Southern Elephant Seal
Mirounga leonina

2025 Red list status
Least Concern
Regional Population Trend
Increasing
Change compared
to 2016
Downlisted
Overview
Mirounga leonina – (Linnaeus, 1758)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – PHOCIDAE – Mirounga – leonina
Common Names: Southern Elephant Seal, South Atlantic Elephant-seal, Southern Elephant-seal (English), Suidelike Olifantrob (Afrikaans), Elefante Marino del Sur (Spanish; Castilian), Eléphant de mer Austral, Eléphant de mer du sud (French)
Synonyms: Phoca leonina Linnaeus, 1758
Taxonomic Note:
None
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessor: de Bruyn, P.J.N.1
Reviewer: Nicholson, S. K.2
Contributor: da Silva, J.M.3 & Patel, T.2
Institutions: 1University of Pretoria, South Africa,2Endangered Wildlife Trust, 3South African National Biodiversity Institute
Previous Assessors & Reviewers: de Bruyn, N., Bester, M., Oosthuizen, C., Hofmeyr, G.J.G. & Pistorius, P.
Previous Contributors: Kirkman, S., Oosthuizen, H., Meyer, M., Seakamela, M., Lowry, L., Tosh, C., Nicholson, S., Wege, M. & Child, M.F.
Assessment Rationale
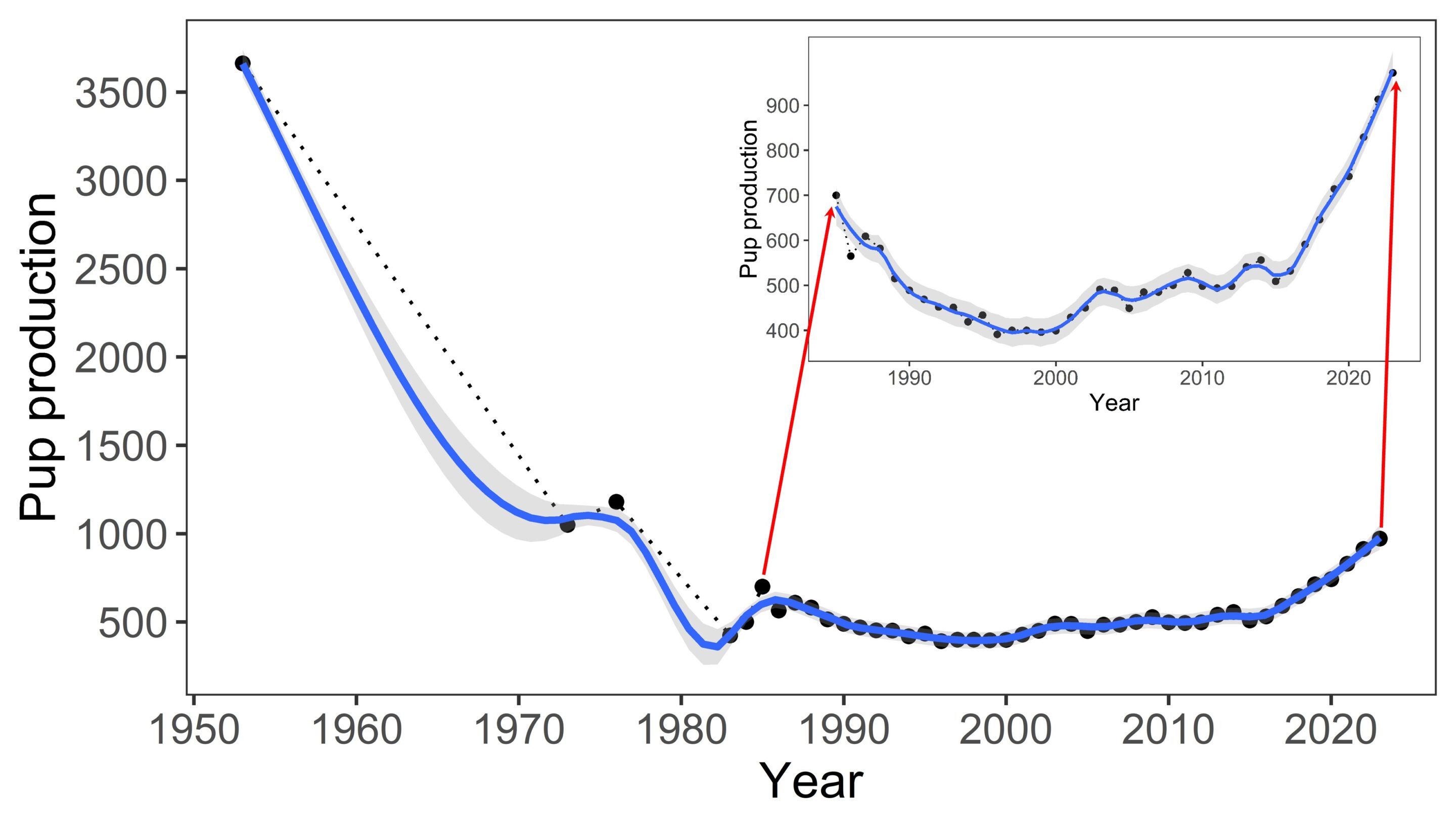
No serious threats have been affecting the land breeding colonies at the Prince Edward Islands in the last 40 years (four generations time). The population on Marion Island, as estimated through annual pup production, declined by 83% between 1951 and 1994, which prompted the Endangered A2b listing in 2004. At its nadir in 1997 annual pup production at Marion Island was 421, but this has gradually increased to around 972 pups in 2023, representing a mature population of 3,062. If females not breeding in that year and neighbouring Prince Edward Island’s population are included (see below), it probably equates to about 5,500 individuals in total for the Prince Edward islands archipelago population. Thus, although the Marion Island population declined by 37% between 1986 and 1994, when observed collectively over the past four generations (1986–2023), the population has shown an increase (from 690 pups in 1986 to 972 pups in 2023) by about 29% and in fact pup production has increased by 47% between 1997 and 2023, providing hope for a sustained positive population trajectory. The pup production has not yet recovered to the ~3,500 estimated at first assessment of the Marion Island population in the early 1950’s, but current production is fast approaching mid-1970’s estimates of ~1,000 pups (Figure 2). A late breeding season survey in November 2023 (the first in two decades) at Prince Edward Island showed a pup production there of 412 (given their close proximity, we assume similar pup production trajectories for the neighbouring Marion and Prince Edward islands).
