Small-spotted Genet
Genetta genetta

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – VIVERRIDAE – Genetta – genetta
Common Names: Small-spotted Genet, Common Genet (English), Kleinkolmuskejaatkat (Afrikaans), Insimba (Ndebele, Swati), Thsipa ya Dithokolo tse Nyenyane (Sepedi), Tshipa, Tshipo e Matheba a Masesane (Sesotho), Nsimba-maxanatsi (Tsonga), Tshipa (Tswana), Tsimba (Venda), Inyhwagi (Xhosa), Insimba Enamabala (Zulu)
Synonyms: afra G. Cuvier 1825; albipes Trouessart 1904; balearica Thomas 1902; barbar Wagner 1841; barbara C.E.H. Smith 1842; bella Matschie 1902; bonapartei Loche 1857; communis Burnett 1830; dongolana Hemprich and Ehrenberg 1832; gallica Oken 1816; grantii Thomas 1902; guardafuensis Neumann 1902; hararensis Neumann 1902; hispanica Oken 1816; isabelae Delibes 1977; leptura Reichenbach 1836; ludia Thomas and Schwann 1906; lusitanica Seabra 1924; melas Graells 1897; neumanni Matschie 1902; peninsulae Cabrera 1905; pulchra Matschie 1902; pyrenaica Bourdelle and Dezillière 1951; rhodanica Matschie 1902; senegalensis J.B. Fischer 1829; tedescoi de Beaux 1924; terraesanctae Neumann 1902; vulgaris Lesson 1827.
Taxonomic Note: There is a high degree of phenotypic variation in this species which resulted in the description of several subspecies; the validity of many of these remains untested, while others may actually represent distinct subspecies or even species (Gaubert et al. 2004, 2005, 2009). An mtDNA-based study suggested at least four distinct geographic lineages within the species circumscribed to the Mediterranean Basin, western Africa (also spread into North Africa), southern Africa and the Arabian Peninsula (Gaubert et al. 2011). The taxonomic status of Genetta felina, a lineage endemic to southern Africa, remains debated (see Gaubert et al. 2004, 2005; Gaubert & Begg 2007).
Based on museum specimens, cryptic hybridisation has been detected in hybrids with Genetta tigrina from the southern provinces of South Africa, in particular the Western Cape and Eastern Cape (Gaubert et al. 2005a). This may confuse attempts to document the species in various landscapes.
The karyotype of G. maculata appears to have undergone more rearrangements than that of G. genetta, of which the latter is an older lineage, and further genetic research at the subspecies level is suggested for the species (Adega et al. 2018).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Carvalho, F.1,2, Power, R.J.3 & da Silva, J.M.4
Reviewers: Do Linh San, E.5 & Patel, T.6
Institutions: 1University of Fort Hare, 2CIBIO – University of Porto, 3North West Province’s Directorate of Biodiversity Management, 4South African National Biodiversity Institute, 5Sol Plaatjie University, 6Endangered Wildlife Trust
Previous Assessors and Reviewers: Carvalho, F., Matolengwe, T., Camps, D., Gaubert, P. & Do Linh San, E.
Assessment Rationale
The Small-spotted Genet is listed as Least Concern as it has a wide distribution within the assessment region, is locally common, and has a very broad habitat tolerance that includes being commensal with human settlements in urban and rural landscapes. Small-spotted Genets are also present in many protected areas across the region.
Regional population effects: The species’ range within the assessment region is continuous with the rest of its southern African range and given the ecological versatility of the species, we suspect that there is dispersal across regional boundaries (although no empirical and genetic data supporting this assertion are available) (Carvalho et al. 2016b). There is likely a strong rescue effect along the boundaries of Namibia, Botswana, and Zimbabwe.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Carvalho F, Power RJ & da Silva JM. 2025. A conservation assessment of Genetta genetta. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species is a widespread species, occurring in North Africa (Morocco, Algeria, Tunisia), and in open and dry savannah zones throughout sub-Saharan Africa in three large disjunct blocks, corresponding roughly to West Africa, East Africa and southern Africa (Skinner and Chimimba 2005; Delibes & Gaubert 2013). It also occurs in coastal regions of Arabia, Yemen and Oman (Harrison & Bates 1991); while records from Palestine are erroneous (Schlawe 1981; Kock 1983). The range of elevation from where it was been recorded varies from the sea level to almost 2,700 m asl. in the Atlas Mountains in Morocco (Delibes and Gaubert 2013). It was introduced in Europe, possibly via Phoenicians’ commercial trade routes (Gaubert 2016), and now occurs in South-Western Europe (Portugal, Spain, France and northwest Italy) (Gaubert et al. 2008; Delibes & Gaubert 2013; Carvalho and Rosalino 2023), where it may have naturalised. It is also present on some of the Balearic Islands (Delibes 1999; Gaubert 2016).
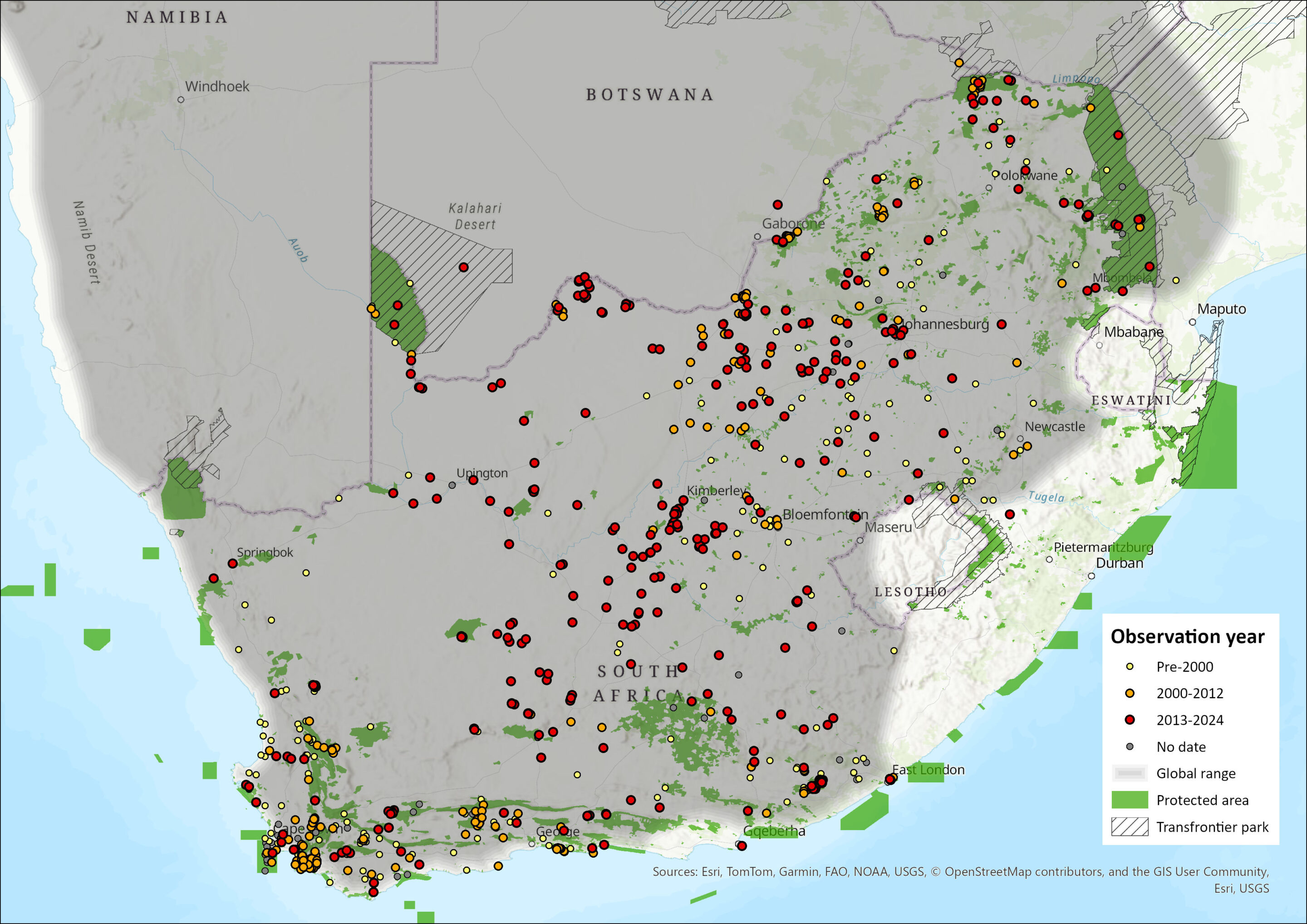
In South Africa, the Small-spotted Genet occurs across most of the country (Skinner and Chimimba 2005), and is common in the Northern, Eastern and Western Cape provinces (Stuart 1981; Carvalho et al. 2024a,b), parts of KwaZulu-Natal (Rowe-Rowe 1992), Gauteng (Kuhn 2011), North West (Power et al. 2019), and in the Free State Province (Lynch 1983; Larivière & Calzada 2001; Skinner and Chimimba 2005; Carvalho et al. 2016b). Although it occurs in Lesotho (Lynch 1994), it is absent from Eswatini (Carvalho et al. 2016b).
