
Steenbok
Raphicerus campestris

2025 Red list status
Least Concern
Regional Population Trend
Stable
change compared
to 2016
No Change
Overview
Raphicerus campestris – (Thunberg, 1811)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Raphicerus – campestris
Common Names: Steenbok (English, Afrikaans), Steinbuck (English), Ingina, Iqina (Ndebele), Iqhina (Ndebele, Zulu), Pudubudu (Sepedi), Phudufudu (Sepedi, Setswana), Thiane (Sesotho), Phuduhudu (Setswana), Mhene (Shona), Lingcina (Swati), Phuluvhulu (Venda), Xipene (Tsonga), Itshabanqa (Xhosa)
Synonyms: Antilope campestris Thunberg, 1811, acuticornis, bourquii, capensis, capricornis, cunenensis, fulvorubescens, grayi, hoamibensis, horstockii, ibex, kelleni, natalensis, neumanni, pallida, pediotragus, rupestris, steinhardti, stigmatus, subulata, tragulus, ugabensis, zukowskyi, zuluensis
Taxonomic Note:
Although various subspecies have been suggested, for example Ansell (1972) recorded eight and Meester et al. (1986) listed five subspecies south of the Sahara Desert, the validity of these subspecies remains questionable. Two commonly accepted subspecies include Raphicerus campestris campestris from southern Africa and R. c. neumanni from East Africa (Kingdon 1997; du Toit 2013). Taxonomic revision of this species is necessary (Skinner & Chimimba 2005).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Russo, I.M.1, Jansen van Vuuren, A.2, Swanepoel, H.2 & da Silva, J. M.3
Reviewer: Anderson, J.4,5
Institutions: 1Cardiff University, Cardiff, Wales, 2Nelson Mandela University, South Africa, 3South African National Biodiversity Institute, 4IUCN SSC Antelope Specialist Group, 5International Conservation Services
Previous Assessors: Birss, C. & Palmer, G.
Previous Reviewer: Du Toit, J.
Previous Contributors: Child, M.F., Relton, C. & Avenant, N.
Assessment Rationale
Listed as Least Concern as the Steenbok is widely distributed (and continues to be well represented in protected areas according to 2012–2013 game counts), considered to be relatively common (for example, 3.78 animals/km2 on two small-livestock farms in the Northern Cape and Free State provinces) and no major threats have been identified within the assessment region. Although local and regional declines are suspected due to bushmeat hunting and competition with livestock outside of protected areas, especially where human densities are high, the population trend is generally stable. The effects of the conversion to wildlife ranching on this species should be monitored across the various bioregions, as Steenbok in arid and fynbos habitats may be vulnerable to increasing competition with introduced extralimital herbivores, and which may represent an emerging threat given the expansion of the wildlife ranching industry. Key interventions include the formation of conservancies and the promotion of permeable fences, and the control of bush encroachment.
Regional population effects: There is dispersal across regions through transfrontier spaces along the northern border of South Africa (including the Kgalagadi and Greater Limpopo Transfrontier Parks). Rescue effects are considered possible but not necessary.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Russo IM, Jansen van Vuuren A, Swanepoel H & da Silva JM. 2025. A conservation assessment of Raphicerus campestris. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and Occurrence
Geographic Range
In Africa, this species occurs in two disjunct areas, one in East Africa, including northern and central Tanzania and into southern Kenya, and the other in southern Africa, intruding marginally into southern Angola and western Zambia (Skinner & Chimimba 2005; du Toit 2013). These regions are separated by the tall, dense Miombo woodlands of central Zambia, northern Mozambique and Malawi (du Toit 2013), and are approximately 1,000 km apart (Skinner & Chimimba 2005). Steenbok are generally absent from forested and thick woodland areas (Skinner & Chimimba 2005). In East Africa, their distribution has declined somewhat as they previously occurred in Uganda, but much of their appropriate habitat has been transformed due to agriculture (East 1999).
