
Smither’s Horseshoe bat
Rhinolophus smithersi

2025 Red list status
Vulnerable
Regional Population Trend
Declining
Change compared
to 2016
Uplisted
Overview
Rhinolophus smithersi – Taylor, Stoffberg, Monadjem, Schoeman, Bayliss & Cotterill, 2012
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – RHINOLOPHIDAE – Rhinolophus – smithersi
Common Names: Smithers’s Horseshoe Bat (English)
Synonyms: No Synonyms
In previous Red List assessments, this species was included within Rhinolophus hildebrandtii. R. hildebrandtii was revised in 2012 (Taylor et al. 2012).
Taxonomic Note:
This species was formerly included in Rhinolophus hildebrandti but acoustic, biogeographical, morphological and molecular data show it to be a distinct species. The isolated population from Zimbabwe may constitute a distinct species from the South African populations but more sampling is required to test this.
Red List Status: VU – Vulnerable C2a(i) (IUCN version 3.1)
Assessment Information
Assessors: Taylor, P.1, Lötter, C.A.2, Balona, J.3 & da Silva, J.M.4
Reviewers: Howard, A.1, Richards, L.5 & Naidoo, T.5
Contributors: Raimondo, D.4 & Patel, T.6
Institutions: 1University of the Free State, 2Inkululeko Wildlife Services (Pty) Ltd, 3Gauteng and Northern Regions Bat Interest Group, 4South African National Biodiversity Institute, 5Durban Natural Science Museum, 6Endangered Wildlife Trust
Previous Assessors and Reviewers: Taylor, P., Jacobs, D., Cohen, L., Kearney, T., MacEwan, K., Richards, L, Schoeman, C., Sethusa, T. & Monadjem, A.
Previous Contributors: Raimondo, D. & Child, M.F.
Assessment Rationale
This species is near endemic to the assessment region, occurring in riparian woodlands across the Limpopo Valley and savannah or forests from the Soutpansberg, Blouberg, and Waterberg Mountains and the Mahabeng Plateau of Limpopo Province. Recent field surveys have shown that, although essentially limited to one province of South Africa (with a small, isolated Zimbabwean population), the species is quite widespread so areas of occupancy and occurrence exceed thresholds for Vulnerable under Criterion B. Colony sizes are small (possibly < 40 individuals) and the species appears to have a scattered occurrence in the landscape since it is limited by the availability of roosting sites, and possibly suitable water sources. There are at least 20 known localities, and a number of unrecorded colonies are likely to exist. Therefore, a population size of 1,000-2,000 individuals within the assessment region is projected. Since the species occurs only in escarpment habitats there are four subpopulations inferred, all of these have fewer than 1,000 mature individuals and there is population decline inferred from ongoing loss of habitat. It is therefore listed as Vulnerable under criterion C.
Regional population effects: This species has intermediate wing loading (Aldridge & Rautenbach 1987; Norberg & Rayner 1987; Schoeman & Jacobs 2008), and thus dispersal capacity is limited. Additionally, as the Zimbabwean population appears to be isolated from the assessment region, we assume no rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: Uplisted, based on inferred population decline from ongoing habitat loss. The number of mature individuals is <1,000 in each subpopulation.