The species is still present in the Northern Cape, Western Cape, in mostly karroid areas (Schurch et al. 2021; Woodgate et al. 2023), Eastern Cape (Smith et al. 2023; Carvalho et al. 2024a, b), North West (Power et al. 2019), mostly Highveld and Kalahari, as well as Free State, north-western Kwazulu-Natal, Gauteng, Limpopo and Mpumalanga (Figure 1). The species was not recorded in 2019 at the Verloren Vallei Nature Reserve in the Mpumalanga Highland grasslands (Loock and Swanepoel 2019), which may be a local absence.
Since the last assessment (Carvalho et al. 2016b), there has been virtually no change in the distribution, though there have been new distribution records on the margins of the species distribution (Figure 1), though it is unclear whether these represent a range expansion.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 3000
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Small-spotted Genet (Genetta genetta) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Palearctic
Occurrence
Countries of Occurrence within southern Africa
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Botswana | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
It is important to consider a species’ ecological and biological traits to determine the likely mechanisms of climate change impact and quantify these using expert knowledge supported by adequate methods for the species under study (Foden & Young 2016). Accordingly, genets as slender body like small carnivores are prone to high thermoregulatory challenges. In a recent telemetry activity study in a semi-arid Albany Thicket landscape (Eastern Cape, South Africa), Small-spotted Genets showed a significant decrease in activity during the dry-cold season (March-September) and stopped totally below 0ºC (Carvalho et al. 2024b). Similarly, during hot days (> 35 º C) due to their nocturnal nature they avoid moving during those days, although some activity might happen near resting sites in dense vegetation areas (Carvalho et al. 2014, 2024b). Thus, genets might rely in dense vegetation cover to counteract harsh weather, but other crucial factors such as preys, resting sites availability and predation risk are interrelated and play a key role in their occurrence and abundance (Carvalho et al. 2024b). Therefore, future studies might be needed to assess whether vegetation cover per se in arid areas might be enough to provide refuges for the species survival. Currently, in the assessment region, no severe effects from global warming are expected for this species so we believe that the population maintains stable numbers across the region since the last assessment.
Population information
The Small-spotted Genet is one of the most common small carnivores in its native range, though detailed data on density in Africa are scarce; in Serengeti, Waser (1980) estimated a density of 1.5 ± 0.37 individuals / km². Another study reported densities of 0.18 individual / km² in Senegal (Sillero-Zubiri & Marino 1997). In Europe, this species is moderately abundant, with increasing populations in France, and densities of 0.3–0.98 individual / km² were recorded in two studies in Spain (Palomares & Delibes 1994; Camps & Llimona 2004). In Portugal, studies from camera-traps and capture–recapture analysis showed a density varying from 0.19–0.92 individual / km² (Sarmento et al. 2010, 2014). The genet is common in suitable habitat throughout the Iberian Peninsula (Palomo & Gisbert 2002), where populations are either stable or slowly increasing (J. Herrero pers. comm. 2007, In: Carvalho et al. 2016b). In Ibiza, habitat is declining and becoming more fragmented, thus this species is suspected to be declining there (Calzada 2007).
In the assessment region few studies have been published regarding density estimations. In the Albany Thicket landscape (Eastern Cape, South Africa) a multi-year telemetry study shows some preliminary results densities of 0.5 individual / km² (Filipe Carvalho, unpublished data).
Population Information
Current population trend: Stable
Continuing decline in mature individuals: Unknown, but probably not
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Severely fragmented: No. Small-spotted Genets have a broad habitat tolerance and can also exist in agricultural and rural landscapes.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
While the Small-spotted Genet is a common carnivore, its population genetic structure and diversity are not well studied. Based on its distribution in the assessment region and its dispersal capabilities, the species is likely a single population (metapopulation), connected with neighbouring countries.
Based on this information, one of the Convention on Biological Diversity’s population genetic indicators can be calculated. This species is a single population that still exists; hence it has all of its populations maintained (1 continuous metapopulation; PM indicator = 1/1 = 1.0).
While density estimates exist for some localities (see Population section), without an AOO it is difficult to estimate an approximate ‘census’ size for this metapopulation and subsequently convert it to obtain a measure of effective population size. Consequently, the Ne 500 indicator cannot be quantified.
Habitats and ecology
Small-spotted Genets are found in a large variety of habitats, but availability of prey and shelters for resting also constitute key factors in habitat selection (Larivière & Calzada 2001; Carvalho et al. 2014). Small-spotted Genets tend to inhabit all types of wooded habitats (deciduous and evergreen) and scrub areas, where they are sometimes associated with rivers and streams (Delibes & Gaubert 2013; Gaubert et al. 2015). They avoid very open habitats (but see Stuart 1981), but may occur in even small fragments of woodland in farmland or near villages, and usually are absent from rainforests, dense woodlands and woodland–moist savannah mosaics (e.g. miombo woodland in Angola) (Pereira & Rodríguez 2010; Delibes & Gaubert 2013). However, ancestrally the Small-spotted Genet is a species might have used open habitats during early invasions, while dispersing (Gaubert et al. 2004). The species generally uses open areas more in arid regions (Stuart 1981; Skinner and Chimimba (2005), but within them, it prefers cover in the form of scrub and trees, especially along watercourses and rock outcrops (Skinner & Chimimba 2005). In the North West province, the species occurs mostly in the Highveld and Kalahari bioregions, while it is marginally in the Bushveld (Power et al. 2019). This alludes to open and dry habitats as preferred habitats. In Kalahari rangelands of the Northern Cape, the abundance of Small Spotted Genets showed a unimodal relationship with increasing shrub cover, and their abundance was maximal at a shrub cover of 17-18% (Blaum et al. 2007).