The southern African distribution extends through suitable habitats southwards from southern Angola and western Zambia, across most of Namibia (with the exception of the extreme arid, coastal regions), throughout Botswana, into central and southern Zimbabwe, southern Mozambique and across most of South Africa (IUCN SSC Antelope Specialist Group 2016). Its distribution is continuous and widespread throughout South Africa with lower densities in areas without suitable cover. Within the assessment region this species is present in all provinces, throughout several habitat types, including grassland, savannah, shrublands and semi-desert, absent only from the southeastern forested regions of the Western Cape, Eastern Cape and KwaZulu-Natal Provinces (du Toit 2013). It is likely that Steenbok once occurred in the lowlands of Lesotho, as they occur along the Caledon River in the eastern Free State (N. Avenant pers. comm. 2016), but were not recorded by Lynch (1994) and subsequent field surveys have failed to detect the species (Ambrose & Talukdar 2000; N. Avenant unpubl. data). Locals in the area have positively identified the species from memory (Sesotho name for Steenbok is Thiane), suggesting it was once present (N. Avenant pers. comm. 2016), but is probably regionally extinct due to overhunting (sensu Lynch 1994).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): 1500
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
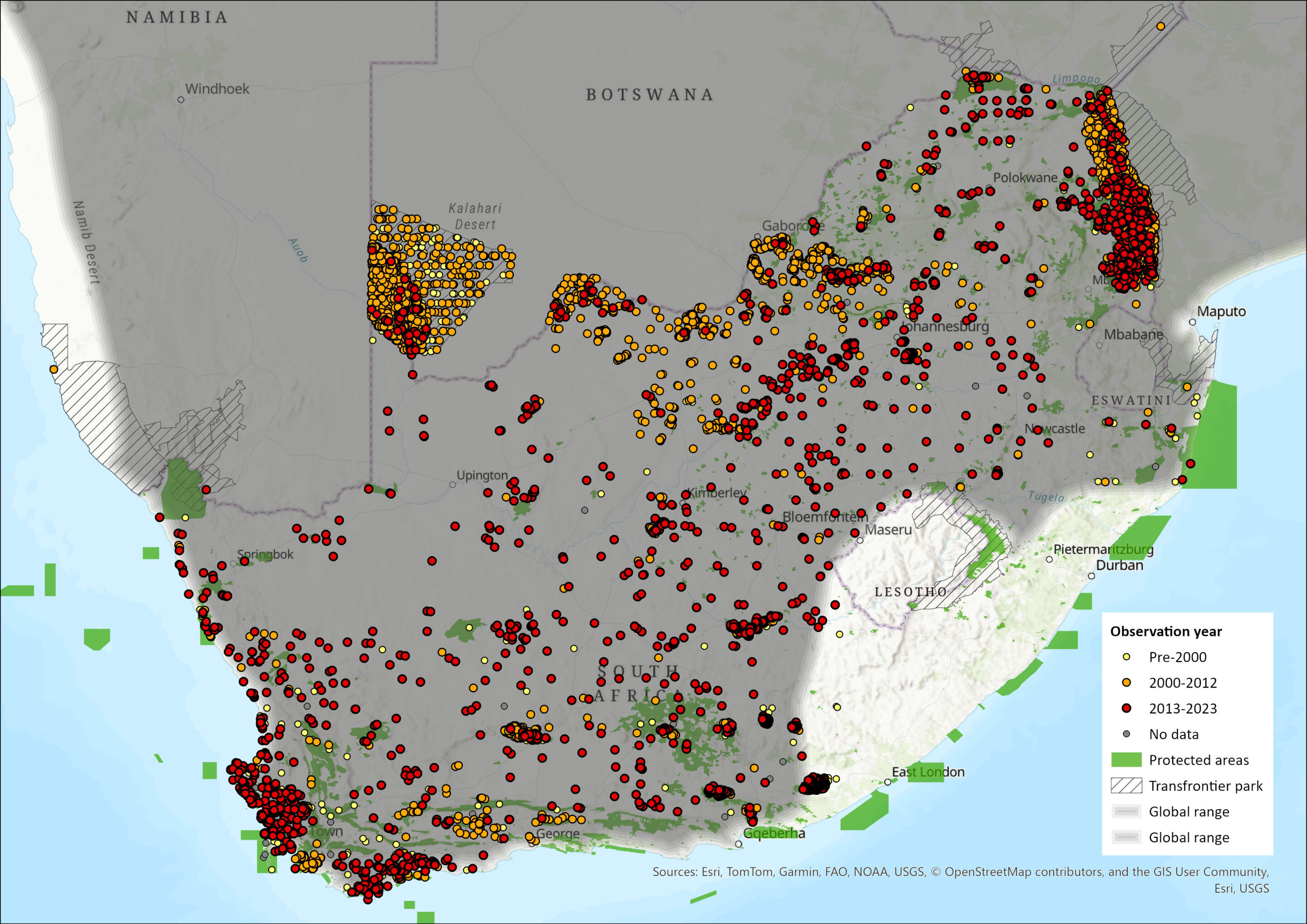
Figure 1. Distribution records for Steenbok (Raphicerus campestris) within the assessment region (South Africa, Eswatini and Lesotho). Note: records are from multiple sources and are not all individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin |
|---|---|---|
| Angola | Extant | Native |
| Botswana | Extant | Native |
| Eswatini | Extant | Native |
| Kenya | Extant | Native |
| Lesotho | Extinct Post-1500 | Native |
| Mozambique | Extant | Native |
| Namibia | Extant | Native |
| South Africa | Extant | Native |
| Tanzania | Extant | Native |
| Uganda | Extinct Post-1500 | Native |
| Zambia | Extant | Native |
| Zimbabwe | Extant | Native |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
The effects of climate change are often intertwined with other anthropogenic effects, making it difficult to evaluate one without the other (Trouwborst & Blackmore 2020). Not only is climate change predicted to alter temperatures, but it is also predicted to cause variability in rainfall (Déqué et al. 2017). Additionally, it is predicted that climate change will alter the frequency and intensity of fires, which will contribute to the alteration of biomes (Department of Environmental Affairs 2015). The changes in vegetation resulting from climate change will