
Southern Hemisphere Fin Whale
Balaenoptera physalus

2025 Red list status
Least Concern
Regional Population Trend
Increasing
Change compared
to 2016
No Change
Overview
Balaenoptera physalus – (Linnaeus, 1758)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BALAENOPTERIDAE – Balaenoptera – physalus
Common Names: Fin Whale, Common Fin Back, Common Fin Whale, Common Rorqual, Fin-backed Whale, Finback, Finfish, Finner, Gibbar, Herring Whale, Razorback, True Fin Whale (English), Vinwalvis (Afrikaans)
Synonyms: Balaena physalus Linnaeus, 1758
There has been a recent new article in support of returning to Artiodactlyla https://link.springer.com/article/10.1007/s10914-021-09572-7
Moreover, the committee on taxonomy of the SMM has adopted it: https://marinemammalscience.org/science-and-publications/list-marine-mammal-species-subspecies/
The subspecies for this species have never been assessed; they are attached here to ensure the full cetacean taxonomy is accounted for in SIS, and to make these available to the Cetacean SG if they decide to assess these taxa in future.
Taxonomic Note:
The Committee on Taxonomy of the Society for Marine Mammalogy provisionally recognises four subspecies: the North Atlantic Fin Whale, Balaenoptera physalus physalus, (Linnaeus, 1758); the North Pacific Fin Whale, Balaenoptera p. velifera; the Southern Fin Whale, B. p. quoyi (Fischer, 1829), which is larger than B. p. physalus, and the Pygmy Fin Whale, B. p. patachonica (Burmeister, 1865), proposed by Clark (2004), which is smaller than B. p. quoyi and has dark baleen (Committee on Taxonomy 2018). However, a more recent population genetic study incorporating increased representation across all subspecies was only able to detect three significant lineages, with no genetic structure detected among the southern hemisphere subspecies (B. p. quoyi and B. p. patachonica) (Perez-Alvarez et al. 2021). They therefore recommend the collapse of B. p. patachonica into B. p. quoyi.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Vermeulen, E.,1 Shabangu, F.1,2 & da Silva, J.3
Reviewer: Patel, T.4
Institutions: 1Mammal Research Institute, University of Pretoria, 2Department of Forestry, Fisheries and the Environment, 3South African National Biodiversity Institute, 4Endangered Wildlife Trust
Previous Assessors: Findlay, K., Elwen, S., Meÿer, M., Oosthuizen, H. & Plön, S.
Previous Reviewer: Plön, S.
Previous Contributors: Relton, C., Child, M.F., Reilly, S.B., Bannister, J.L., Best, P.B., Brown, M., Brownell Jr., R.L., Butterworth, D.S., Clapham, P.J., Cooke, J.G., Donovan, G. & Zerbini, A.N.
Assessment Rationale
Most of the global decline is attributable to the major decline in the Southern Hemisphere. Most specifically, between the 1930s and 1960s Fin Whales were severely overexploited by commercial whaling in the Southern Ocean. There is no recent data documenting the current population status of this species, since the surveys that have been conducted in the Southern Hemisphere do not cover their entire summer distribution. There is no indication that they have recovered to levels anywhere near those prior to exploitation (which was estimated at 200,000), however the population is expected to be increasing.
The analysis in this assessment estimates that the global population has declined by more than 70% over three generations mainly due to commercial whaling (1935–2013). Since the cessation of whaling, they are regularly observed in polar waters where there do not appear to be any current major threats. The national assessment for this species is considered in line with that of the global assessment, and since the majority of the decline is attributable to the Southern Hemisphere, this species is listed as Endangered A1d. However, more current data on population size and trends for the Southern Ocean are needed and this species should be reassessed once such data become available. Additionally, the ingestion of microplastics by this species has recently been documented as an emerging threat to populations elsewhere in the world and should be monitored within the assessment region.
Regional population effects: Fin Whales are highly migratory and wide-ranging. There are no barriers to dispersal, thus rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Vermeulen E, Shabangu F & da Silva JM. 2025. A conservation assessment of Balaenoptera physalus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Although rare in coastal waters, this species is located within all major ocean basins worldwide, including the Mediterranean. Similar to Blue (Balaenoptera musculus), Minke (Balaenoptera acutorostrata) and Humpback (Megaptera novaeangliae) Whales, southern Fin Whales spend summers in the Antarctic, where they gather at feeding grounds, exhibiting a circumpolar distribution. Sightings data and historical catches revealed that in winter they migrate to middle latitudes, usually between 40°S–60°S in the southern Indian and Atlantic Oceans, but are rare in the Tropics, aside from areas of cooler tropical water, such as the South Pacific (50–65°S) (Miyashita et al. 1996, IWC 2006a). During the winter months, they migrate to lower latitudes, reason for which they were once included in the catches of the Saldanha Bay and Durban whaling operators (Skinner and Chimimba 2005). Based on patterns of seasonal abundance, Fin Whales might not migrate much further north than Durban (Bannister and Gambell 1965). Abundance peaks in June/July on both the east and west coasts of South Africa (Bannister and Gambell 1965, Best 1967). According to catch data, migrations appear to occur along the edge of the continental shelf. Migratory whales were frequently caught off South Georgia (Southern Atlantic) but Moore et al. (1999) found that they no longer commonly occur in this region. The extent of their winter distribution is not well-known, as it is difficult to track Fin Whales as they don’t travel along coastlines, but rather in deep, open oceans. Catch records revealed that they were common off southern Africa in winter, but as Southern Ocean stocks declined due to commercial whaling, their frequency depreciated off southern Africa, suggesting that this was a popular wintering area for the migrating population (Best 2003). Although the majority of catches occurred in South African waters in the early 20th century, whaling expanded into regions off Angola, Congo and Mozambique (Best 1994). Their current distribution is considered extensive throughout South Africa’s EEZ (Exclusive Economic Zone).
