increased drought, will likely impact the species distribution, as seen elsewhere (Adams and Hayes 2008).
Population Information
Sundevall’s Leaf-nosed Bat is considered a common species, with colonies in the hundreds reported within the assessment region and it is well represented in museums with over 900 specimens examined in Monadjem et al. (2020). The species is extremely gregarious, and forms colonies ranging from less than a dozen to huge colonies of more than 1,000 individuals where there is adequate roosting space (Kock et al. 2008; Monadjem et al. 2020). Occasionally, males may roost solitarily (Monadjem et al. 2020; De Jong et al. 2025).
Current population trend: Stable
Continuing decline in mature individuals? None reported
All individuals in one subpopulation: Uncertain, however see population genetic section
Number of subpopulations: 1 (sensu Patterson et al. 2020)
Extreme fluctuations in the number of subpopulations: Not suspected
Continuing decline in number of subpopulations: No
Number of mature individuals in largest subpopulation: >500
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Unlikely
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Unlikely
Probability of extinction in the wild within 100 years: Unlikely
Population Genetics
According to recent phylogenetic studies aimed at uncovering cryptic diversity within the Hipposideros caffer complex using mitochondrial and nuclear DNA markers (Patterson et al. 2020; Baldwin et al. 2021), individuals within the assessment region form a distinct clade with other individuals from Eswatini and Mozambique (H. Caffer 4: Patterson et al. 2020), which appears to more broadly extend to individuals from Kenya and Senegal (Baldwin et al. 2021). Due to the limited sample sizes used in the studies, it is not possible to determine whether the Mozambique, Eswatini and South African bats exists as a separate ESU; however distinct haplotypes were detected between Mozambique and South Africa (Patterson et al. 2020), suggesting a closer examination of the population genetic structure would be beneficial. Considering this is a common, gregarious species, capable of dispersing fairly widely, it is likely that individuals within South Africa form a single metapopulation. Based on this assumption, the large number of colonies identified and the large size of several colonies (e.g. > 1000), population numbers are likely to greatly exceed 5000 individuals, which translates to an Ne > 500 for the species.
Habitats and ecology
The species is generally associated with savannah, bushveld and/or coastal forests, near to rivers and other water sources (Taylor 2000). Nearby caves, sinkholes, rock fissures, hollow trees or cavities (including those created by humans, such as mines and culverts) are essential for roosting during the day (Happold 1987; Monadjem et al. 2020). Roosts are commonly shared with other species, including Nycteris thebaica in Eswatini (Monadjem et al. 2020). They are able to utilise man-made structures as roosting sites, having been found in disused sheds (De Jong et al. 2025) and within the wall of the Pongoloport (Jozini) Dam (L.R. Richards, pers obs). The species is a clutter-edge forager and feeds predominantly on Lepidoptera, with smaller quantities of Trichoptera and Coleoptera (Monadjem et al. 2020). It has low wing-loading (Monadjem et al. 2020).
This species exhibits seasonal breeding: in KwaZulu-Natal, copulation was found to take place in April, although embryonic development was delayed during winter until the end of September, and females gave birth to a single young in early December (Rautenbach 1997; Skinner & Chimimba 2005). Due to this seasonally delayed embryonic development, the gestation period of this species in KwaZulu-Natal was found to be nearly 100 days longer (~220 days) than populations from the tropics (Bernard & Meester 1982).
Ecosystem and cultural services: The species’ feeding ecology makes them important regulators of insect populations (Boyles et al. 2011; Kunz et al. 2011; Taylor et al. 2017). Bats particularly feed on species which damage crops, and agricultural areas with bats require less pesticides (Kunz et al. 2011).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Marginal | – |
| 5.1. Wetlands (inland) -> Wetlands (inland) – Permanent Rivers/Streams/Creeks (includes waterfalls) | – | Marginal | – |
| 5.4. Wetlands (inland) -> Wetlands (inland) – Bogs, Marshes, Swamps, Fens, Peatlands | – | Marginal | – |
Life History
Generation Length: Unknown
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: Mean forearm length = 4.79 ± 0.15 cm; mean total length = 8.26 ± 0.42 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Mean forearm length = 4.71 ± 0.16 cm; mean total length = 8.10 ± 0.59 cm (Monadjem et al. 2020
Longevity: Unknown
Average Reproductive Age: Unknown
Maximum Size (in cms): Females forearm length = 5.10 cm; Males forearm length = 4.71 cm
Size at Birth (in cms): Unknown
Gestation Time: Unknown
Reproductive Periodicity: Seasonal monoestry (Bernard & Meester 1982)
Average Annual Fecundity or Litter Size: One
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Uncertain, however the low wing loading suggests limited dispersal capabilities.