inevitably alter the distribution of fauna (Boone 2019). Documented changes in species richness and ecological structure provide significant evidence to indicate the effects of climate change on mammalian herbivores (Badgley 2008). Threats such as habitat loss are accelerated as a result of climate change as certain habitats inhabited by Steenbok become more suitable for agricultural practices (Guo et al. 2017, Curtis 2013, Powell et al. 2007, Pote et al. 2006, Hannah et al. 2013). The influence of climate change as well as the economic climate on farmers and rural communities may lead to an increase in habitat transformation which could be detrimental to the species, especially the transformation of already vulnerable areas (Curtis 2013, Pote et al. 2006). This is concerning considering that small portions of ecosystems of conservation concern occur within protected areas (Curtis 2013, Guo et al. 2017, Pote et al. 2006), resulting in large portions of these ecosystems relying on the management practices of private landowners.
The effect of climate change on a species is dependent on their exposure, sensitivity, resilience and adaptability (Boone 2019). Changes in body condition (Parker et al. 2009), population sizes and reproductive rates (Ellis and Swift 1998) are some of the potential short-term effects that ungulates may face. Even though Steenbok are well adapted to a variety of ecosystems we can predict that they will face habitat loss which may result in population declines and changes in distribution for some localised populations.
Population
A global estimated population size of 600,000 individuals was recorded by East (1999), however, this is considered an underestimate due to the constraints associated with aerial surveys. In areas where Steenbok are common, ground surveys provided density estimates of 0.3–1.0/km² (East 1999) and distance sampling methods produced estimates of 3.78/km² on Benfontein Game Farm and two small-livestock farms in the eastern Northern Cape and western Free State Provinces (Stenkewitz et al. 2010). Similarly, field surveys in the North West Province confirmed an abundant population in the Kalahari vegetation types (Power 2014). However, lower densities are expected in areas without suitable vegetation cover and reliable estimates of population density are currently unavailable across most of its range, due to the cryptic nature of this species (du Toit 2013). Within the assessment region, habitat for this species is fairly continuous and widespread, thus there is suspected to be only one major subpopulation throughout the region. The population may only be declining in certain areas due to hunting by local settlements and farmers. Overall, the population is suspected to be stable.
