Southern Tree Hyrax
Dendrohyrax arboreus

2025 Red list status
Endangered
Regional Population Trend
Declining
Change compared
to 2016
No Change
Overview
Dendrohyrax arboreus – (A. Smith, 1827)
ANIMALIA – CHORDATA – MAMMALIA – HYRACOIDEA – PROCAVIIDAE – Dendrohyrax – arboreus
Common Names: Southern Tree Hyrax, Eastern Tree Dassie, Eastern Tree Hyrax (English), Boomdassie, Boomdas (Afrikaans), Umqha (isiZulu), Imbila ye ma Hlatsi (siSwati), Perere (Swahili), Baumschliefer, Waldschliefer (German), Daman d’Arbre (French)
Synonyms: Hyrax arboreus A. Smith, 1827
Taxonomic Note:
There are eight subspecies listed, of which only one, D. a. arboreus, occurs in the assessment region (Bothma 1971). This subspecies is endemic to South Africa.
Red List Status: EN – Endangered, C2a(i) (IUCN version 3.1)
Assessment Information
Assessors: Venter, J1., Ehlers Smith, Y2,3.& da Silva, J.4
Reviewer: Raimondo, D.4 & Patel, T.5
Institutions: 1Nelson Mandela University,2Ezemvelo KZN Wildlife,3University of KwaZulu-Natal,4South African National Biodiversity Institute, 5Endangered Wildlife Trust
Previous Assessors: Gaylard, A., Venter, J. & Ehlers Smith, Y
Previous Reviewer: Child, M.F.
Previous Contributors: Roxburgh, L. & Rushworth, I.
Assessment Rationale
This arboreal species is restricted to mature and intact forests within the assessment region in the Eastern Cape and KwaZulu-Natal provinces of South Africa. The estimated extent of occurrence (EOO) is 49,232 km2 and the area of occupancy (AOO), using all suitable forest patches within the EOO, is estimated to be a maximum of 1,598 km2 (SANBI 2018). However, patch occupancy is suspected to be significantly lower, since Southern Tree Hyraxes require specific den-tree species. Surveys undertaken between 2013–2015 reveal that many ostensibly suitable patches are unoccupied, possibly due to encroaching human pressures (for example, disturbance) and habitat degradation (see below). Using only forest patches within 10 km of an occurrence record collected since 2010 yields a minimum AOO estimate of 566 km2.
Although many forests are well protected, there is an inferred continuing decline in the population from forest patch loss and forest quality degradation, especially along the coast, through agricultural and human settlement expansion. For example, there was a 19.7% loss of natural habitat in KwaZulu-Natal from 1994 to 2011 due to agriculture, timber plantations, settlement expansion and mining (with an average loss of 1.2% per year), with the losses mainly occurring in small (< 0.5 ha) forest patches. There was also an estimated 7% loss of natural vegetation in the Indian Ocean Coastal Belt Biome over a 16 year period (three generations) up to 2015, which may impact its ability to adapt to climate change as suitable forest patches shrink and become isolated further. Additionally, from 2000–2013, there was a 5.6% and 6.3% rate of urban expansion in KwaZulu-Natal and the Eastern Cape, respectively, which will increase the frequency of secondary threats such as tree harvesting and poaching, and thus lead to ongoing habitat degradation. As Southern Tree Hyraxes rely on relatively few tree species for nesting and food, the selective removal of such favoured species could disproportionately impact the population. Recent landcover change data for KwaZulu-Natal province as a whole over the period from 2014 to 2020 indicates that there was a 4.5% transformation of natural habitat (DFFE 2022).
This is a low-density, selective species and thus extrapolating known density estimates across the entire forest patch can overestimate population size. However, based on estimated densities in four forest patches (3–7 individuals / km2), mature population size ranges from 750 (confirmed occupied patches at 50% mature structure) to 7,573 (assuming all forest patches occupied at 73% mature structure) with a most likely estimate of 1,094–1,761 individuals. As most forest patches are fragmented, with limited Southern Tree Hyrax dispersal between them, isolated patches are construed as subpopulations with the largest patch/subpopulation estimated to contain 136–465 mature individuals. Due to their small population size and ongoing population decline we list this species as Endangered under C2a(i). Key interventions for this species to prevent further loss or degradation of forest patches include protected area expansion to connect remaining forest patches, metapopulation management, education and awareness in local communities, and harvest regulation of critical den-tree species needed by this species. We urge further monitoring and surveys to confirm current patch occupancy, density and population trends. This species should be reassessed once these data are available.
Regional population effects: There is a disjunct distribution between the South African population and the rest of its range and, because of the isolation from populations elsewhere, no rescue effects in case of local extinctions are possible. Even within South Africa, the patches of forest within which they still occur are not contiguous, and they cannot disperse far without a forest corridor.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Venter J, Ehlers Smith Y & da Silva JM. 2025. A conservation assessment of Dendrohyrax arboreus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
They are dependent on well-developed woodland or forest, and the clearing of such areas for agriculture has led to a major range reduction (Skinner & Chimimba 2005). As such, they are patchily distributed in east, central and southern Africa, and have not been recorded from Namibia, Botswana, Zimbabwe or the Limpopo Province of South Africa (Skinner & Chimimba 2005; Milner & Gaylard 2013). Confined and isolated subpopulations are located south of the Zambezi River, Mozambique, and in the Eastern Cape and KwaZulu-Natal provinces of South Africa (Jennings & Jennings 1993; Milner & Gaylard 2013). The subspecies D. a. arboreus is endemic to South Africa.