Congregatory: Can roost in colonies ranging from a handful of individuals to several thousand and may co-occur with other species (Wright 2009).
Systems
System: Terrestrial
General Use and Trade Information
Not known to be utilised or traded in any form.
Local Livelihood: None reported.
National Commercial Value: May provide essential ecosystem services in the form pest insect population control, particularly pest moth species.
International Commercial Value: May provide essential ecosystem services in the form pest insect population control in respect of exported produce.
End Use: None reported
Is there harvest from captive/cultivated sources of this species? None reported
Harvest Trend Comments: N/A
Threats
Human disturbance at roosting sites (such as religious ceremonies by local communities and cave tourism) has been highlighted as a threat to Sundevall’s Leaf-nosed Bat, although this is unlikely to cause significant declines of this species across its distribution.
A recent initiative to physically close abandoned mines to prevent illegal mining operations is an emerging threat, as it reduces available roost sites and can cause a mass mortality event if bats are roosting at the time of closure. Or Recent developments by the Department of Mineral and Petroleum Resources to seal unused mine shafts from illegal miners could potentially cause mass mortality of this species.
It is known to roost in hollows in baobab trees (Wright 2009); the recent announcement of the Musina-Makhado Special Economic Zone (MMSEZ) project may displace and pose a potential threat to tree hollow-roosting colonies in the proposed area of development.
Conservation
Although, no specific conservation measures are reported to be in place for this species, it is likely that it would benefit from protection of key roost sites. The species occurs in many protected areas throughout its range in the assessment region, including large reserves such as Kruger National Park, iSimangaliso Wetland Park, and Vhembe Biosphere Reserve (Monadjem et al. 2020; ACR 2024). The number of mature individuals appears to be in decline elsewhere in its range (Richards et al. 2020).
Recommendations for land managers and practitioners:
- Retain natural vegetation within mixed agricultural land-use areas to promote the presence of bats and the associated biological pest control services.
- Reduce the use of pesticides in agricultural landscapes.
- Do not disturb known day or night roosts.
- Protection or restricted access to abandoned mines harbouring colonies.
Research priorities:
- Taxonomic resolution of the H. caffer-ruber species complex.
- Long-term monitoring of known colonies to determine colony size and possible fluctuations.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
Bibliography
ACR. 2024. African Chiroptera Report. 2024. AfricanBats NPC, Pretoria. i – xv; 1 – 1397.
Adams, R.A. and Hayes, M.A. 2021. The importance of water availability to bats: climate warming and increasing global aridity. In 50 years of bat research: foundations and new frontiers (pp. 105-120). Cham: Springer International Publishing.
Baldwin, H.J., Vallo, P., Ruiz, A.T., Anti, P., Nkrumah, E.E., Badu, E.K., Oppong, S.K., Kalko, E.K., Tschapka, M. and Stow, A.J. 2021. Concordant patterns of genetic, acoustic, and morphological divergence in the West African Old World leaf‐nosed bats of the Hipposideros caffer complex. Journal of Zoological Systematics and Evolutionary Research, 59(6), pp.1390-1407.
Bernard RTF, Meester JAJ. 1982. Female reproduction and the female reproductive cycle of Hipposideros caffer caffer (Sundevall, 1846) in Natal, South Africa. Annals of the Transvaal Museum 33: 131–144.
Boyles JG, Cryan PM, McCracken GF, Kunz TH. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
De Jong, G.J., Melville, H.I. and Richards, L.R. 2024. The insectivorous bat assemblage in Telperion Nature Reserve, Mpumalanga, South Africa: species inventory and call library. Acta Chiropterologica, 26(2), pp.315-327. Happold, D.C.D. 1987. The Mammals of Nigeria. Oxford University Press, London, UK.