Small-spotted Genets are also sometimes found in proximity to people and human buildings (e.g. Admasu et al. 2004) and are a common sight in rural and urban gardens (Carvalho et al. 2016). They can live in areas without trees, but then in most cases these areas correspond to rocky outcrops where they rest in rock crevices (Stuart 1981; Livet & Roeder 1987; Larivière & Calzada 2001; Skinner and Chimimba 2005). They rest mainly in hollow trees and in burrows dug by other animals (e.g. Aardvark Orycteropus afer, Springhares Pedetes capensis, Hares (Lepus sp.) etc.) (Skinner and Chimimba 2005; Delibes & Gaubert 2013), or crevices in rocky areas (Skinner and Chimimba 2005). In Europe, several authors found that they select and are especially abundant in oak (Quercus spp.) forests, but they are also often present in olive (Olea europaea) groves, riparian copses, ash (Fraxinus spp.) groves and scrublands, being rare or absent in open areas, marsh and cereal crops (Carvalho et al. 2011, 2014, 2016; Delibes & Gaubert 2013). The latter habitats may however be crossed during dispersal, whenever riparian or hedgerow corridors are present (Carvalho et al. 2016a). In some Spanish regions, genets also prefer pine forests and ravine areas (Camps & Alldredge 2013). In Europe, studies on resting ecology pointed out a great prevalence of hollow trees (> 48%) with peaks of more than 70% during the wet season (autumn–winter), followed by nests in treetops or under shrubs and dens in riverbanks under shrubs or tree roots (Carvalho et al. 2014). Sporadically, genets also use rock crevices and human buildings (Skinner and Chimimba 2005; Camps 2011; Carvalho et al. 2014). Genets select riverine areas for their latrine sites and generally select less rugged areas too for this (Carvalho et al. 2024a).
Small spotted genet at !Khamab Kalahari Reserve (Photo courtesy of Panthera, circa 2016).
Small-spotted Genets feed mainly on small mammals, but will also take birds, other small vertebrates, insects and fruits (Smithers 1971; Stuart 1981; Matolengwe 2010; Delibes & Gaubert 2013). Rodents, birds and insects are fed upon by the species (Smithers 1971; Stuart 1981). The diet was said to comprise vertebrates from 43% (Smithers 1971) to 50% (Stuart 1981).
In South Africa, one broad-based study on diet has been done (Stuart 1981), while there has only been one genet- specific study done (Matolengwe 2010), based on scat analysis, of this species. In the Great Fish River Nature Reserve (GFRNR) (Eastern Cape Province), its diet consisted essentially, in terms of percentage of occurrence, of mammals (rodents and Macroscelidae; 93%), followed by arthropods (79%) and plant materials (exclusively grass; 42%). As supplementary prey items, fruits, birds and reptiles were recorded, but not amphibians or fish (Matolengwe 2010). Regarding the relative volume (RV) of food remains in the scats, results were similar with mammals being the main food category (66%) followed by arthropods (22%) and plant material (8%), respectively. The RV of some prey varied significantly across seasons, with higher volumes of arthropod remains recorded in spring and summer, and of reptile remains in summer (Matolengwe 2010), suggesting of seasonal diet shifts there. However, we caution that the above study was conducted in an area where Small-spotted Genet occurs in sympatry with Cape Genet (Genetta tigrina), and therefore results could be based on scat samples from both species.
In North Africa, Small Spotted Genets also revealed a preference for mammals and arthropods, followed by birds and plants. They also showed a seasonal dietary variation with arthropods peaking in summer, mammals and birds in winter, and high-energy plants in autumn (Amroun et al. 2014). Several dietary studies have been conducted in Europe and overall, it appears that the Small-spotted Genet’s diet is flexible and highly opportunistic. The diet varies being almost specialised on predation of Wood Mouse (Apodemus sylvaticus) in areas where this prey is abundant, to more generalist where other mammals, reptiles, amphibians, fish, crabs, arthropods, fruits, grass and eggs are also consumed (Virgós et al. 1999; Rosalino & Santos-Reis 2002). The genet’s diet in Europe also varies seasonally with birds and amphibians eaten more frequently in spring and winter, fruits in summer and autumn, and reptiles and arthropods in summer (Virgós et al. 1999; Rosalino & Santos-Reis 2002).
Small-spotted Genets are preyed upon by Black-backed Jackals (Lupulella mesomelas), though at very low frequencies (Bagniewska and Kamler 2014). Although they are preyed upon (Skinner and Chimimba 2005; Bagniewska and Kamler 2014), the species is never completely excluded by other large carnivores, as there is often fine-scaled temporal separation that allows coexistence of the species (de Satge et al. 2017; Vissia and Van Langevelde 2022; Smith et al. 2023). There is temporal separation by the Small-spotted Genet and diurnal mongooses. The species may show lower spatial co-occurrence with larger competitors such as African Wildcat (Felis lybica) for instance (De Satge et al. 2017).
The Small-spotted Genet is mostly nocturnal (Smithers 1971; Stuart 1981; Skinner and Chimimba 2005; De Satge et al. 2017; Vissia and Van Langevelde 2022; Carvalho et al. 2024b). In Europe, studies confirmed that Small-spotted Genets are nocturnal with peaks just after sunset and before sunrise (Camps & Llimona 2004; Camps 2008). Sometimes juveniles can be active during the day (Palomares & Delibes 1994; Larivière & Calzada 2001). Some studies in Ethiopia and Kenya reported nocturnal activity during the whole night but with more incidences in the first hours after sunset (Ikeda et al. 1982, 1983; Waser 1980). In South Africa, radio-tracked genets were also crepuscular, starting activity on average around 18h00 (but as early as 16h30) and ceasing around 04h00 (but as late as 07h20). Genets were active between 5 and 13 hours and travelled over 1.7–7.9 km per night, depending on the sex and season (Matolengwe et al. 2011). Recently, the activity of 15 Small-spotted Genets was studied through telemetry at the Great Fish River Nature Reserve (Eastern Cape) and the species maintained a nocturnal activity year-round (Carvalho et al. 2024b). However, they decreased significantly the activity during the cold-dry season (Mar-Sep), reflecting the thermoregulatory hypothesis where species with an elongated body like genets, which suggests a decreased activity under extreme cold weather conditions to conserve energy (Carvalho et al. 2024b). Females were less active than males, possibly due to their smaller home ranges, especially during the cold-dry season (Carvalho et al. 2024b).
Home ranges in Africa seem to be smaller than in Europe and range from 0.34–0.75 km², with no intersexual differences (Ikeda et al. 1982, 1983; Waser 1980). In Portugal, a study yielded an average size of 3.9 km² with no differences among sexes (Carvalho et al. 2014; Carvalho 2015). At the GFRNR (Eastern Cape), for the Small-spotted Genet males the annual home range was 3.62 km² which was larger than females with 1.97 km² (Filipe Carvalho, Unpublished data). These results might reflect local habitat conditions but also can reflect the different sampling effort conducted, which in the GFRNR was based in intensive multi year-round study and a large number of locations were collected, compared to the old studies that were based in few animals tracked and shorter periods.