The well monitored Marion Island population (likely mirrored in the neighbouring Prince Edward Island population) has shown steady increase in pup production since the mid-1990’s. Temporary reduction between 1983 and 1997 was close to the threshold for applying the Near Threatened listing for the A criterion (20–25% reduction; IUCN Standards and petitions Subcommittee 2014). However, with current positive population trajectory and no major threats that could cause rapid population decline, we list the national population of Southern Elephant Seals as Least Concern. It is also worth noting that we do not fully understand the cause/s of the past decline, albeit attributed to food limitation. The causes of changes in food availability remains poorly understood. In all likelihood, climate-related impacts and oceanographic changes have influenced prey availability and distribution as well as spatial distribution of Southern Elephant Seal foraging efforts.
Regional population effects: The global range is continuous and connected by movement of individuals between islands, with potential for augmentation or rescue of locally declining or extinct subpopulations. There is connectivity with the Îles Crozet but also with distant Îles Kerguelen (see Oosthuizen et al. 2011).
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: Sustained positive trajectory in pup production at Marion Island (likely mirrored at neighbouring Prince Edward Island), collectively the only breeding site for the species within the Region, has resulted in a change from Near Threatened (previous assessment) to Least Concern (this assessment).
Red List Index
Red List Index: Downlisted
Recommended citation: de Bruyn PJN. 2025. A conservation assessment of Mirounga leonina. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Southern Elephant Seals have a circumpolar distribution in the Southern Hemisphere. Although they reach the Antarctic continent and even very high latitude locations such as Ross Island, they are most common north of the seasonally shifting pack ice, especially in Subantarctic waters where most rookeries and haul-outs are located. The northern-most breeding locality for the species is Gough Island, southern Atlantic, although this population is probably heading for local extinction (pup production decreased from c. 18 to c.2 / year; Bester et al. 2001, Jones et al. 2020).
Some pups are also born on the Antarctic continent. Southern Elephant Seals prefer sandy and cobble beaches, but will haul-out on sea ice, snow and rocky terraces and regularly rest (but especially moult) above the beach in tussock grass, other vegetation, and mud wallows. At sea, females and males tend to disperse to different feeding grounds, although there is large individual variation in foraging site fidelity, and some overlap between the sexes and ages.
Within the assessment region, this species breeds on Prince Edward Island and Marion Island. Movement at sea is considerable and, even though breeding colonies are disjunct or fragmented, there is movement of individuals between colonies to ensure gene flow (see section below).