Recent passive acoustic results indicate that Fin Whales are present off the west coast from April to November with a peak in June/July, indicating that whales might be overwintering and/or breeding in this region (Shabangu et al. 2019; Letsheleha et al. 2022). Around the Prince Edward Islands, Fin Whale calls are detected year-round with peak occurrence in winter (Shabangu et al. 2024). The Prince Edward Islands seem to be a suitable year-round habitat for this species. In Antarctica, this species has a negative correlation with sea ice and thus occur there in summer and autumn (Shabangu et al. 2020).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): 0
Depth Upper Limit (in metres below sea level): 359
Depth Zone: Mesopelagic
Map
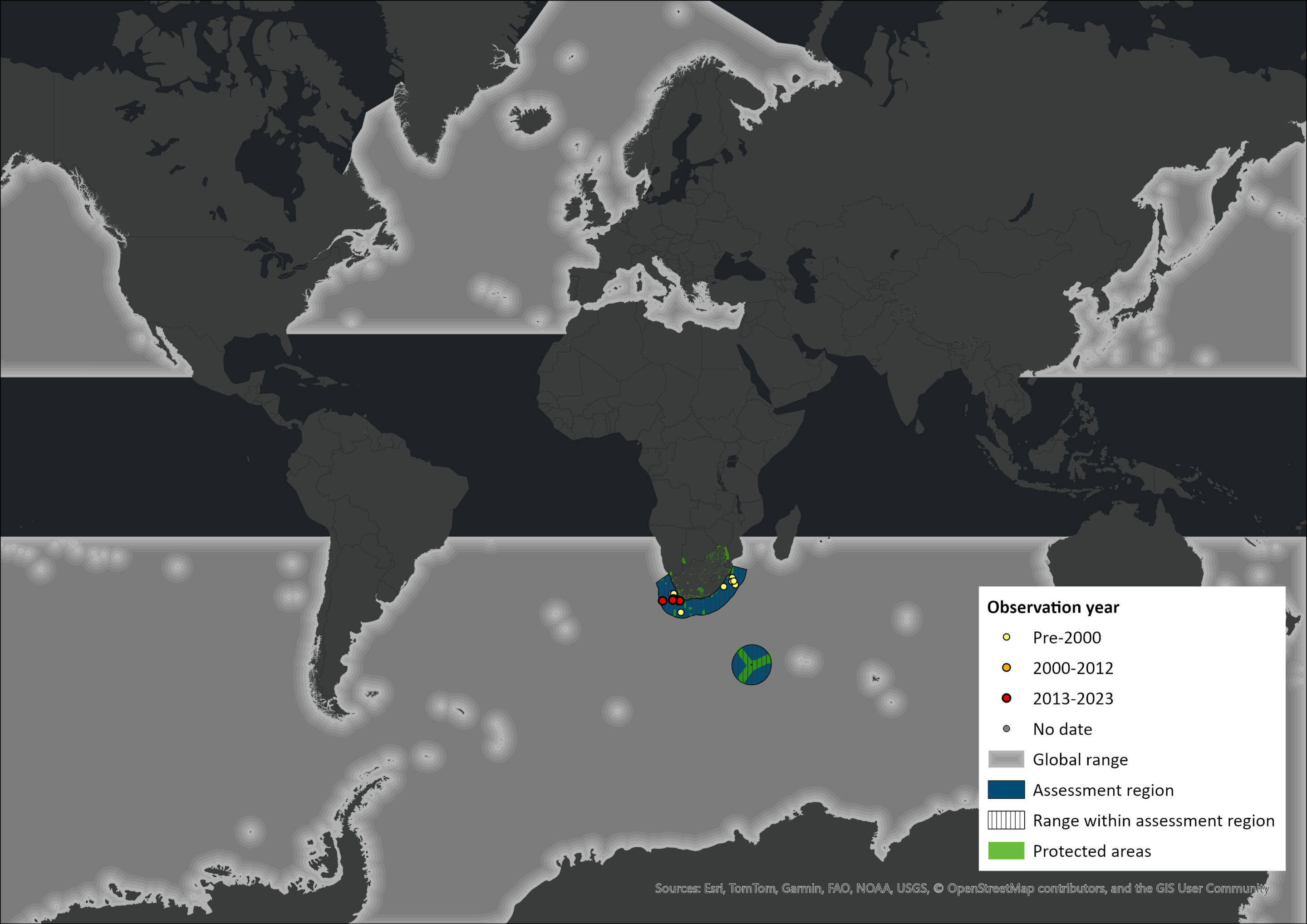 Figure 1. Distribution records for Fin Whale (Balaenoptera physalus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Fin Whale (Balaenoptera physalus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Antarctic, Australasian, Indomalayan, Nearctic, Neotropical, Oceanian, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Algeria | Extant | Native | – | Resident |
| Angola | Extant | Native | – | Resident |
| Antarctica | Extant | Native | – | Resident |
| Argentina | Extant | Native | – | Resident |
| Australia | Extant | Native | – | Resident |
| Belgium | Extant | Native | – | Resident |
| Bermuda | Extant | Native | – | Resident |
| Bouvet Island | Extant | Native | – | Resident |
| Brazil | Extant | Native | – | Resident |
| Cabo Verde | Extant | Native | – | Resident |
| Canada | Extant | Native | – | Resident |
| Chile | Extant | Native | – | Resident |
| China | Extant | Native | – | Resident |
| Congo | Extant | Native | – | Resident |
| Congo, The Democratic Republic of the | Extant | Native | – | Resident |
| Croatia | Extant | Native | – | Resident |
| Cyprus | Extant | Native | – | Resident |
| Denmark | Extant | Native | – | Resident |
| Ecuador | Extant | Native | – | Resident |
| Egypt | Extant | Native | – | Resident |
| Falkland Islands (Malvinas) | Extant | Native | – | Resident |
| Faroe Islands | Extant | Native | – | Resident |
| Fiji | Extant | Native | – | Resident |
| France | Extant | Native | – | Resident |
| French Southern Territories | Extant | Native | – | Resident |
| French Southern Territories -> Kerguelen | Extant | Native | – | Resident |
| Gabon | Extant | Native | – | Resident |
| Germany | Extant | Native | – | Resident |
| Gibraltar | Extant | Native | – | Resident |
| Greece | Extant | Native | – | Resident |
| Greenland | Extant | Native | – | Resident |
| Heard Island and McDonald Islands | Extant | Native | – | Resident |
| Iceland | Extant | Native | – | Resident |
| India | Extant | Native | – | Resident |
| India -> Andaman Is. | Extant | Native | – | Resident |
| India -> Laccadive Is. | Extant | Native | – | Resident |
| India -> Nicobar Is. | Extant | Native | – | Resident |
| Indonesia | Extant | Native | – | Resident |
| Iran, Islamic Republic of | Extant | Native | – | Resident |
| Iraq | Extant | Native | – | Resident |
| Ireland | Extant | Native | – | Resident |
| Isle of Man | Extant | Native | – | Resident |
| Israel | Extant | Native | – | Resident |
| Italy | Extant | Native | – | Resident |
| Japan | Extant | Native | – | Resident |
| Korea, Democratic People’s Republic of | Extant | Native | – | Resident |
| Korea, Republic of | Extant | Native | – | Resident |
| Lebanon | Extant | Native | – | Resident |
| Libya | Extant | Native | – | Resident |
| Madagascar | Extant | Native | – | Resident |