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| Yes | – | From hunting in some areas and loss of habitat to agriculture and deterioration of grasslands due to scrub encroachment. |
Current population trend: Stable
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: One major subpopulation throughout the assessment region.
Severely fragmented: No
Extreme fluctuations: Steep decline in Hluhluwe-Imfolozi Park from an estimated 235 in 1982 to 3 in 2014 (Cromsigt et al. 2017).
Continuing decline in number of subpopulations: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
The complete nuclear (Chen et al. 2019) and mitochondrial genome (mitogenome) (Hassanin et al. 2012) for R. campestris has been assembled, and after comparisons with the mitogenomes of Cape (R. melanotis) and Sharpe’s (R. sharpei) Grysbok, it was found that the Steenbok is more closely related to Sharp’s Grysbok (R. sharpei is a sister lineage to R. campestris) than the other two species are to each other (de Jager and Lorenzen 2023). This supports previous findings based on the cytochrome b gene only (Mathee and Robinson 1999). In contrast, a consensus tree of eight genes (mitochondrial and nuclear genes) confirmed the sister species relationship between R. melanotis and R. sharpei to the exclusion of R. campestris (Bärmann et al. 2013). While subspecies have been proposed within Steenbok, these have not been validated using molecular methods. Moreover, no population genetic study has been published on the species within the assessment region; therefore, the number and extent of genetically distinct subpopulations is unknown. However, it is assumed animals in the assessment region are comprised of a single metapopulation that extents into adjacent countries. Accordingly, a historical effective population size (Ne) estimated at over 120,000 individuals based on animals from Botswana (de Jager, pers comm, June 2024) is thought to be representative of the genetic diversity historically present in South Africa. This far exceeds the 500 threshold for assigning populations as genetically healthy and stable. However, more contemporary estimates of Ne are needed to confirm whether the species has remained genetically healthy over time.
Habitats and Ecology
Steenbok are well adapted to a range of habitat types, from semi-desert regions to mesic mountainous moorlands, including high altitude areas, such as Mt. Kenya (3,500 m asl) (du Toit 2013). They are generally absent from forests and thick woodland areas (Skinner & Chimimba 2005). Steenbok are also sometimes associated with transitional or disturbed areas due to the formation of novel habitats that they occupy (Winterton, Van Wilgen, & Venter, 2020) Within the Overberg landscape in the Western Cape of South Africa, steenbok have similar occupancy estimates in transformed agricultural landscapes and in De Hoop Nature reserve (Swanepoel, 2021). They also utilise agricultural areas such as crops and pastures as part of their resource selection in these transformed landscapes (Jansen van Vuuren 2021, Swanepoel, 2021). Occurring commonly in the drier grasslands, shrublands and savannahs of southern Africa, this species often favours heavily grazed regions with a high concentration of forbs. These areas frequently form around water sources, although they are largely water-independent (IUCN SSC Antelope Specialist Group 2016). However, in Botswana, Steenbok are known to dig up roots, rhizomes and succulent bulbs to meet their water requirements in dry conditions (Haim & Skinner 1991). Well adapted to dry habitats, this species exhibits low metabolic rates and high overall minimal thermal conductance, allowing it to conserve water when exposed to high temperatures and extreme environmental conditions (Haim & Skinner 1991). Being one of the smallest ruminant browsers, and thus having high mass-specific metabolic needs, Steenbok are reliant on the year-round availability of high-quality forage resources, including geophytes, berries, flowers, green browse material, and indehiscent pods when green foliage is scarce during the dry season (du Toit 2013). In the Kruger National Park (KNP), they depend heavily on the pods of Acacia tortilis in dry conditions (du Toit 1993) and so their key vegetation types include thorn thickets where they find both food and shelter from predators. In the Overberg region steenbok diets were found to contain mainly anthropogenic food sources such as crops or “opslag” and were supplemented with naturally occurring Fynbos such as Hermannia spp. (Jansen van Vuuren 2021, Jansen van Vuuren et al. 2022). Steenbok is a highly adaptable species and can survive in human-modified landscapes providing there is sufficient food resources (Jansen van Vuuren et al. 2022).