Wandering and vagrant Southern Elephant Seals reach southern Africa (with 1–10 sightings per year off South African coastlines).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): Diving beyond 2000m
Depth Upper Limit (in metres below sea level): Must breathe above the surface
Depth Zone: (Not specified)
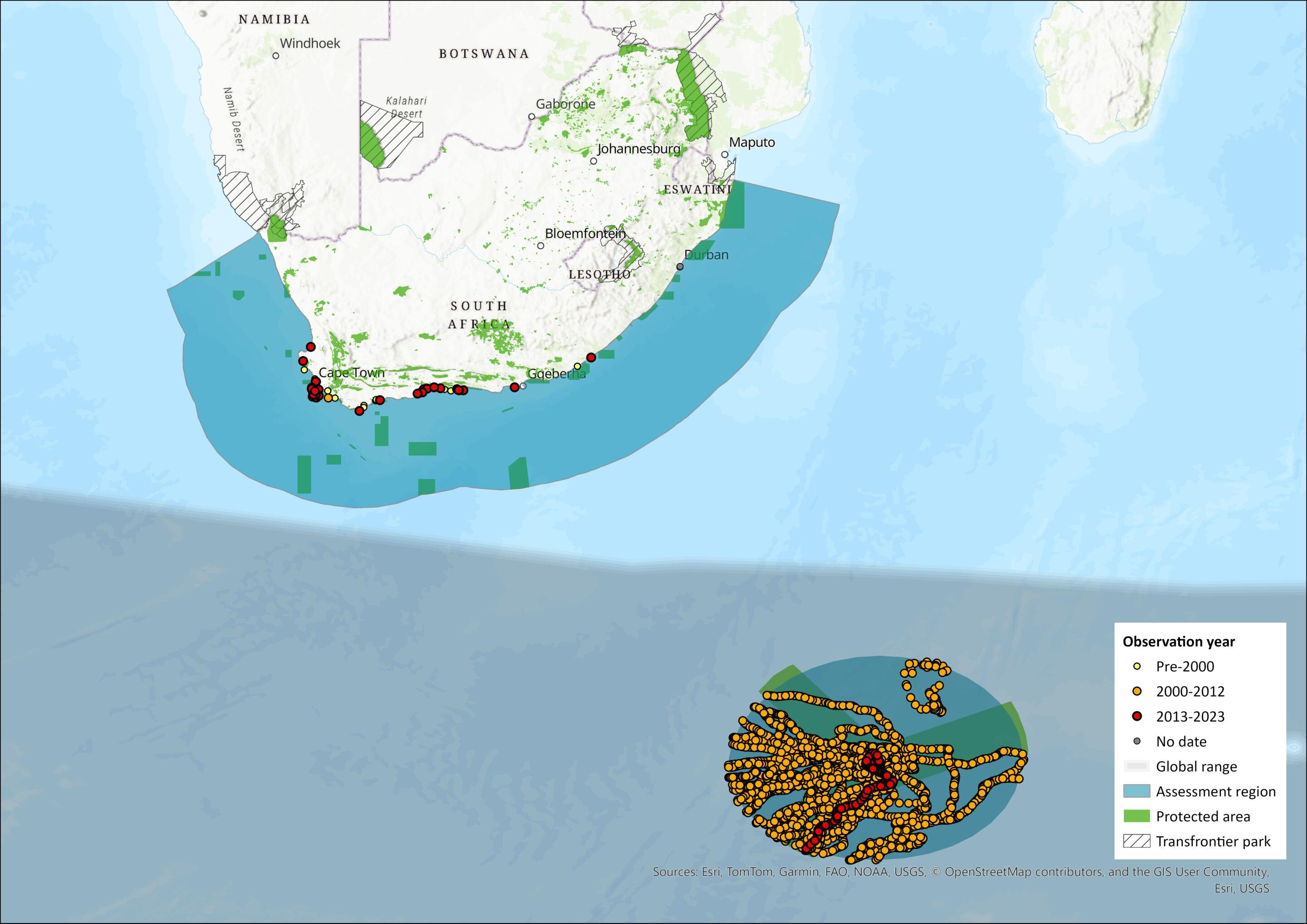
Map
Figure 1. Distribution records for Southern Elephant Seal (Mirounga leonina) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Antarctic, Australasian, Neotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Vagrant | – | – |
| Antarctica | Extant | Native | – | – |
| Argentina | Extant | Native | – | – |
| Australia | Extant | Native | – | – |
| Australia -> Macquarie Is. | Extant | Native | – | – |
| Australia -> New South Wales | Presence Uncertain | Vagrant | – | – |
| Australia -> South Australia | Presence Uncertain | Vagrant | – | – |
| Australia -> Victoria | Presence Uncertain | Vagrant | – | – |
| Bouvet Island | Extant | Native | – | – |
| Brazil | Extant | Vagrant | – | – |
| Chile | Extant | Native | – | – |
| Falkland Islands (Malvinas) | Extant | Native | – | – |
| French Southern Territories | Extant | Native | – | – |
| French Southern Territories -> Crozet Is. | Extant | Native | – | – |
| French Southern Territories -> Kerguelen | Extant | Native | – | – |
| Heard Island and McDonald Islands | Extant | Native | – | – |
| Mauritius | Extant | Vagrant | – | – |
| Mozambique | Extant | Vagrant | – | – |
| Namibia | Extant | Vagrant | – | – |
| New Zealand | Extant | Native | – | – |
| New Zealand -> Antipodean Is. | Extant | Native | – | – |
| New Zealand -> Chatham Is. | Presence Uncertain | Vagrant | – | – |
| New Zealand -> North Is. | Presence Uncertain | Vagrant | – | – |
| New Zealand -> South Is. | Presence Uncertain | Vagrant | – | – |
| Oman | Extant | Vagrant | – | – |
| Peru | Extant | Vagrant | – | – |
| Saint Helena, Ascension and Tristan da Cunha | Extant | Native | – | – |
| Saint Helena, Ascension and Tristan da Cunha -> Tristan da Cunha | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| South Africa -> Marion-Prince Edward Is. | Extant | Native | – | – |
| South Georgia and the South Sandwich Islands | Extant | Native | – | – |
| Uruguay | Extant | Vagrant | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified) Southern Ocean (circumpolar)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
The possible effects of global climate and associated oceanographic change on Southern Elephant Seals are not well understood. Learmonth et al. (2006) suggested that while the effects of global climate change are uncertain, the species is likely to decline as a result of habitat and ecosystem changes. For example, predicted reduction in sea ice habitats due to continued climate warming will impact the distribution of food and breeding habitat for the species (Hindell et al. 2017). Several recent studies have linked climate-mediated ecosystem changes to variability in demographic parameters for female and male southern elephant seals (Volzke et al. 2021, 2024 and references therein). However, predictions are not clear yet regarding how these effects will impact the size or trajectory of populations. Certainly, despite evident changes in the climate associated with the sub-Antarctic region in general and the Prince Edward Islands region specifically (Nel et al. 2023) the local population of southern elephant seals are currently thriving (Christian et al. 2024).
