Spotted Hyaena
Crocuta crocuta

2025 Red list status
Near threatened
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Crocuta crocuta – (Erxleben, 1777)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – HYAENIDAE – Crocuta – crocuta
Common Names: Spotted Hyaena, Laughing Hyaena (English), Gevlekte Hiëna (Afrikaans), Ipisi Enamabala (Ndebele), Impisi (Ndebele, Xhosa, Zulu), Phiri (Sepedi, Sesotho, Setswana), Sentawana, Leliritshwana (Sesotho), Phiri-thamaga, Leholo, Masonokwane (Setswana), Isandawane, Ingcuka-cheya (Xhosa)
Synonyms: Canis crocuta Erxleben, 1777
Taxonomic notes: None
| Red List Status |
| NT – Near Threatened C2a(ii) |
| (IUCN version 3.1) |
Assessment Information
Assessors: Jansen van Vuuren, A1; Nicholson, S.2; Adams, E.C.2 & da Silva, J.M.3
Reviewers: Ferreira, S.4
Contributor: Weise, F.5,6
Institutions: 1Nelson Mandela University; 2Endangered Wildlife Trust; 3South African National Biodiversity Institute, 4SANParks;5Hyaena Specialist Group, 6Zambian Carnivore Programme
Previous Assessors and Reviewers: Hunnicutt, A., Power, J., Lerm, L., Nicholson, S., Mills, M., Camacho, G., Dalerum, F. & Child, M.F.
Previous Contributors: Druce, D., Ferreira, S.M., Hootan, C. & Kraft, C.
Assessment Rationale
The estimated minimum population size ranges from 7,687–8,369 individuals in ten protected areas across the assessment region (estimated primarily through call-up surveys; counts conducted between 2008 and 2014), which yields a total estimated mature population size of 4,228–4,603 individuals (assuming a 55% mature population structure). This total is likely to be an underestimate as not all data from protected areas (statutory and private) are available or known. The largest group occurs in Kruger National Park (KNP) (6,998–7,680 adults and subadults; 2015 count) and KTP (375 individuals – 80 on the South African side; 2009 count). Thus, in KNP alone, there were at least an estimated 3,849–4,224 adults and subadults in 2015 (note that estimates were made using the call-up method, therefore population counts are underestimated), which is estimated to be a genuine increase from 2005 (1,722–1,961 mature individuals) due to increasing prey biomass. However, once further data have been generated, population trends should be measured over three generations (c. 17 years) on a national level.
Additionally, reintroductions throughout their former range, especially into privately protected areas, in South Africa (for example, in the Eastern Cape) are expanding the area of occupancy and increasing the national population size. The previous assessment in 2004 estimated population size as 3,138 individuals (1,725 mature individuals). The population thus appears to have been stable over the past decade at > 1,500 mature individuals but inconsistent surveys and lack of long-term subpopulation trends hamper this conclusion. Additionally, at least two major groups in KwaZulu-Natal have exhibited net declines over the past seven to 12 years of 40–68%. Thus, we list as Near Threatened C2a(ii), under a precautionary purview, as there is a continuing decline in at least some protected areas (and possibly outside protected areas too) and the proportion of mature individuals in one subpopulation (KNP) ranges from 91–95% when working with reliable estimates of population sizes (but is likely to be slightly less as not all subpopulation data are available). More comprehensive, accurate and long-term estimates of subpopulation sizes and trends are needed. This species should be reassessed once such data are available.
Localised threats include persecution, poaching for the traditional medicine trade and trophy hunting (an emerging threat), which may be causing declines in some subpopulations, particularly in KwaZulu-Natal Province. Such threats should be monitored and quantified. Similarly, we urge further collation of long-term subpopulation data, especially outside protected areas, to quantify population trends more accurately. The success of the transfrontier parks has ensured that the population is stable. However, the majority of their range is fragmented and the rest of the population (besides KNP and KTP) exists in small, isolated subpopulations. Interventions to combat snaring and unsustainable persecution or trophy hunting, combined with continued reintroduction into private protected areas, are recommended.
Regional population effects: There is dispersal between the assessment region and neighbouring countries, particularly across KTP, Greater Limpopo Transfontier Park (GLTP) and Greater Mapungubwe Transfrontier Conservation Area (GMTFCA). However, the number of immigrants is not suspected to be enough to significantly enhance the population.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Jansen van Vuuren A, Nicholson S, Adams EC & da Silva JM. 2025. A conservation assessment of Crocuta crocuta. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Throughout the continent, Spotted Hyaenas have a wide distribution in regions south of the Sahara, but it has, however, shrunk considerably as a consequence of human disturbance (Skinner & Chimimba 2005). In Eswatini, they occur in the lowveld at Mlawula and Hlane National Parks (Skinner & Chimimba 2005). There is no confirmed evidence of their occurrence in Lesotho (Grab & Nash, 2022).
Presently, within the assessment region, the species is largely confined to protected areas. The free-roaming range extends patchily across the lowveld of Mpumalanga and Limpopo provinces (including KNP), and into the Limpopo valley, where they occur in the GMTFCA, and other reserves, such as De Beers Venetia Limpopo Nature Reserve (Venetia), Vhembe, and surrounding farms. In the North West Province they were reintroduced onto Madikwe Game Reserve where some residents were already present (Hofmeyr 1997), and they occur naturally outside the park in the Dwarsberg range (Power 2014), where they were absent in the 1970s from this area (Rautenbach 1982), perhaps having dispersed from Botswana (Power 2014). Vagrants from Botswana occasionally enter the country across the Molopo fossil riverbed in western North West Province (Power 2014), and the Northern Cape Province (Stuart 1981), while they are resident in KTP (Mills 1990; Skinner & Chimimba 2005). There have also been recent sightings of a free-roaming clan in the Waterberg, Limpopo Province (F. Dalerum and C. Jordaan pers. comm. 2015).