Steenbok of both sexes remain within stable home ranges throughout the year. Territories in the Kuise Valley of the Namib were measured as 0.58 km² (Cloete 1983), while in KNP the home ranges of two females were approximately 0.62 km² (du Toit 1990, 1993). They are generally solitary (Skinner & Chimimba 2005), but are not asocial, and are occasionally seen foraging in pairs while the female is in oestrus, or in small groups consisting of an adult male, female and her offspring (Cohen 1997). Steenbok are considered largely diurnal (Stenkewitz et al. 2010), and activity peaks in the early mornings and late afternoons in warm, summer conditions (Walther 1990; Nowak 1991; Skinner & Chimimba 2005). However, nocturnal activity is not uncommon, especially during hot, dry conditions. In fact, in KNP during the dry season, foraging activity constituted 57% of the night, while the majority of diurnal activity included resting and ruminating, and only 33% foraging (du Toit 1993). Giving birth at any time of the year and usually producing a single young, Steenbok exhibit a gestation period of about 168–173 days (Hofmeyr & Skinner 1969).
Ecosystem and cultural services: Although it remains to be investigated, Steenbok consume fruits and pods as an important component of their diet and so it is to be expected that they are significant agents of seed dispersal. Additionally, this is an important prey species for predators such as the Cheetah (Acinonyx jubatus) and Martial Eagle (Polemaetus bellicosus). They are an important component in the roll-out of the “holistic approach” to Damage Causing Animal (DCA) management through the maintenance of natural prey species.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
|---|---|---|---|
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.4. Shrubland -> Shrubland – Temperate | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 4.4. Grassland -> Grassland – Temperate | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: Females attain sexual maturity at about seven months
Age at Maturity: Male: Probably 10-12 months
Size at Maturity: Female: 10.9 kgs
Size at Maturity: Male: 11.3 kgs
Longevity: up to nine years in captivity
Average Reproductive Age: (Not specified)
Maximum Size (in cms): Horn length 19 cms
Size at Birth: Birth weight about 1 kg
Gestation Time: Around 170 days
Reproductive Periodicity: Year-round with birth peak shortly before the rains
Average Annual Fecundity or Litter Size: One
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Territorial
Congregatory: Solitary
Systems
System: Terrestrial
General Use and Trade Information
This species is used, both legally and illegally, by biltong hunters and for bushmeat respectively. Additionally, it is targeted by international trophy hunters, as one of the “Tiny 10” small antelope, but this occurs at low levels. However, there is little national or international commercial value of this species, and there is limited translocation of this species within the game industry, possibly due to its persistence in the landscape. Translocations across bioregional boundaries are actively discouraged through conservation legislation.
In parts of its range (such as mesic savannah regions), wildlife ranching and the private sector may have a positive effect on this species, as the conversion from livestock to wildlife ranches is speculated to have improved habitat conditions and decreased persecution rates. However, this is not always the case, and across much of its range, particularly within arid and fynbos regions, game farming and the introduction of competing species, particularly extralimital and exotic species, may cause local declines of Steenbok. Additionally, the establishment of game farms with high-quality impermeable fences poses a substantial threat to gene flow. Due to its abundance and low commercial value, interest in captive breeding is insignificant. This species is also considered to be density dependent and self-regulating. Land owners are encouraged to monitor persistence, density and number of adult males when hunting off-takes are considered.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Illegal bushmeat hunting. | Yes | Trophy hunting, biltong and live sales. |
National Commercial Value: Yes
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | – | – | – |
| 15. Sport hunting/specimen collecting | – | true | – | – |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: Trophy hunting, illegal bushmeat hunting and live sales.