Red List Index
Red List Index: Uplisted
Recommended citation: Taylor P, Lötter CA, Balona J & da Silva JM. 2025. A conservation assessment of Rhinolophus smithersi. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species is known to occur in two isolated areas, northern Zimbabwe (two localities including the Lutope-Ngolanola confluence just south of the Zambezi Escarpment, and one near Mvurwi town; Inkululeko Wildlife Services unpubl. data), and the Limpopo Province of South Africa (≥20 localities) where it is associated with the Limpopo Valley and the Soutpansberg, Blouberg, Waterberg Mountains and the Mahabeng Plateau (Taylor et al. 2012, 2013; Inkululeko Wildlife Services unpubl. data). Accurate delimitation of this species’ range is subject to further collecting and reappraisal of existing museum material previously assigned to R. hildebrandti (Taylor et al. 2012). The estimated extent of occurrence within the assessment region is 91,146 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 212 m asl (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 1,372 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
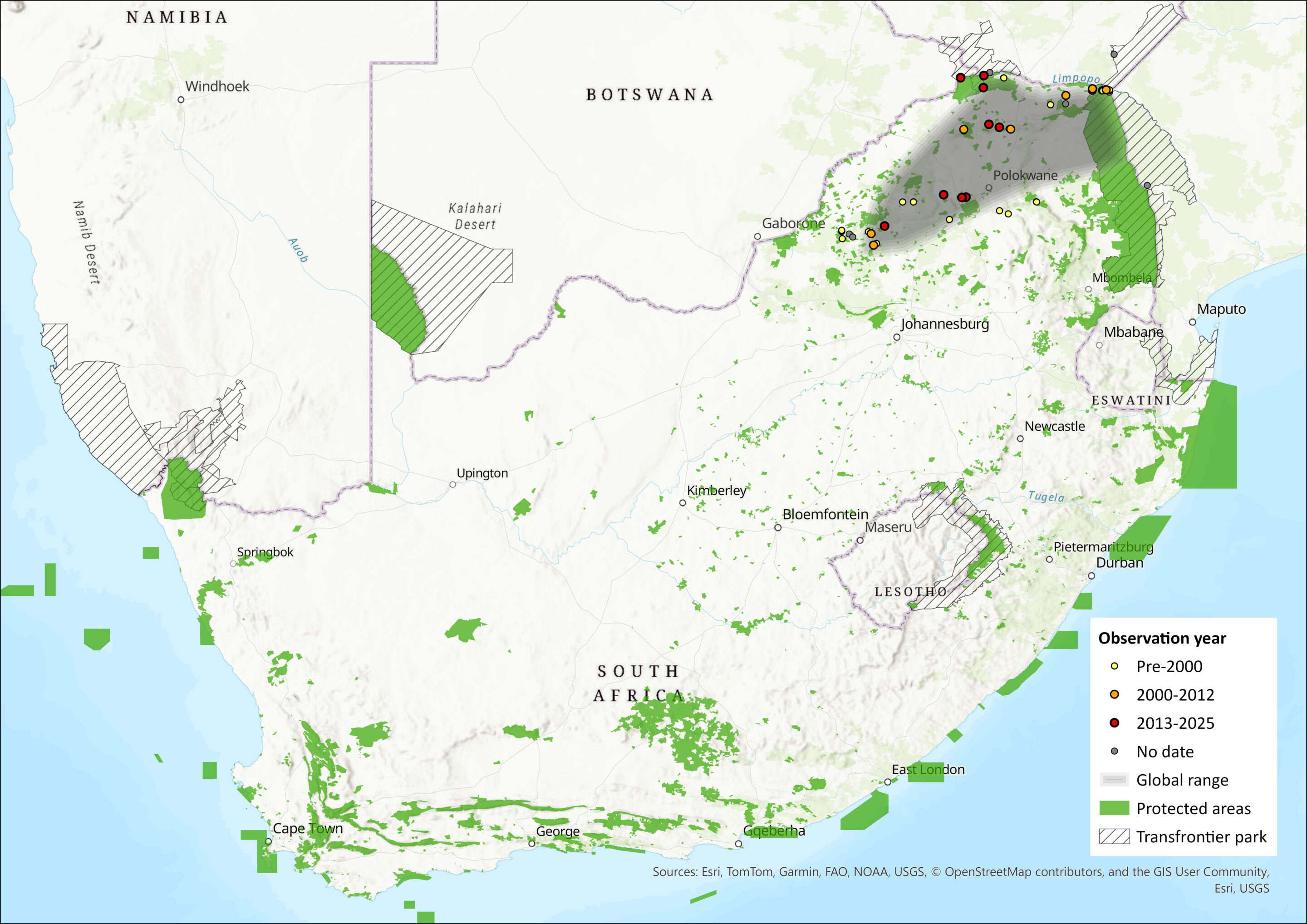 Figure 1. Distribution records for Smithers’s Horseshoe Bat (Rhinolophus smithersi) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Smithers’s Horseshoe Bat (Rhinolophus smithersi) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| South Africa | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
In a review study on the impact and implications of climate change for bats, Sherwin et al. (2012) identified seven risk factors for bats encompassing three broad aspects: 1) biogeography – small range size, high latitude or high altitude range and a range occupying a geographic area likely to become water stressed; 2) foraging niche – frugivory and species restricted to aerial hawking; and 3) dispersal ability – species with restricted dispersal behaviour. These identified risk factors suggest that R. smithersi might be vulnerable to climate change if its distribution range and dispersal capability are limited. R. smithersi also seems to be dependent on water sources for drinking and, therefore, extreme droughts (such as recently in Limpopo Province) could have a significant negative impact on this species.