Small-spotted Genets are solitary but may form pairs during the breeding season (Larivière & Calzada 2001; Delibes & Gaubert 2013). They are territorial, with male home ranges including that of several females but no other adult males, and adult female home ranges do not overlap (Larivière & Calzada 2001; Zuberogoitia et al. 2002; Carvalho 2015; Carvalho et al. 2018). Similar to what is observed in other genet species, intraspecific communication is mainly done through olfaction, notably with scats deposited at conspicuous latrines (Palomares 1993; Espírito-Santo et al. 2007), in particular habitats that offer sufficient cover (Carvalho et al. 2024a). These are usually strategically located in the animals’ home range boundaries, such as large tree branches and high rocks, where they are less likely to be washed away and the scent might be more effectively spread (Larivière & Calzada 2001; Espírito-Santo et al. 2007; Carvalho et al. 2024a). Genetta genetta can use several calls, where the most important one permits contact among individuals and plays an important role in the mother–offspring relationships and during the mating period (Livet & Roeder 1987; Larivière & Calzada 2001).
In southern Africa, Small-spotted Genets have been reported to breed seasonally (Skinner and Chimimba 2005), where pregnancies are reported to span from September (Stuart 1981) and October (Smithers 1971; Stuart 1981), to January (Rautenbach 1978), and February (Smithers 1971).
Ecosystem and cultural services: The species has been reported to prey upon House Rats (Rattus rattus) (Smithers and Wilson 1979, In: Skinner and Chimimba 2005), so there is potential that they can be useful in agricultural and domestic households. Therefore, given its reliance on small rodents as the main energetic intake, the species is likely to play a role in controlling rodent populations and potential vectors of zoonosis. It may also play a secondary role in seed dispersal (Carvalho et al. 2016b)
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.4. Forest -> Forest – Temperate | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.4. Shrubland -> Shrubland – Temperate | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? No
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Body parts use
G.genettais occasionally eaten by people in some localities, and body parts are used for medicinal purposes, while skins may be used for the manufacture of karosses in southern Africa (Delibes & Gaubert 2013; Nieman et al. 2018). The use of the Small-spotted Genet has been documented in Eastern Cape (Simelane & Kerley 1998), Gauteng (Whiting et al. 2013) and Western Cape (Nieman 2018; Nieman et al. 2020). The species appeared in 20% of herbalist shops in the Eastern Cape (Simelane & Kerley 1998). At the Faraday Market in Gauteng in 2005, traders interviewed were stock body parts of this species for sale, while in some cases, it was generically referred to (Whiting et al. 2013). At the Faraday market, 28.1% of traders had genet on offer (n = 36 traders (Whiting et al. 2013). The genet skin can be used decoratively as clothing and can confer high community status (Nieman et al. 2018) and can be sold for R50-R1500 (Nieman 2018). An entire genet carcass, or its oil, can be used to protect against evil spirits (Nieman et al. 2018). The entire carcass of a genet can sell for R100-R2000, while the oil only can sell for R400 (Nieman 2018). Genets are mostly used for traditional medicine in the Western Cape, followed by bushmeat and human-wildlife conflict (Nieman 2018). In the Western Cape, the Genetta had a Cultural Significance Index (CSI) of 6.56, which was moderate amongst several others that are a lot more important such as the Leopard (Panthera pardus) for instance was scored at 15 (Nieman 2018). There may be regional importance of a species when it comes to medicinal uses, which may be a function of its regional abundance.
In North Africa, the species is also hunted for its fur for decorative purposes (Cuzin 2003).
Within the assessment region, there were few reports of this species being utilised as either bushmeat, traditional medicine, or for clothing and it was suspected that such localised use does not have a negative impact on the population (Carvalho et al. 2016b), which may still be the case, but the importance of the use of the species is higher than what was previously reported.
Captive keeping
Several species of genets are kept as pets by people (Carvalho et al 2016b). In the North West Province, there has only been one case, at least since 2020, where ‘genets’ have been kept illegally, though it may have been easy to identify the correct species, this was unfortunately not done (North West Biodiversity Management Directorate records, 2020-2023). The species is very endearing, and they are likely kept frequently, but the scale of this is not significant and neither is it widespread.
Unlike in the USA, it does not seem to be popular in southern Africa (Carvalho et al. 2016). We suspect that a large majority of pet genets originate from captive breeding programmes rather than from the wild (Carvalho et al. 2016). Although the exact number of Small-spotted Genets kept as pets both globally and in the assessment region is unknown, the provinces in South Africa do insist on permits to keep the species, so there would be a record of that. In the North West for instance, there are 6 facilities with the species and at least 10 animals present across them (North West Directorate of Biodiversity Management records, 2021-2023).
Trophy hunting
The species is hunted in South Africa, and since it is provincially protected, permits are issued for this.
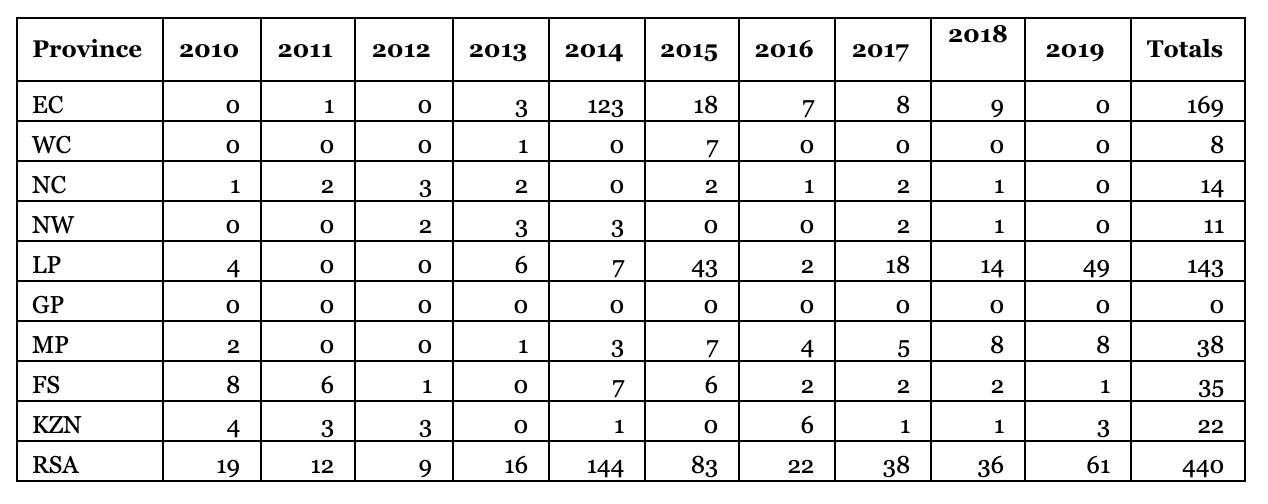
Table 1. Summary of hunted offtakes of the Small Spotted Genet from the Professional Hunters register (SANBI, 2010-2019).
The Eastern Cape and Limpopo have the highest number of offtakes for this species (Table 1). It is unusual that there are more hunts of the species in KwaZulu-Natal and Mpumalanga, which are marginal provinces for the species, while there is fewer use in the arid provinces of Northern Cape and North West (Table 1). The use of the species may track the prevalence of Professional Hunters verse the abundance of the species. Other than the peak in use of the species in 2014, there has a been a slightly increasing trend in use of the species by hunters since the last assessment period (prior to 2016).