In northern KwaZulu-Natal Province, they occur in the larger game reserves (Deane 1962; Whateley & Brooks 1978; Whateley 1981): Mkhuze Game Reserve, Hluhluwe- iMfolozi Park (HiP), Ndumo Game Reserve, Ithala Nature Reserve and Tembe Elephant Park. In the Eastern Cape Province, they have been reintroduced into the Greater Addo Elephant National Park and Pumba Reserve (Hayward et al. 2007a, 2007b). The reintroduction was done to restore a historically extirpated population (Stuart 1981; Hayward et al. 2007b). In general, this species has been reintroduced for ecotourism purposes across most provinces, with most reintroductions being in the Eastern Cape.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
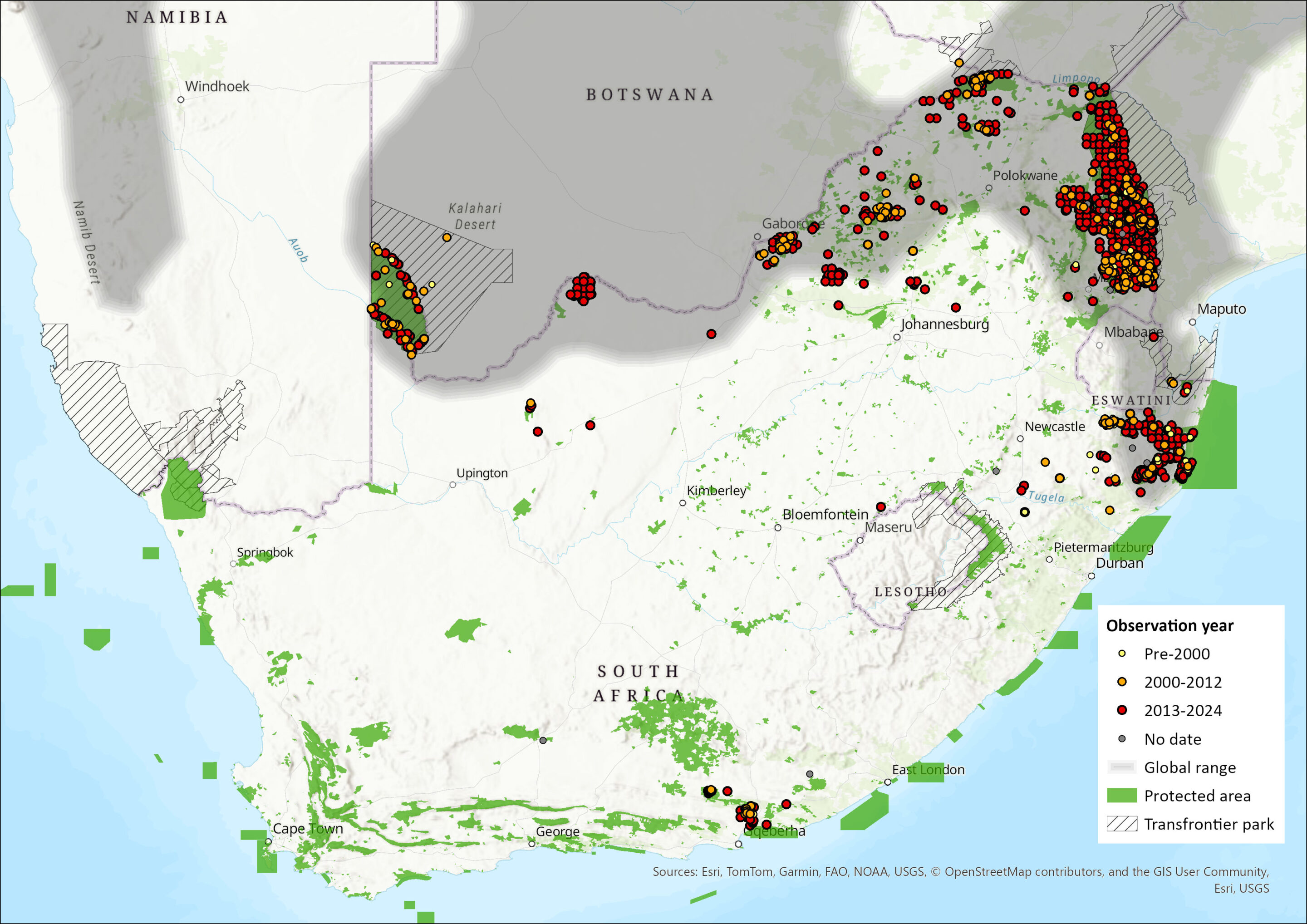
Map
Figure 1. Distribution records for Spotted Hyaena (Crocuta crocuta) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Algeria | Possibly Extinct | Native | – | – |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Djibouti | Extant | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Eritrea | Possibly Extinct | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Possibly Extinct | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Not only is climate change predicted to affect global temperatures, but it is also expected to cause variability in rainfall (Déqué et al. 2017). Rainfall has a significant impact on the populations and habitat suitability of Spotted Hyaenas because of the influence it has on prey populations which affect their feeding behaviour, demography, recruitment and human conflict intensity (Kolowski & Holekamp 2009).
Cooper, Holekamp & Smale (1999) found that changes in rainfall had an immediate effect on the feeding behaviour of hyaenas as it affected prey availability and the dynamics of migration. Predators have better access to prey during periods of low rainfall as prey are significantly easier to hunt when body condition and anti-predator defences are on the decline (Knight 1995; Owen-Smith & Mills 2008). Inversely, predators experience increased body condition, fecundity and survival rates during periods of low rainfall (Ferreira & Viljoen, 2022).