Population information
This species is relatively widespread within the northern parts of the assessment region. However, colony sizes are very small (just a few individuals) and the species appears to have a scattered occurrence in the landscape as, for example, it is rarely collected with harp traps or recorded with bat detectors during surveys of the Soutpansberg. While Smithers (1971) recorded colonies of over 100 individuals of R. hildebrandti in Botswana, we assume the colony structure of R. smithersi is closer to that of R. cohenae, which roosts in colonies of up to 40 individuals. This would yield an overall population estimate of >800 individuals occurring in ≥20 localities in the assessment region. While it is likely that some localities have been missed, we assume that the overall mature population ranges from between 1,000 and 2,000 mature individuals. This inference is supported by the observation that it is limited by the availability of roosting sites and possibly suitable water sources. There are assumed to be four subpopulations in the assessment region, the first is from the Waterberg region, the second from the Mahabeng Escarpment (where assumed to occur but no records), the third occurs in the Blouberg and Soutpansberg mountains and the fourth occurs along the Limpopo Valley between Mapungubwe and Pafuri in the northern Kruger National Park. None of these subpopulations is suspected to have more than 1,000 mature individuals. The population is inferred to be declining due to habitat loss to mining and land transformation and the rate of loss of habitat is predicted to accelerate over the next 20 years linked to the development of the Musina-Mukhado Special Development Zone. Further monitoring is necessary to determine population size and trend more accurately.
Current population trend: Inferred to be declining.
Continuing decline in mature individuals: Yes
Number of subpopulations: Four, with >20 colonies.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: 40
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
A phylogenetic study found this species to be minimally differentiated from R. hildebrandtii based on mitochondrial markers. However, this could be due to historical introgression (Demos et al. 2019). To better investigate evolutionary history of these species, an assessment using nuclear markers is recommended.
Habitats and ecology
This species occurs in miombo savannah on Karoo Sandstone, and in diverse riparian woodland fringes of the Lutope and Ngolanola rivers as well as along the Crocodile River near Thabazimbi (Inkululeko Wildlife Services unpubl. data) and the Limpopo River at Pafuri (Taylor et al. 2012). It appears to be locally quite widespread in the Soutpansberg, Blouberg and Waterberg ranges in Limpopo Province as well as in the Limpopo Valley (≥20 localities known), where it is dependent on natural caves (such as sandstone cliffs or hollows in baobabs) or man-made underground cavities such as old mine adits (Pearl 1994; Taylor et al. 2013). One roost in the Soutpansberg comprised an underground spring which had been opened to the surface artificially. This species is insectivorous.
Ecosystem and cultural services: Insectivorous bats are important regulators of insect populations (Boyles et al. 2011; Kunz et al. 2011).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | Yes |
| 7.1. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Caves | – | Suitable | Yes |
| 7.2. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Other Subterranean Habitats | – | Suitable | Yes |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Sexes combined: Forearm mean = 6.35 cm (Monadjem et al. 2020)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Unknown (likely to be limited).
Congregatory: Colonies may comprise over 100 individuals (Monadjem et al. 2020).
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no known major threats to this species at present as much of its range occurs throughout the Soutpansberg, Blouberg and Waterberg ranges of Limpopo Province where human impacts and habitat transformation are not yet severe. The threat of extensive planned coal, platinum, natural gas (fracking) and other mining developments over much of the Limpopo Valley and the foothills of the Soutpansberg and Waterberg mountains and the Mahabeng Plateau (Desmet et al. 2013), however, could impact heavily on subpopulations through roosting and foraging habitat loss, noise, air and water pollution and water abstraction leading to degradation of riparian habitats. The development of the Musina – Makhado Special Economic Zone (which was sanctioned by government; clearing for coal mining close to Tshipise has commenced in mid-2025) will result in extensive clearing of habitat for industrial development and coal mining which will result in a huge estimated loss of 4,500 Baobab trees, important roost sites. At the site of the Venetia Diamond Mine in the same area, R smithersi was found to be absent in the mining site, although present in surrounding natural sites (Taylor et al. 2020; Cory-Toussaint et al. 2022).