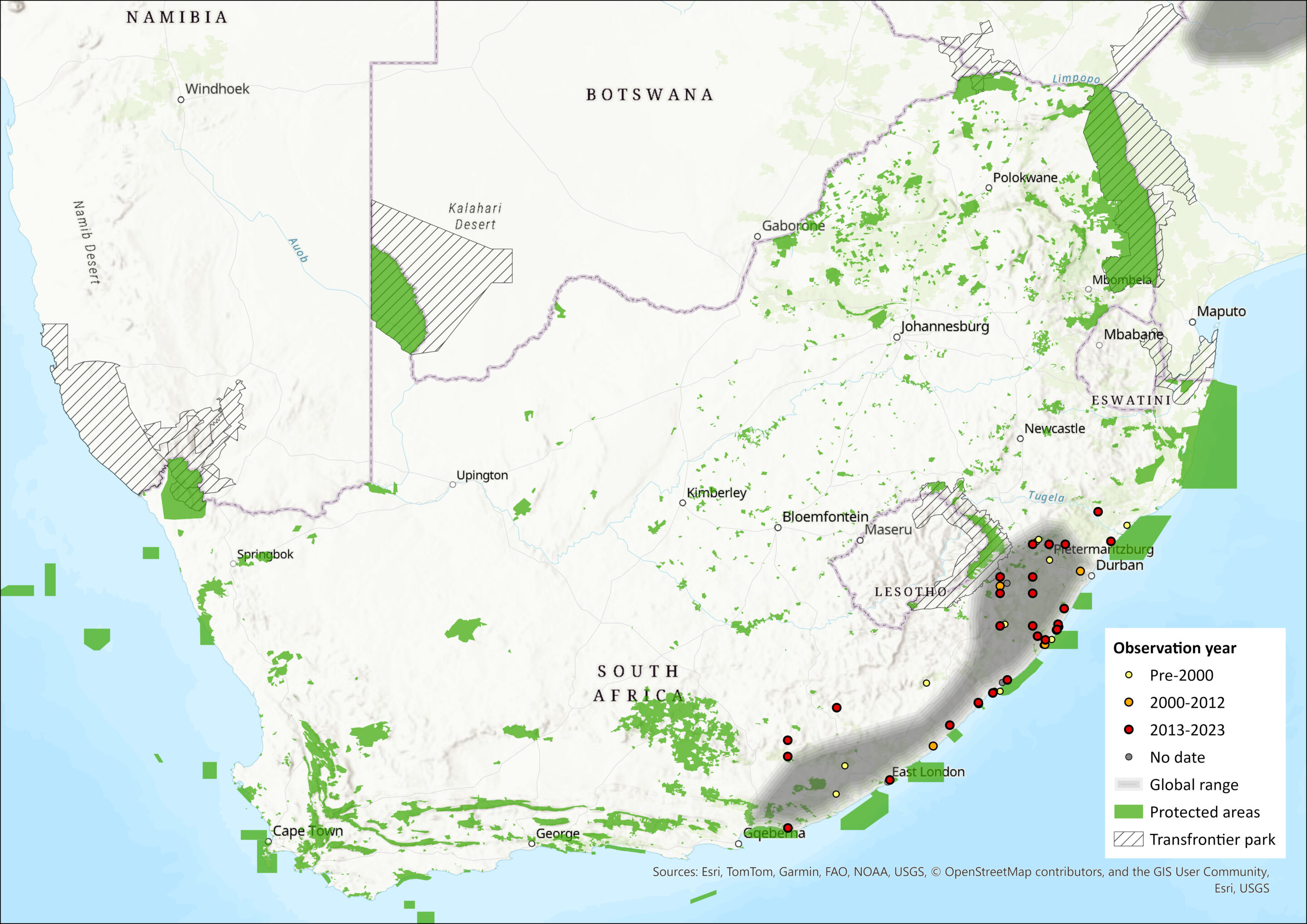
Within the assessment region, they occur in the forests and (marginally) thickets along the coasts of the Eastern Cape and KwaZulu-Natal, with the south-western range limit being the coastal Alexandria Forest towards the Sundays River in the Eastern Cape (Figure 1). There is little known about its historical distribution but it was presumed to once occur more extensively before habitat loss. Remaining forest patches are heavily fragmented and it is suspected that there is little dispersal between patches. They can colonise suitable patches only up to a maximum of 0.9 km away from the “mainland” patch, and the probability of patch occupancy is zero for distances between patches in excess of 1.5 km (Lawes et al. 2000). Furthermore, experimental data confirm that Southern Tree Hyraxes can only exist in intact forests, with density dropping off in lightly disturbed forests and completely absent from formerly logged forests with intense hunting pressure (Topp-Jorgensen et al. 2008). Corroborating this, recent data from unsuccessful call backs and anecdotal reports from landowners who have stopped hearing Southern Tree Hyraxes in various forest patches in southern KwaZulu-Natal indicate absence of the species from forest patches previously thought to be occupied (Y. Ehlers-Smith unpubl. data). Blue Duikers (Philantomba monticola) and Southern Tree Hyraxes most often co-occur in forest patches (Lawes et al. 2000), and the presence of one may be an indicator of the other’s presence.