In the North West Province, hunting of the species is associated with the Kalahari area, where it is secondary to other more high-profile species hunting, where there has been an appreciable demand (NWPG 2016; 2017). At the time of the last assessment, of the hunting applications that have been received, 67 % have been approved (n = 12), and an average of 2.7±1.5 (1-4) were being hunted per year, which was not excessive at the level of the district (NWPG 2017). Since 2020, there have been 11 hunting applications for the species, and 7 of these have been approved, owing inter alia to the revised quota policy for these species (North West Directorate of Biodiversity Management Records, 2020-2023). Here, the species can only be hunted between April and September, and only one animal per property per year would be allowed.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Subsistence hunting for food, medicine or skins. | Yes | Selling of individuals as pets (or breeding stock). |
National Commercial Value: Yes
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | – | – |
| 3. Medicine – human & veterinary | true | true | – | – |
| 10. Wearing apparel, accessories | true | – | – | – |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Increasing (mostly in the USA).
Threats
Overall, we suspect there are currently no major threats to Small-spotted Genets within the assessment region as no significant changes occurred since the last assessment (Carvalho et al. 2016b).
Medicinal use and body parts trade
The suspected localised bushmeat and traditional medicine use in the assessment region (Delibes & Gaubert 2013) is expected to be more important than what was previously reported (Carvalho et al. 2016b), as the species is culturally important in trade (Nieman et al. 2018), though there are no indications of declines in any areas. In Europe, G. genetta used to be trapped for its fur (Livet & Roeder 1987; Delibes 1999).
Persecution due to conflict
Small-spotted Genets are sometimes persecuted by farmers as a response to poultry predation (Rautenbach 1978; Stuart 1981; Larivière & Calzada 2001; Skinner and Chimimba 2005). Genets, even if not specified at species level (see Stuart 1981), are implicated in predation on poultry (Rautenbach 1978; Stuart 1981; Skinner and Chimimba 2005) and birds kept in aviaries (Stuart 1981). In the Western Cape, genets were sometimes involved in occasional human-wildlife conflict (Nieman 2018). The true impacts however arising from persecution may be low and likely have residual impacts in the overall population numbers in the assessment region.
In the North West Province, from 2020 to 2023, there have been 21 cases where ‘genets’ have been responded to, and of these, two were said to be of the specific species. The cases involved poultry predation, and various non-conflict related issues, but species identity was not always confirmed (North West Biodiversity Management Directorate records, 2020-2023).
Wire Snares
Genets are frequently entrapped in wire snares intended for bushmeat poaching in the Boland Mountain Complex of the Western Cape (Nieman 2018). It is likely that larger species are targeted for bushmeat, and thus genets are immune to most noose circumferences of wire snare sets, as a default situation. Again, the possible impacts of this type of practice might be neglected for the overall Small-spotted Genet population.
Trophy Hunting
Between 2015 and 2016, across 624 camera trap nights focussed on the species, and others, none were recorded across 4 sites, including even control sites, hence a moratorium was implemented for the year (NWPG 2016), and a cautious quota implemented the following year (NWPG 2017). In the North West, a seasonal restriction was suggested for them to only be hunted between May and September (NWPG 2016). Trophy hunting may be too limiting to have any meaningful impact as a threat to the species at the global level, although these practices might lead to local populations.
Roadkill
The extent of road mortality on G. genetta populations is unknown, although individuals of this species have been recorded in the Endangered Wildlife Trust’s road collision database (Carvalho et al. 2016).
Small-spotted Genet roadkill in an agricultural landscape of the North West, August 2023 (John Power)
There are several roadkill records for the species from Northern Cape (Bullock et al. 2011) and North West (NWPG 2024). In the Northern Cape, from January to September in 2007, 12 road mortality surveys were carried out along the R360 main road for a total distance of 2796 km (Bullock et al. 2011). On these transects, two Small Spotted Genet road kills were discovered (Bullock et al. 2011). This may then have been a small number.
In the province to the east, and more recently, from 2020 to 2023, 15 Small-spotted Genet road kills have been recorded across the whole province. This is while driving non-systematic routes of 85, 078 km, with no clear trend across the time period (NWPG 2024, and earlier annual reports). On a systematic roadkill searching route connecting the towns of Lichtenburg, Ottoshoop and Mahikeng (150 km circuit), from 2021 to 2024, where the circuit was driven 100 times, no Small-spotted Genet road kills were recorded (NWPG 2024, and earlier annual reports). However, earlier, from 2020 t0 2021, in a more localised circuit around the Mafikeng Game Reserve (approx.40 km), 8 Small-spotted Genet kills were recorded on 79 driven routes (NWPG 2022). This translates to a kill rate of 1 genet/ 10 days, which may be relatively high, and be a significant edge effect on a protected area, although the true impacts of these numbers can only be assessed if the densities inside the reserve are known.
Roadkill may represent a significant local threat, but given the species persistence (Figure 1), it may be nullified by a rescue effect from areas not impacted by roads across the assessment region.
In Portugal 65% of the road killed animals are subadults, which may affect their persistence in a medium-long term (Carvalho 2015; Carvalho et al. 2018). Similar assessments on the age and number of the genet road kills should be also registered, whenever possible, across the assessment region.
Habitat degradation and land-use change
In Ibiza, the Genet is threatened by the loss and fragmentation of habitat caused by urbanisation and infrastructure, and tourism development (Carvalho et al. 2016b). The ability of genets to live close to humans and their domestic animals could have implications for disease transmission (Admasu et al. 2004).
Generally, in Karoo landscapes, land-use, in terms of protection, has had a minimal influence on Small-spotted Genet abundance (Schurch et al. 2021; Woodgate et al. 2023). Schurch et al. (2021) compared mammal species abundance on different rangelands in the Karoo of the Nuweveldberg of the Western Cape. Interestingly, Small-spotted Genet relative abundance had decreased over a four-year period by 25% on wildlife friendly farms (no lethal control, human shepherding) when compared to traditional farms (with lethal control, and intermittent rotational grazing). Furthermore, there may even be local absences of small predators such as Small-spotted Genet was shown (Schurch et al. 2021). This was related to overgrazing and increased shrub cover (Blaum et al. 2007; Schurch et al. 2021), or by-catch in lethal control aimed at larger mesopredators (Schurch et al. 2021). On Kalahari rangelands, where mesopredators (Black-backed Jackal, African Wildcat, and Caracal (Caracal caracal)) are extensively controlled, Small-spotted Genets are positively affected (Blaum et al. 2009). Furthermore, on Kalahari rangelands, abundance of Small-spotted Genet was lowest on farms where stocking rate was high (Blaum et al. 2009). This was related to increasing shrub densities, in response to overgrazing, and maximal abundances of Small Spotted Genets are attained at intermediate levels of shrub covers (Blaum et al. 2007). In terms of livestock management, Small-spotted Genets can tolerate moderate stocking rates of livestock on Kalahari rangelands, but they tend to decline when in excess of between 4-5 LSU/km², which is often when low prey availability is evident (Blaum et al. 2009).
A Rapid Vulnerability Approach (RVA) was done on Genetta sp in the Western Cape, and it scored a 7, while the most threatened species was a 10 and that was the leopard (Nieman 2018).