| Malaysia | Extant | Native | – | Resident |
| Malta | Extant | Native | – | Resident |
| Mauritius | Extant | Native | – | Resident |
| Mauritius -> Rodrigues | Extant | Native | – | Resident |
| Mexico | Extant | Native | – | Resident |
| Monaco | Extant | Native | – | Resident |
| Morocco | Extant | Native | – | Resident |
| Mozambique | Extant | Native | – | Resident |
| Namibia | Extant | Native | – | Resident |
| Netherlands | Extant | Native | – | Resident |
| New Caledonia | Extant | Native | – | Resident |
| New Zealand | Extant | Native | – | Resident |
| Norway | Extant | Native | – | Resident |
| Oman | Extant | Native | – | Resident |
| Pakistan | Extant | Native | – | Resident |
| Peru | Extant | Native | – | Resident |
| Philippines | Extant | Native | – | Resident |
| Portugal | Extant | Native | – | Resident |
| Russian Federation | Extant | Native | – | Resident |
| Réunion | Extant | Native | – | Resident |
| Saint Helena, Ascension and Tristan da Cunha | Extant | Native | – | Resident |
| Saint Helena, Ascension and Tristan da Cunha -> Tristan da Cunha | Extant | Native | – | Resident |
| Saint Pierre and Miquelon | Extant | Native | – | Resident |
| Saudi Arabia | Extant | Native | – | Resident |
| Seychelles | Extant | Native | – | Resident |
| Seychelles -> Aldabra | Extant | Native | – | Resident |
| Slovenia | Extant | Native | – | Resident |
| South Africa | Extant | Native | – | Resident |
| South Georgia and the South Sandwich Islands | Extant | Native | – | Resident |
| Spain | Extant | Native | – | Resident |
| Sri Lanka | Extant | Native | – | Resident |
| Svalbard and Jan Mayen | Extant | Native | – | Resident |
| Sweden | Extant | Native | – | Resident |
| Syrian Arab Republic | Extant | Native | – | Resident |
| Tunisia | Extant | Native | – | Resident |
| Türkiye | Extant | Native | – | Resident |
| United Arab Emirates | Extant | Native | – | Resident |
| United Kingdom of Great Britain and Northern Ireland | Extant | Native | – | Resident |
| United States of America | Extant | Native | – | Resident |
| Venezuela, Bolivarian Republic of | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
| Presence | Origin | Formerly Bred | Seasonality | |
| 18. Arctic Sea | Extant | Native | – | Resident |
| 21. Atlantic – northwest | Extant | Native | – | Resident |
| 27. Atlantic – northeast | Extant | Native | – | Resident |
| 31. Atlantic – western central | Extant | Native | – | Resident |
| 34. Atlantic – eastern central | Extant | Native | – | Resident |
| 37. Mediterranean and Black Sea | Extant | Native | – | Resident |
| 41. Atlantic – southwest | Extant | Native | – | Resident |
| 47. Atlantic – southeast | Extant | Native | – | Resident |
| 48. Atlantic – Antarctic | Extant | Native | – | Resident |
| 51. Indian Ocean – western | Extant | Native | – | Resident |
| 57. Indian Ocean – eastern | Extant | Native | – | Resident |
| 58. Indian Ocean – Antarctic | Extant | Native | – | Resident |
| 61. Pacific – northwest | Extant | Native | – | Resident |
| 67. Pacific – northeast | Extant | Native | – | Resident |
| 71. Pacific – western central | Extant | Native | – | Resident |
| 77. Pacific – eastern central | Extant | Native | – | Resident |
| 81. Pacific – southwest | Extant | Native | – | Resident |
| 87. Pacific – southeast | Extant | Native | – | Resident |
| 88. Pacific – Antarctic | Extant | Native | – | Resident |
Climate change
There is not much information specific on the effects of climate change on fin whales.
Population