A study conducted in KNP by Ferreira and Viljoen (2022) used the 2015/2016 droughts to assess the potential effects of climate variability on large carnivores. They found that Spotted Hyaenas increased or maintained their abundances 18 months after a drought.
High rainfall negatively affects recruitment in Spotted Hyaenas as a result of an increase of juvenile mortality (Watts & Holekamp 2009). Some theories behind this response are juveniles drowning in den flooding incidents and in increase in disease transmission during high rainfall periods (Watts & Holekamp 2009). However, Watts & Holekamp (2009) indicate that it is more likely that the influence of rainfall on recruitment is from retaliatory killings of either the juveniles, or starvation (when their mothers are killed) rather than being solely from den flooding or disease infection.
Population increases following periods of drought result in an increase of young dispersal males. These dispersal males are likely to depredate on livestock outside the park boundaries, resulting in conflict with humans (Ferreira & Viljoen 2022). Livestock depredation is strongly and positively correlated with rainfall, and local pastoralists kill hyaenas in retaliation of livestock depredation (Abade et al. 2014).
Population information
The global population is between 27,000 and 47,000 individuals (Bohm & Höner 2015). Most subpopulations in protected areas in southern Africa are considered stable, whereas many subpopulations in eastern and western Africa, even in protected areas, are declining, mostly due to incidental snaring and poisoning (Hofer et al. 1996; Hofer & Mills 1998a). The encounter rate of these carnivores in some areas is extremely low due to dense vegetation and their nocturnal behaviour, which can make estimating population size difficult.
Within the assessment region, we estimate a population size of 7,687–8,369 individuals in ten protected areas (2008–2015 counts). Frank (1986a) calculated the mean composition of clans as 16.7 adult males, 22 adult females, 13.7 subadults and 18 cubs. This yields an estimated mature population size of 4,228–4,603 individuals using a 55% mature population structure. However, this may be an underestimate of mature individuals as two clans in KNP have an average of 89% (range: 69–100%) and 73% (range: 63–100%) individuals > 14 months (assuming a c. 12 month age of maturity) during 31 and 15 months of monitoring (L. Belton pers. comm. 2016). Using 89% as the mature population structure yields a mature population of 6,841–7532 individuals. Additionally, the mature population size is an underestimate as 1) data were not available for some protected areas (either statutory or private) containing Spotted Hyaena subpopulations; and 2) most estimates were call-up rather than capture-recapture surveys. Estimating subpopulation size through capture–recapture methods (using camera traps) may be a more effective survey method in smaller areas than call-ups. For example, the subpopulation in Phinda Private Game Reserve, where the known subpopulation is 27 adult hyaenas, was estimated as 24 ± 5.6 individuals using capture-recapture compared to 19 ± 5 individuals using call-up surveys (C. Hootan unpubl. data; 2012 count). The corollary of this finding is that call-up surveys may thus underestimate subpopulation size and the total population of Spotted Hyaenas may be larger than currently estimated. However, Ferreira and Funston (2016) show that a calibrated call-up survey increases the precision of estimates and adjusts to non-response similarly to distance sampling. The previous assessment estimated a population size of 3,138 (Friedmann & Daly 2004), which yields a mature population size of at least 1,569 (using a 50% mature population structure). Thus, we infer the national population to have been stable at above 1,500 mature individuals over the past decade. However, the KwaZulu-Natal subpopulations are showing localised declines (see below).
The two major subpopulations of Spotted Hyaena within the assessment region exist in KNP and KTP. Using call-up surveys, Mills et al. (2001) estimated Spotted Hyaena abundance in KNP as 2,668 individuals (surveys conducted between 1984 and 1989). Similarly, using call-up surveys, Ferreira et al. (2010, 2013) estimated the subpopulation to range from 2,000 to 5,340 individuals with a most likely size of 3,667 (3,443–3,891) individuals in 2008 (Ferreira & Funston 2016). This was a slight increase from the 2005/2006 count of 3,348 (3,131–3,566) individuals (Ferreira & Funston 2016). Friedmann and Daly (2004) stated the KNP subpopulation as being 2,500 individuals while Bohm and Höner (2015) quote it as ranging between 1,300 and 3,900 individuals. The most recent and accurate estimate is 7,339 (6,998–7,680) individuals (S. Ferreira unpubl. data) in 2015. This is thought to be a genuine increase (along with an increase in Lion Panthera leo numbers) between 2005 and 2015 as the same methodology was used and is correlated with an increase in prey biomass (Ferreira & Funston 2016; S. Ferreira pers. comm. 2016). The KNP subpopulation does not appear to be threatened but could decline through the outbreak of diseases and/or climatic changes (Ferreira & Funston 2016). The subpopulation on the South African side of KTP is inferred to have remained stable at c. 80 adults since 1984 (Mill 1990), while an estimate for all of KTP is c. 375 individuals (2009 count; Ferreira et al. 2013). KNP holds the majority of mature individuals within the assessment region, ranging from 91% to 95% of the total population (using the KNP subpopulation range and including all of the KTP subpopulation or the South African side only).
