
Stanley’s Serotine
Laephotis stanleyi

2025 Red list status
Data Deficient
Regional Population Trend
Unknown
Change compared
to 2016
New Assessment
Overview
Laephotis stanleyi – Goodman, Kearney, Michèle, Ratsimbazafy & Hassanin, 2017
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – VESPERTILIONIDAE – Laephotis – stanleyi
Common Names: Stanley’s Serotine Bat (English)
Synonyms: Neoromicia stanleyi Goodman et al. 2017. Eptesicus melckorum Hayman & Hill 1971, in part. Eptesicus (Neoromicia) melckorum Koopman 1993, in part. Neoromicia melckorum Simmons 2005, in part. Neoromicia cf. melckorum Monadjem et al. 2010. Pipistrellus (Neoromicia) melckorum Hill & Harrison 1987, in part. Pipistrellus cf. melckorum Rautenbach et al. 1993. Pipistrellus cf. melckorum Kearney 2013a.
Taxonomic Note:
This previously unknown species was known as Neoromicia. cf. melckorum, before being named as a formal species by Goodman et al. in 2017. The species is named in honour of William Stanley, the mammal collection manager at the Field Museum of Natural History who conducted decades of research on African small mammals. The holotype specimen was collected in 2009 in the Okavango Delta of Botswana by South Africans, Dr Teresa Kearney and Ernst C.J Seamark. More recent research has now placed it in the genus Laephotis (Monadjem et al. 2020a) along with L. capensis which it previously formed a species complex with. Monadjem et al. 2020 conducted a comprehensive taxonomic revision of pipistrelle-like bats in East Africa and, among other findings, provided molecular evidence that Neoromicia stanleyi more appropriately belongs to the genus Laephotis rather than Neoromicia.
Red list status: DD – Data Deficient (IUCN version 3.1)
Assessment Information
Assessors: Howard, A.1, da Silva, J.M.2 & Taylor, P.1
Reviewer: Sathyan, R.3
Institutions: 1University of the Free State, 2South African National Biodiversity Institute, 3University of the Western Cape
Assessment Rationale
This species is listed as Data Deficient on the Global IUCN Red List with a total of 13 known localities in southern Africa and only 2 records of Laephotis stanleyi reported within the assessment region (Jacobs 2019; Monadjem et al. 2020a). It has only been recorded in the northern parts of Kruger National Park in South Africa which is assumed to be the southern part of its distribution range (Goodman et al. 2017; Monadjem et al. 2020a). The EOO for this species is 1,77 km2. The population size, movements and reproduction biology of this species is currently unknown. Thus, the species is also listed as Data Deficient within the assessment region.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: New assessment
Regional Distribution and occurrence
Geographic Range
All records of this species in the assessment region are only known from the far northeastern corner of Kruger National Park, Limpopo Province where it was recorded at multiple camps in the mid-1980s (Goodman et al. 2017). The estimated extent of occurrence within the assessment region is 1,77 km².
In addition to South Africa, the species has been recorded in Botswana, Zambia and Zimbabwe and may occur in Malawi (Goodman et al. 2017; Monadjem et al. 2020b). The type locality for this species is from the Okavango Delta, Gomoti River, Chitabe (950 m asl) in Botswana where it was captured in April 2009 with the holotype an adult male (Goodman et al. 2017).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
