Subantarctic Fur Seal
Arctocephalus tropicalis

2025 Red list status
Least Concern
Regional Population Trend
Declining
Change compared
to 2016
No Change
Overview
Arctocephalus tropicalis – (J.E. Gray, 1872)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – OTARIIDAE – Arctocephalus – tropicalis
Common Names: Subantarctic Fur Seal, Amsterdam Island Fur Seal (English), Subantarktiese Pelsrob (Afrikaans), Inja Lolwandle Enobuso Obumhlophe (isiXhosa), Otarie d’Australie, Arctocéphale d’Australie (French)
Synonyms: Gypsophoca tropicalis J.E. Gray, 1872; Arctocephalus elegans Peters, 1876; Arctocephalus tropicalis Peters 1876 ssp. tropicalis; Arctophoca tropicalis Peters 1876
Taxonomic Note:
This species was referred to formerly as Arctocephalus elegans, A. gazella and A. tropicalis tropicalis. It is now known as A. tropicalis (Repenning et al. 1971; Rice 1998). In 2011 the Taxonomy Committee of the Society for Marine Mammalogy revised the genus of this, and many other species of fur seals to Arctophoca based on evidence presented in Berta and Churchill (2012). However, in 2013, and based on genetic evidence presented in Nyakatura and Bininda-Emonds (2012), this change was considered to be premature, and these species were returned to the genus Arctocephalus pending further research (Committee on Taxonomy 2013).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: de Bruyn, P.J.N.1 & Hofmeyr, G.J.G.2
Reviewer: Kerley, G.I.H.3
Contributor: da Silva, J.M.4 & Patel, T. 5
Institutions: 1University of Pretoria, 2Port Elizabeth Museum at Bayworld, 3Nelson Mandela University,4South African National Biodiversity Institute, 5Endangered Wildlife Trust
Previous Assessors & Reviewers: Hofmeyr, G.J.G., Wege, M., de Bruyn, N. & Bester, M.N.
Previous Contributors: Kirkman, S., Oosthuizen, H., Meyer, M., Seakamela, M., Lowry, L., Page-Nicholson, S. & Child, M.F.
Assessment Rationale
Subantarctic Fur Seals are found at a single population within South Africa’s extended territory, at the Prince Edward Islands. The total population here was (at last assessment) estimated at ~108,000 individuals., However recent population assessments show an inflexion from positive (up to 2007) to a current negative population trend (Wege et al. 2016; MIMMP unpubl. 2023). Pup production in the assessment area was estimated at 14,130 at Prince Edward Island itself in 2008/2009 (Bester et al. 2009) and 8,312 (CI: 7,983–8,697) at neighbouring Marion Island in the 2012/2013 summer. Th pup population declined by 46% between 2004 (~ 15,260 pups) and 2013 (~ 8,312), mirrored by a 58%–60% decline at selected rookeries that had been counted annually between 2007 and 2015. The total Marion Island pup production continued to decline between 2013 (~ 8,312) and 2023 (~ 7,500) but at a lower rate. However, these trends are based on two counts spaced 10 years apart. The annual pup production counts representing a smaller portion of the Marion Island coastline (direct beach counts along a strip of the north and eastern coastline, and capture-mark-recapture at Cape Davis rookery) paint a more intricate fluctuating but fairly stable picture.
Population decline at Marion Island was highest at high-density rookeries despite negligible changes in female attendance patterns, pup mortality or the median pupping date over the last 35 years. Pup growth and weaning mass at Marion Island remain substantially higher than at the largest population of this species in the world, at Gough Island, South Atlantic Ocean (Oosthuizen et al. 2016). It remains unclear why the Marion Island population has been declining for the past two decades and ongoing research is attempting to assess the cause/s. However, abundance on Marion Island, while having declined by roughly 49% between 2004 and 2023, has remained fairly stable when observed over a wider timeframe (i.e. the past four generations – 1989: 8,684 pups; 2023: 7,500 pups). No recent estimates of the pup production at neighbouring Prince Edward Island are available. However, although a net population decline was observed, the PEIs population remains well above 10,000 mature individuals within the assessment region and thus the Least Concern listing remains. The species should be reassessed if the declining population trend continues.
Although there are no major threats facing this species at present, climate change may become a concern if prey species dwindle or re-distribute. Furthermore, commercial exploitation of fish in their feeding range has the potential to influence the environment of these seals even though it does not target their predominant prey species. Subantarctic Fur Seals experienced a population bottleneck during the 19th and early 20th centuries that may have reduced genetic variation, and which may render this species vulnerable to disease or climate change. Protection is provided for this species at the Prince Edward Islands under the Sea Birds and Seals Protection Act (Act No. 46 of 1973). Furthermore, the Prince Edward Islands were proclaimed a Special Nature Reserve in 1995 under the South African Environmental Conservation Act (No. 73 of 1989) and a Marine Protected Area in 2013.