Conventionally, the Internation Whaling Commission (IWC) has assessed Fin Whales across the Southern Hemisphere within six key management areas, which are divided longitudinally, approximately between 50° and 70° wide. For Fin Whales, these management areas possess little biological distinction (Donovan 1991), as they are based primarily on Humpback Whale breeding and feeding grounds. Since the majority of hunting occurred within breeding grounds in lower latitudes rather than feeding areas in the high latitudes, most population estimates are described from feeding aggregations. It is probable that some whales within particular feeding grounds overlap and intermingle with other subpopulations across their breeding grounds in winter.
Between 1905 and 1976, more than 725,000 Fin Whales were documented as caught in the Southern Hemisphere (IWC 2006b). This figure, although possibly unreliable, was based on a number of sources, including catch per unit whaling trends, and sightings by Japanese scouting vessels. More recently, from data collected between 1966 and 1979, the IWC (1995) estimated the overall summer Fin Whale population south of 30°S at 18,000, and at 15,000 from data collected between 1979 and 1988. These results were produced by extrapolating population estimates for the region south of 60°S to the area south of 30°S from international surveys conducted by the International Decade of Cetacean Research (IDCR) and data from Japanese scouting ships. However, these estimates were considered rather imprecise and to be an overestimation of the population. Thus, using alternate methods, the same data produced estimates of 8,387 for 1966–1979 and 15,178 for 1979–1988 (IWC 1996). In agreement, Best (2003) suggested that the previous estimates may have been overly optimistic and proposed a decline of 89–97% between 1954 and 1975 for Fin Whales at the South African winter whaling grounds. There is no recent data documenting the current population status of this species, since the surveys that have been conducted in the Southern Hemisphere do not cover their entire summer distribution. However, Branch and Butterworth (2001) estimated Fin Whale abundance south of 60°S in the Southern Ocean for the period 1991/92–1997/98 as 5,500, an increase from the period 1985/86 – 1990/91, which was estimated as 2,100. Importantly, these results should not be recognized as estimates for the entire Southern Hemisphere population. Other data sources cover only limited areas and lack time-series to estimate trends. Estimates include 3,180 (CV 0.57) for the Scotia Sea, 1,492 (CV 0.57) for the Antarctic Peninsula, 4,898 (95%CI 2,221-7,575) for the Drake Passafe, 94 (95% CI 0-2010) for the Brainsfield Strait, 528 (95%CI 156-1,782) for Elephant Island, 796 (95%CI 249-2,541) for South Orkney Islands (IWC, 2024).
As an incredibly popular species of the Southern Hemisphere whaling industry following the Second World War, 28,761 Fin Whales were reportedly taken during the 1960/61 whaling season in Antarctica. Around this period, the global Fin Whale population was estimated to have declined from 400,000 to around 84,000, and as a result this species has been formally protected since 1976. The recovery rate (if any) of this species currently remains inconclusive, though Brown (1973) suggests that they may have a 37-year recovery period. However, the generation period of a non-depleted Fin Whale population is estimated at 25.9 years (Taylor et al. 2007).
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Current population trend: Increasing
Continuing decline in mature individuals: No
Number of mature individuals in population: Unknown, but possibly > 5,500.