In KwaZulu-Natal, the subpopulations appear to be exhibiting mixed trends. In HiP, while a call-up survey in 2003 and 2004 revealed a subpopulation size of 327 and 331 individuals respectively (J. Graf unpubl. data), repeat surveys (using the same methods) in 2010, 2013 and 2015 estimated 301, 126 and 108 individuals respectively (D. Druce unpubl. data), which indicates a 68% decline over 12 years. The decline could be real or spurious (hyaenas not responding to the call-ups) but is suspected to be genuine (D. Druce pers. comm. 2016). Population declines were also seen in Mkhuze Game Reserve with density estimates from capture-recapture data decreasing from 12.1 individuals per 100 km2 in 2008 to 7.2 individuals per 100 km2 in 2015 (A. Hunnicutt unpubl. data), which represents a 40% decline over seven years. Conversely, the subpopulation at Phinda Private Game Reserve has been shown to be increasing between 2007 and 2014 (using capture-recapture surveys) as subpopulation estimates for 2007, 2009, 2011, 2012 and 2014 are 11, 18, 30, 24 and 46 respectively, although this may be due to dispersal into the reserve from other areas (A. Hunnicutt unpubl. data). In Ndumo Game Reserve, seven individuals have been positively identified, of which three were cubs (L. Lerm unpubl. data; 2010 survey). Spotted Hyaenas have been reintroduced to Tswalu Kalahari Private Game Reserve in 2019 (Van Dyk 2022). This species has been reintroduced to the Waterberg in 2019 (Annear et al. 2023). There is a subpopulation on Venetia Limpopo Nature Reserve, but numbers are uncertain (D. MacFadyen pers. comm. 2016). Subpopulation trends should be estimated over three generations, where generation length has been calculated as 13.8 years (Pacifici et al. 2013), which yields a 41.5 year three generation period; or 5.7 years (Bohm & Höner 2015), which yields a 17-year three-generation period. We accept the latter estimate.
Groups outside protected areas in the Limpopo and North West provinces are probably stable too with the potential to be naturally re-colonised by emigrants from protected areas or immigration from Botswana. For example, Power (2014) noted the existence of a free-roaming group on the periphery of a reserve in the Dwarsberg hills, which are either escapees from Madikwe Game Reserve or immigrants from Botswana.
Population Information
Current population trend: Stable, but with localised declines.
Continuing decline in mature individuals: No overall, but localised declines.
Number of mature individuals in population: 4,228-4,603
Number of mature individuals in largest subpopulation: 3,849-4,224
Number of Subpopulations: At least 10
Severely fragmented: No. Most subpopulations are fenced adjacent to the Kgalagadi and Greater Limpopo Transfrontier Parks.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Analysis of the paleogenomes for the Spotted Hyaena and Eurasian Cave Hyaena was able to investigate the phylogeographic relationships between these two species which are thought to have diverged approximately 2.5 Mya (Yang et al. 2020). Evidence of bidirectional gene flow between the continents was detected (through introgression of several loci indicating substantial admixture) likely complicating prior taxonomic classifications. While no population genetic study has been conducted to date on the Spotted Hyaena, the paleogenome will be useful in future population genomics studies.
Despite the lack of population genetic information, knowledge that animals (free-roaming and in reserves) are capable of moving between areas, with immigration from neighbouring countries also possible, it is likely the species exists as a genetic metapopulation with some level of gene flow present across the range, likely showing a pattern of isolation by distance. If this is correct, the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF’s) complementary genetic indicator – proportion of populations maintained (PM) – can be estimated and receive an indicator value of 1.0 (1/1 populations remaining). A population genetic study is needed to verify this. Additionally, knowledge on the origins of the animals used for reintroductions into reserves would be highly informative, as would the inclusion of genetic material from museum specimens in areas where they were extirpated.
Assuming a single metapopulation and considering a population size of 7,687–8,369, the effective population size (Ne) for the species is estimated to exceed 768-2,510 individuals (based on a conversion ratio of Ne/Nc of 0.1-0.3). This would indicate that the species is genetically healthy and stable. The headline genetic indicator in the GBF – proportion of populations within a species with Ne > 500 – would therefore receive a score of 1.0 (1/1 populations with Ne > 500). If additional genetically distinct subpopulations exist, this estimate would be reduced. It is therefore highly recommended a population genetic study be undertaken to obtain a more precise estimate of the genetic structure and diversity within the species.
Habitats and ecology
Although the Spotted Hyaena is predominantly a savannah species, it has been found to occur in most habitat types including semi-desert, thicket, open woodland and dense dry woodlands (Skinner & Chimimba 2005). In many parts of its range, it occurs in close association with human habitation.
Population densities based on systematic censuses vary substantially from 0.006–1.7 individual(s) / km² across Africa (Frank et al. 1995; Hofer & Mills 1998a, 1998b). Spotted Hyaenas are slow breeders that repopulate areas gradually (Henschel 1986), which was demonstrated when hyaenas were culled from certain areas in KNP (Smuts 1979). Spotted Hyaenas societies are more complex than other carnivore species. They are social animals which live in multi-generational social groups called clans which can vary in size. In the Serengeti, clan sizes can be as large as 47 individuals (Skinner & Chimimba 2005); while in the nearby Masai Mara, clan sizes could reach as high as 79 (Watts & Holekamp 2009). Clan sizes range from 9 to 18 individuals in the lowveld of Limpopo and Mpumalanga (Henschel 1986; Henschel & Skinner 1987), and in KNP densities can be 0.02–0.2 individual / km² (Mills et al. 2001). In KwaZulu-Natal, clan sizes are c. 14, in HiP with densities of 0.36 individuals / km² (Whateley 1981); and in Mkhuze, clan sizes are 6–7, and densities are at 0.13 individual / km² (Skinner et al. 1992). In arid areas, such as KTP, the mean clan sizes are 8 (range 3–12), and densities lower at 0.9 individual / 100 km² (Mills 1984a, 1990). Capture–recapture surveys have estimated a density of 8.8 hyaenas / 100 km2 in Phinda Private Game Reserve, KwaZulu-Natal Province (2014 survey; A. Hunnnicutt unpubl. data) and 6.7 hyaenas / 100 km2 in Mkhuze Game Reserve, KwaZulu-Natal Province (A. Hunnicutt unpubl. data; 2015 survey). In 2021, the minimum population size of Spotted Hyaenas in the Addo Elephant National Park was 31 individuals (excluding cubs) (Chake 2023). By 2023, the population in the main camp section of the Addo Elephant National Park was calculated to be 11.13 (± SE 1.96) individuals / 100 km2, and an abundance of 37 (± SE 6.44) individuals (Visagie et al. 2024). Further population estimates have recently been recorded in Platjan, within the Limpopo province of South Africa, where the density was recorded to be 0.22 individuals / 100 km2 with approximately nine individuals (Faure et al. 2022). The variation in clan sizes could reflect the variation in the prevailing ecological conditions, particularly with respect to Lion populations and prey densities. Spotted Hyaena clans are composed of multiple adult females, their immature offspring, and immigrant males (Watts & Holekamp 2009). The clan is led by a dominant female called the matriarch and generally in the clan, females dominate even the males (Frank 1986b; Watts & Holekamp 2009). Sexual dimorphism is evident as females are larger and heavier than males and even have the resemblance of male external genitalia. Generally, females will remain with their natal clan while most males will emigrate from their natal clans after they become reproductively mature, usually occurring from 2–5 years of age (Holekamp et al. 2012). The mating system is highly polygynous and only the behaviourally dominant male mates, though all other resident males regularly court females (Frank 1986b). The species is a non-seasonal breeder but exhibits a peak in reproduction in the late summer (Skinner & Chimimba 2005). Females generally have litters of one or two cubs (Holekamp et al. 2012). Cubs remain in the clan’s den for 9–14 months and are generally weaned when they are 12–18 months, later than most other carnivores (Holekamp et al. 2012).