Regional population effects: Subantarctic Fur Seals have a continuous global range in the Southern Ocean (Wynen et al. 2000) and there is potential for immigrants from other subpopulations in the sub-Antarctic region to augment or re-colonise the local Prince Edward Island subpopulation in case of decline or extinction. This especially since the species is known to range widely, with vagrants being recorded far from existing haulouts (Hofmeyr and Amir 2010, Zanre and Bester 2011, Bester 2021). While there is currently no direct evidence of immigration to the Prince Edward Islands, this is most probably due to limited research effort in terms of marking individuals. Hence, the Least Concern listing is supported.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citations: de Bruyn PJN & Hofmeyr GJG. 2025. A conservation assessment of Arctocephalus tropicalis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Subantarctic Fur Seals are widely-distributed in the southern hemisphere. They breed on sub-Antarctic islands north of the Antarctic Polar Front (SCAR 2008), including both islands in the Prince Edward Islands Archipelago (Bester et al. 2003; Wege et al. 2016). At sea they are wide-ranging (de Bruyn et al. 2009; Wege et al. 2019). Vagrants have been recorded widely, including on the South African coast (Shaughnessy & Ross 1980; Bester 1989), on the East African coast as far north as Kenya and Tanzania (Hofmeyr & Amir 2010, Butynski et al. 2025) and on the West African coast as far north as Gabon (Zanre & Bester 2011). The largest single population of the species exists at Gough Island in the South Atlantic Ocean, although the last comprehensive (albeit not complete) island counts there were done in the late 1970’s (Bester et al. 2006).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
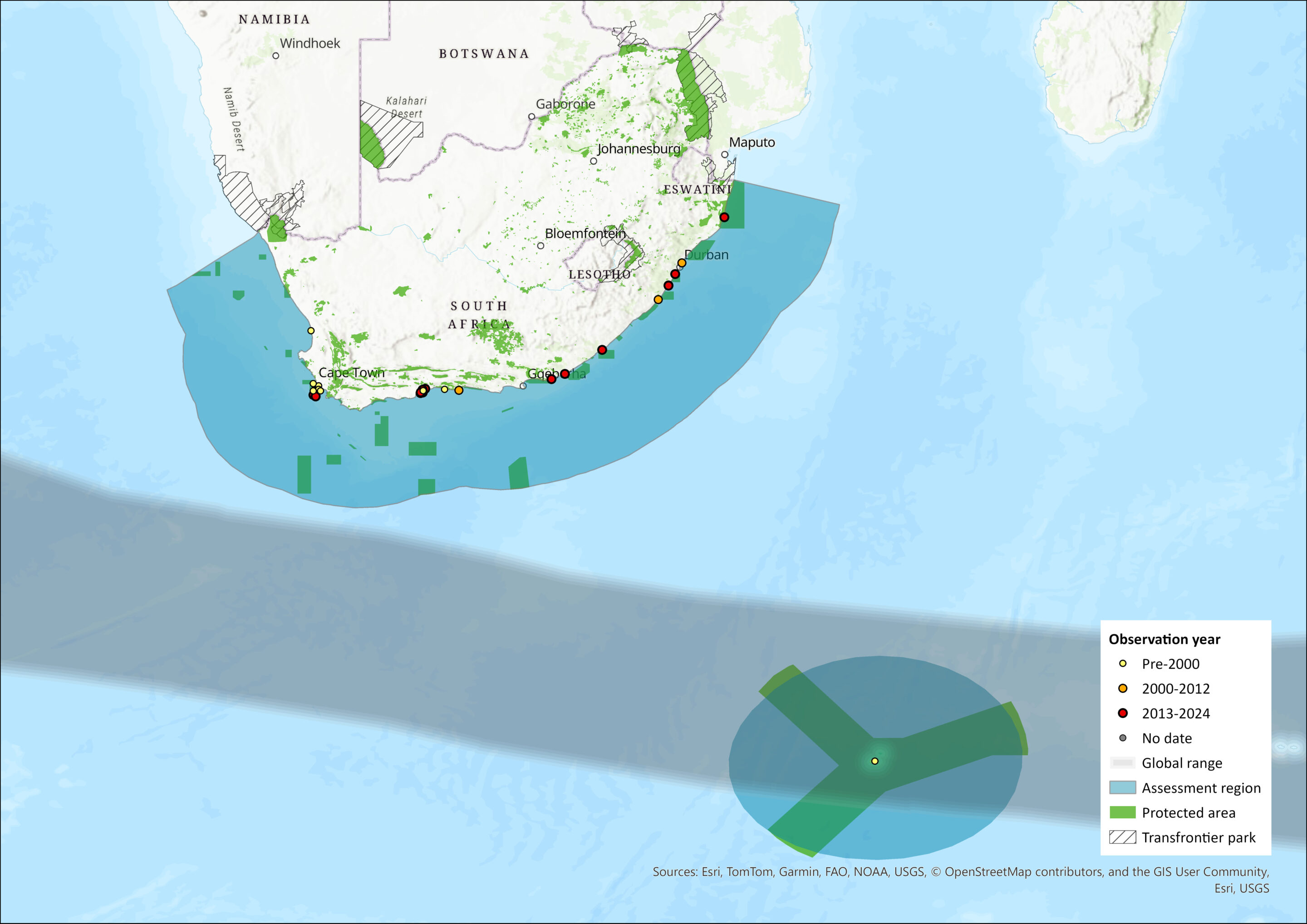
Map
Figure 1. Distribution records for Subantarctic Fur Seal (Arctocephalus tropicalis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Antarctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Vagrant | – | – |
| Antarctica | Extant | Vagrant | – | – |
| Argentina | Extant | Native | – | – |
| Australia | Extant | Vagrant | – | – |
| Australia -> Macquarie Is. | Extant | Native | – | – |
| Australia -> New South Wales | Presence Uncertain | Vagrant | – | – |
| Australia -> South Australia | Presence Uncertain | Vagrant | – | – |
| Australia -> Victoria | Presence Uncertain | Vagrant | – | – |
| Australia -> Western Australia | Presence Uncertain | Vagrant | – | – |
| Bouvet Island | Extant | Vagrant | – | – |
| Brazil | Extant | Vagrant | – | – |
| Chile | Extant | Vagrant | – | – |
| Chile -> Juan Fernández Is. | Extant | Vagrant | – | – |
| Comoros | Extant | Vagrant | – | – |
| French Southern Territories | Extant | Native | – | – |
| French Southern Territories -> Amsterdam-St. Paul Is. | Extant | Native | – | – |
| French Southern Territories -> Crozet Is. | Extant | Native | – | – |
| French Southern Territories -> Kerguelen | Extant | Vagrant | – | – |
| Heard Island and McDonald Islands | Extant | Native | – | – |
| Kenya | Extant | Vagrant | ||
| Madagascar | Extant | Native | – | – |
| Mauritius | Extant | Vagrant | – | – |
| Mozambique | Extant | Vagrant | – | – |
| Namibia | Extant | Vagrant | – | – |
| New Zealand | Extant | Vagrant | – | – |
| New Zealand -> Antipodean Is. | Presence Uncertain | Vagrant | – | – |
| New Zealand -> South Is. | Presence Uncertain | Vagrant | – | – |
| Isle Réunion | Extant | Vagrant | ||
| Saint Helena, Ascension and Tristan da Cunha | Extant | Native | – | – |
| Saint Helena, Ascension and Tristan da Cunha -> Tristan da Cunha (incl. Gough Island) | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| South Georgia and the South Sandwich Islands | Presence Uncertain | Vagrant | – | – |