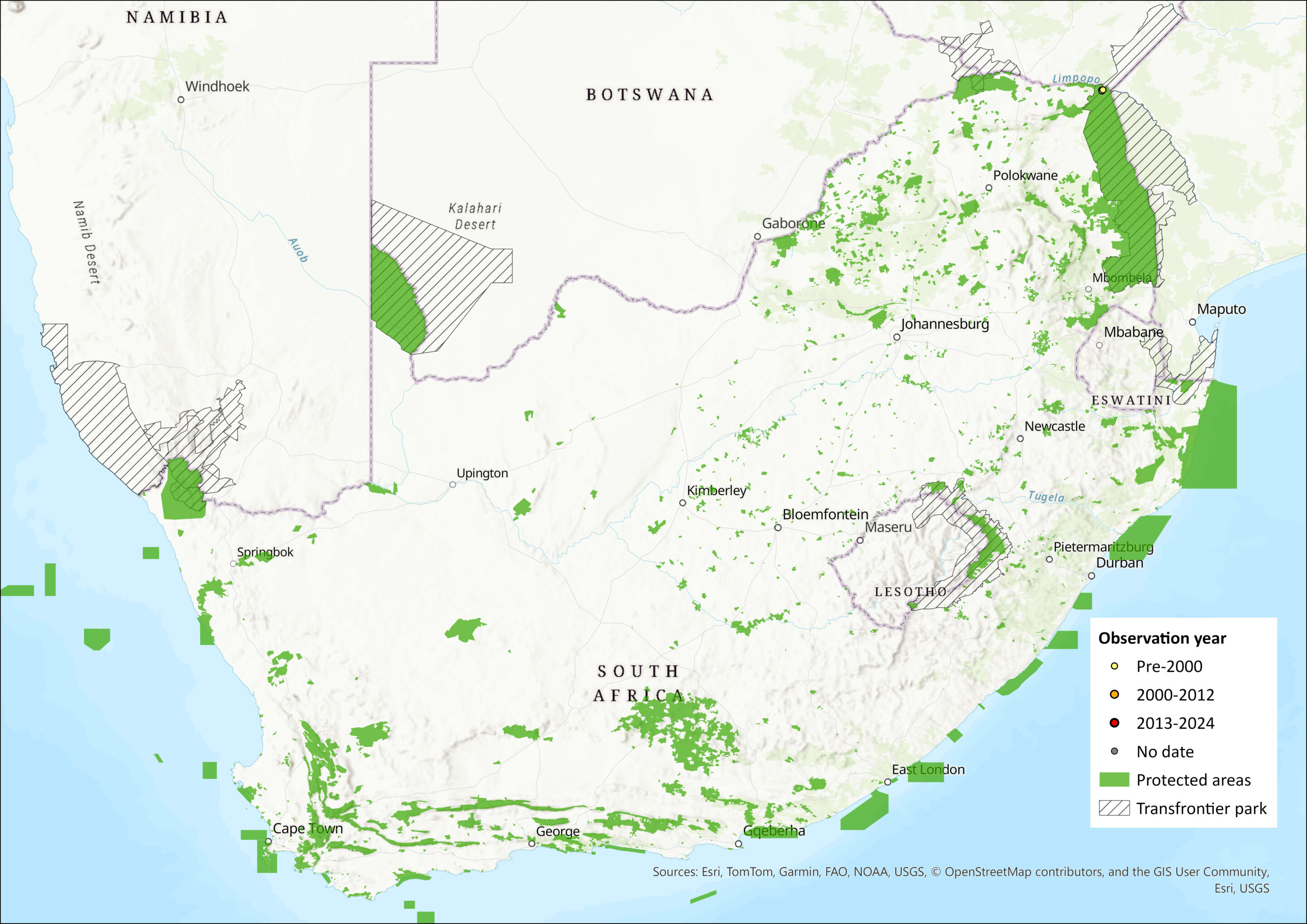 Figure 1. Distribution records for Stanley’s Serotine Bat (Laephotis stanleyi) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Stanley’s Serotine Bat (Laephotis stanleyi) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Savannah
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Botswana | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
No formal study on the direct effects of climate change on this species has yet been conducted. However, due to the increasing temperatures across most of the species’ distribution, the roosting preferences and foraging areas are predicted to shift (Pacifici et al. 2018). Most studies on the effects of climate change on bats are based on predictive species distribution modelling, thus there is a lack of empirical studies measuring behavioural, physiological, phenological or genetic responses to extreme and seasonal climatic changes, especially in the Global South (Festa et al. 2023; Pio et al. 2014). Globally, there have been documented declines in bat populations, species richness and distributions in relation to water availability with increasing global aridity which may become a growing concern as heat waves and maximum temperatures are expected to increase over much of sub-Saharan Africa (Adams & Hayes 2021; Mbokodo et al. 2020; Pacifici et al. 2018).
Recent ecological niche-modelling research underscores that temperature and precipitation are the most influential environmental determinants of bat distributions across South Africa. Under projected climate change scenarios, many savanna and grassland associated bat species are expected to experience significant range contractions. Given that Laephotis stanleyi is presumed to inhabit these biomes, it is likely to be especially vulnerable to shifts in rainfall regimes and rising temperatures, which may impair insect prey availability and diminish suitable roosting and foraging environments (Schoeman & Jacobs 2011; Cooper-Bohannon et al. 2016).
Population information
The population size, movements and reproduction biology of this species is currently unknown.
Current population trend: Unknown
Continuing decline in mature individuals: Unknown
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: Unknown
All individuals in one subpopulation: Assumed to form a single population
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Recent phylogenetic analyses have led to a taxonomic revision of the genus Laephotis, expanding it to include both long-eared species traditionally placed in Laephotis and certain short-eared species previously assigned to Neoromicia. Laephotis stanleyi, recently described, is resolved as sister to the broader Laephotis group comprising both long- and short-eared taxa. This reclassification is supported by molecular phylogenetics, bacular morphology, and skull morphometrics, suggesting that ear length may evolve rapidly and is not a reliable trait for defining generic boundaries. All members of this revised genus, including L. stanleyi, are endemic to sub-Saharan Africa, including Madagascar (Monadjem et al. 2021).