While it was previously thought that Spotted Hyaenas were typically scavengers, studies show that they are efficient and active predators (Hayward 2006). A meta-analysis study by Hayward (2006) on the dietary preferences of the Spotted Hyaena found that the species prefer prey species within a body mass range of 56–182 kg – similar to that of Lions – revealing a 69% overlap in preferred prey. Results from Hayward (2006) illustrate the flexible and unselective nature of Spotted Hyaena predation; this characteristic is likely the reason for the species’ success, despite a large degree of overlap with Lions. Kudu (Tragelaphus strepsiceros), Blue Wildebeest (Connochaetes taurinus), Red Hartebeest (Alcelaphus buselaphus caama), Waterbuck (Kobus ellipsiprymnus) and Buffalo (Syncerus caffer) are common prey items (Hayward 2006). In the woodlands of the KNP, about half their diet is obtained from kills they make (Mills & Biggs 1993), while elsewhere, in KTP for example, over 60% their diet is comprised of their own kills, of which this comprises 73% of the meat eaten (Mills 1984b). Spotted hyaenas are highly adaptable in their diet and have even been observed feeding on passerine birds in Namibia (Portas & Krofel 2024). Spotted Hyaenas do also frequently steal kills from other carnivores such as Wild Dogs (Lycaon pictus) (Gorman et al. 1998), Cheetah and Brown Hyaena (Parahyaena brunnea) (Mills 1990), as well as Leopards (Panthera pardus) (Mills & Biggs 1993), and thus influence their persistence.
Ecosystem and cultural services: Spotted Hyaenas have been referred to as keystone predators (Holekamp 2006; Trinkel 2009). As Spotted Hyaenas are typically cursorial predators, they tend to select prey that are weak, injured or sick. This supposedly increases the health of prey populations in an ecosystem. Generally, Spotted Hyaenas can survive in ecosystems in which most larger predators cannot; if Spotted Hyaenas were to disappear from an area, it indicates that a system has been severely degraded (Holekamp 2006; Trinkel 2009).
They minimise the spread of disease by devouring carcasses quickly (sensu Butler & du Toit 2002; Ogada et al. 2012), and they have been said to be important in reducing the prevalence of anthrax in KNP to which they are apparently not susceptible (Pienaar 1969).
They feature extensively in African folklore, as they are associated with witchcraft, and are often viewed as the preferred mode of transport for witches. They are also thought to be shape shifters, to rob graves for bodies, and steal livestock. Folklore has ascribed them to be “hermaphroditic” since the time of Aristotle, due to the masculanised genitalia in the females (Neaves et al. 1980; van Jaarsveld & Skinner 1987; Frank 1996), which is a by-product of high levels of circulating testosterone (Racey & Skinner 1979; van Jaarsveld & Skinner 1991), needed to socially dominate other clan members (Mills 1990; Frank 1996).
The whooping call of this species is distinctive (Skinner & Chimimba 2005), and a depiction of a classic African evening, and its haunting call is appealing to visitors in parks and game reserves.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Marginal | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 4.7. Grassland -> Grassland – Subtropical/Tropical High Altitude | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
In Nigeria, Spotted Hyaenas have been found in traditional medicines (Sodeinde & Soewu 1999). In Guinea-Bissau, Spotted Hyaena skins were found for sale in markets (Sá et al. 2012). Within the assessment region, this species is illegally hunted and persecuted outside protected areas as a damage causing animal, and is occasionally controlled by the state authorities. It is also in high demand in traditional medicine (tails, bones, fur, glands, and organs), especially as it is strongly associated with witchcraft (Bohm & Höner 2015). Spotted Hyaenas are easily caught in snares either deliberately or incidentally. In Mpumalanga, many animals outside formally protected areas have been in contact with snares (G. Camacho unpubl. data).