Using both historical and recent records, we estimate the extent of occurrence to be 54,711 km2. Within this, the area of occupancy (AOO) is estimated to be a maximum of 1,598 km2 based on all remaining forest patches. Both mangrove forest patches and all forest patches smaller than the minimum estimated critical patch size for the species of 0.06 km2 (below which local extinction is expected to occur) (Lawes et al. 2000) were excluded, leaving 1549 km2 (N = 2187 patches). The minimum observed AOO, based only on forest patches within 10 km of recent (post-2010) records, is estimated to be 566 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 4000
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Southern Tree Hyrax (Dendrohyrax arboreus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | Resident |
| Burundi | Extant | Native | – | Resident |
| Congo, The Democratic Republic of the | Extant | Native | – | Resident |
| Kenya | Extant | Native | – | Resident |
| Malawi | Extant | Native | – | Resident |
| Mozambique | Extant | Native | – | Resident |
| Rwanda | Extant | Native | – | Resident |
| South Africa | Extant | Native | – | Resident |
| Tanzania, United Republic of | Extant | Native | – | Resident |
| Uganda | Extant | Native | – | Resident |
| Zambia | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
The Tree Hyrax vulnerability to climate change can be approached from the habitat level as the species is linked to the fate of the fragmented forest biome. Colyn et al. (2020) examined forest habitat contraction patterns across different altitude ranges for forest specialist bird species. However, many of their findings are pertinent to the effects of climate change on habitat persistence for Tree Hyrax. Their results show that Mistbelt and Afrotemperate (Mid to high-altitude, 800-1,800m) forests habitats are predicted to experience severe contractions, whereas coastal lowland and scarp forests had contraction rates in only 8.6% of core habitats. Their model also showed some expansion to occur at highest altitudes (Afromontane forest >2,100m) though moderate (305 ha). Lower altitudinal coastal forests (0-200m) saw greater expansion in area (1,961 ha; Colyn et al. 2020). However, despite habitat suitability for lowland forest persistence – it should not be considered as a standalone factor. Land-use within the coastal belt will be the most determining variable to consider, given the rate of land conversion for residential, commercial, tourism and leisure activities. Secondary regrowth forest currently is not afforded the same protection as forests that lie within the Department of Forestry Fisheries and Environment’s official data layers.