Population
The worldwide population of Southern Elephant Seals was estimated to be 650,000 in the mid-1990s. No recent integrated estimate is available throughout the entire distribution.
Traditionally, three distinct provinces/populations have been distinguished: South Georgia, Macquarie and Îles Kerguelen. However, Elephant Seals breeding at Península Valdes on the mainland of South America and at the Falkland (Malvinas) Islands are now thought to be a distinct province/population from those at South Georgia (see Genetics section). Similarly, the Elephant Seal populations at the Prince Edward Islands and Îles Crozet are also considered to be distinct from those at Îles Kerguelen and Heard Island, and the small subpopulation at Gough Island may also be distinct. Additionally, some colonies have unknown affiliations (e.g. Bouvet). Thus, there may be at least five and perhaps more, distinct breeding populations, although their foraging areas overlap quite extensively at sea (Reijnders et al. 1993; Bailleul et al. 2007; Biuw et al. 2007) and we suspect there is gene flow between the subpopulations (Reisinger & Bester 2010).
Due to the strict conservation status of Prince Edward Island access is sporadic and difficult. Population monitoring of Southern Elephant Seals is therefore based on annual censuses conducted on Marion Island which hosts the bulk of the Prince Edward Islands population. Adult females haul out synchronously to reach a peak on 15 October during which island-wide counts of females and pups are conducted. Based on life table analyses it has been estimated that the entire population can be estimated through multiplying pup numbers by a conversion factor of 3.15 (Pistorius et al. 1999). However, this is based on the assumption that all females (past primiparity) breed annually, which is not the case (de Bruyn et al. 2011). In fact, about half seem to breed annually, so the correction factor based on 972 pups (2023) would give us around 3,062 animals from all sex-age categories annually, but because of skipping we would actually have a higher number in the mature population.
Within the assessment region, two periods of population decline are recorded: hunting pressure during the 1900s significantly reduced population size, which then recovered until the 1950s, but then saw a sharp decline of 83% between 1951 and c. 1994/1997 (Pistorius et al. 1999; McMahon et al. 2009), for largely unknown reasons although the “food limitation hypothesis” (suspected loss of prey availability, the drivers of which remain speculative) has received most support (Pistorius et al. 1999). Between 1986 and 1994 the population declined by 37% (2,120 [690 pups born] to 1,330 [437 pups born] individuals, Pistorius et al. 1999). Over the last four generations (1984/1986 – 2023), the population decline firstly decelerated to 20% (from 690 pups to 552 pups) and in fact pup production (based on direct counts) has increased by 47% between 1997 and 2023 (from 421 to 972 pups), with signs of continued exponential recovery for a sustained positive population trajectory (Figure 2).
The population remained stable between 1993 and 1999 (Pistorius et al. 2004 but see McMahon et al. 2009) and has since shown an increase; numbering about 3,062 individuals (972 pups born) in 2023 at Marion Island alone (Pistorius et al. 2011). By utilizing total peak breeding season counts at Marion Island and including conversion factors from pup numbers, the mature archipelago population (acknowledging skipping of breeding seasons by many mature females), including the 2023 opportunistic survey at the smaller Prince Edward Island (pup production of 412), numbers between 3,000 and 5,500 individuals (Mammal Research Institute, unpubl. data).
Figure 2: Population count (pup production) of the Southern Elephant Seal (Mirounga leonina) between 1950 and 2023.
Population Information
Current population trend: Increasing
Continuing decline in mature individuals? No
Extreme fluctuations in the number of subpopulations: No
Continuing decline in number of subpopulations: No
All individuals in one subpopulation: No
Number of mature individuals in largest subpopulation: 3062
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
The global population of Southern Elephant Seals is divided into four genetically distinct subpopulations or “stocks”, namely the 1) Peninsula Valdés – Argentina, 2) South Georgia, 3) Kerguelen, and 4) Macquarie stocks (Slade et al. 1998; Hoelzel et al. 2001). The elephant seals on the Prince Edward Islands (PEI and Marion) form part of the Kerguelen or South Indian Ocean stock.