Given its history of being able to colonise areas outside its distribution range (Gaubert et al. 2008; 2009), this propensity may assist in, when needing to replace losses from the various threats outlined.
Conservation
Compared with mammals worldwide, the overall conservation status of small carnivores in Africa appears relatively favourable (Do Linh San et al. 2013). Within the assessment region, no major and immediate conservation interventions are necessary, however regular distribution and abundance assessment should be performed (Carvalho et al. 2016b). Education should be used to raise the profile of this species and encourage farmers to live with rather than against genets, especially because this species mostly relies on rodents and arthropods (Smithers 1971; Matolengwe 2010; Amroun et al. 2014).
Marketing and awareness campaigns can also be used to position the presence of this species as a point of pride for urban and rural landowners, and conservationists should encourage better land management to facilitate genet conservation. The most important conservation area for the species is likely the Kgalagadi Transfrontier Park, with the Kruger National Park also being important. Other important conservation areas are the Augrabies Falls, Mokala, Karoo, Mountain Zebra and Greater Addo Elephant National Parks. Important provincial nature reserves are the Great Fish Nature Reserve and Molopo Nature Reserves – to name a few.
Recommendations for land managers and practitioners:
- In order to reduce the risk of collisions with vehicles (Larivière & Calzada 2001; Carvalho et al. 2014), mitigation measures such as road fencing and improvement of habitat near road crossing structures (for example, underpasses) should be carried out whenever possible (Collinson et al. 2015; Carvalho et al. 2016a, 2018).
Research priorities:
- Establish and/or update the distribution range across the country.
- Initiate a study to understand the ecological separation between different species which can then be used to predict distribution.
- Collect representative specimens and place in appropriate institutions for training and collect DNA to establish population structure, especially given hybridisation adds extra complexity (Gaubert et al. 2005b).
- The differentiation between G. genetta and the putative (cryptic) species G. felina. Indeed, Small-spotted Genets from the southernmost distribution of their southern African range have longer, brighter hairs and darker markings than their northernmost congeners (Gaubert et al. 2004, 2005). This may reflect phenotypic plasticity due to climatic variability across the species’ range or genuine evolutionary processes. Further investigations are in progress and needed to clarify the status of G. felina in southern Africa.
- Population monitoring should take place. Small-spotted Genet abundance and occurrence has been determined from spoor in the Kalahari (Blaum et al. 2008), while camera trapping, even as by-catch (Hargey 2022), or as part of multi-species studies has its merits to report on populations (Schurch et al. 2021; Smith et al. 2023; Woodgate et al. 2023).
- Population estimates based on individual recognition, and spatially explicit capture recapture (see Sarmento et al. 2010; 2014), should be prioritised for South African studies.
- Given the plethora of European studies on the species (Carvalho et al. 2016b), there must be more studies conducted in the assessment region.
- Research initiated at the Great Fish Nature Reserve in the Eastern Cape (see Matolengwe 2010, et al. 2011; Carvalho et al. 2024a,b), should expand into other related aspects.
Encouraged citizen actions:
- Report sightings of any genet species on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas, as well as to Emmanuel Do Linh San (emmanuel.dolinhsan@gmail.com). GPS locations and photographs would be of great assistance.
Bibliography
Admasu, E., Thirgood, S.J., Bekele, A. and Laurenson, M.K. 2004. A note on the spatial ecology of African civet Civettictis civetta and common genet Genetta genetta in farmland in the Ethiopian Highlands. African Journal of Ecology 42: 160-162.
Amroun M, Bensidhoum M, Delattre P, Gaubert P. 2014. Feeding habits of the common genet (Genetta genetta) in the area of Djurdjura, north of Algeria. Mammalia 78: 35–43.
Bagniewska JM, Kamler JF. 2014. Do black-backed jackals affect numbers of smaller carnivores and prey? African Journal of Ecology. 52(4):564-567.
Blaum N, Engeman RM, Wasiolka B, Rossmanith E. 2008. Indexing small mammalian carnivores in the southern Kalahari, South Africa. Wildlife Research. 35(1):72-79.
Blaum N, Rossmanith E, Popp A, Jeltsch F. 2007. Shrub encroachment affects mammalian carnivore abundance and species richness in semiarid rangelands. Acta Oecologica. (1):86-92.
Blaum N, Tietjen B, Rossmanith E. 2009. Impact of livestock husbandry on small‐and medium‐sized carnivores in Kalahari savannah rangelands. The Journal of Wildlife Management. (1):60-67.
Bullock KL, Malan G, Pretorius MD. 2011. Mammal and bird road mortalities on the Upington to Twee Rivieren main road in the southern Kalahari, South Africa. African Zoology 46(1):60-71.
Calzada J. 2007. Gineta. Genetta genetta. In: Palomo LJ, Gisbert J, Blanco JC. (ed.), Atlas y Libro Rojo de los Mamíferos Terrestres de España, pp. 330–332. DGB-SECEM-SECEMU, Madrid. Spain.
Camps D. 2008. Activity patterns of adult common genets Genetta genetta (Linnaeus, 1758) in northeastern Spain. Galemys 20: 47–60.
Camps D. 2011. Resting site selection, characteristics and use by the common genet Genetta genetta (Linnaeus 1758). Mammalia 75: 23–29.
Camps, D. and Alldredge, J.R. 2013. Multi-scale habitat use and selection of common genet Genetta genetta (Viverridae, Carnivora) in a Mediterranean environment. Mammalia 77: 285–295.
Camps, D. and Llimona, F. 2004. Space use of common genets Genetta genetta in a Mediterranean habitat of Northeastern Spain: differences between sexes and seasons. Acta Theriologica 49(4): 491-502.
Carvalho F, Galantinho A, Mira A. 2011. Factors affecting small and middle-sized carnivore occurrence and abundance in Mediterranean agricultural landscapes: case studies in southern Portugal. In: Rosalino LM, Gheler-Costa C. (ed.), Middle-sized Carnivores in Agricultural Landscapes, pp. 39–67. Nova Science Publishers, New York, USA.
Carvalho, F., Carvalho, R., Mira, A. and Beja, P. 2014. Use of tree hollows by a Mediterranean forest carnivore. Forest Ecology and Management 315: 54–62.
Carvalho F. 2015. Assessing and mitigating road effects on functional landscape connectivity: a case study with common genets (Genetta genetta) in a Mediterranean context. Ph.D. Thesis. University of Évora.
Carvalho F, Carvalho R, Mira A, Beja P. 2016a. Assessing landscape functional connectivity in a forest carnivore using path selection functions. Landscape Ecology 31: 1021–1036.
Carvalho F, Matolengwe T, Camps D, Gaubert P, Do Linh San E. 2016b. A conservation assessment of Genetta genetta. In Child MF, Roxburgh L, Do Linh San E, Raimondo D, Davies-Mostert HT, editors. The Red List of Mammals of South Africa, Swaziland and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Carvalho F, Lourenco A, Carvalho R, Alves PC, Mira A, Beja P. 2018. The effects of a motorway on movement behaviour and gene flow in a forest carnivore: joint evidence from road mortality, radio tracking and genetics. Landsc Urban Plan 178:217–227.