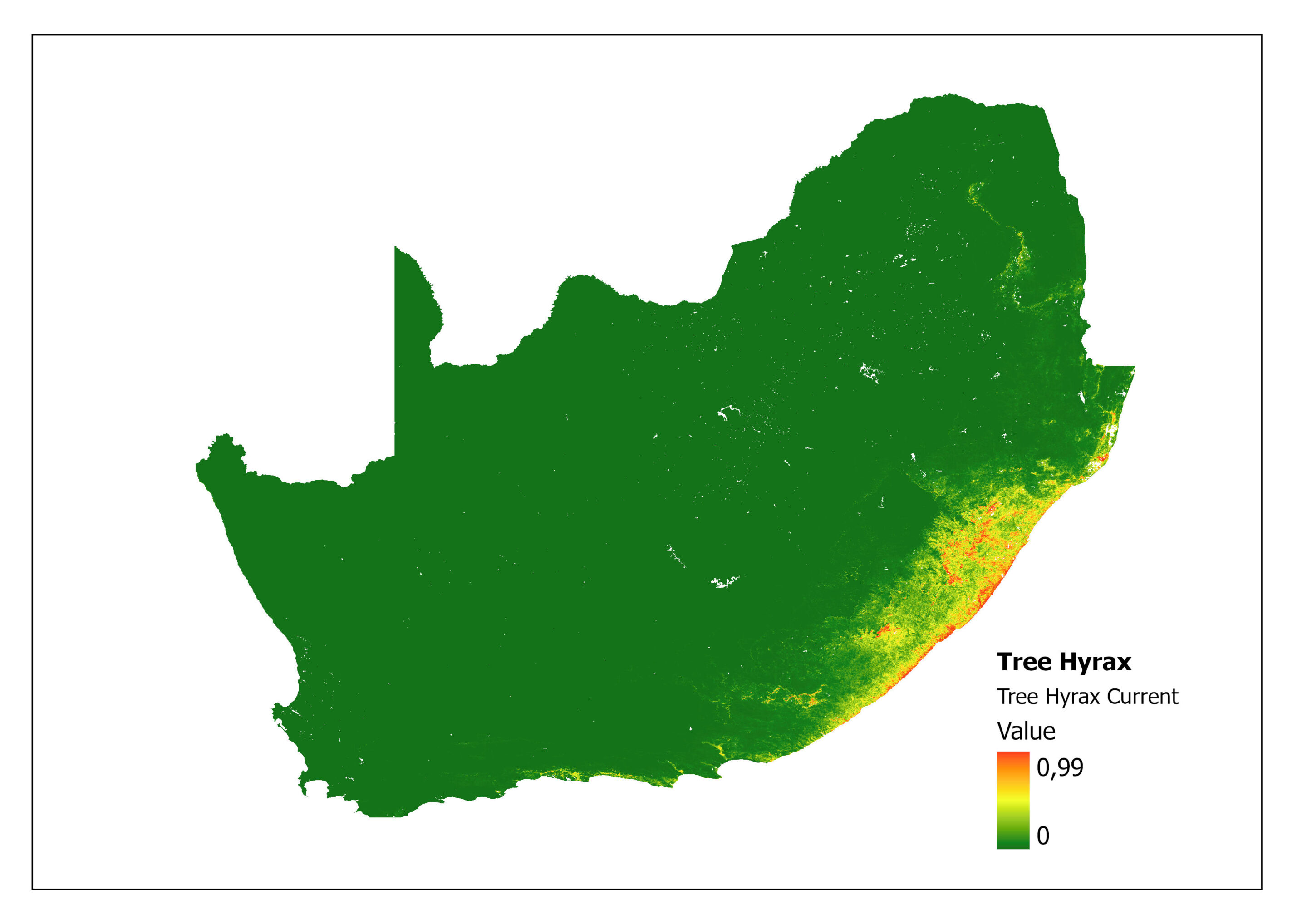
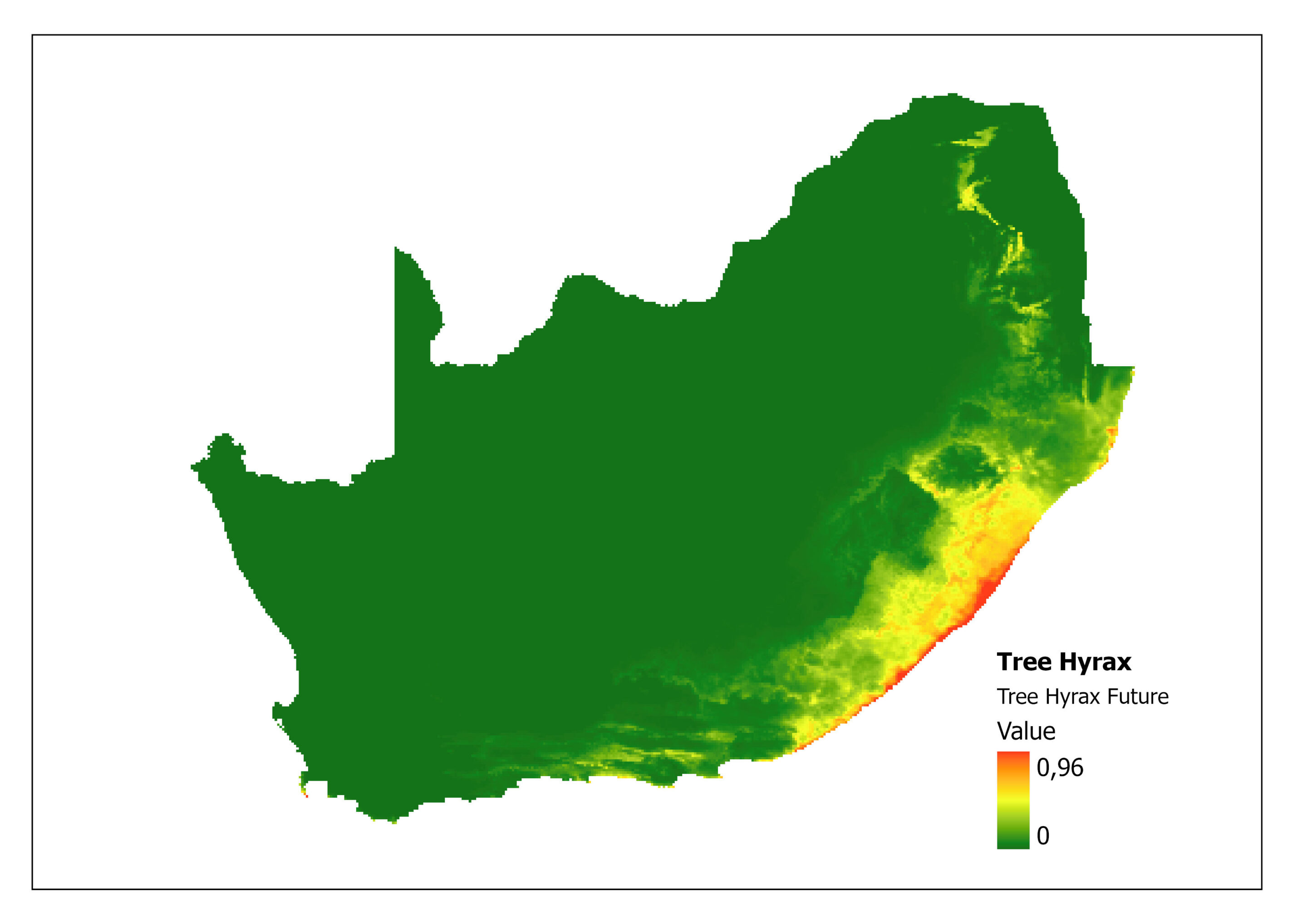
In addition, the impacts of climate change on Tree Hyrax can be explored through MaxENT (Maximum Entropy) species distribution modelling (Ehlers Smith Y, unpublished). A prediction specifically modelled for this species compares the current niche (Figure 2) with the future predictive niche (based on future predictive climate conditions) (Figure 3) and supports the coastal shift for environmental suitability. A major limitation of this analysis is that the rarified species localities provide few background points (43) as training points to calculate the model.
Figure 2: A MaxEnt (Maximum Entropy) species distribution map for Dendohyrax arboreus based on current environmental conditions. The higher the value, the higher the likely distribution of the species.
Figure 3: A MaxEnt (Maximum Entropy) predictive species distribution model for Dendohyrax arboreus in South Africa based on future environmental conditions. The higher the value, the higher the likely distribution of the species.
Population information
Southern Tree Hyraxes spend long periods inactive in the high canopy or in tree holes, and so often escape notice. In southern Africa, relative density has been estimated by means of counts of latrines in cavity trees (Catch Per Unit Effort, Gaylard 1994): 0.07 ± 0.29 latrines / man hour searching were found in three forests within the Eastern Cape, which can be roughly translated as 0.07 ± 0.29 individual / ha, as single individuals generally use only one latrine. In undisturbed forests of the Udzungwa Mountains in Tanzania, latrine density was estimated at 2.6 latrines / ha (Topp-Jorgensen et al. 2008). Similarly, it was estimated that at least 20 individuals occurred in the Gxalingenwa Forest Reserve, KwaZulu-Natal, in 2003, based on the assumption that they do not call more than once every 2 minutes (I. Rushworth unpubl. data), which equates to 0.03 individual / ha based on forest size. However, we caution that these may even be overestimates, as patch size of the forest is not a good proxy for abundance. Rather, the habitat quality is an important predictor of abundance, where the density of key den-trees is critical (Gaylard 1994). In Kenya, density estimates were 0-13 animals / ha of remaining forest, based on the size of remaining forest fragments and estimated group size (Rosti et al. 2022). Estimated territory of each group was 2 ha (Rosti et al. 2022).