R.smithersi was amongst five bat species recorded for the first time at Gatkop Cave (compared to surveys in the 1960s and 1980s), Limpopo Province, in December 2011 when thousands of M. natalensis use the cave, which may indicate a loss of other roost sites in the region (Kearney et al. 2016). Possible future renewable energy development especially in association with mines in Limpopo Province could disturb roosts of this species, but due to the clutter and clutter-edge foraging habits of this species, it is regarded as having a “Low” risk of fatality from wind turbines (MacEwan et al. 2020).
Suppression of fire, together with over-grazing of cattle and game, and climate change (sensu Sherwin et al. 2013) has resulted in serious bush encroachment of savannahs across much of its range (for example, the western Soutpansberg) which has been shown to have a negative effect on biodiversity generally (for example, Taylor et al. 2016). In the eastern Soutpansberg, afforestation and alien plant invasions have considerably altered natural habitats (sensu Armstrong & van Hensbergen 1996; Armstrong et al. 1998). Limpopo Province is extremely drought-prone and water-stressed and is projected to experience climate change (Erasmus et al. 2002; Boko et al. 2007). Since the species seems to be dependent on water sources for drinking, extreme droughts in the area have had potentially devastating effects on wildlife generally.
Conservation
In the assessment area, the species is recorded from many protected areas, including Greater Mapungubwe Transfrontier Conservation Area, Kruger National Park, Nwanedi Nature Reserve, Blouberg Nature Reserve, Wolkberg Wilderness Area and Legalameetse Nature Reserve. Not counting the Zimbabwean population, the majority of the species’ range within South Africa is included within two UNESCO Biosphere Reserves, the Vhembe and Waterberg Biosphere Reserves. Defining and refining core and buffer areas is a critical part of the ongoing management of these reserves. Roosting sites of bats should be included in determining the location of such zones within biosphere reserves. This means that important bat underground (natural and man-made) roosts (including those of R. smithersi) should be included wherever possible in core or buffer areas where developments (including mining) would have to be regulated. Such conservation zonation plans (including Strategic Environmental Frameworks, EMFs) would inform planning by Provincial Nature Conservation (including the protected areas expansion strategy) and municipal Integrated Development Plans (IDPs), thereby affording protection to roosting sites. Renewable energy development must avoid encroachment into the prescribed buffers around confirmed and potential roosts of this species (see MacEwan et al. 2020).
Recommendations for land managers and practitioners:
- Landowners should implement best land-use management practices to maintain sustainability and limit disturbance at roosting sites. To achieve this, ecological advisory services and training are needed.
- Renewable energy development must avoid encroachment into the prescribed buffers around confirmed and potential roosts of this species (see MacEwan et al. 2020).
Research priorities:
- Field surveys and niche modelling to more accurately delimit distribution range, and to identify key roost sites for protection.
- Further vetting of R. hildebrandti specimens to more accurately delimit distribution range.
- Investigating patterns of movement to establish level of demographic and genetic exchange between colonies and quantifying the effects on transformation/fragmentation on such processes. This would also enable a more accurate estimate of population size and trend.
Encouraged citizen actions:
- Limit disturbance to roost sites.
- Avoid or limit the use of pesticides/insecticides for agricultural and household purposes as this can potentially negatively affect the prey base and lead to poisoning of individuals of this species and bats in general.
Bibliography
Aldridge, H.D.J.N. and Rautenbach, I.L. 1987. Morphology, echolocation and resource partitioning in insectivorous bats. The Journal of Animal Ecology 56: 763–778.
Armstrong, A.J., Benn, G., Bowland, A.E., Goodman, P.S., Johnson, D.N., Maddock, A.H. and Scott-Shaw, C.R. 1998. Plantation forestry in South Africa and its impact on biodiversity. The Southern African Forestry Journal 182: 59–65.