It occasionally occurs on game farms and wildlife ranches: based on telephonic questionnaires, for the North West Province it has a prevalence of 1.4% (N = 214 farms), but the distribution is highly localised (Power 2014), while in the Limpopo valley it is higher at 13% (N = 115 farms), with the majority of reporting for this species being in the Alldays–Pont Drift area, adjacent the Mapungubwe area (Endangered Wildlife Trust unpubl. data). However, this species is not tolerated for the most part on managed game ranches due its impact on high-value game species.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| No | – | Yes | Sold at muthi markets as traditional medicine. Used for commercial trophy hunting. |
National Commercial Value: No
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 3. Medicine – human & veterinary | true | – | – | – |
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
While they are well protected in some conservation areas, subpopulations outside protected areas are subject to human persecution through shooting, trapping and poisoning. Such animals are often captured and kept illegally and subsequent escapees can cause problems (C. Kraft pers. comm. 2016). Similarly, in areas adjacent to protected areas, human-wildlife conflict and associated persecution may threaten local subpopulations (Wittemyer et al. 2008). Additionally, due to the high prevalence of snares in protected areas, it is likely that Spotted Hyaenas are caught as bycatch in snares that are set for bushmeat. This is an important cause of adult mortality in the Serengeti, where snares kill around 400 adult Spotted Hyaenas each year and are responsible for more than half of all adult mortality (Hofer et al. 1996). In KwaZulu-Natal Province, the Ndumo subpopulation is similarly susceptible to poaching where an individual was caught in a snare in 2010 (L. Lerm unpubl. data). In Mkhuze Game Reserve, KwaZulu-Natal Province in 2015, 18.3% of all Spotted Hyaenas caught on camera trap surveys were found to have neck snares or neck snare wounds (A. Hunnicutt unpubl. data). Snares have a severe impact on this species as it has a keen sense of smell and is often bycatch in areas where snares are set for antelope. Human-wildlife conflict could become a larger issue in the KNP, as juveniles and subadults frequently visit anthropogenic sites (Belton et al. 2018). Across Africa, the most prominent causes of anthropogenic induced mortality is poisoning, government lethal control, persecution, roadkill and trophy hunting (McFadden 2022). In South Africa, the leading anthropogenic cause of mortality in Spotted Hyaenas was roadkill (McFadden 2022).
Conservation
Spotted Hyaenas are conserved in a number of protected areas across their southern African range (Skinner & Chimimba 2005), where, in South Africa, KNP and KTP are the most important. A large number of medium sized-game reserves have subpopulations in five provinces in South Africa. Conservation of Spotted Hyaenas should focus on expanding protected areas, especially through transfrontier areas and/or conservation corridors (for example, in northern KwaZulu-Natal; Smith et al. 2008) and creating a managed metapopulation in smaller protected areas. Where the species is found outside of protected areas (or in regions where they occur on the boundaries of protected areas), conflict mitigation measures should be explored to encourage landowners and communities to protect game/livestock rather than persecute predators, such as through the use of livestock guarding dogs (Rust et al. 2013; McManus et al. 2015). Private game farms and protected areas should be encouraged to form conservancies and create areas large enough to sustain large subpopulations to enhance the resilience of the population overall. Public education campaigns should be continued to dispel the myriad myths about this species and to curb their use in traditional medicine, as well as to discourage landowners and communities from shooting/snaring Spotted Hyaenas. Conservation authorities should work with stakeholders to capture ‘perceived’ problem individuals and simulate dispersals to protected areas where they are needed (for example, reserves in North West Province; Power 2014; and Tswalu Kalahari Reserve in Northern Cape Province).
Recommendations for land managers and practitioners:
- Conduct snare removal on private land.
- Implement conflict mitigation measures to protect livestock/game and deter predators.
- Develop a Biodiversity Management Plan and metapopulation strategy to ensure that reintroductions and translocations contribute to Spotted Hyaena conservation and genetic diversity. From a tourism point of view, their distinctive calls are an added drawcard to any given area
Research priorities:
- Comprehensively determine national population size and distribution of free-roaming Spotted Hyaenas, protected subpopulations and occupancy on wildlife ranches.
- Quantify the loss of Spotted Hyaenas to snares, persecution and trophy hunting and determine subpopulation trends.
- Determine the empirical impact of Spotted Hyaenas to livestock/game loss and generate evidence for the effectiveness of interventions.
- Population genetics in order to understand the genetic health of the species.
- Further research to assess the species risk to climate change.
Encouraged citizen actions:
- Contribute to citizen science projects within protected areas (by reporting sightings locations and pride numbers), and report sighting data on virtual museum platforms (for example, iNaturalist and MammalMAP) outside of protected areas (although care must be taken to not include captive animals occurring in predator camps, safari parks, zoos).
- Do not purchase Spotted Hyaena products.
- Landowners should drop fences to form conservancies and reintroduce hyaenas as a tourism draw card.
Bibliography
Abade L, MacDonald DW, Dickman AJ. 2014. Using landscape and bioclimatic features to predict the distribution of lions, leopards and spotted hyaenas in Tanzania’s Ruaha landscape. PLoS One 9(5): e96261.
Annear, E., Minnie, L., Andrew, K., & Kerley, G. I. 2023. Can smaller predators expand their prey base through killing juveniles? The influence of prey demography and season on prey selection for cheetahs and lions. Oecologia, 201(3), 649-660.
Belton, L. E., Cameron, E. Z., & Dalerum, F. 2018. Spotted hyaena visitation at anthropogenic sites in the Kruger National Park, South Africa. African Zoology, 53(3), 113-118.
Bohm T, Höner OR. 2015. Crocuta crocuta. The IUCN Red List of Threatened Species 2015.
Butler JRA, du Toit JT. 2002. Diet of free-ranging domestic dogs (Canis familiaris) in rural Zimbabwe: implications for wild scavengers on the periphery of wildlife reserves. Animal Conservation 5: 29–37.
Chake P. 2023. Population estimates of spotted hyenas in Addo Elephant National Park In SANParks research report 21/22. URL: https://www.sanparks.org/wp-content/uploads/2022/11/Research-Report-2021-2022.pdf.