| South Georgia and the South Sandwich Islands -> South Georgia | Presence Uncertain | Vagrant | – | – |
| Tanzania | Extant | Vagrant |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
| Presence | Origin | Formerly Bred | Seasonality | |
| 47. Atlantic – southeast | Extant | Native | – | – |
| 51. Indian Ocean – western | Extant | Native | – | – |
| 57. Indian Ocean – eastern | Extant | Native | – | – |
| 58. Indian Ocean – Antarctic | Extant | Native | – | – |
| 81. Pacific – southwest | Extant | Native | – | – |
| 87. Pacific – southeast | Extant | Native | – | – |
Climate change
Direct effects of climate-mediated change to the ecology of the species within its current distribution range remain unclear, although there is some evidence for increased extralimital dispersal of vagrants driven by climatic variability (Oliveira et al. 2024). Although the population at the Prince Edward islands archipelago has shown a sharp decrease over the past two decades, the drivers thereof cannot unequivocally be attributed to climate change within the region (Oosthuizen et al. 2016). Continued monitoring of the Prince Edward Islands population and especially added research into their foraging responses to environmental changes (e.g. Wege et al. 2019) is required.
Population
Globally, the total population was estimated to be greater than 400,000 animals in the early 2000s (SCAR 2008). Recent indications based on the scant available population assessment data, show a possible stabilisation or even slight decrease in population numbers at between 300,000 and 400,000 animals. Subantarctic Fur Seals breed at numerous sites on eight islands or island groups. Some 99% of Subantarctic Fur Seals breed at three of these sites (Gough Island, Prince Edward Islands and Amsterdam Island). About 63% of global pup production takes place at Gough Island (Bester et al. 2006) and a further 11% at Amsterdam Island (Guinet et al. 1994). The Prince Edward Islands host some 25% of global pup production (SCAR 2008; Bester et al. 2009; Wege et al. 2016). While all other populations were considered to be either stable or increasing in the early 2000’s (SCAR 2008), new assessments are required.
Within the assessment region, approximately 14,130 pups were born on Prince Edward Island during the 2008/2009 summer (Bester et al. 2009), 8,312 (CI: 7,983–8,697) on Marion Island in 2012/2013 (Wege et al. 2016). The most recent assessment for Marion Island is ~7,500 pups born in 2023 (MIMMP unpubl. data). Recent estimates for Prince Edward Island do not exist, but given the proximity of Marion and Prince Edward islands, similar trends in pup production are assumed. Thus, there are currently probably between 15,000 20,000 pups born each year within the assessment region, which can be extrapolated to a total population of around 90,000 individuals based on the ratio of births to total numbers of 1:4.8 (Kerley 1987). While both components of the population increased steadily to the early 21st century, Marion Island pup production declined by 6.4% between 2003/2004 and 2012/2013 (Wege et al. 2016) and continued to decline slightly until 2023 (MIMMP unpubl. data). This represents a 49% decline in the pup population between 2004 (~15,260 and 2023 (~ 7,500) and is mirrored by a 58%–62% decline at rookeries counted annually (2007–2023). This decline is not an anomaly but due to a real decline evidenced by continuous annual decreases (2007–2023) (Wege et al. 2016; MIMMP unpubl. data). It is unlikely that this slowed growth is a result of interspecific competition for breeding space with the sympatric Antarctic Fur Seal (A. gazella), as the Subantarctic Fur Seals prefer to breed on boulder/jumbled rocky beaches (Bester 1982), whereas Antarctic Fur Seals prefer small-pebble beaches backed by vegetated slopes (Kerley 1984).