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Strong genetic differentiation has been detected in the Northern Hemisphere, between whales of the North Pacific (NP) and North Atlantic (NA) (Bérubé et al. 1998; Archer et al. 2013; Cabrera et al. 2019). Based on new genetic evidence, Archer et al. (2019) formally described fin whales in the North Pacific as a new subspecies, B. p. velifera. As a result. four subspecies, supported by genetic differentiation, are accepted by the Committee on Taxonomy (Taxonomy, 2020).
Due to the limited geographic representation, especially within the southern hemisphere, in most of these studies, Perez-Alvarez et al. (2021) conducted another population genetic study across the entire species range, boosting representation from previously underrepresented areas. Their study showed three main global lineages, not four as previously identified. The study also found an absence of recurrent female mediated gene flow between hemispheres; although, rare dispersal events were detected revealing past migrations. The absence of genetic structure within the southern hemisphere suggests the existence of one subspecies within the Southern Hemisphere (B. p. quoyi) pending additional investigations supporting the subspecific designation of B. p. patachonica (e.g., genetic, ecological, behavioral, morphological). Based on this evidence, the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF’s) complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (all populations remaining). This is assuming no distinct subpopulations have gone extinct.
Due to limited current knowledge on population size and the fact that a genetic bottleneck likely transpired due to whaling, applying a general conversion ratio of Ne/Nc between 0.1-0.3 based on only population estimates could provide an overestimation of the effective population size for this population. This is because even if the species is showing signs of recovery through increased population numbers, levels of inbreeding made be elevated and would influence effective population size estimates (e.g. showing reduced genetic diversity, genetic erosion). Consequently, it would be ill advised to use proxies to quantify the GBF’s headline genetic indicator – proportion of populations with an Ne > 500. Should fresh tissue be difficult to acquire within the assessment region, it is highly recommended that a population genetic study be conducted using samples collected within the past 25 years (i.e., approximate generation length of the species) to more accurately estimate the genetic health and diversity of this potential subpopulation.
Habitats and ecology
Fin Whales are only known to form large aggregations within feeding grounds or while migrating but are usually present within schools of up to five individuals. Females with calves usually separate themselves from the rest of the group, and pregnant females are known to arrive at feeding grounds earlier than the rest of the group and leave earlier towards the end of the season (Skinner and Chimimba 2005). They are the fastest of all baleen whales and have been recorded swimming at speeds of approximately 20 knots over substantial distances. While feeding, Fin Whales engulf up to 70 tonnes of seawater into an elastic throat pouch, and filter food, in what has been declared the greatest biomechanical action exhibited by any animal (Brodie 1993). Southern Hemisphere Fin Whales consume mostly euphausiid prey but may fortuitously engulf fish simultaneously during feeding.
During migrations through waters off Durban to wintering grounds, whales caught had mostly empty stomachs. Only 12.7% contained food, which mostly consisted of euphasid remains (Euphausia recurva and Thysanoessa gregaria), but also amphipods, copepods, needlefish and megalopa larvae (Bannister and Baker 1967). On South Africa’s West Coast (at Donkergat), the stomachs of 35.8% of caught whales held food. Again, the majority were euphasiids (E. recurva, E. lucens, E. spinifera and T. gregaria), and to a lesser degree copepods and amphipods (Best 1967). It is uncertain whether they rely predominantly on blubber for energy during winter, or whether they actively locate food. Results have confirmed that Fin Whales are thinner at the beginning of summer when they return to their Antarctic feeding grounds. Furthermore, no feeding associated Fin Whale call, 40 Hertz pulse, was detected off the west coast of South Africa despite extended detection of their socialising call, the 20 Hertz pulse (Shabangu et al. 2019; Letsheleha et al. 2022). The Fin Whale 40 Hertz pulse was detected year-round around the Prince Edward Islands (Shabangu et al. 2024), indicating that Fin Whale feed and reside in that sub-Antarctic region.
On the contrary, in the northwest Atlantic, Overholz and Nicolas (1979) described Fin Whales feeding on American Sand Lance/Sand Eel (Ammodytes americanus); additionally in waters off Newfoundland, 80–90% of the diet of Fin Whales was thought to consist of Capelin (Mallotus villosus). The inter-annual variability associated with Capelin abundance suggests that Fin Whales probably feed opportunistically on this species when its availability increases.
Following a gestation period of 11 to 12 months, Fin Whale calves are born within temperate and subtropical waters in winter (typically between April and June). Newly born calves are usually around 6.4 m in length, and are generally dependent on their mother’s milk for the first seven months of life, before reaching a length of approximately 11.5 m. Females in the Southern Hemisphere become sexually mature after reaching a length of 20.0 m, and usually produce a calf every two years, while Northern Hemisphere individuals reach sexual maturity at shorter body lengths (Ohsumi et al. 1958).