Kock, D., Amr, Z., Mickleburgh, S., Hutson, A.M., Bergmans, W. & Aulagnier, S. 2008. Hipposideros caffer. The IUCN Red List of Threatened Species 2008: e.T10115A3166805. http://dx.doi.org/10.2305/IUCN.UK.2008.RLTS.T10115A3166805.en. Downloaded on 16 February 2016.
Kunz TH, Braun de Torrez E, Bauer D, Lobova T, Fleming TH. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Molur S, Marimuthu G, Srinivasulu C, Mistry SH, Hutson AM, Bates PJ, Walker S, Padma Priya K, Binu Priya A.R. 2002. Status of South Asian Chiroptera: Conservation Assessment and Management Plan (C.A.M.P.) Workshop Report. Zoo Outreach Organisation, IUCN SSC Conservation Breeding Specialist Group South, Asia and WILD, Coimbatore, India.
Mammal Diversity Database. 2025. Mammal Diversity Database (Version 2.2) Zenodo. https://doi.org/10.5281/zenodo.15007505
Monadjem, A., Richards, L., Taylor, P. J.,Denys, C.,Dower, A. and Stoffberg, S. 2013. Diversity of Hipposideridae in the Mount Nimba massif, West Africa,and the taxonomic status of Hipposideros lamottei. Acta Chiropterologica 15(2): 341-352.
Monadjem, A., Shapiro, J.T., Mtsetfwa, F., Reside, A.E. and McCleery, R.A. 2017. Acoustic call library and detection distances for bats of Swaziland. Acta Chiropterologica, 19(1), pp.175-187.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis, Second Edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Patterson, B.D., Webala, P.W., Lavery, T.H., Agwanda, B.R., Goodman, S.M., Peterhans, J.C.K. and Demos, T.C. 2020. Evolutionary relationships and population genetics of the Afrotropical leaf-nosed bats (Chiroptera, Hipposideridae). ZooKeys, 929, p.117.
Rautenbach N. 1997. Sundevall’s leaf-nosed bat Hipposideros caffer. Page 261 in Mills G, Hes L, editors. The Complete Book of Southern African Mammals. Struik Publishers, Cape Town, South Africa.
Richards, L.R., Cooper-Bohannon, R., Kock, D., Amr, Z.S.S., Mickleburgh, S., Hutson, A.M., Bergmans, W. & Aulagnier, S. 2020. Hipposideros caffer. The IUCN Red List of Threatened Species 2020: e.T80459007A22094271. https://dx.doi.org/10.2305/IUCN.UK.2020-2.RLTS.T80459007A22094271.en. Accessed on 18 July 2025.
Simmons, N.B. 2005. Order Chiroptera. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World, pp. 312-529. The Johns Hopkins University Press, Baltimore, MD, USA.
Skinner J.D. and Chimimba C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Swartz, E., Weier, S.M., Pretorius, M.E. and Keith, M., 2022. Natural vegetation edges promote bat activity in macadamia orchards in northeastern South Africa. African Journal of Wildlife Research, 52(1).
Taylor, P. J. 2000. Bats of South Africa. University of Natal Press, Pietermaritzburg, South Africa.
Taylor, P.J., Matamba, E., Steyn, J.N.K., Nangammbi, T., Zepeda-Mendoza, M.L. and Bohmann, K., 2017. Diet determined by next generation sequencing reveals pest consumption and opportunistic foraging by bats in macadamia orchards in South Africa. Acta Chiropterologica, 19(2), pp.239-254.
Vallo, P.,Guillen-Servent, A. Benda, P., Pires, D. B. and Koubek, P. 2008. Variation of mitochondrial DNA in the Hipposideros caffer complex (Chiroptera: Hipposideridae) and its taxonomic implications. Acta Chiropterologica 10(2): 193-206.
Wright, G.S. 2009. Hipposideros caffer (Chiroptera: Hipposideridae). Mammalian Species, (845), pp.1-9.