The Prince Edward Island component changed very little between 2001/2002 and 2008/2009 (0.3% mean annual decline, Bester et al. 2009), compared to the previous natural population increase of 9.3% per annum between 1987/1988 and 2001/2002. However, it is unknown whether there has been a more recent decline mirroring that at Marion Island. The population decline at Marion Island was highest at high-density rookeries despite negligible changes in female attendance patterns, pup mortality, weaning mass or median pupping date over the last 35 years (Oosthuizen et al. 2016; Wege et al. 2016; MIMMP unpubl. data). It is currently unclear why this population is declining and further research is needed to assess the cause of decline and determine whether it will continue.
Generation length is estimated as 10.7 years (Pacifici et al. 2013), yielding a 41-year four-generation window (1983-2024). On Marion Island, the 2023 total population estimate of pups (~7,500) is only slightly less than that of 1989 when the population was still increasing exponentially (8,684 pups, Wilkinson & Bester 1990). Thus, there has been only slight net decline over four generations.
Population Information
Current population trend: Declining
Continuing decline in mature individuals? Yes
Number of mature individuals in population: 400,000 (global)
Number of mature individuals in largest subpopulation: 107,000 (assessment region).
All individuals in one subpopulation: (Not specified)
Number of subpopulations: One (both Marion and Prince Edward Island) subpopulation.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Commercial sealing in the Southern Ocean during the 18th and 19th centuries reduced the abundance of the Subantarctic Fur Seal to very low levels. At Marion Island the population was also hunted by commercial sealers, with the last harvest taking place in 1921 (Kerley 1987), after which the population was too low to remain commercially viable (Condy 1978). Rand (1956) estimated that only 167 pups were born at Marion Island in 1951. Such dramatic population reductions can have consequences for the genetic profiles of recovering populations. However, Maboko (2009) indicated that genetic diversity is high in the Subantarctic Fur Seal population at the Prince Edward Islands, suggesting that the population bottleneck did not reduce genetic variation. Wynen et al. (2001) assessed post sealing mitochondrial DNA variation in several A. tropicalis Southern Ocean Island groups and showed that the Prince Edward islands remnant population likely was the source for repopulation of Macquarie and Crozet islands. This study indicated high levels of genetic variation and significant lineage structure within A. tropicalis.; however, these lineages showed no geographical association, indicating no spatial population structure. Consequently, this species is thought to exist as a metapopulation. Further assessment of the population genetics of Subantarctic Fur Seals is needed.
Based population estimates provided above (i.e., 90,000 individuals) and applying a Ne/Nc conversion ratio between 0.1-0.3, the effective population size can be estimated for the population at around 9,000-27,000. This far exceeds the Ne 500 threshold for a stable and healthy population. However, given that this species experienced a bottleneck, these are likely to be overestimates of Ne.
Habitats and ecology
Subantarctic Fur Seals are highly sexually dimorphic with adult males reaching 1.8 m in length and mases of 70 – 165 kg, while adult females grow to 1.52 m in length and masses of 25 – 67 kg (Laws 1993). Pups are some 4 kg at birth (Laws 1993,). While females attain sexual maturity at 4-6 years of age (Bester 1995, males are only socially mature at 10-11 years.
The preferred terrestrial habitat of Subantarctic Fur Seals is rough beaches with jumbled boulders and rocks, which provide exposure to prevailing winds and sources of shade for thermoregulation (Bester 1982). Subantarctic Fur Seals are polygynous with males defending territories during the summer breeding haulout (Bester 1981, Kerley 1983a). After a gestation of some 51 weeks, pups are born between late October and early January with the median pupping date in mid-December. Parturition takes place up to six days after adult females haul out during the breeding season. Mating occurs eight to twelve days later (Bester 1981, Kerley 1983a, Goldsworthy 1999). Adult females wean their pups at approximately 11 months of age, undertaking a cycle of foraging trips and periods of attending their pups during this time. These increase from 6-10 days during the first month, to 23-28 days by the 11th month (Goldsworthy 1999, Georges and Guinet 2000, Kirkman et al. 2002).
Subantarctic Fur Seals exploit pelagic habitat for foraging, diving primarily less than 100 m. However, dives of as deep as 208 m have been recorded (Georges et al. 2000). Foraging behaviour differs substantially between populations (Kirkman et al. 2002, de Bruyn et al. 2009, Wege 2013, Wege et al. 2016). The diet of Subantarctic Fur Seals consists primarily of a variety of species of myctophid and notothenid fish, and cephalopods (Bester and Laycock 1985, Klages and Bester 1998, Makhado et al. 2013).