Deane NN. 1962. The spotted hyaena (Crocuta crocuta). Lammergeyer 2: 26–43.
Cooper SM, Holekamp KE, Smale L. 1999. A seasonal feast: long-term analysis of feeding behaviour in the spotted hyaena (Crocuta crocuta). African Journal of Ecology 37: 149–160.
Déqué M, Calmanti S, Christensen OB, Aquila AD, Maule CF, Haensler A, Nikulin G & Teichmann C. 2017. A multi-model climate response over tropical Africa at +2°C. Climate Services, 7, 87–95.
Faure, J. P. B., Swanepoel, L. H., Cilliers, D., Venter, J. A., & Hill, R. A. 2022. Estimates of carnivore densities in a human-dominated agricultural matrix in South Africa. Oryx, 56(5), 774-781.
Ferreira S, Gaylard, A, Greaver, C, Hayes, J, Cowell C, Ellis G. 2013. Animal abundances in Parks 2012/2013. Scientific Services, SANParks, Skukuza, South Africa.
Ferreira S, Gaylard, A, Greaver, C, Herbst M. 2010. Animal abundances in Parks 2009/2010. Scientific Services, SANParks, Skukuza, South Africa.
Ferreira SM, Funston PJ. 2016. Population estimates of spotted hyaenas in the Kruger National Park, South Africa. African Journal of Wildlife Research 46: 61–70.
Ferreira SM and Viljoen P. 2022. African Large Carnivore Population Changes in Response to a Drought.”African Journal of Wildlife Research 52(1).
Frank LG, Holecamp KE, Smale L. 2015. Dominance, demography and reproductive success of female spotted hyaenas. In: Sinclair ARE, Arcese P. (ed.), Serengeti II: Dynamics, Management and Conservation of an Ecosystem, pp. 364–384. University of Chicago Press, Chicago, USA.
Frank LG. 1986a. Social organization of the spotted hyaena (Crocuta crocuta). I. Demography. Animal Behaviour 34: 1500–1509.
Frank LG. 1986b. Social organization of the spotted hyaena Crocuta crocuta. II. Dominance and reproduction. Animal Behaviour 34: 1510–1527.
Frank LG. 1996. Female masculinization in the spotted hyena: endocrinology, behavioral ecology, and evolution. In: Gittleman JL (ed.), Carnivore Behavior, Ecology, and Evolution, pp. 78–131. Cornell University Press, New York, USA.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. Conservation Breeding Specialist Group (SSC/IUCN) and Endangered Wildlife Trust, Parkview, South Africa.
Gorman ML, Mills MG, Raath JP, Speakman JR. 1998. High hunting costs make African wild dogs vulnerable to kleptoparasitism by hyaenas. Nature 391: 479–481.
Grab SW and Nash D J. 2022. But what silence! No more gazelles…: Occurrence and extinction of fauna in Lesotho, southern Africa, since the late Pleistocene.” Quaternary International 611-612: 87-101.
Hayward MW et al. 2007a. Practical considerations for the reintroduction of large, terrestrial, mammalian predators based on reintroductions to South Africa’s Eastern Cape Province. The Open Conservation Biology Journal 1: 1–11.
Hayward MW. 2006. Prey preferences of the Spotted Hyaena (Crocuta crocuta) and degree of dietary overlap with the Lion (Panthera leo). Journal of Zoology 270: 606-614.
Hayward, M.W., Adendorff, J., O’Brien, J., Sholto-Douglas, A., Bissett, C., Moolman, L.C., Bean, P., Fogarty, A., Howarth, D., Slater, R. and Kerley, G.I.H. 2007. The reintroduction of large carnivores to the Eastern Cape Province, South Africa: an assessment. Oryx 41: 205-214.
Henschel JR, Skinner JD. 1987. Social relationships and dispersal patterns in a clan of spotted hyaenas Crocuta crocuta in the Kruger National Park. South African Journal of Zoology 22: 18–24.
Henschel JR. 1986. Socio-ecology of a spotted hyaena Crocuta crocuta clan in the Kruger National Park. Ph.D. Thesis. University of Pretoria.
Hofer, H. and Mills, M.G.L. 1998a. Worldwide distribution of Hyaenas. In: M.G.L. Mills and H. Hofer (eds), Hyaenas. Status Survey and Conservation Action Plan, pp. 39-63. IUCN/SSC Hyaena Specialist Group. IUCN, Gland, Switzerland and Cambridge, UK.
Hofer, H. and Mills, M.G.L. 1998b. Population size, threats and conservation status of hyaenas. In: M.G.L. Mills and H. Hofer (eds), Hyaenas. Status Survey and Conservation Action Plan, pp. 64-79. IUCN/SSC Hyaena Specialist Group. IUCN, Gland, Switzerland and Cambridge, UK.
Hofer, H., Campbell, K.L.I., East, M.L. and Huish, S.A. 1996. The impact of game meat hunting on target and non-target species in the Serengeti. In: V.J. Taylor and N. Dunstone (eds), The exploitation of mammal populations, pp. 117-146. Chapman and Hal, London, UK.
Hofmeyr M. 1997. Operation Phoenix: the restocking of Madikwe Game Operation Phoenix: the restocking of Madikwe Game Reserve. North West Parks Board, Rustenburg, South Africa.
Holekamp KE, Smith JE, Strelioff CC, Van Horn RC, Watts HE. 2012. Society, demography and genetic structure in the spotted hyena. Molecular Ecology 21: 613–632.
Holekamp KE. 2006. Spotted hyenas. Current Biology 16: R944–R945.
Knight MH. 1995. Drought-related mortality of wildlife in the southern Kalahari and the role of man. African Journal of Ecology, 33, 377–394.
Kolowski J M and Holekamp KE. 2006. Spatial, temporal, and physical characteristics of livestock depredations by large carnivores along a Kenyan reserve border. Biological Conservation 128(4): 529-541.