Threats
No major threats have been identified for this species, however, Steenbok are locally susceptible to predation by domestic dogs (Canis familiaris) and subsistence hunters who frequently capture and kill juveniles in particular (when they are found lying alone in cover) for bushmeat (Lynch 1994; du Toit 2013; IUCN SSC Antelope Specialist Group 2016). Although these threats are not suspected to cause range-wide population declines, they can result in local declines or even local extinction (for example, Lesotho). This is likely to apply in South Africa when land tenure is changed from private ownership to communally occupied land and also if the practice of “Taxi Hunting” is not stopped. The increasing decline in capacity of the state-run conservation agencies and their inability to control illegal hunting is a deep concern (Patel et al. 2023).
Habitat fragmentation through the erection of impermeable fences in the development of wildlife or livestock ranches may inhibit gene flow for this species, as well as other small antelopes and similar sized animals. Additionally, overstocking and mismanagement of livestock or wildlife ranches leading to overgrazing and declines in habitat and forage quality may threaten the success of this species. The development of wildlife ranches is likely to affect local Steenbok subpopulations disparately across different bioregions. Within arid regions and the Fynbos Biome, herbivores are historically less diverse and numerous, and ecological niches are narrow (compared to savannah regions); thus, the introduction of extralimital herbivores into these regions is likely to increase competition for local Steenbok populations. Also, loss of grasslands due to bush encroachment caused by utilisation by excessive wildlife numbers and inappropriate management of fire (J Anderson pers comm).
Current habitat trend: Stable, although continued human settlement and habitat conversion for agriculture has caused some local decline in habitat for this species. However, in parts of the Savannah Biome, the expansion of wildlife ranching may lead to an increase in suitable habitat.
Conservation
Steenbok are widespread and occur extensively within protected areas and private ranch lands. Injudicious translocation of this species needs to be addressed: reintroductions and translocations need to source animals from the same ecotypic range. Habitat connectivity across different vegetation types is essential to maintain gene flow and clinal variation within the species. Thus, the formation of conservancies and the promotion of permeable fences is required to ensure ecologically resilient subpopulations of Steenbok.
Monitoring numbers of Steenbok is important for the development of management strategies, investigating population dynamics, and understanding the relationships between predators and prey (Stenkewitz et al. 2010). Due to its cryptic and solitary nature, this species is not suitable for harvesting in any feasible meat-production system, and should not be promoted as a viable species in the wildlife-based rural economy as a source protein. It is likely that Steenbok harvesting would exhibit low financial feasibility and would cause counter-productive disturbance to local Steenbok subpopulations.
Recommendations for land managers and practitioners:
- Promote this species as a natural forage species for indigenous predators as part of the “holistic approach” to the management of damage-causing animals. Apply genetic conservation principles in the management of this ecotypic species.
- Where feasible and practical, ensure that fences are permeable particularly on conservancies/stewardship sites.
- Monitor persistence through the collection of sighting records.
- Monitor population density in areas where this species is hunted.
- Determine the level of clinal variation at a landscape scale through genetic research: collect and bank genetic samples to support genetic research.
- Prevent the unselective “Taxi hunting” with dogs that takes place primarily in KwaZulu-Natal.
Research priorities:
- Effects of wildlife ranching on this species across various bioregions.
- Extent of habitat loss due to expanding agriculture and human settlement.
- Extent of grassland habitat loss by bush encroachment resulting from overgrazing and poor fire management.
- The impact of “Taxi hunting” with dogs in small antelope populations in KwaZulu-Natal.
- Functional properties of Steenbok in seed dispersal.
- Investigations into the relationship between this species and other forage species and damage causing animals, relating specifically to the “holistic approach” to DCA management.