Ecosystem and cultural service: Subantarctic Fur Seals are important top predators in the Southern Ocean being an important component of the trophic system. As such they are a potential indicator species. Furthermore, they are a link between the marine environment and terrestrial habitats on Subantarctic islands, enriching coastal areas through faeces and moult hair (Smith 2008). However, disturbance of terrestrial areas and the import of nutrient can lead to the spread of invasive plants (Hausman et al. 2013).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 9.1. Marine Neritic -> Marine Neritic – Pelagic | – | Suitable | Yes |
| 10.1. Marine Oceanic -> Marine Oceanic – Epipelagic (0-200m) | – | Suitable | Yes |
| 12.1. Marine Intertidal -> Marine Intertidal – Rocky Shoreline | – | Suitable | Yes |
| 13.1. Marine Coastal/Supratidal -> Marine Coastal/Supratidal – Sea Cliffs and Rocky Offshore Islands | – | Suitable | Yes |
Life History
Generation Length: 10.7 years
Age at Maturity: Female or unspecified: ~3-5 years
Age at Maturity: Male: Sexually mature at 3-4 years, socially mature at 10-11 years
Size at Maturity (in cms): Female: 152 cm
Size at Maturity (in cms): Male: 180 cm
Longevity: 23 years (females), 18 years (males)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): 180 cm
Size at Birth (in cms): (Not specified)
Gestation Time: ~ 51 weeks
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: Single pup, very rarely twins
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Inshore and pelagic foraging, diving within the top few hundred meters of the water column
Congregatory: Highly social at breeding sites, possibly predominantly solitary at sea
Systems
System: Terrestrial, Marine
General Use and Trade Information
The species is not traded nor under threat of future trade as it is largely protected by the isolation of its distribution and by legislation. Protection is provided for this species at the Prince Edward Islands under the Sea Birds and Seals Protection Act (Act No. 46 of 1973).
Local Livelihood: No
National Commercial Value: None
International Commercial Value: None
End Use: None
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: No harvest
Threats
Commercial sealing during the 18th and 19th centuries overexploited all species of fur seals and reduced Subantarctic Fur Seals to the brink of extinction by the beginning of the 20th century. Since then, populations have increased rapidly and much of the former range has been re-occupied. This population bottleneck has potentially reduced their genetic variation and may render this species vulnerable to disease or climate change (Wynen et al. 2000; Kovacs et al. 2012). However, genetic diversity remains relatively high for the species (Wynen et al. 2000) and the Prince Edward Islands population (Maboko 2009). Fur seals are also at risk of mass mortality from infectious diseases, though the isolation of their island habitat affords some species a higher degree of protection from disease (Lavigne & Schmitz 1990; Chown et al. 1998). The transfer of pathogens from invasive species, such as the House Mouse (Mus musculus) may potentially lead to mass mortality events (de Bruyn et al. 2008). Recent Southern Hemisphere avian influenza (H5N1) outbreak events are cause for concern (Banyard et al. 2024), with the disease being detected in various species of seabirds at Marion Island (Duvenage 2024). This virus has caused mass mortalities of pinnipeds elsewhere (Alava et al. 2024, Campagna et al. 2024). A further disease of concern is rabies, which has recently become endemic in Cape Fur Seals (Van Helden 2024). Subantarctic Fur Seals are regular vagrants to the South African coast (Shaughnessy & Ross 1980, Bester 1989) and could potentially come into contact with Cape Fur Seals, which have also been recorded on Marion Island (Kerley 1983b). The return of infected vagrants to the Southern Ocean may result in the further distribution of the disease.
Commercial exploitation of fish in their feeding range has the potential to influence the environment of these seals (Hanchet et al. 2003) even though it does not target predominant prey species (Klages & Bester 1998; Makhado et al. 2013). While few fisheries exploit waters occupied by Subantarctic Fur Seals, these may expand in the future and have potentially detrimental trophic impacts (Hanchet et al. 2003). Entanglement in marine debris occurs at the Prince Edward Islands but incidences are low at 0.24% for this species and the sympatric Antarctic Fur Seal combined for the period 1996–2001 (Hofmeyr et al. 2002). Tourist visits to Subantarctic Fur Seal haul-out sites are rare and thought to cause minimal disturbance (Shirihai 2002; Hofmeyr & Bester 2008). Tourism does not currently take place at the Prince Edward Islands (Prince Edward Islands Management Plan 2010).
Subantarctic Fur Seals are sympatric with Antarctic Fur Seals at the Prince Edward Islands (Bester et al. 2006) and the Îles Crozet (Guinet et al. 1994), and with both Antarctic Fur Seals and New Zealand Fur Seals at Macquarie Island (Goldsworthy 1999), leading to hybridisation at all three sites. Levels of hybridisation at the Prince Edward Islands are very low and no threat to the species at this site and are thought to have declined over time (Hofmeyr et al. 1997).
The recent decline in pup production is possibly linked to density-dependent factors but to what extent has not been determined (Wege et al. 2016). It is unknown if this is a consequence of the potentially detrimental effects of climate change (Chown et al. 1998; Learmonth et al. 2006; Kovacs et al. 2012).