The genetic bottleneck legacy that is well documented for the Northern Elephant Seal (M. angustirostris) has not been mirrored by the southern species as a whole (as evidenced by allozymes, mitochondrial DNA, minisatellite DNA, microsatellite DNA and immune system genes; Hoelzel et al. 1993; 1999).
The highly philopatric breeding habits of the species reinforce the genetic differentiation between major stocks. However, given the ability of the species to range over massive distances (primarily for foraging), some instances of breeding dispersal (and thus gene flow) between stocks have been reported (e.g. Fabiani et al. 2003; Reisinger & Bester 2010). There is no evidence of population structure within the subpopulations located in the assessment region. Consequently, they are considered a single metapopulation. Based on this information, the Convention on Biological Diversity’s Global Biodiversity Framework’s complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (1/1 population remaining).
While contemporary estimates of effective population size (Ne) are not available, it can be inferred from the available population estimates (3 062-5 500; see Population section). Using a conversion ratio of 0.1-0.3 for Ne/Nc, Ne is approximated between 306 and 1 650. These estimates assume the metapopulation exists in isolation (if not this could elevate estimates) and previously bottlenecks have not had a major impact (if not, estimates could be reduced). Given this uncertainty, it is difficult to quantify the GBF’s headline indicator – proportion of species with an effective population size (Ne) greater than 500. It is highly recommended that Ne be estimated from genetic data to clarify this uncertainty.
Habitats and ecology
The Southern Elephant Seal is the largest pinniped species. Adult males typically reach 4.5 m and a maximum of 5.8 m in length, and weigh 1,500–3,000 kg, with a maximum weight of about 4,000 kg. Adult females are similar in size and weight to Northern Elephant Seal females weighing 350–600 kg with exceptionally large females reaching 800 kg. Newborn pups are about 1.3 m and 40–50 kg. Pups are born in a long woolly black lanugo coat that is shed at about 3 weeks of age, to reveal a silver grey counter-shaded coat that is yellowish grey ventrally.
Females reach sexual maturity between 3 and 5 years and males reach sexual maturity at an age of 5 years. However, few males breed until they reach social maturity at an age of least 10 years (Jones 1981). At Marion Island, rather anomalously, many males commence breeding at 7 or 8. Ninety percent of males die before the age of 10 years while 90% of females die before the age of 14 years (McCann 1980; Pistorius et al. 1999).
Elephant Seals have an annual cycle with two well-defined pelagic phases, with transitions being marked by moult and reproduction. Adult males and females come ashore to reproduce from August to October. This species provides one of the most extreme examples of polygyny among mammals on land, but recent evidence suggests an alternative strategy in the species with some females mating pelagically (de Bruyn et al. 2011). Although females are capable of annual pup production, many females skip breeding seasons (de Bruyn et al. 2011). The social units are harems, each held by a single dominant male that monopolises access to around 60 sexually receptive females (at Marion Island) for a period of approximately two months. Early in the breeding season males establish dominance hierarchies on beaches via impressive displays, which include rearing up on the hindquarters and lifting almost two-thirds of the body straight up to fight with a peer or issue vocal challenges to nearby bulls.
Vocalisations include a booming, loud call of the adult male in the breeding season, variously called a bubbling roar, a harsh rattling sound, and a low-pitched series of pulses with little variation in frequency. Adult females have a high-pitched yodelling call which they use when distressed, and to call their pups. They will also utter a low pitch, sputtering growl. Pups call to their mothers with a sharp bark or yap, which is also used when interacting with other seals.
Southern Elephant Seals spend a large percentage of their lives at sea (McIntyre et al. 2010) and only return to land to give birth, breed and moult, although especially young animals sometimes engage in a little-understood winter rest haulout. At sea, they range far from their rookeries and predominantly feed between the Subantarctic Convergence and the northern edge of the pack ice, south of the Antarctic Convergence. Adult males typically venture further south than females, and are known to forage at the seaward edge of the Antarctic continental shelf. However, at Marion Island they forage pelagically over very deep water, unlike many of the other populations that feed benthically or over much shallower water (McIntyre et al. 2011).
Foraging Elephant Seals combine exceptionally deep diving with long-distance travelling, covering millions of square kilometres while traversing a wide range of oceanographic regions during periods of up to seven months at sea (McIntyre et al. 2010). The seals spend most of their at-sea time in particular water masses that include frontal systems, currents and shifting marginal ice-edge zones. Studies of foraging locations suggest that seals are sensitive to fine-scale variation in bathymetry and ocean surface properties (sea-ice concentration and sea surface temperature) (McIntyre et al. 2011; Tosh et al. 2015).
Although not well understood, prey consists predominantly of squid and fish. Antarctic Notothenia fishes are thought to be important prey when these seals are near the Antarctic continental shelf. Most feeding by females occurs in deep ocean areas at mid-water depths (McIntyre et al. 2010). Juvenile southern elephant seals from Marion Island present a large proportion of sub-Antarctic krill (up to 76%) in their diet (Lübcker et al. 2017).
Prince Edwards Islands have been proclaimed a Marine Protected Area, although human traffic due to research activities may have a minor and spatially limited disturbance effect on Marion Island.