Ecosystem and cultural services: Marine mammals integrate and reflect ecological variation across large spatial and long temporal scales, and therefore they are prime sentinels of marine ecosystem change; migratory mysticete whales may be used to investigate broadscale shifts in ecosystems (Moore 2008).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 9.1. Marine Neritic -> Marine Neritic – Pelagic | – | Suitable | Yes |
| 10.1. Marine Oceanic -> Marine Oceanic – Epipelagic (0-200m) | – | Suitable | Yes |
Life History
| Generation Length | Justification | Data Quality |
| 25.9 | – | – |
Age at Maturity: Female or unspecified: 6-12 years
Age at Maturity: Male: 4-11 years
Size at Maturity (in cms): Female: 2,000
Size at Maturity (in cms): Male: 1,740
Longevity: 85-90 years
Average Reproductive Age: 10 years
Maximum Size (in cms): 26,000
Size at Birth (in cms): 640
Gestation Time: 11-12 months
Reproductive Periodicity: 2-3 years
Average Annual Fecundity or Litter Size: 1
Natural Mortality: Between 0.04 and 0.06
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? Yes
Movement Patterns
Movement Patterns: Partial and full migrants
Congregatory: up to six individuals
Systems
System: Marine
General Use and Trade Information
Extensive commercial whaling of this species has ceased but is known to continue on a minor scale in the North Atlantic and Antarctic. Japanese scientific whaling in the Southern Hemisphere is set at 50 Fin Whales per year for human consumption. During the whaling era, Fin Whale blubber and baleen were used for products such as lamp oil, margarine, cooking oil, candles, soaps, cosmetics, corsets, umbrellas and tennis racquets, while whale meat was sold for human consumption, animal feed and fertiliser.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| No | – | – | – |
National Commercial Value: Yes
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | – | true | – | – |
| 15. Sport hunting/specimen collecting | – | true | – | – |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: (Not specified)
Threats
Before the commencement of modern whaling in the late 19th century, Fin Whales were mostly safe from human exploitation due to their speed and evasiveness. However, modern whaling, characterised by larger ships with diesel engines and helicopters to locate and overtake target animals, resulted in a major global decline of Fin Whales in the 20th century. Since 1976, Fin Whales in the Southern Hemisphere and North Pacific have been protected, and whaling in the North Atlantic ceased in 1990, aside from small-scale catches off Greenland. Japanese scientific whaling in Antarctica recommenced in 2005, where 10 whales were taken during both the 2005–2006 and 2006–2007 summer seasons, and with a proposal to take 50 individuals per year during the seasons to follow (IWC 2006a). In the waters off Iceland, commercial whaling resumed in 2006, with nine individuals of this species taken that same year. The likelihood that the commercial exploitation of this species will return to the historically high levels is low, owing to the limited demand for whale products. However, bycatch in fishing gear may be a minor threat to Fin Whales. For example, off the eastern coast of the United States, four mortalities and severe injuries were reported for Fin Whales between 2000 and 2004 (Cole et al. 2006). On average, the IWC records one Fin Whale caught in fishing nets per year. The severity of this threat within the assessment region is unknown.
Of the other large whale species, Fin Whales are one of the most commonly reported in collisions with ships (Laist et al. 2001, 2014). For example, off the east coast of the United States, five Fin Whales were fatally injured by ships between 2000 and 2004 (Cole et al. 2006). Additionally, in the Mediterranean, Fin Whale-vessel collisions appear to be an important source of mortality; however, it is not suspected to have any major effect on the population (Panigada et al. 2006). Similarly, within the oceans off South Africa, collisions with vessels are not deemed a major threat to this species.
A potential emerging threat to Fin Whales is the ingestion of microplastics, which has been documented in the blubber of stranded Fin Whales in the Mediterranean where Fossi et al. (2012) found 56% of planktonic samples contained microplastic samples. Fin Whales are sensitive to microplastic pollution due to their filter-feeding activity and have been proposed as an indicator species for this threat (Fossi et al. 2014). The severity of this threat within the assessment region is unknown.
Underwater noise from marine traffic is another threat to this species off the west coast of South Africa affecting the communication distance and acoustic behaviour where they vocalise more frequent to counter the increasing low frequency noise (Shabangu et al. 2022). These whales behave differently around the Prince Edward Islands where there is little to no marine traffic, and they do not respond to the increasing natural noise (Shabangu et al. unpublished data). Overall, the acoustic occurrence of Fin Whales off the west coast of South Africa (Shabangu et al. 2019; Letsheleha et al. 2022) and around the Prince Edward Islands (Shabangu et al. unpublished data) are predominantly influenced by environmental conditions, making this species vulnerable for climate change.
Overall, no current major threats to this species have been identified for Southern Hemisphere stocks, though climate change may severely impact food sources in the Antarctic, resulting from a decline in sea ice. For example, recent research predicts that sea ice in the Southern Hemisphere will reduce by more than 40% in the next century, thus affecting food resources for whales in the form of Euphausiacea spp. (Burns and Baker 2000).
Current habitat trend: Declining in the quality of feeding grounds off Antarctica, as a result of the effects of climate change (Burns & Baker 2000).
Conservation
Large-scale commercial whaling of Fin Whales in the North Pacific and Southern Hemisphere was deemed illegal by the IWC in 1976. In 1982 the IWC adapted the commercial whaling moratorium, which set all catch limits for commercial whaling to zero from 1986. The Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) listed Fin Whales on Appendix I, however this does not relate to Iceland, Norway or Japan. This species is also listed on Appendices I and II of the Convention on the Conservation of Migratory Species of Wild Animals (CMS). Within the assessment region, no specific interventions are necessary, but we recommend continuous systematic population monitoring to estimate population size and trend.