Conservation
Subantarctic Fur Seals live in some of the most remote oceanic areas and breed on many of the most isolated islands on earth. All of the breeding islands are managed as protected areas or parks by the governments that claim these territories. Seals on the Prince Edward Islands are protected by the South African Seabirds and Seals Protection Act (Prince Edward Islands Management Plan 2010), the Marine Living Resources Act (Act no. 18 of 1998; Policy on the management of seals, seabirds and shorebirds. (Government Gazette No. 30534, 2007) and the Threatened Or Protected Species Regulations (Government Gazette No. 40876 of 2017) of the National Environmental Management: Biodiversity Act (10/2004). The islands themselves are a Special Nature Reserve (de Villiers & Cooper 2008) and part of a Marine Protected Area (DEA 2013), and thus the breeding grounds within the assessment region are secure. Special management areas restrict visitor access to specific areas, including all of Prince Edward Island itself and most of Marion Island (Prince Edward Islands Management Plan 2010). Tourism does not take place in the Prince Edward Islands (Prince Edward Islands Management Plan 2010). No specific interventions are required at present, but the population should continue to be monitored.
Recommendations for land managers and practitioners:
- Continuation of monitoring and research programme conducted to date (over the past 4 decades) by the Marion Island Marine Mammal Programme previously under the auspices of the Mammal Research Institute, Department of Zoology and Entomology, University of Pretoria, and now managed through the South African Polar Research Infrastructure. Especially continued assessment of population trends and foraging behaviour, given the continuing declining population trend.
Research priorities:
- Assess the causes of the identified inflexion in population trajectory, from positive to negative, at Marion Island.
- Determine the effect of global climate change on the foraging and breeding behavior of this species.
- Assess the effects of local fisheries on prey populations.
- Implement a programme to assess the occurrence of Avian H5N1 virus in PEI population and develop contingency plans should the population be affected.
Encouraged citizen actions:
- Foraging ecology research is progressing but has been impeded by the financial investment that is required for satellite telemetry. Aid in funding this aspect can be securely provided through http://www.givengain.com/cause/4655/.
- Due to the isolation of Antarctic Fur Seal habitat, citizen actions are limited. However, citizens can report potential sightings of vagrants on virtual museum platforms (for example, iNaturalist and MammalMAP).
Bibliography
Alava, J.J., Tirapé, A., Denkinger, J., Calle, P., Rosero R, P., Salazar, S., Fair, P.A. and Raverty, S. 2024. Endangered Galápagos sea lions and fur seals under the siege of lethal avian flu: a cautionary note on emerging infectious viruses in endemic pinnipeds of the Galápagos Islands. Frontiers in Veterinary Science, 11, p.1457035.
Banyard, A.C., Bennison, A., Byrne, A.M., Reid, S.M., Lynton-Jenkins, J.G., Mollett, B., De Silva, D., Peers-Dent, J., Finlayson, K., Hall, R. and Blockley, F. 2024. Detection and spread of high pathogenicity avian influenza virus H5N1 in the Antarctic Region. Nature Communications, 15(1), p.7433.
Berta, A. and Churchill, M. 2012. Pinniped taxonomy: review of currently recognized species and subspecies, and evidence used for their description. Mammal Review 42: 207-234.
Bester, M. N. 1981. Seasonal changes in the population composition of the fur seal Arctocephalus tropicalis at Gough Island. South African Journal of Wildlife Research 11: 49-55.
Bester, M. N. 1982. Distribution, habitat selection and colony types of the Amsterdam Island fur seal Arctocephalus tropicalis at Gough Island. Journal of Zoology (London) 196: 217-231.
Bester, M. N. 1989. Movements of southern elephant seals and subantarctic fur seals in relation to Marion Island. Marine Mammal Science 5: 257-265.
Bester, M. N. 1995. Reproduction in the female subantarctic fur seal, Artocephalus tropicalis. Marine Mammal Science 11(3): 362-375.
Bester, M.N. 2021. Vagrant sub-Antarctic fur seal at tropical Ascension Island, South Atlantic Ocean. Polar Biology, 44(2), pp.451-454.
Bester, M. N., Ryan, P. G. and Dyer, B. M. 2003. Population numbers of fur seals at Prince Edward Island, Southern Ocean. African Journal of Marine Science 25: 549-554.
Bester, M. N., Wilson, J. W., Burle, M.-H. and Hofmeyr, G. J. G. 2006. Population trend of Subantarctic fur seals at Gough Island. South African Journal of Wildlife Research 36: 191-194.
Bester, M.N. and Laycock P.A. 1985. Cephalopod prey of the Sub-Antarctic fur seal, Arctocephalus tropicalis, at Gough Island. In: W.R. Siegfried, P.R.Condy, R.M. Laws (ed.), Antarctic nutrient cycles and food webs, pp. 551-554. Springer-Verlag, Berlin.
Bester, M.N., Ryan, P.G. and Visagie, J. 2009. Summer survey of fur seals at Prince Edward Island, southern Indian Ocean. African Journal of Marine Science 31: 451-455.
Butynski, T.M., Mathews, P. and de Jong, Y.A. 2025. The Subantarctic Fur Seal Arctocephalus tropicalis (Carnivora: Pinnipedia: Otariidae) on the Coast of East Africa: A Review. Journal of East African Natural History, 114(1), pp.1-7.
Campagna, C., Uhart, M., Falabella, V., Campagna, J., Zavattieri, V., Vanstreels, R.E. and Lewis, M.N. 2024. Catastrophic mortality of southern elephant seals caused by H5N1 avian influenza. Marine Mammal Science, 40(1).
Chown, S. L., Gremmen, N. J. M. and Gaston, K. J. 1998. Ecological biogeography of the Southern Ocean Islands: species-area relationships, human impacts, and conservation. American Naturalist 152: 562-575.