Subantarctic islands (the pebble or sand beaches in particular) are vital haul-out habitats for breeding and moult in the species.
Ecosystem and cultural services: Southern Elephant Seals are major consumers in the Southern Ocean, playing a critical role as indicators of change in the ecosystems. As ubiquitous visitors to land, these apex predators provide opportunity for dedicated long term life history studies by field researchers. They are therefore a useful model species for assessment of demographic change, drivers of change and consequences thereof within this ecosystem. As top predators within the Prince Edward Islands ecosystem, they form important links in the islands’ marine food web. Through import of large quantities of nutrients from the ocean when they breed and moult on the islands they markedly enhance soil and plant nutrient status in the vegetated areas in which they occur (Smith 2008).
In addition their value in the historical “sealing” industry is useful for understanding socio-political and economic drivers of 19th and 20th century human endeavours into unchartered territories in the southern latitudes of our world.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 9.1. Marine Neritic -> Marine Neritic – Pelagic | – | Suitable | Yes |
| 10.1. Marine Oceanic -> Marine Oceanic – Epipelagic (0-200m) | – | Suitable | Yes |
| 10.2. Marine Oceanic -> Marine Oceanic – Mesopelagic (200-1000m) | – | Suitable | Yes |
| 10.3. Marine Oceanic -> Marine Oceanic – Bathypelagic (1000-4000m) | – | Marginal | – |
| 12.1. Marine Intertidal -> Marine Intertidal – Rocky Shoreline | – | Suitable | Yes |
| 12.2. Marine Intertidal -> Marine Intertidal – Sandy Shoreline and/or Beaches, Sand Bars, Spits, Etc | – | Suitable | Yes |
| 12.3. Marine Intertidal -> Marine Intertidal – Shingle and/or Pebble Shoreline and/or Beaches | – | Suitable | Yes |
| 13.1. Marine Coastal/Supratidal -> Marine Coastal/Supratidal – Sea Cliffs and Rocky Offshore Islands | – | Suitable | Yes |
Life History
Generation Length: 4-10 years
Age at Maturity: Female or unspecified: Earliest primiparity at 3 yrs, but most females start breeding at 4 yrs
Age at Maturity: Male: Sexual maturity as early as 5 yrs, but social maturity ~7 yrs
Size at Maturity (in cms): Female: 2.0 – 3.2m
Size at Maturity (in cms): Male: 4.5 – 5.8m
Longevity: Females: max 24 yrs. Males: max 14 yrs
Average Reproductive Age: Females: 4 yrs; Males: 7 yrs
Maximum Size (in cms): Females 320cm; Males 580cm
Size at Birth (in cms): ~130cm standard length
Gestation Time: 1 year including several months of delayed blastocyst implantation
Reproductive Periodicity: minimum 1 year, but many females skip breeding years
Average Annual Fecundity or Litter Size: 1
Natural Mortality: ~50% in first year, decreasing to <10% thereafter
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Far ranging (>1000kms), deep diving (>1000m) pelagic interspersed with terrestrial haul out.
Congregatory: Polygynous mating system harem formation on beaches during breeding season. Other haulout events (moult, winter) singly, or more generally in small (2-5 individuals) to very large huddles (20-40 individuals).
Systems
System: Marine
General Use and Trade Information
Southern Elephant Seals were historically commercially harvested, starting in the early 19th century and not ending completely until 1964 at South Georgia. They were prized for their large quantity of blubber that could be rendered to fine, valuable oil. This hunting caused precipitous declines. However, this species is no longer commercially harvested.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are few threats and conflicts today, as Southern Elephant Seals live far from human population centres and have minimal interactions with commercial fisheries but are affected to various degrees by entanglement in fishing gear (Hofmeyr et al. 2002; Campagna et al. 2007; Jepsen & de Bruyn 2019). Intensive fishing could potentially deplete important prey stocks. However, relatively little is known about their feeding habits. There is no evidence that past declines for animals breeding in the Indian and Pacific Oceans are related to fisheries in the Southern Ocean. However, development of new (and growth of existing) fisheries at high latitudes could have a significant impact on Elephant Seal populations.
Southern Elephant Seals that haul out at mainland sites could come in contact with feral dogs and other terrestrial carnivores and be exposed to a variety of diseases including morbilliviruses (Bester 2014) or rabies (Winkler & Parker 2024).
Small population sizes facilitate minor threats such as predation and genetic drift, which may threaten local breeding colonies. Reisinger et al. (2011) refuted the claim that predation from Killer Whales (Orcinus orca) is a current threat at the Prince Edward islands, although such predation undoubtedly plays some regulatory role on this population. Furthermore, there is immigration into the Prince Edward Islands population from elsewhere (particularly the French Islands to the east), shown to contribute markedly to the Prince Edward Island population (Christian et al. 2024). Changes in Elephant Seal numbers at distant islands can therefore impact local population growth.
Conservation
Any future exploitation within the Antarctic Treaty area (south of 60ºS) would be regulated by the Convention for the Conservation of Antarctic Seals. Listed on CITES Appendix II.