Although more demographic research is needed, we estimate the mature population structure to be c. 50%, as they are mostly solitary and exist as one adult female and her young occupying a single shelter (Skinner & Chimimba 2005). The average litter size is two (Rudnai 1984), and thus a typical group may comprise solely two adults and two juveniles. Thus, maximum mature population size, based on all available forest patches, may range from 2,209–5,187 individuals. Minimum mature population size (using confirmed occupied patches) is estimated to be 750–1,760 individuals. The latter population size range is likely to be more realistic considering that many sites identified as suitable habitat in southern KwaZulu-Natal are not currently occupied (based on surveys from 2013-2016; Y. Ehlers-Smith unpubl. data). In East Africa (Rwanda), where they can be locally abundant, estimated density using mark-recapture is 13.4 individuals / ha (10.3 adults and 3.8 juveniles) in undisturbed montane forest (Milner & Harris 1999). Using a 73% mature population structure would lead to a mature population estimate for the assessment region of 1,094–7,573 individuals, with a most likely population size of 2,570 individuals. Further density estimates are needed for the assessment region to calculate population size more accurately. However, density estimates vary greatly depending on the method used. Topps-Jorgensen et al. (2008) suggest using plot counts of calling individuals at dusk to estimate density, as these counts seem to vary less than latrine counts and daytime calling.
Unfortunately, there are no other baseline data or more recent density estimates in the same areas from which to estimate direct populations trends. For example, although the population in KwaZulu-Natal, especially Karkloof forest and other Afromontane forest patches, was deemed to be secure (Friedmann & Daly 2004), these data were collected in 1995 (Lawes et al. 2000), and are thus outdated. Recent surveys are urgently needed to confirm Southern Tree Hyrax patch occupancy and population trends.
As their habitats are fragmented, with limited dispersal and gene flow between patches, we suspect that large individual forest patches may be construed as isolated subpopulations (Lawes et al. 2000). The generation length is estimated to be 5.6 years (Pacifici et al. 2013), which equates to a 16.7 year three generation period.
There is ongoing pressure on their habitat and this species is also directly targeted for bushmeat, as a result the population is suspected to be declining.
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| Yes | Estimated | Due to loss of large nesting trees and potential bushmeat poaching in and around forests. |
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: 465
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
To date, no population of phylogenetic genetic studies have been undertaken on this species within or including animals from the assessment region; however, a fine scale molecular study would be greatly beneficial in identifying the genetic structure and health of the species within the assessment region. In the past, when the forest was more contiguous (not in patches), it is likely the species existed as a single metapopulation. However, it is possible that genetic structure currently exists given their dependence on forest patches and the discontinuous nature of the current forest ecosystem. Due to this uncertainty, it is not possible to quantify the proportion of populations (PM) indicator. However, an indication of the Convention on Biological Diversity’s headline genetic indicator (the proportion of populations with an effective population size > 500) is possible. Based on density estimates and niche models for suitable habitats, it is presumed (albeit with low accuracy) that approximately 2,570 individuals exist within the assessment region. Assuming an effective population size to census size ratio (i.e., Ne/Nc) of 0.1, the species as a single metapopulation would possess an Ne of 275, which already falls short of the threshold of 500, which would indicate a genetically healthy and stable population. The estimated value suggests the species could be at genetic risk. If the species experienced a severe bottleneck, this estimate is likely to be far less. And if genetic structure exists within the species, the projection is far more concerning. A population genetic assessment is highly recommended to better understand the health of this species.
Habitats and ecology
Tree Hyraxes occur in Afromontane, scarp and coastal forests of the KwaZulu-Natal and Eastern Cape provinces. At the western coastal limit, they occur in milkwood-dominated coastal forests between Alexandria and the Sundays River, as milkwoods (Mimosops caffra and Sideroxylon inerme) are ideal den and forage trees (Gaylard 1994). Throughout their range, they are dependent on tree cavities, epiphytes or dense matted forest vegetation for shelter during the day. Primarily, they need den trees – tree species that form cavities. For example, in the Pirie Forest, Eastern Cape, only Schotia latifolia, Rhus chirindensis, Andrachne ovalis, Podocarpus falcatus, Strychnos decussata, Cordia caffra and Sideroxylon inerme were used, and were also a preferred food source (Gaylard 1994; Milner & Gaylard 2013). They also prefer trees with multiple cavity entrances (Gaylard & Kerley 2001). Latrines are located in the low fork of a tree or on the ground beside the trunk. Thus, a decrease in numbers in southern Africa has been attributed to loss of structure within habitat, rather habitat loss outright (Castley & Kerley 1993). They are selective browsers, and the species selected are not related to their abundance but may instead be correlated with predator avoidance and the energetics of an arboreal lifestyle (Gaylard & Kerley 1997). Unlike other hyrax species, they are predominantly solitary, with only one adult or an adult female and her young occupying a shelter. They are arboreal and nocturnal (Skinner & Chimimba 2005). The biology and life-history of the species is summarized by Milner and Gaylard (2013).
Ecosystem and cultural services: They are an important prey species for forest predators, such as Crowned Eagles (Stephanoeatus cornatus) (Boshoff et al. 1994). This species has the potential to become a flagship species of eastern forests for biodiversity stewardship programmes.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.4. Forest -> Forest – Temperate | Resident | Suitable | Yes |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | Resident | Marginal | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | Resident | Suitable | Yes |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | Resident | Suitable | Yes |
| 2.2. Savanna -> Savanna – Moist | Resident | Suitable | No |
| 6. Rocky areas (eg. inland cliffs, mountain peaks) | Resident | Suitable | No |
Life History
Generation Length: (Not specified)
Age at Maturity – Female or unspecified: 20 – 30 months
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 7.5 – 8 months
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: 1 – 3
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Hunted (through snaring) for bushmeat, traditional medicine (“hyraceum” is used primarily for gastrointestinal complaints), traditional beliefs (to make one shy and timid) perhaps their pelts (for karosses). They are thought to have been hunted in the former Transkei and Ciskei and East-Griqualand (Friedmann & Daly 2004; Opperman et al. 2018; Sosibo et el. 2022).