- Map the genetic composition of the southern African population to ensure the development of effective and informed decisions relating to reintroductions and translocations of this species.
- The collection of distribution information to monitor the persistence of this species in the landscape across the Western Cape, as well as ad hoc information currently being collected and stored by CapeNature. This information is used for bioregional planning and to determine data gaps in the Western Cape.
Encouraged citizen actions:
- Report sightings and roadkills on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Landowners should ensure that disturbance of this species and its young is kept to a minimum, particularly with regards to domestic dogs.
- Create conservancies to broaden habitat available for the species.
- Install permeable fences.
- Understand and support the concept of genetic conservation with regards to ecotypic species and their management.
- Report illegal hunting to provincial conservation authorities.
- Submit hunting returns (to enable higher confidences in calculating impacts of hunting and evaluating bag limit size).
Bibliography
Ambrose D, Talukdar S. 2000. Biological Diversity in Lesotho. National Environment Secretariat, Maseru, Lesotho.
Ansell, W.F.H. 1972. Part 2, 15 Family Artiodactyla. In: J. Meester and H.W. Setzer (eds), The Mammals of Africa: An Identification Manual, pp. 1-84. Smithsonian Institution Press, Washington, DC, USA.
Badgley, C., Barry, J.C., Morgan, M.E., Nelson, S.V., Behrensmeyer, A.K., Cerling, T.E. and Pilbeam, D., 2008. Ecological changes in Miocene mammalian record show impact of prolonged climatic forcing. Proceedings of the National Academy of Sciences, 105(34), pp.12145-12149.
Bärmann EV, Rössner GE, Wörheide G. 2013. A revised phylogeny of Antilopini (Bovidae, Artiodactyla) using combined mitochondrial and nuclear genes. Mol Phylogenet Evol. 67(2):484–493.
Boone, R.B., 2019. Weather and climate impacts on browsing and grazing ungulates. The ecology of browsing and grazing II, pp.197-213.
Cloete G. 1983. Etho-ecological aspects of the steenbok (Raphicerus campestris (Thunberg, 1811)) in the Namib desert, South West Africa. M.Sc. Thesis. University of the Orange Free State, Bloemfontein, South Africa
Cohen M. 1997. Steenbok Raphicerus campestris. In: Mills G, Hes L (ed.), The Complete Book of Southern African Mammals, pp. 267. Struik Publishers, Cape Town, South Africa.
Cromsigt. J.P.G.M, S. Archibald and N. Owen-Smith (2017). Conservating Africa’s Mega-Diversity in the Anthropocene: The Hluhluwe-Imfolozi Park Story. 2017. Cambridge University Press. 406pp.
Curtis, O. E. (2013). Management of Critically Endangered renosterveld fragments in the Overberg, South Africa. Department of biological sciences, University of Cape Town. Dotor of Philosophy.
Department of Environmental Affairs. 2015. Climate Change Adaptation Plans for South African Biomes (ed. Kharika, J.R.M., Mkhize, N.C.S., Munyai, T., Khavhagali, V.P., Davis, C., Dziba, D., Scholes, R., van Garderen, E., von Maltitz, G., Le Maitre, D., Archibald, S., Lotter, D., van Deventer, H., Midgely, G. and Hoffman, T). Pretoria.
Déqué M, Calmanti S, Christensen OB, Aquila AD, Maule CF, Haensler A, Nikulin G & Teichmann C. 2017. A multi-model climate response over tropical Africa at +2°C. Climate Services, 7, 87–95.
East, R. (compiler). 1999. African Antelope Database 1998. IUCN, Gland, Switzerland and Cambridge, UK.
Guo, D., et al. (2017). “Impact of the Future Changing Climate on the Southern Africa Biomes, and the Importance of Geology.” Journal of Geoscience and Environment Protection 05(07): 1-9.
Haim, A. and Skinner, J.D. 1991. A comparative study of metabolic rates and thermoregulation of two African antelopes, the steenbok Raphicerus campestris and the blue duiker Cephalophus monticola. Journal of Thermal Biology 16: 145-148.