Carvalho F, Rosalino LM. 2023. Genetta genetta geneta. In: Mathias ML, Fonseca C, Rodrigues L, Grilo C, Lopes-Fernandes M, Palmeirim JM, Santos-Reis M, Alves PC, Cabral JA, Ferreira M, Mira A, Eira C, Negrões N, Paupério J, Pita R, Rainho A, Rosalino LM, Tapisso JT, Vingada J (eds) Livro Vermelho dos Mamíferos de Portugal Continental. FCiências.ID, ICNF, Lisboa, pp. 228–229.
Carvalho F, Ziko BA, Mrubata Z, Xhobani M, Plaatjie A, Do Linh San E. 2024a. Multi-scale habitat modelling of latrine site selection by sympatric carnivores in an Albany Thicket landscape, South Africa. Landscape Ecology. 39(3):1-9.
Carvalho F, Galantinho A, Sommers MJ, Do Linh San E. 2024b. Influence of season, sex, and interspecific interactions on the diel activity patterns of two sympatric African small carnivores. Scientific Reports, 14:29701.
Collinson W, Parker D, Patterson-Abrolat C, Alexander G, Davies-Mostert H. 2015. Setjhaba SA, South Afrika: a South African perspective on an emerging transport infrastructure. In: van der Ree R, Smith DJ, Grilo C. (ed.), Handbook of Road Ecology, pp. 439–447. Wiley, Oxford, UK.
Cuzin, F. 2003. Les grands mammifères du Maroc méridional (Haut Atlas, Anti-Atlas et Sahara) : Distribution, écologie et conservation. Ph.D. Thesis, Laboratoire de Biogéographie et Écologie des Vertébrés, École Pratique des Hautes Études, Université Montpellier II.
Delibes, M. 1999. Genetta genetta. In: A.J. Mitchell-Jones, G. Amori, W. Bogdanowicz, B. Kryštufek, P.J.H. Reijnders, F. Spitzenberger, M. Stubbe, J.B.M. Thissen, V. Vohralík and J. Zima (eds), The Atlas of European Mammals, pp. 352-353. Academic Press, London, UK.
Delibes, M. and Gaubert, P. 2013. Genetta genetta Common Genet (Small-spotted Genet). In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. V. Carnivores, Pangolins, Equids and Rhinoceroses, pp. 223-229. Bloomsbury, London, UK.
De Satgé J, Teichman K, Cristescu B. 2017. Competition and coexistence in a small carnivore guild. Oecologia 184:873-884.
Do Linh San E, Ferguson AW, Belant JL, Schipper J, Hoffmann M, Gaubert P, Angelici FM, Somers MJ. 2013. Conservation status, distribution and species richness of small carnivores in Africa. Small Carnivore Conservation 48:4-18.
Espírito-Santo C, Rosalino LM, Santos-Reis M. 2007. Factors affecting the placement of common genet latrine sites in a Mediterranean landscape in Portugal. Journal of Mammalogy 88: 201–207.
Foden WB, Young BE. 2016. IUCN SSC Guidelines for Assessing Species’ Vulnerability to Climate Change. Version 1.0. Occasional Paper of the IUCN Species Survival Commission No. 59. Cambridge, UK and Gland, Switzerland: IUCN Species Survival Commission. x+114pp.
Gaubert P, Begg CM. 2007. Re-assessed molecular phylogeny and evolutionary scenario within genets (Carnivora, Viverridae, Genettinae). Molecular Phylogenetics and Evolution 44: 920–927.
Gaubert P, Carvalho F, Camps D, Do Linh San E. 2015. Genetta genetta. The IUCN Red List of Threatened Species 2015.
Gaubert P. 2016. Fate of the mongooses and the genet (Carnivora) in Mediterranean Europe: none native, all invasive? In: Angelici FM. (ed.), Problematic Wildlife: A Cross-Disciplinary Approach, pp. 295–314. Springer, Cham, Switzerland.
Gaubert, P., Del Cerro, I., Godoy, J.A. and Palomares, F. 2009. Early phases of a successful invasion: mitochondrial phylogeography of the common genet (Genetta genetta) within the Mediterranean Basin. Biological Invasions 11: 523-546.
Gaubert, P., Fernandes, C.A., Bruford, M.W. and Veron, G. 2004. Genets (Carnivora, Viverridae) in Africa: an evolutionary synthesis based on cytochrome b sequences and morphological characters. Biological Journal of the Linnean Society 81: 589-610.
Gaubert, P., Jiguet, F., Bayle, P. and Angelici, F.M. 2008. Has the common genet (Genetta genetta) spread into south-eastern France and Italy? Italian Journal of Zoology 75: 43-57.
Gaubert, P., Machordom, A., Morales, A., López-Bao, J.V., Veron, G., Amin, M., Barros, T., Basuony, M., Djagoun, C.A.M.S., Do Linh San, E., Fonseca, C., Geffen, E., Ozkurt, S.O., Cruaud, C., Couloux, A. and Palomares, F. 2011. Comparative phylogeography of two African carnivorans presumably introduced into Europe: disentangling natural versus human-mediated dispersal across the Strait of Gibraltar. Journal of Biogeography 38: 341-358.
Gaubert, P., Taylor, P.J. and Veron, G. 2005a. Integrative taxonomy and phylogenetic systematics of the genets (Carnivora, Viverridae, genus Genetta): a new classification of the most speciose carnivoran genus in Africa. In: B.A. Huber, B.J. Sinclair and K.-H. Lampe (eds), African Biodiversity: Molecules, Organisms, Ecosystems, pp. 371-383. Springer, New York, USA.
Gaubert P, Taylor PJ, Fernandes CA, Bruford MW, Veron G. 2005b. Patterns of cryptic hybridization revealed using an integrative approach: a case study on genets (Carnivora, Viverridae, Genetta spp.) from the southern African subregion. Biological Journal of the Linnean Society. 86(1):11-33.
Hargey, A. 2022. Estimating leopard density in a coastal protected area of the Western Cape, South Africa. Msc thesis, University of Cape Town, Cape Town.
Harrison, D.L. and Bates, P.J.J. 1991. The Mammals of Arabia. Harrison Zoological Museum, Sevenoaks, UK.
Ikeda H, Izawa M, Baba M, Takeishi M, Doi T, Ono Y. 1983. Range size and activity pattern of three nocturnal carnivores in Ethiopia by radio-telemetry. Journal of Ethology 1: 109–111.
Ikeda H, Ono Y, Baba M, Doi I, Iwamoto T. 1982. Ranging and activity patterns of three nocturnal viverrids in Omo National Park, Ethiopia. African Journal of Ecology 20: 179–186.
Kock, D. 1983. Identifizierung der Palastina-Genetten von J. Aharoni als Vormela peregusna (Guldenstaedt, 1770). Zeitschrift für Säugetierkunde 48: 381-383.
Kuhn, B.F. 2014. A preliminary assessment of the carnivore community outside Johannesburg, South Africa. South African Journal of Wildlife Research 44(1): 95-98.
Larivière S, Calzada J. 2001. Genetta genetta. Mammalian Species 680: 1–6.
Livet F, Roeder JJ. 1987. Encyclopédie des carnivores de France: la genette (Genetta genetta, Linnaeus, 1758). Société Française pour l’Étude et la Protection des Mammifères 17: 1–33.
Loock D, SwanepoeL L. 2019. Assessing local patterns in carnivore occupancy and richness by camera trapping: a monitoring of diversity at Verloren Vallei Nature Reserve, Dullstroom, Mpumalanga. Wildlife & Resource Association (WRA), Bloemfontein.