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Bushmeat and traditional medicine. | – | – |
National Commercial Value: No
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | – | – | – |
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: All harvested individuals are wild.
Threats
The main threats to Southern Tree Hyraxes within the assessment region are habitat quality degradation and direct disturbance through tree removal. While elsewhere in Africa, forest habitat loss continues to be the primary threat, forests in South Africa are well protected and current rates of absolute forest loss have been low since the 1990s (see below). However, the legacy of past forest loss through agricultural, industrial and residential expansion (Lawes et al. 2004) is that remaining forest patches are highly fragmented, making Southern Tree Hyraxes more susceptible to current ongoing threats. Many forest patches are also too small to maintain viable subpopulations (Lawes et al. 2000). Currently, ongoing habitat loss of natural vegetation in the matrix between forest patches is inferred to be increasing the frequency of both fuelwood harvesting and poaching.
Southern Tree Hyraxes rely on relatively few tree species for nesting and food, and thus the selective removal of these species (through either commercial controlled logging or informal harvesting) disproportionately impacts the population. For example, Gaylard and Kerley (1997) cautioned that, because most tree species utilised by the species occur at low densities, removal of stems for fuelwood could lead to a loss of key resource areas (both dens and forage areas) and thus inhibit population growth. The felling of mature Yellowwood (mostly Afrocarpus falcatus) and other canopy trees may be particularly damaging (Gaylard & Kerley 2001). Mahogany trees (Trichilia dregeana and T. emetica) are important species on the south coast of KwaZulu-Natal because of an association with a wood-borer beetle that creates suitable cavities (Y. Ehlers-Smith pers. obs. 2015). In the Udzungwa Mountains of Tanzania, Tree Hyrax density decreased significantly from little disturbed (closed canopy; 17.3 ± 1.9 individuals / ha) to moderately disturbed forest areas (more open canopy; 11.7 ± 1.4 individuals / ha) and individuals were completely absent in heavily hunted and formerly logged areas (Topp-Jorgensen et al. 2008).
The lack of arboreal pathways in open forest may increase vulnerability to hunting by snares or terrestrial predators, as they are forced to use the ground more frequently (Topp-Jorgensen et al. 2008). In Gxalingenwa Forest Reserve, KwaZulu-Natal, approximately 6,500 building poles and 1,500 tonnes of fuel wood are harvested annually (I. Rushworth unpubl. data 2003) from these forests, which is leading to over-exploitation of the medium-sized classes of tree. Large forest trees are also being burned out and felled for fuel and construction material. Model output, based on empirical data from the Karkloof forest region of KwaZulu-Natal, corroborates these findings and indicates that Southern Tree Hyraxes are sensitive to disturbances caused by wood removal and burning (Lawes et al. 2000): occupancy was greater in patches with low wood removal, but declined significantly with high wood removal (0.3% probability of occurrence in a 10 ha patch under high wood removal). Similarly, frequent burning of small forest patch edges significantly decreased their likelihood of occupancy by Southern Tree Hyraxes (0.08% probability of occurrence in a 10 ha patch under high levels of burning). Similar studies suggest that small-scale wood harvesting may not alter canopy structure and, as long as key den trees are not removed, this should not affect Southern Tree Hyraxes. For example, in a study assessing the impact of human utilisation of woodlands and sand forests in Maputaland, while utilisation decreased the average stem diameter of trees, the height structure of the forest remained largely the same (Gaugris & Van Rooyen 2010). A further study has concluded that such understorey harvesting at low levels of harvesting does not affect the regeneration of canopy species and will not detrimentally affect the overall species composition of scarp forest (Boudreau & Lawes 2005). Similarly, Louw (2010) found that, while creating small gaps from harvesting pole-sized trees will not affect forest dynamics and species composition, harvesting in excess of eight adjacent trees, and so creating larger gaps, will potentially lead to successional shifts and alternate states in the ecosystem. Thus, harvest management should be regulated accordingly.
Southern Tree Hyraxes did not exhibit a negative response to livestock disturbance or general disturbance (e.g. past logging activities) in the Karkloof forest complex (Lawes et al. 2000). This is in contrast with Friedmann and Daly (2004), who asserted overgrazing was a threat, and Topp-Jorgensen (2008), who found zero occupancy by Southern Tree Hyraxes in previously logged patches. However, other land uses are suspected to be detrimental to Southern Tree Hyraxes. For example, if a forest patch is surrounded by plantations, it is half as likely to be occupied by Southern Tree Hyraxes than by Blue Duikers (Lawes et al. 2000). Informal sand mining on the east coast of the Eastern Cape is also causing major disturbance and forest degradation. Informal logging as well as large scale collection of firewood in this area is a further major factor in forest degradation.
Southern Tree Hyraxes are vulnerable to human disturbance and encroachment. For example, hunting with the use of dogs is common practice in the forests of the former Transkei, and has a negative effect on the population. In areas in KwaZulu-Natal, where Southern Tree Hyraxes were known to exist previously, they have now disappeared – probably due to anthropogenic disturbance (the south coast is a popular tourist destination).
The most recent study undertaken by Sosibo et al. (2022) showed that there is ongoing pressure on both the species and its preferred habitat. Sixteen percent of respondents that participated in community questionnaires (n=303) around the iNgeli, Creighton (KwaZulu-Natal) and Umtata (Eastern Cape) southern mistbelt forest clusters indicated that Tree Hyrax was being hunted for consumptive use or for financial gain. The highest reporting rate was from the iNgeli forest area. Opperman et al. (2018) found that 40% of male respondents from Pirie forest complex, Eastern Cape, reported to have hunted Tree Hyrax as some point in their lives. They also reported the harvesting of tree species that are utilised by Tree Hyrax either as dens or as food sources.