Recommendations for managers and practitioners:
- Systematic monitoring: design and implement a monitoring programme (acoustic and sightings) that can detect population size and trends.
- Implement vessel speed reduction during peak whale occurrence to reduce underwater noise.
Research priorities:
- Estimate current population size, distribution and trend.
- Quantify current and future threats which may impact this species or its habitat.
- Continue passive acoustic monitoring in South African waters.
- Stable isotope research to identify food source links.
Encouraged citizen actions:
- Whale-watching operators could contribute to photo-ID catalogues and behavioural observations.
- Report strandings to relevant authorities.
- Participate as volunteers in Fin Whale research projects.
- Reduce marine pollution.
Bibliography
Archer, F. I., Morin, P. A., Hancock-Hanser, B. L., Robertson, K. M., Leslie, M. S., Bérubé, M., et al. 2013. Mitogenomic phylogenetics of fin whales (Balaenoptera physalus spp.): genetic evidence for revision of subspecies. PLoS One 8:e63396. doi: 10.1371/journal.pone.0063396.
Archer, F. I., Brownell, R. L. Jr., Hancock-Hanser, B. L., Morin, P. A., Robertson, K. M., Sherman, K. K., et al. 2019. Revision of fin whale Balaenoptera physalus (Linnaeus, 1758) subspecies using genetics. J. Mammal. 100, 1653–1670. doi: 10.1093/jmammal/gyz121.
Bannister JL, Baker A de C. 1967. Observations on food and feeding of baleen whales at Durban. Norsk Hvalfangsttid 54: 78–82.
Bannister JL, Gambell R. 1965. The succession and abundance of fin, sei and other whales off Durban. Norsk Hvalfangsttid 54: 45–60.
Bérubé, M., Aguilar, A., Dendanto, D., Larsen, F., Notarbartolo Di Sciara, G., Sears, R., et al. 1998.. Population genetic structure of North Atlantic, Mediterranean Sea and Sea of Cortez fin whales, Balaenoptera physalus (Linnaeus, 1758): analysis of mitochondrial and nuclear loci. Mol. Ecol. 7, 585–599. doi: 10.1046/j.1365-294x.1998.00359.x.
Best PB. 1967. Distribution and feeding habits of baleen whales off the Cape Province. Republic of South Africa, Department of Commerce and Industries, Division of Sea Fisheries.
Best, P. B. 1994. A review of the catch statistics for modern whaling in southern Africa, 1908-1930. Reports of the International Whaling Commission 44: 467-485.
Best, P.B. 2003. How low did they go? An historical comparison of indices of abundance for some baleen whales on the Durban whaling ground. International Whaling Commission Scientific Committee doc. SC/55/SH18.
Branch, T.A. and Butterworth, D.S. 2001. Estimates of abundance south of 60°S for cetacean species sighted frequently on the 1978/79 to 1997/98 IWC/IDCR-SOWER sighting surveys. Journal of Cetacean Research and Management 3(3): 251-270.
Brodie PF. 1993. Noise generated by the jaw actions of feeding fin whales. Canadian Journal of Zoology 71: 2546–2550.
Brown SG. 1973. Whale marking – progress report 1972. Reports of the International Whaling Commission.
Burns WC, Baker DJ. 2000. From the harpoon to the heat: Climate change and the International Whaling Commission in the 21st century. Journal of International Wildlife Law & Policy 3: 50– 72.
Cabrera, A. A., Hoekendijk, J. P. A., Aguilar, A., Barco, S. G., Berrow, S., Bloch, D., et al. (2019). Fin whale (Balaenoptera physalus) mitogenomics: a cautionary tale of defining sub-species from mitochondrial sequence monophyly. Mol. Phylogen. Evol. 135, 86–97. doi: 10.1016/j.ympev.2019.02.003.
Clarke, R. 2004. Pygmy fin whales. Marine Mammal Science 20(2): 329-334.
Cole, T., Hartley, D. and Garron, M. 2006. Mortality and serious injury determinations for large whale stocks along the eastern seaboard of the United States 2000-2004.
Donovan, G.P. 1991. A review of IWC stock boundaries. Reports of the International Whaling Commission 13: 39-68.
Fossi MC, Coppola D, Baini M, Giannetti M, Guerranti C, Marsili L, Panti C, de Sabata E, Clò S. 2014. Large filter feeding marine organisms as indicators of microplastic in the pelagic environment: The case studies of the Mediterranean basking shark (Cetorhinus maximus) and fin whale (Balaenoptera physalus). Marine Environmental Research 100: 17–24.
Fossi MC, Panti C, Guerranti C, Coppola D, Giannetti M, Marsili L, Minutoli R. 2012. Are baleen whales exposed to the threat of microplastics? A case study of the Mediterranean fin whale (Balaenoptera physalus). Marine Pollution Bulletin 64(2374–2379).
IUCN (International Union for Conservation of Nature). 2013. Balaenoptera physalus. Available at: http://www.iucnredlist.org. (Accessed: 21 February 2016).
IWC. 1995. Report of the scientific committee. Report of the International Whaling Commission 45: 53–221.