More finescale population genetic work has unfortunately not been undertaken. While it is assumed these bats form a single population, insufficient information exists on population size or structure to make any informed inference. Further research and surveys are needed to gain better insight into the population size, movements and reproduction of this species.
Habitats and ecology
There is limited information on the habitat requirements or roost selection sites for this species. It has been recorded in miombo woodland and riparian habitat but is sparsely distributed in these low elevation, hot wooded habitats (Monadjem et al. 2020a). Insect prey of Coleoptera, Hemiptera, Diptera and Lepidoptera orders were reported in the diet of this insectivorous species (Aldridge & Rautenbach 1987; Fenton et al. 1998). It is assumed to require crevices, typical of the genus and may be overlooked in acoustic surveys due to the one known echolocation call reported of 39-41 kHz overlapping with the much more common and widespread L. capensis (Kearney et al. 2013; Monadjem et al. 2020).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
Life History
Generation Length: Unknown
Age at Maturity: Female or unspecified: Unknown
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Female: Forearm 3.4-3.9 cm
Size at Maturity (in cms): Male: Forearm 3.4-3.9 cm
Longevity: Unknown
Average Reproductive Age: Unknown
Maximum Size (in cms): 9.9cm total length
Size at Birth (in cms): Unknown
Gestation Time: Unknown
Reproductive Periodicity: Unknown
Average Annual Fecundity or Litter Size: Presumed one based on average for other species in this genus
Natural Mortality: Unknown
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Unknown
Congregatory: Unknown
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
The threats to this species are currently unknown but presumably buffered within the protected areas of occurrence. General threats of insect prey decline, loss of roosting sites and foraging habitat is predicted to be of growing concern.
Conservation
In the assessment area, the species is only present in the protected area of Kruger National Park. Key interventions for this species include field and acoustic surveys targeted at recording this species as well as habitat conservation.
Recommendations for land managers and practitioners:
- Report discoveries of roost sites and ensure roosts are protected.
Research priorities:
- Determine the range of echolocation call parameters of this species to confirm distribution and occurrence.
- Field surveys to discover new roost sites and investigate habitat ecology and reproductive biology.
- Investigate the feasibility of using eDNA and non-invasive techniques to distinguish L. stanleyi from L. capensis.
- Monitoring of known subpopulations to establish population trend.
- More comprehensive studies are required to assess possible threats to the species. For example, there are no data available for the types of roosting sites utilised by this species, which undermines the ability to predict which sites may be vulnerable to human disturbance or habitat alteration. Like other insectivorous bats, the use of pesticides in agricultural landscapes may diminish its prey base.
Encouraged citizen actions:
- Limit disturbance to roost sites.
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP).
Bibliography
Aldridge HDJN, Rautenbach IL. 1987. Morphology, echolocation and resource partitioning in insectivorous bats. The Journal of Animal Ecology 56: 763–778.
Brinkley ER, Weier SM, Parker DM, Taylor PJ. 2021. Three decades later in the northern Kruger National Park: multiple acoustic and capture surveys may underestimate the true local richness of bats based on historical collections. Hystrix, the Italian Journal of Mammalogy 32(2), doi:10.4404/hystrix-00319-2020.
Fenton, M.B., Cumming, D.H.M., Rautenbach, I.L., Cumming, G.S., Cumming, M.S., Ford, G.S., Taylor, R.D., Dunlop, J.M., Hovorka, M.D., Johnston, D.S., Portfors, C.V., M.C. Kalcounis and Z. Mahlanga. 1998. Bats and the loss of tree canopy in African woodland. Conservation Biology 12:339-407.
Goodman, S.M., Kearney, T., Ratsimbazafy, M.M. and Hassanin, A. 2017. Description of a new species of Neoromicia (Chiroptera: Vespertilionidae) from southern Africa: a name for ‘N. cf. melckorum’. Zootaxa, 4236(2), pp.351-374
Jacobs, D. 2019. Neoromicia melckorum. The IUCN Red List of Threatened Species 2019: e.T44922A22047486. https://dx.doi.org/10.2305/IUCN.UK.2019-3.RLTS.T44922A22047486.en. Accessed on 21 July 2024.
Monadjem A, Demos T., Dalton D., Webala P., Musila S., Kerbis Peterhans J., Patterson B. 2020a. A revision of Pipistrelle-like bats (Mammalia: Chiroptera: Vespertilionidae) in East Africa with the description of new genera and species, Zoological Journal of the Linnean Society 25.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis, 2nd EdUniversity of Witwatersrand Press, Johannesburg.
Pacifici, M., Visconti, P. and Rondinini, C. 2018. A framework for the identification of hotspots of climate change risk for mammals. Global Change Biology, 24(4), pp.1626-1636.