Armstrong, A.J. and van Hensbergen, H.J. 1996. Impacts of afforestation with pines on assemblages of native biota in South Africa. South African Forestry Journal 175: 35-42.
Boko, M., Niang, I., Nyong, A., Vogel, C., Githeko, A., Medany, M., Osman-Elasha, B., Tabo, R. and Yanda, P. 2007. Africa. Climate Change 2007: Impacts, Adaptation and Vulnerability. In: M. L. Parry, O. F. Canziani, J. P. Palutikof, P. J. van der Linden, and C. E. Hanson (eds), Contribution of Working Group II to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, pp. 433–467. Cambridge University Press, Cambridge, UK.
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Cory-Toussaint, D. and Taylor, P.J. 2022. Anthropogenic light, noise, and vegetation cover differentially impact different foraging guilds of bat on an opencast mine in South Africa. Frontiers in Ecology and Evolution 10, doi: 10.3389/fevo.2022.752665
Demos, T.C., Webala, P.W., Bartonjo, M. and Patterson, B.D. 2018. Hidden Diversity of African Yellow House Bats (Vespertilionidae, Scotophilus): Insights From Multilocus Phylogenetics and Lineage Delimitation. Frontiers in Ecology and Evolution 6: https://doi.org/10.3389/fevo.2018.00086.
Desmet, P.G., Holness, S., Skowno, A. and Egan, V.T. 2013. Limpopo Conservation Plan v.2. Technical Report. Contract Number EDET/2216/2012. Limpopo Department of Economic Development, Environment & Tourism (LEDET) by ECOSOL GIS.
Erasmus, B.F.N., van Jaarsveld, A.S., Chown, S.L., Kshatriya, M. and Wessels, K.J. 2002. Vulnerability of South African animal taxa to climate change. Global Change Biology 8: 679–693.
Kearney, T.C., Keith, M. and Seamark, E.C.J. In press. New records of bat species using Gatkop Cave in the maternal season. Mammalia.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
MacEwan, K., Sowler, S., Aronson, J. and Lötter, C. 2020. South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities. Edition 5. South African Bat Assessment Association. South Africa.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Norberg, U.M. and Rayner, J.M. 1987. Ecological morphology and flight in bats (Mammalia; Chiroptera): wing adaptations, flight performance, foraging strategy and echolocation. Philosophical Transactions of the Royal Society B: Biological Sciences 316: 335–427.
Pearl, D.L. 1994. Rhinolophus hildebrandti. Mammalian Species Archive 486: 1–3.
Schoeman, M.C. and Jacobs, D.S. 2008. The relative influence of competition and prey defenses on the phenotypic structure of insectivorous bat ensembles in southern Africa. PLoS One 3: e3715.
Sherwin, H.A., Montgomery, W.I. and Lundy, M.G. 2013. The impact and implications of climate change for bats. Mammal Review 43: 171–182.
Smithers, R.H. 1971. The mammals of Botswana. Museum Memoir No. 4. The Trustees of the National Museums of Rhodesia, Salisbury.
Taylor, P. J., Nelufule, M., Parker, D. M., Cory Toussaint, D., Weier, S. M. 2020. The Limpopo River exerts a powerful but spatially limited effect on bat communities in a semi-arid region of South Africa. Acta Chiropterologica 22(1): 75–86, doi: 10.3161/15081109ACC2020.22.1.007.
Taylor, P.J., Nengovhela, A., Linden, J. and Baxter, R.M. 2016. Past, present, and future distribution of Afromontane rodents (Muridae: Otomys) reflect climate-change predicted biome changes. Mammalia in press.
Taylor, P.J., Sowler, S., Schoeman, M.C. and Monadjem, A. 2013. Diversity of bats in the Soutpansberg and Blouberg Mountains of northern South Africa: complementarity of acoustic and non-acoustic survey methods. South African Journal of Wildlife Research 43: 12–26.
Taylor, P.J., Stoffberg, S., Monadjem, A., Schoeman, M.C., Bayliss J. and Cotterill, F.P.D. 2012. Four new bat species (Rhinolophus hildebrandtii complex) reflect Plio-Pleistocene divergence of dwarfs and giants across an Afromontane archipelago. PLoS ONE 7(9): e41744. doi:10.1371/journal.pone.0041744.