Condy, P.R. 1978. Distribution, abundance and annual cycle of fur seals (Arctocephalus spp.) on the Prince Edward Islands. S. Afr. J. Wildl. Res. 8: 159-168.
de Bruyn PJN, Bastos ADS, Eadie C, Tosh CA, Bester MN. 2008. Mass Mortality of Adult Male Subantarctic Fur Seals: Are Alien Mice the Culprits? PLoS One 3: 3757.
de Bruyn, P.J.N., Tosh, C.A., Oosthuizen, W.C., Bester, M.N. and Arnould, J.P.Y. 2009. Bathymetry and frontal systems interactions influence seasonal foraging movements of lactating subantarctic fur seals from Marion Island. Marine Ecology Progress Series 394: 263-276.
de Villiers MS, Cooper J. 2008. Conservation and Management. In: Chown SL, Froneman PW (ed.), The Prince Edward Islands: Land-Sea Interactions in a Changing Ecosystem, pp. 301–330. Sun Press, African Sun Media, Stellenbosch, South Africa.
Department of Environmental Affairs. 2013. Prince Edward Islands declared a Marine Protected Area.
Georges, J.-Y., Tremblay, Y. and Guinet, C. 2000. Seasonal diving behaviour in lactating subantarctic fur seals on Amsterdam Island. Polar Biology 23: 59-69.
Duvenage, E. 2024. Avian flu suspected on South Africa’s remote Marion Island. Nat. Afr, 10.
Georges, J.Y. and Guinet, C. 2000. Early mortality and perinatal growth in the subantarctic fur seal (Arctocephalus tropicalis) on Amsterdam Island. Journal of Zoology, 251(3), pp.277-287.
Goldsworthy, S. D. 1999. Maternal attendance behaviour of sympatrically breeding Antarctic and subantarctic fur seals, Arctocephalus spp., at Macquarie Island. Polar Biology 21: 316-325.
Guinet, C., Jouventin, P. and. Georges, J.-Y. 1994. Long term population changes of fur seals Arctocephalus gazella and Arctocephalus tropicalis on subantarctic (Crozet) and subtropical (St. Paul and Amsterdam) Islands and their possible relationship to El Nino Southern Oscillation. Antarctic Science 6(4): 473-478.
Hanchet, S., Horn, P. and Stevenson, M. 2003. Fishing in the ice: is it sustainable? Water & Atmosphere 11: 24–25.
Haussmann NS, Rudolph EM, Kalwij JM, McIntyre T. 2013. Fur seal populations facilitate establishment of exotic vascular plants. Biological Conservation 162: 33–40.
Hofmeyr, G. J. G., De Maine, M., Bester, M. N., Kirkman, S. P., Pistorius, P. A. and Makhado, A. B. 2002. Entanglement of pinnipeds at Marion Island, Southern Ocean, 1991-2001. Australian Mammalogy 24: 141-146.
Hofmeyr, G.J.G. and Amir, O.A. 2010. Vagrant Subantarctic fur seal on the coast of Tanzania. African Zoology 45: 144-146.
Hofmeyr, GJG., Bester, MN. & Jonker, FC. 1997. Changes in population sizes and distribution of fur seals at Marion Island. Polar Bio 17:150–158
Hofmeyr, G.J.G. and Bester M.N. 2008. Subantarctic Islands. In: M. Lück (ed.), Encyclopaedia of Tourism and Recreation in Marine Environments, pp. 456-457. CABI, Wallingford, UK.
Kerley GIH. 1984. The relationship between two species of fur seals Arctocephalus tropicalis (Gray) and A. gazella (Peters) on Marion Island. M.Sc. Thesis. University of Pretoria.
Kerley, G. I. H. 1983a. Comparison of seasonal haulout patterns of fur seals Arctocephalus tropicalis and A. gazella on Subantarctic Marion Island. South African Journal of Wildlife Research 13: 71-77.
Kerley, G.I.H. 1983b. Record for the Cape fur seal Arctocephalus pusillus pusillus from subantarctic Marion Island. S. Afr. J. Zool. 18:139-140.
Kerley, G.I.H. 1987. Arctocephalus tropicalis on the Prince Edward Islands. In: J.P. Croxall and R.L. Gentry (eds), Status, Biology and Ecology of Fur Seals; Proceedings of an International Symposium and Workshop, Cambridge, England, 23-27 April 1984. NOAA Technical Report NMFS. 51:61-64.
Kirkman, S.P., Bester, M.N., Hofmeyr, G.J.G., Pistorius, P.A. and Makhado, A.B. 2002. Pup growth and maternal attendance patterns in Subantarctic fur seals. African Zoology 37: 13-19.
Klages, N. T. W. and Bester, M. N. 1998. Fish prey of fur seals Arctocephalus spp. at subantarctic Marion Island. Marine biology 131: 559-566.
Kovacs, K.M., Aguilar, A., Aurioles, D., Burkanov, V., Campagna, C., Gales, N.J., Gelatt, T., Goldsworthy, S.D., Goodman, S.J., Hofmeyr, G.J.G., Härkönen, T., Lowry, L., Lydersen, L., Schipper, J., Sipilä, T., Southwell, C., Thompson, D. and Trillmich, F. 2012. Global threats to pinnipeds. Marine Mammal Science 28: 414-436.