Hannah, L., Roehrdanz, P.R., Ikegami, M., Shepard, A.V., Shaw, M.R., Tabor, G., Lu, Z., Marquet, P.A. and Hijmans, R.J. 2013. Climate change, wine, and conservation. Proceedings of the National Academy of Sciences of the United States of America 110: 6907-6912.
Hofmeyr JM, Skinner JD. 1969. A note on ovulation and implantation in the steenbok and the impala. Proceedings of the South African Society of Animal Production 8: 175.
IUCN SSC Antelope Specialist Group. 2016. Raphicerus campestris.The IUCN Red List of Threatened Species 2016: e.T19308A50193533
Jansen van Vuuren, A., 2021. The feeding ecology and habitat selection of small antelopes in the Overberg Renosterveld, Western Cape, MSc Dissertation, Department of Conservation Management, Faculty of Science, Nelson Mandela University, George Campus, Madiba Drive, George, South Africa.
Jansen van Vuuren A., Fritz H,, Venter J.A. 2022. Five small antelope species diets indicate different levels of anthrodependence in the Overberg Renosterveld, South Africa. African Journal of Ecology 60: 917-925.
Kingdon, J. 1997. The Kingdon Field Guide to African Mammals (first edition}. Academic Press, San Diego, California, USA.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
Matthee CA, Robinson TJ. 1999. Cytochrome b phylogeny of the family Bovidae: resolution within the Alcelaphini, Antilopini, Neotragini, and Tragelaphini. Mol Phylogenet Evol. 12(1):31–46.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum , Pretoria, South Africa.
Patel. T, Cowan, O., Little, I., Friedmann, Y. & Blackmore, A. 2023. The State of Provincial Reserves in South Africa, Challenges and Recommendations. https://ewt.org.za/resources/cpsu-programme/
Pote, J.M., Shackleton, C.M., Cocks, M.L., & Lubke, R.A. (2006). Fuelwood harvesting and selection in Valley Thicket, South Africa. Journal of Arid Environments, 67, 270-287.
Powell, M.B., Mills, A.J., & Marais, C. (2007). CARBON SEQUESTRATION AND RESTORATION : CHALLENGES AND OPPORTUNITIES IN SUBTROPICAL THICKET.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Stenkewitz U, Herrmann E, Kamler JF. 2010. Distance sampling for estimating springhare, cape hare and steenbok densities in South Africa. South African Journal of Wildlife Research 40: 87-92.
Walther FR. 1990. Duikers and dwarf antelopes. In: Parker SP (ed.), Grzimek’s Encyclopedia of Mammals, pp. 325-343. McGraw-Hill, New York, USA.
Swanepoel, H.I.J. 2021. The impact of transformed landscapes on the resource selection and occupancy of specialized browsing species in a lowland fynbos and renosterveld ecosystem. MSc Dissertation, Department of Conservation Management, Faculty of Science, Nelson Mandela University, George Campus, Madiba Drive, George, South Africa.
du Toit JT. 1990. Home range – body mass relations: a field study on African browsing ruminants. Oecologia 85: 301-303.
du Toit JT. 1993. The feeding ecology of a very small ruminant, the steenbok (Raphicerus campestris). African Journal of Ecology 31: 35-48.
du Toit, J. T. 2013. Raphicerus campestris Steenbok. In: J. S. Kingdon and M. Hoffmann (eds), The Mammals of Africa, pp. 311-314.. Bloomsbury, London, UK.
Trouwborst, A. and Blackmore, A., 2020. Hot dogs, hungry bears, and wolves running out of mountain—international wildlife law and the effects of climate change on large carnivores. Journal of International Wildlife Law & Policy, 23(3), pp.212-238.
Winterton, D. J., Van Wilgen, N. J., & Venter, J. A. (2020). Investigating the effects of management practice on mammalian co-occurrence along the West Coast of South Africa. PeerJ, 2020(1),