Seals on the Prince Edward Islands are protected by virtue of these islands’ status as a special nature reserve and also by the South African Seabirds and Seals Protection Act (PEIMP 2010), and thus the breeding grounds within the assessment region are secure.
Recommendations for land managers and practitioners:
- Continuation of monitoring and research programme conducted to date (over the past 4 decades) by the Marion Island Marine Mammal Programme under the auspices of the Mammal Research Institute, University of Pretoria, and recently the South African Polar Research Infrastructure (SAPRI).
Research priorities:
- Understanding cause of decline since the 1950s, and reasons for current increase.
- Understanding the causes and consequences of skipping behaviour in breeding female southern elephant seals.
- Understanding individual temporal and spatial variation in foraging behaviour.
- The effect of global climate change on the foraging and breeding behaviour of this species.
- The incredible physiological feats shown by the species, for example deep diving, breath holding and fasting abilities, provide challenging questions to enhance our broader understanding of ecophysiology, environmental adaptation and evolution in mammals more generally.
Encouraged citizen actions:
- Foraging ecology research is limited by the financial investment that is required for satellite telemetry. Aid in funding this aspect can be securely provided through: http://www.givengain.com/cause/4655/
Bibliography
Bailleul, F., Charrassin, J. B., Monestiez, P., Roquet, F., Biuw, M. and Guinet, C. 2007. Successful foraging zones of southern elephant seals from the Kerguelen Islands in relation to oceanographic conditions. Philosophical Transactions of the Royal Society B, Biological Sciences 362: 2169-2181.
Bester M. 2014. Marine Mammals – Natural and Anthropogenic Influences. In: Freedman B. (ed.), Global Environmental Change, pp. 167–174. Springer, Dordrecht, Netherlands.
Bester MN, Möller H, Wium J, Enslin B. 2001. An update on the status of southern elephant seals at Gough Island. South African Journal of Wildlife Research 31: 68–71.
Biuw, M., Boehme, L., Guinet, C., Hindell, M., Costa, D., Charrassin, J. B., Roquet, F., Bailleul, F., Meredith, M., Thorpe, S., Tremblay, Y., McDonald, B., Park, Y. H., Rintouul, S. R., Bindoff, N., Boebel, M., Crocker, D., Lovell, P., Nicholson, J., Monks, F. and Fedak, M. A. 2007. Variations in behavior and condition of a Southern Ocean top predator in relation to in situ oceanographic conditions. Proceedings of the National Academy of Sciences of the United States of America 104: 13705-13710.
Campagna, C., Falabella, V. and Lewis, M. 2007. Entanglement of southern elephant seals in squid fishing gear. Marine Mammal Science 23(2): 414-418.
Christian, M., Oosthuizen, W.C., Bester, M.N. and de Bruyn, P.N. 2024. Robustly estimating the demographic contribution of immigration: Simulation, sensitivity analysis and seals. Journal of Animal Ecology, 93(5), pp.632-645.
De Bruyn, P.J.N., Tosh, C.A., Bester, M.N., Cameron, E.Z., McIntyre, T. and Wilkinson, I.S. 2011. Sex at sea: alternative mating system in an extremely polygynous mammal. Animal Behaviour 82: 445–451.
Fabiani, A., Hoelzel, A.R., Galimberti, F. and Muelbert, M.M. 2003. Long-range paternal gene flow in the southern elephant seal. Science, 299(5607), pp.676-676.
Hindell, M. A., M. Sumner, S. Bestley, S. J. Wotherspoon, R. G.Harcourt, M.-A. Lea, R. Alderman, and C. R. McMahon. 2017. Decadal Changes in Habitat Characteristics Influence Population Trajectories of Southern Elephant Seals. Global Change Biology 23: 5136–50. https://doi.org/10.1111/gcb.13776
Hoelzel, A.R., Halley, J., O’Brien, S.J., Campagna, C., Arnborm, T., Le Boeuf, B., Ralls, K. and Dover, G.A. 1993. Elephant seal genetic variation and the use of simulation models to investigate historical population bottlenecks. Journal of Heredity, 84(6), pp.443-449.
Hoelzel, A.R., Stephens, J.C. and O’Brien, S.J. 1999. Molecular genetic diversity and evolution at the MHC DQB locus in four species of pinnipeds. Molecular Biology and Evolution, 16(5), pp.611-618.
Hoelzel, A.R., Campagna, C. and Arnbom, T. 2001. Genetic and morphometric differentiation between island and mainland southern elephant seal populations. Proc Royal Soc B 268:325-332.
Hofmeyr, G. J. G., De Maine, M., Bester, M. N., Kirkman, S. P., Pistorius, P. A. and Makhado, A. B. 2002. Entanglement of pinnipeds at Marion Island, Southern Ocean, 1991-2001. Australian Mammalogy 24: 141-146.
Jones, E. 1981. Age in relation to breeding status of the male southern elephant seal, Mirounga leonina (L.), at Macquarie Island. Australian Wildlife Reseach 8: 327-334.