Conservation
The species is present in many large protected areas across the assessment region, including the Alexandria Forest in the Woody Cape section of Addo Elephant National Park, and have been observed in a few protected areas in the former Transkei (for example, Dwesa and Hluleka Nature Reserves, as well as Manubi State Forest; Eastern Cape Parks and Tourism Agency unpubl. data in 2011 and 2012). The species has also been observed previously in the Matiwane Range Forests north of Mthatha (Hayward et al. 2005), as well as the Karkloof and Balgowan forests (Lawes et al. 2000). In KwaZulu-Natal, they have recently been confirmed present in Gxalingenwa, and are thought to exist in Vernon Crookes, Umtamvuna, Oribi Gorge and Mbumbazi Nature Reserves.
Forests are formally protected within the assessment region, so conservation interventions should focus on mitigating disturbance and encroachment of remaining forest patches. Protected area expansion and biodiversity stewardship schemes could be used to connect isolated forest patches, but effectively connecting patches is highly unlikely as patches are too far apart and separated by incompatible land uses like sugar cane plantations (KwaZulu-Natal) and dairy farms (Eastern Cape). The primary interventions should thus be 1) to limit harvesting of large den tree species; 2) to enforce existing legislation to reduce settlement sprawl and disturbance around forest patches; and 3) to develop and implement education and awareness campaigns.
Selective logging even within intact forests must be minimised, as Southern Tree Hyrax make dens in certain species which are also those that tend to be logged; for example, yellowwood and mahogany (Gaylard 1994). These species often are also the species that they feed on, presumably to minimise distance travelled between dens and foraging areas (Gaylard & Kerley 1997). Conservationists should also work with local communities to regulate the harvesting of pole-sized trees in forests. For example, recent research suggests that small-scale harvesting of understorey trees (around 11% of available stems) should be sustainable and not alter forest structure if fewer than eight trees are harvested adjacently in any one area (Boudreau & Lawes 2005; Boudreau et al. 2005; Louw 2010). It has also been suggested that selective logging could help finance tree hyrax conservation by logging only tree species not used for shelter or food (Gaylard & Kerley 2001). This will have to be assessed on a forest-by-forest basis. For example, in Gxalingenwa Forest, once the forest has recovered, some level of sustainable utilisation may be considered, probably in 20–25 years (I. Rushworth pers. comm. 2003).
If this species is to have a greater chance of long-term survival, then management of the matrix is important (Lawes et al. 2000). Reducing disturbance in the matrix may indirectly decrease the isolation effect, thereby improving the ability of Southern Tree Hyraxes to colonise small patches. Enforcing existing laws established to protect forests is critical for achieving this. This includes both enforcing regulations on trespassers within forests, as well as zoning regulations to reduce settlement sprawl, especially holiday homes in coastal areas. Biodiversity stewardship schemes should be used to protect remaining patches of forest.
Some areas require significant work with local communities and landowners to inform people about the species and its requirements, which should include information on sustainable harvesting of the correct tree species and appropriate fire regimes. Snare removal should also be a priority. Anecdotal observations suggest that people are not aware of their existence and, because of their vocalisation at night, people find them threatening because they do not know that they are harmless herbivores (Y. Ehlers-Smith pers. obs. 2015).
Recommendations for land managers and practitioners:
- Conservation authorities should develop harvesting strategy guidelines for understorey trees, and engage with local communities to develop and implement co-management agreements. Small-scale wood removal is predicted not to affect Southern Tree Hyrax occupancy (Lawes et al. 2000). The size of the forest patch is not as important as its quality – determined by the presence and abundance of cavity-forming trees.
- Conservation authorities should enforce regulations on developments that potentially impact on the habitat integrity of forests.
- Further field surveys are needed to improve estimates of population numbers and status in all regions.
- Undertake studies and institute monitoring to ascertain the options to use carbon market financing to protect and monitor forests.
Research priorities:
- Further field studies are needed to determine the distribution of this species in forest patches within the matrix between protected areas. Plot counts of calling individuals at dusk should be used to estimate density (Topp-Jorgensen et al. 2008). This will help to improve the accuracy of current distribution and subpopulation size estimates.
- More research and systematic reviews are needed to determine the net effects of forest land cover change on this species.
- Further research should be undertaken on isolation distances, dispersal capacity, connectivity and metapopulation dynamics to inform conservation planning.
- Quantifying the rates and effects of bushmeat poaching on subpopulations is required.
- Eastern Cape Parks and Tourism Agency is conducting ad hoc camera trap surveys focusing on protected areas and other state forests with no information.
Encouraged citizen actions:
- Report sightings of this species, especially outside protected areas, on virtual museum platforms (for example, iNaturalist and MammalMAP).
- Keep pets away from trees with cavities (Port Alfred and coastal villages especially, where Tree Hyraxes are prone to predation by dogs and cats).
- Visit forest patches and document deterioration and apply pressure to government agencies to do more to conserve/protect forests.
- Do not cut down any cavity trees in your garden unnecessarily (milkwoods are protected species, so should not be felled anyway).
Bibliography
Berliner, D. and Desmet, P. 2007. Eastern Cape Biodiversity Conservation Plan: Technical Report. Department of Water Affairs and Forestry, Pretoria, South Africa.
Boshoff AF, Palmer NG, Vernon CJ, Avery G. 1994. Comparison of the diet of crowned eagles in the savanna and forest biomes of south-eastern South Africa. South African Journal of Wildlife Research 24: 26-31.