McFadden, S. 2022. Mortality Analysis and Threats Assessment for the Spotted Hyena (Crocuta crocuta). Master’s thesis, The University of North Carolina at Chapel Hill.
McManus JS, Dickman AJ, Gaynor D, Smuts BH, Macdonald DW. 2015. Dead or alive? Comparing costs and benefits of lethal and non-lethal human–wildlife conflict mitigation on livestock farms. Oryx 49: 687-695.
Mills MG. 1984a. The comparative behavioural ecology of the brown hyaena Hyaena brunnea and the spotted hyaena Crocuta crocuta in the southern Kalahari. Koedoe 27: 237–247.
Mills MG. 1984b. Prey selection and feeding habits of the large carnivores in the southern Kalahari. Koedoe 27: 281–294.
Mills MGL, Biggs HC. 1993. Prey apportionment and related ecological relationships between large carnivores in Kruger National Park. In: Dunstone N, Gorman ML. (ed.), Symposia of the Zoological Society of London, pp. 253–268. Zoological Society of London, London, UK.
Mills MGL, Juritz JM, Zucchini W. 2001. Estimating the size of spotted hyaena (Crocuta crocuta) populations through playback recordings allowing for non-response. Animal Conservation 4: 335–343.
Mills, M.G.L. 1990. Kalahari hyaenas: the comparative behavioural ecology of two species. Unwin Hyman, London.
Neaves WB, Griffin JE, Wilson JD. 1980. Sexual dimorphism of the phallus in spotted hyaena (Crocuta crocuta). Journal of Reproduction and Fertility 59: 509–513.
Ogada DL, Torchin ME, Kinnaird MF, Ezenwa VO. 2012. Effects of vulture declines on facultative scavengers and potential implications for mammalian disease transmission. Conservation Biology 26: 453–460.
Owen-Smith N & Mills MG (2008). Predator–prey size relationships in an African large-mammal food web. Journal of Animal Ecology, 77, 173–183.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Pienaar, U. de V. 1969. Predator-prey relationships amongst the larger mammals of the Kruger National Park. Koedoe 12: 108.
Portas, R., & Krofel, M. 2024. Spotted hyena (Crocuta crocuta) predation on passerine birds in Namibia. Food Webs, 38, e00340.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Racey PA, Skinner JD. 1979. Endocrine aspects of sexual mimicry in spotted hyaenas Crocuta crocuta. Journal of Zoology 187: 315–326.
Rautenbach, I. L. 1982. Mammals of the Transvaal. Ecoplan monograph, N.S. Supplemento 1: 111-211.
Rust NA, Whitehouse-Tedd KM, MacMillan DC. 2013. Perceived efficacy of livestock-guarding dogs in South Africa: implications for cheetah conservation. Wildlife Society Bulletin 37: 690–697.
Skinner JD, Funston PJ, van Aarde RJ, Van Dyk G, Haupt MA. 1992. Diet of spotted hyaenas in some mesic and arid southern African game reserves adjoining farmland. South African Journal of Wildlife Research 22: 119.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smith RJ et al. 2008. Maputaland centre of endemism using biodiversity, economic and threat data. Biological Conservation 141: 2127–2138.
Smuts GL. 1979. Diet of lions and spotted hyaenas assessed from stomach contents. South African Journal of Wildlife Research 9: 19–25.
Sodeinde OA, Soewu DA. 1999. Pilot study of the traditional medicine trade in Nigeria. TRAFFIC Bulletin 18: 35–40.
Stuart, C.T. 1981. Notes on the mammalian carnivores of the Cape Province, South Africa. Bontebok 1: 1-58.
Sá, R.M.M., Ferreira da Silva, M.J., Sousa, F.M. and Minhós, T. 2012. The trade and ethnobiological use of chimpanzee body parts in Guinea-Bissau: implications for conservation. Traffic Bulletin 24: 31-34.
Trinkel M. 2009. A keystone predator at risk? Density and distribution of the spotted hyena (Crocuta crocuta) in the Etosha National Park, Namibia. Canadian Journal of Zoology 87: 941–947.
Watts, H.E. and Holekamp, K.E. 2009. Ecological determinants of survival and reproduction in the spotted hyena. Journal of Mammalogy 90: 461-471.
Whateley A. 1981. Density and home range of spotted hyaenas in Umfolozi Game Reserve, Natal. Lammergeyer 31: 15–20.
Wittemyer, G., Elsen, P., Bean, W.T., Burton, A.C.O. and Brashares, J.S. 2008. Accelerated human population growth at protected area edges. Science 321: 123-126.
Van Dyk, G. 2022. The Hyenas that call Tswalu home. URL: https://tswalu.com/the-hyenas-that-call-tswalu-home/. Date accessed: 25/02/2025.
van Jaarsveld AS, Skinner JD. 1987. Spotted hyaena monomorphism: an adaptive “phallus”. South African Journal of Science 83: 612–615.
van Jaarsveld, A.S. and Skinner, J.D. 1991. Plasma androgens in spotted hyaenas (Crocuta crocuta): influence of social and reproductive development. Journal of Reproduction and Fertility 93: 195–201.
Visagie, M., Davis, R. S., Venter, J. A., & Honiball, T. L. 2024. Using spatial capture‐recapture models to estimate spotted hyaena (Crocuta crocuta) population density and assess the influence of sex‐specific covariates on space use and detection probability. Conservation Science and Practice, 6(9), e13214.
Yang, C., Li, F., Xiong, Z., Koepfli, K. P., Ryder, O., Perelman, P., Li, Q., & Zhang, G. 2020. A draft genome assembly of spotted hyena, Crocuta crocuta. Scientific data, 7(1), 126.