Learmonth, J.A., Macleod, C.D., Santos, M.B., Pierce, G.J., Crick, H.Q.P. and Robinson, R.A. 2006. Potential effects of climate change on marine mammals. Oceanography and Marine Biology: An Annual Review 44: 431-464.
Lübcker, N., Reisinger, R.R., Oosthuizen, W.C., de Bruyn, P.N., Van Tonder, A., Pistorius, P.A. and Bester, M.N. 2017. Low trophic level diet of juvenile southern elephant seals Mirounga leonina from Marion Island: a stable isotope investigation using vibrissal regrowths. Marine Ecology Progress Series, 577, pp.237-250.
McCann, T. S. 1980. Population structure and social organization of southern elephant seals Mirounga leonina (L). Biological Journal of the Linnean Society 14: 133-150.
McIntyre T, Ansorge IJ, Bornemann AH, Plötz J, Tosh CA, Bester MN. 2011. Elephant seal dive behaviour is influenced by ocean temperature: implications for climate change impacts on an ocean predator. Marine Ecology Progress Series 441: 257–272.
McIntyre, T., deBruyn, P.J.N., Ansorge, I.J., Bester, M.N., Bornemann, H., Plötz, J. and Tosh, C.A. 2010. A lifetime at depth: vertical distribution of southern elephant seals in the water column. Polar Biology 33: 1037-1048.
McMahon, C.R., Bester, M.N., Hindell, M.A., Brook, B.W. and Bradshaw, C.J.A. 2009. Shifting trends: detecting environmentally mediated regulation in long-lived marine vertebrates using time-series data. Oecologia 159: 69-82.
Nel W, Hedding DW, Rudolph EM. 2023. The sub-Antarctic islands are increasingly warming in the 21st century. Antarctic Science, 35(2):124-126. doi:10.1017/S0954102023000056
Oosthuizen WC, Bester MN, Tosh CA, Guinet C, Besson D, de Bruyn PJN. 2011. Dispersal and dispersion of southern elephant seals in the Kerguelen Province Southern Ocean. Antarctic Science 23: 567–577.
Pistorius PA, Bester MN, Kirkman SP. 1999. Survivorship of a declining population of southern elephant seals, Mirounga leonina, in relation to age, sex and cohort. Oecologia 121: 201–211.
Pistorius, P. A., Bester, M. N., Lewis, M., Taylor, F. E., Campagna, C. and Kirkman, S. P. 2004. Adult female survival, population trend, and the implication of early primiparity in a capital breeder, the southern elephant seal (Mirounga leonina). Journal of Zoology (London) 263(2): 107-119.
Pistorius, P.A., de Bruyn, P.J.N. and Bester, M.N. 2011. Population dynamics of southern elephant seals: a synthesis of three decades of demographic research at Marion Island. African Journal of Marine Science 33: 523-534.
Reijnders, P., Brasseur, S., van der Toorn, J., van der Wolf, P., Boyd, I., Harwood, J., Lavigne, D. and Lowry, L. 1993. Seals, fur seals, sea lions, and walrus. Status survey and conservation action plan. IUCN Seal Specialist Group.
Reisinger, R.R. and Bester, M.N. 2010. Long distance breeding dispersal of a southern elephant seal. Polar Biology 33: 1289-1291.
Reisinger, R.R., de Bruyn, P.J.N. and Bester, M.N. 2011. Predatory impact of killer whales on pinniped and penguin populations at the Subantarctic Prince Edward Islands: fact and fiction. Journal of Zoology, London 285: 1-10.
Slade, R.W., Moritz, C., Hoelzel, A.R. and Burton, H.R. 1998. Molecular population genetics of the southern elephant seal Mirounga leonina. Genetics 149:1945–1957.
Smith VR. 2008. Energy flow and nutrient cycling in the Marion Island terrestrial ecosystem: 30 years on. Polar Record 44: 211–226.
Tosh CA, de Bruyn PN, Steyn J, Bornemann H, van den Hoff J, Stewart BS, Plötz J, Bester MN. 2015. The importance of seasonal sea surface height anomalies for foraging juvenile southern elephant seals. Marine Biology 162: 2131–2140.
Volzke, S., C. R. McMahon, M. A. Hindell, H. R. Burton, and S. J.Wotherspoon. 2021. Climate Influences on Female Survival in a Declining Population of Southern Elephant Seals (Mirounga Leonina). Ecology and Evolution 11: 11333–44. https://doi.org/10.1002/ece3.7919
Volzke, Sophia, JaimieB. Cleeland, Clive R. McMahon, Mark A. Hindell,Stuart P. Corney, and Simon J. Wotherspoon. 2024.Modeling Climate Influences on Male Survival in the Highly Polygynous Southern Elephant Seal (Mirounga Leonina). Ecosphere 15(6): e4904. https://doi.org/10.1002/ecs2.490414
Winkler, M.P. and Parker, S. 2024. Rabies in seals: visitors to Cape Town marine areas urged to be alert. Journal of Travel Medicine, 31(8), p.taae106.