Lynch CD. 1983. Mammals of the Orange Free State. Memoirs van die Nasionale Museum, Bloemfontein 18: 1-218.
Lynch CD. 1994. The mammals of Lesotho: order Carnivora: family Canidae. Navorsinge van die Nasionale Museum: Research of the National Museum 10(4): 199-209.
Matolengwe T, Mbatyoti A, Somers MJ, Do Linh San E. 2011. Spatio-temporal ecology of spotted genets (Genetta genetta) in the Great Fish River Reserve (Eastern Cape, South Africa): a preliminary analysis. Poster presented at the 11th African Small Mammal Symposiumvvvv. Kwaluseni, Swaziland.
Matolengwe T. 2010. Diet of the small-spotted genet, Genetta genetta (Carnivora: Viverridae), in the Great Fish River Reserve (South Africa). M.Sc. Thesis. University of Fort Hare.
Milton SJ, Short S, Dean WRJ. 2022. Decline in whistling rat (Parotomys brantsii) density: Possible response to climate change in the Karoo, South Africa. African Journal of Ecology, 60(4): 969-979.
Nieman WA. 2018. Culture, Conflict, and Cuisine: A Quantitative Assessment of Terrestrial Vertebrate Off-Take at the Human Wildlife Interface. Msc thesis, University of Stellenbosch, Stellenbosch.
Nieman, W.A., Leslie, A.J. and Wilkinson, A., 2018. The Use of Animals and Animal-Derived Constituents in African Traditional Medicine and Other Cultural Applications: Townships in the Western Cape Province. Culture, Conflict and Cuisine: A Quantitative Assessment of Terrestrial Vertebrate Off-Take at the Human-Wildlife Interface, p.215.
NWPG (North West Provincial Government) 2016. Report examining the status of small carnivores and the detriment of hunting them in the Molopo Bushveld of the Dr Ruth Segomotsi Mompati District of the North West. Department of Rural, Environment and Agricultural Development, North West Provincial Government, Mahikeng.
NWPG (North West Provincial Government) 2017. The status of free ranging carnivores in Bokone Bophirima and impacts of hunting them. Department of Rural, Environment and Agricultural Development, North West Provincial Government, Mahikeng.
NWPG (North West Provincial Government) 2022. The impact of roadkill on mammal populations in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
NWPG (North West Provincial Government) 2024 The impact of roadkill on mammal populations in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Palomares F. 1993c. Faecal marking behaviour by free-ranging common genets Genetta genetta and Egyptian mongooses Herpestes ichneumon in southwestern Spain. Zeitschriftt für Säugetierkunde 58: 225–231.
Palomo, L.J. and Gisbert, J. 2002. Atlas de los mamíferos terrestres de España. Dirección General de Conservación de la Naturaleza. SECEM-SECEMU, Madrid, Spain.
Pereira, M. and Rodríguez, A. 2010. Conservation value of linear woody remnants for two forest carnivores in a Mediterranean agricultural landscape. Journal of Applied Ecology 47: 611–620.
Power RJ, Van Straaten A, Schaller R, Mooke M, Boshoff T, Nel HP. 2019. An inventory of mammals of the North West Province, South Africa. Annals of the Ditsong Museum of Natural History 8: 6-29.
Rautenbach IL. 1978. The mammals of the Transvaal. Phd thesis, University of Natal, Pietermaritzburg.
Rosalino LM, Santos-Reis M. 2002. Feeding habits of the common genet Genetta genetta (Carnivora: Viverridae) in a semi-natural landscape of central Portugal. Mammalia 66: 195–205.
Rowe-Rowe DT. 1992. The Carnivores of Natal. Natal Parks Board, Pietermaritzburg, South Africa.
Sarmento, P., Cruz, J., Eira, C. and Fonseca, C. 2014. A spatially explicit approach for estimating space use and density of common genets. Animal Biodiversity and Conservation 37: 23–33.
Sarmento, P.B., Cruz, J.P., Eira, C.I. and Fonseca, C. 2010. Habitat selection and abundance of common genets Genetta genetta using camera capture-mark-recapture data. European Journal of Wildlife Research 56: 59–66.
Schlawe, L. 1981. Material, Fundorte, Text- und Bildquellen als Grundlagen für eine Artenliste zur Revision der Gattung Genetta G. Cuvier, 1816. Zoologische Abhandlungen (Dresden) 37: 85-182.
Schuette P, Wagner AP, Wagner ME, Creel S. 2013. Occupancy patterns and niche partitioning within a diverse carnivore community exposed to anthropogenic pressures. Biological Conservation 158: 301–312.
Schurch MP, McManus J, Goets S, Pardo LE, Gaynor D, Samuels I, Cupido C, Couldridge V, Smuts B. 2021. Wildlife-friendly livestock management promotes mammalian biodiversity recovery on https://doi.org/10.3389/fcosc.2021.652415a semi-arid Karoo farm in South Africa. Frontiers in Conservation Science 2:6.
Sillero-Zubiri, C. and Marino, J. 1997. The status of small carnivore species in Niokolo-Koba National Park, Senegal. Small Carnivore Conservation 17: 15-19.
Simelane TS, Kerley GI. 1998. Conservation implications of the use of vertebrates by Xhosa traditional healers in South Africa. South African Journal of Wildlife Research (4):121-126.
Skinner JD. Chimimba CT. 2005. The mammals of the southern African subregion. Cambridge University Press, Cambridge.
Smith K, Venter JA, Peel M, Keith M, Somers MJ. 2023. Temporal partitioning and the potential for avoidance behaviour within South African carnivore communities. Ecology and Evolution. 13(8):e10380.
Smithers RHN. 1971. The mammals of Botswana. Museum Memoir No 4. Trustees of the National Museum and Monuments, Salisbury.
Stuart CT. 1981. Notes on the mammalian carnivores of the Cape Province. Bontebok 1: 1 – 58.
Virgós E, Llorente M, Cortés Y. 1999. Geographic variation in genet (Genetta genetta) diet: a literature review. Mammal Review 29: 119–128.
Vissia S, Van Langevelde F. 2022. The effect of body size on co‐occurrence patterns within an African carnivore guild. Wildlife Biology (2):e01004.
Waser, P.M. 1980. Small nocturnal carnivores: ecological studies in the Serengeti. African Journal of Ecology 18: 167-185.
Wentzel J, Gall C, Bourn M, De Beer J, Du Plessis F, Fosgate GT. 2021 Carnivore detection at the domestic/wildlife interface within Mpumalanga Province, South Africa. Animals. 11(9):2535.
Whiting MJ, Williams VL, Hibbitts TJ. 2013. Animals traded for traditional medicine at the Faraday market in South Africa: species diversity and conservation implications. In:Alves RRN, Rosa IL, editors. Animals in traditional folk medicine: implications for conservation. Springer-Verlag Berlin Heidelberg, Pages 421-473.
Woodgate Z, Drouilly M, Distiller G, O’Riain MJ. 2023. The effect of multi-use landscapes on mammal assemblages and its implication for conservation. Land.12(3):599.https://doi.org/10.3390/land12030599.
Zuberogoitia I, Zabala J, Garin I, Aihartza J. 2002. Home range size and habitat use of male common genets in the Urdaibai biosphere reserve, Northern Spain. Zeitschriftt für Säugetierkunde 48: 107–113.