Bothma, J. du P. 1971. Order Hyracoidea. In: J. Meester & H.W. Setzer (eds) The Mammals of Africa: An identification manual. Smithsonian Institution Press, Washington, DC.
Boudreau S, Lawes MJ. 2005. Small understorey gaps created by subsistence harvesters do not adversely affect the maintenance of tree diversity in a sub-tropical forest. Biological Conservation 126: 279-286.
Boudreau, S., Lawes, M.J., Piper, S.E. and Phadima, L.J. 2005. Subsistence harvesting of pole-size understorey species from Ongoye Forest Reserve, South Africa: Species preference, harvest intensity, and social correlates. Forest Ecology and Management 21: 149-165.
Castley, J.G. and Kerley, G.I.H. 1993. Trends in indigenous forest patch size in the North-Eastern Cape. In: D.A. Everard (ed.), The Relevance of Island Biogeographic Theory in Commercial Forestry, pp. 148-149. Forum Report, FRD Environmental, Pretoria.
Colyn, R.B., Ehlers Smith, D.A., Ehlers Smith, Y.C., Smit‐Robinson, H. and Downs, C.T., 2020. Predicted distributions of avian specialists: A framework for conservation of endangered forests under future climates. Diversity and Distributions, 26(6), pp.652-667.
Department of Forestry and Fisheries and the Environment (DFFE). 2022. South African National Land-Cover 2014/ 2020 Change assessment Report.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. Conservation Breeding Specialist Group (SSC/IUCN) and Endangered Wildlife Trust, Parkview, South Africa.
Gaugris JY, Van Rooyen MW. 2010. Woody vegetation structure in conserved versus communal land in a biodiversity hotspot: a case study in Maputaland, South Africa. South African Journal of Botany 76: 289-298.
Gaylard A, Kerley GI. 1997. Diet of tree hyraxes Dendrohyrax arboreus (Hyracoidea: Procaviidae) in the Eastern Cape, South Africa. Journal of Mammalogy 78: 213-221.
Gaylard A, Kerley GIH. 2001. Habitat assessment for a rare, arboreal forest mammal, the tree hyrax Dendrohyrax arboreus. African Journal of Ecology 39: 205-212.
Gaylard, A. 1994. The tree hyrax Dendrohyrax arboreus as a rare forest vertebrate: habitat and diet. M.Sc. Thesis, University of Port Elizabeth.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Hayward MW, White RM, Mabandla KM, Bukeye P. 2005. Mammalian fauna of indigenous forest in the Transkei region of South Africa: an overdue survey. South African Journal of Wildlife Research 35: 117-124.
Jennings BH, Jennings PJ. 1993. The distribution of the tree dassie (Dendrohyrax arboreus arboreus) in the Eastern Cape. Bontebok 8: 35-37.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Lawes MJ, Macfarlane DM, Eeley HA. 2004. Forest landscape pattern in the KwaZulu-Natal midlands, South Africa: 50 years of change or stasis? Austral Ecology 29: 613-623.
Lawes, M.J., Meakin, P.E. and Piper, S.E. 2000. Patch occupancy and potential metapopulation dynamics of three forest mammals in fragmented Afromontane forest in South Africa. Conservation Biology 14: 1088-1098.
Louw SL. 2010. The effect of the spatial scale of tree harvesting on woody seedling establishment and tree dynamic at Ongoye Forest Reserve. M.Sc. Thesis. University of KwaZulu-Natal, Pietermaritzburg, South Africa.
Milner, J.A. and Gaylard, A. 2013. Dendrohyrax arboreus Southern Tree Hyrax. In: J.S. Kingdon, D.C.D. Happold, M. Hoffmann, T. M. Butynski, M. Happold and J. Kalina (eds), Mammals of Africa. Vol. I. Introductory Chapters and Afrotheria. , pp. 152-155. Bloomsbury Publishing, London.
Milner, J.M. and Harris, S. 1999. Habitat use and ranging behaviour of tree hyrax, Dendrohyrax arboreus, in the Virunga Volcanoes, Rwanda. African Journal of Ecology 37: 281-294.
Opperman, E. J., Makunga, N. P., & Cherry, M. I. (2018). Community harvesting of trees used as dens and for food by the tree hyrax (Dendrohyrax arboreus) in the Pirie forest, South Africa. Koedoe: African Protected Area Conservation and Science, 60(1), 1-9.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Rosti, H., Heiskanen, J., Loehr, J. et al. 2022. Habitat preferences, estimated abundance and behavior of tree hyrax (Dendrohyrax sp.) in fragmented montane forests of Taita Hills, Kenya. Sci Rep 12, 6331. https://doi.org/10.1038/s41598-022-10235-7
Rudnai J. 1984. Suckling behaviour in captive Dendrohyrax arboreus (Mammalia: Hyracoidea). South African Journal of Zoology 19: 121-123.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Sosibo, M. T., Ehlers Smith, Y. C., Ehlers Smith, D. A., & Downs, C. T. (2022). Some perspectives on the use and value of Southern Mistbelt Forests to surrounding rural communities in northern Eastern Cape, and southern KwaZulu-Natal, South Africa. African Journal of Wildlife Research, 52(1), 103-121.
South African National Biodiversity Institute 2018. The Vegetation Map of South Africa, Lesotho and Swaziland, Mucina, L., Rutherford, M.C. and Powrie, L.W. (Editors), Online, http://bgis.sanbi.org/SpatialDataset/Detail/18, Version 2018
Topp-Jorgensen JE, Marshall AR, Brink H, Pedersen UB. 2008. Quantifying the response of tree hyraxes (Dendrohyrax validus) to human disturbance in the Udzungwa Mountains, Tanzania. Tropical Conservation Science 1: 63-74.