International Whaling Commission. 1996. Report of the subcommittee on Southern Hemisphere baleen whales. Report of the International Whaling Commission 46: 117-138.
International Whaling Commission. 2006. Report of the Scientific Committee. Journal of Cetcaean Research and Management 8: 49.
International Whaling Commission. 2006. The IWC Summary Catch Database.
International Whaling Commission 2024. Report of the Scientific Committee (SC69B) – Bled, Slovenia 22 April – 3 May 2024.
Laist DW, Knowlton AR, Pendleton D. 2014. Effectiveness of mandatory vessel speed limits for protecting North Atlantic right whales. Endangered Species Research 23: 133-147.
Laist, D. W., Knowlton, A. R., Mead, J. G., Collet, A. S. and Podesta, M. 2001. Collisions between ships and whales. Marine Mammal Science 17(1): 35-75.
Letsheleha IS, Shabangu FW, Farrell D, Andrew RK, la Grange PL, Findlay KP. 2022. Year-round acoustic monitoring of Antarctic blue and fin whales in relation to environmental conditions off the west coast of South Africa. Marine Biology 169: 41. https:// doi. org/ 10.1007/ s00227- 022- 04026-x
Miyashita, T., Kato, H. and Kasuya, T. 1996. Worldwide Map of Cetacean Distribution Based on Japanese Sighting Data. National Research Institute of Far Seas Fisheries.
Moore SE. 2008. Marine mammals as ecosystem sentinels. Journal of Mammalogy 89: 534-540.
Moore, M. J., Berrow, S. D., Jensen, B. A., Carr, P., Sears, R., Rowntree, V. J., Payne, R. and Hamilton, P. K. 1999. Relative abundance of large whales around South Georgia (1979-1998). Marine Mammal Science 15(4): 1287-1302.
Nishiwaki M. 1972. General Biology. In: Ridgeway SH. (ed.), Mammals of the Sea, pp. 3–200. Charles C. Thomas, Springfield, USA.
Ohsumi S, Nishiwaki M, Hibiya T. 1958. Growth of fin whale in the Northern Pacific. Scientific Reports of the Whales Research Institute 13: 97–133.
Overholtz W. J. and Nicolas J. R. 1979. Apparent feeding by the fin whale Balaenoptera physalus and the humpback whale Megaptera novaeangliae on the American sand lance Ammodytes americanus in the northwest Atlantic. US Fisheries Bulletin 88(4): 687-696.
Panigada, S., Pesante, G., Zanardelli, M., Capoulade, F., Gannier, A. and Weinrich, M. 2006. Mediterranean fin whales at risk from fatal ship strikes. Marine Pollution Bulletin 52: 1287-1298.
Pérez-Alvarez MJ, Kraft S, Segovia NI, Olavarría C, Nigenda-Morales S, Urbán RJ, Viloria-Gómora L, Archer F, Moraga R, Sepúlveda M, Santos-Carvallo M, Pavez G and Poulin E (2021) Contrasting Phylogeographic Patterns Among Northern and Southern Hemisphere Fin Whale Populations With New Data From the Southern Pacific. Front. Mar. Sci. 8:630233. doi: 10.3389/fmars.2021.630233.
Reilly SB et al. 2013. Balaenoptera physalus. The IUCN Red List of Threatened Species 2013.
Shabangu, F. W., Findlay, K. P., Yemane, D., Stafford, K. M., van den Berg, M., Blows, B. & Andrew, R. K. 2019. Seasonal occurrence and diel calling behaviour of Antarctic blue whales and fin whales in relation to environmental conditions off the west coast of South Africa. Journal of Marine Systems 190: 25-39. https://doi.org/10.1016/j.jmarsys.2018.11.002
Shabangu, F. W., Andrew, R. K., Yemane, D., and Findlay, K. P. (2020). Acoustic Seasonality, Behaviour and Detection Ranges of Antarctic Blue and Fin Whales Under Different Sea Ice Conditions Off Antarctica. Endangered Species Research 43: 21–37. doi: 10.3354/esr01050
Shabangu FW, Yemane D, Best G, Estabrook BJ. 2022. Acoustic detectability of whales amidst underwater noise off the west coast of South Africa. Marine Pollution Bulletin 184: 114122. (doi:10.1016/j.marpolbul.2022.114122(doi:10.1016/j.marpolbul.2022.114122)
Shabangu, F.W., Munoz, T., Van Uffelen, L., Estabrook, B.J., Yemane, D., Stafford, K.M., Branch, T.A., Vermeulen, E., van den Berg, M.A., Lamont, T., 2024. Diverse baleen whale acoustic occurrence around two sub-Antarctic Islands: A tale of residents and visitors. Scientific Reports 14: 21663. https://doi.org/10.1038/s41598-024-72696-2.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor, B.L., Chivers, S.J., Larese, J. and Perrin, W.F. 2007. Generation length and percent mature estimates for IUCN assessments of cetaceans. NOAA, Southwest Fisheries Science Center Administrative Report LJ-07-01. La Jolla, California.
Taxonomy, C. O. 2020. List of Marine Mammal Species and Subspecies. Lawrence, KS: Society for Marine Mammalogy.