Lavigne D.M. and Schmitz, O.J. 1990. Global warming and increasing population densities: a prescription for seal plagues. Marine Pollution Bulletin 21: 280-284.
Laws, R. 1993. Antarctic Seals. Cambridge University Press.
Learmonth, J.A., Macleod, C.D., Santos, M.B., Pierce, G.J., Crick, H.Q.P. and Robinson, R.A. 2006. Potential effects of climate change on marine mammals. Oceanography and Marine Biology: An Annual Review 44: 431-464.
Maboko, V.J. 2009. Genetic diversity and hybridisation estimates of Arctocephalus tropicalis and A. gazella from Marion Island. University of Pretoria (South Africa).
Makhado, A.B., Bester, M.N., Somhlaba, S., Crawford, R.J.M. 2013. The diet of the subantarctic fur seal Arctocephalus tropicalis at Marion Island. Polar Biology 36: 1609–1617.
Nyakatura, K. and Bininda-Emonds, O.R.P. 2012. Updating the evolutionary history of Carnivora (Mammalia): a new species-level supertree complete with divergence time estimates. BMC Biology 10: 12.
Oliveira, C.D., Guimarães, M., Schroeder, L., Zagonel-Oliveira, M., Da Silva, G.A.M., Borges-Martins, M., Danilewicz, D., Prado, J.H.F., Schossler, V., Botta, S. and Secchi, E.R. 2024. Patterns of occurrence of the sub-Antarctic fur seal Arctocephalus tropicalis (Gray 1872) in Southern Brazil: climatic and environmental associations. Polar Biology, 47(1), pp.53-71.
Oosthuizen, W.C., de Bruyn, P.N., Wege, M. and Bester, M.N. 2016. Geographic variation in subantarctic fur seal pup growth: linkages with environmental variability and population density. Journal of Mammalogy, 97(2), pp. 347-360.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and
Rand, R.W. 1956. Notes on the Marion Island fur seal. Proc. Zoo!.Soc. Land. 126:65-82.
Repenning, C.A., Peterson, R.S. and Hubbs, C.L. 1971. Contributions to the systematics of the southern fur seals, with particular reference to the Juan Fernández and Guadalupe species. In: W.H. Burt (ed.), Antarctic Pinnipedia, pp. 1-34. Antarctic Research Series 18, American Geophysical Union, New York, USA.
Rice, D.W. 1998. Marine Mammals of the World: Systematics and Distribution. Society for Marine Mammalogy, Special Publication Number 4, Lawrence, Kansas.
Scientific Committee for Antarctic Research – Expert Group on Seals. 2004. Scientific Committee for Antarctic Research – Expert Group on Seals Report.
Shaughnessy, P. D. and Ross, G. J. B. 1980. Records of the subantarctic fur seal (Arctocephalus tropicalis) from South Africa with notes on its biology and some observations of captive animals. Annals of the South African Museum 242: 71-89.
Shirihai, H. 2002. A complete guide to Antarctic wildlife. Alula Press.
Smith VR. 2008. Energy flow and nutrient cycling in the Marion Island terrestrial ecosystem: 30 years on. Polar Record 44: 211–226.
Van Helden, L. 2024. Rabies in the Western Cape: Seals, Dogs and Jackals. South Africa.
Wege M. 2013. Maternal foraging behaviour of Subantarctic fur seals from Marion Island. M.Sc. Thesis. University of Pretoria.
Wege, M., Etienne, M.P., Chris Oosthuizen, W., Reisinger, R.R., Bester, M.N. and de Bruyn, P.N. 2016. Trend changes in sympatric Subantarctic and Antarctic fur seal pup populations at Marion Island, Southern Ocean. Marine Mammal Science, 32(3), pp.960-982.
Wege, M., de Bruyn, P.N., Hindell, M.A., Lea, M.A. and Bester, M.N. 2019. Preferred, small-scale foraging areas of two Southern Ocean fur seal species are not determined by habitat characteristics. BMC ecology, 19(1), p.36.
Wilkinson, IS. & Bester, MN. 1990. Continued population increase in fur seals, Arctocephalus tropicalis and A. gazella, at the Prince Edward Islands. South African Journal of Antarctic Research 20: 58–63.
Wynen, L. P., Goldsworthy, S. D., Guinet, C., Bester, M. N., Boyd, I. L., Gjertz, I., Hofmeyr, G. J. G., White, R. W. G. and Slade, R. W. 2000. Post sealing genetic variation and population structure of two species of fur seals (Arctocephalus gazella and A. tropicalis). Molecular Ecology 9: 299-314.
Wynen, L.P., Goldsworthy, S.D., Insley, S.J., Adams, M., Bickham, J.W., Francis, J., Gallo, J.P., Hoelzel, A.R., Majluf, P., White, R.W. and Slade, R. 2001. Phylogenetic relationships within the eared seals (Otariidae: Carnivora): implications for the historical biogeography of the family. Molecular phylogenetics and evolution, 21(2), pp.270-284.
Zanre, R. and Bester, M.N. 2011. Vagrant Subantarctic fur seal in the Mayumba National Park, Gabon. African Zoology 46: 185-187.