Sloggett’s Vlei Rat
Otomys sloggetti

2025 Red list status
Least Concern
Regional Population Trend
Increasing
Change compared
to 2016
No Change
Overview
Otomys sloggetti – Thomas, 1902
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Otomys – sloggetti
Common Names: Sloggett’s Vlei Rat, Ice Rat, Sloggett’s Karoo Rat (English), Sloggett se Rot, Sloggett se Karoorot (Afrikaans) Leboli-leqhoa (Sotho)
Synonyms: Myotomys sloggetti (Thomas, 1902)
Taxonomic Note: Although Otomys sloggetti is considered to be defined by primitive dental characters, its position on the phylogeny of Otomyinae remains unresolved (Taylor et al. 2004).
Red List Status: LC – Least Concern
Assessment Information
Assessors: Taylor, P.1 & da Silva, J.M.2
Reviewer: Oosthuizen, M.3
Institutions: 1University of the Free State,2South African National Biodiversity Institute,3University of Pretoria
Previous Assessors and Reviewers: Pillay, N., Taylor, P., Baxter, R. & Child, M.F.
Previous Contributors: Monadjem, A., Wilson, B., MacFadyen, D., Avenant, N., Avery, M. & Palmer, G.
Assessment Rationale
This high-altitude endemic is listed as Least Concern because it has a relatively wide distribution within the assessment region, occurs in several protected areas, including the Maloti-Drakensberg Transfrontier Conservation Area, and because it either occurs in inaccessible habitats unlikely to be transformed, or it benefits for example from overgrazing and soil erosion in Lesotho (Mokotjomela et al. 2009). There are no known threats that could cause rapid population decline. Climate change is not suspected to be an emerging threat. Conversely, density has been estimated to have increased threefold in the Lesotho Drakensberg between 1994 and 2006 possibly due to warmer temperatures. Thus, we list it as Least Concern. However, continuing habitat degradation from overgrazing, as well as any other identified minor threats, must be monitored.
Red List Index
Red List Index: No change
Recommended Citation: Taylor P & da Silva JM. 2025. A conservation assessment of Otomys sloggetti. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
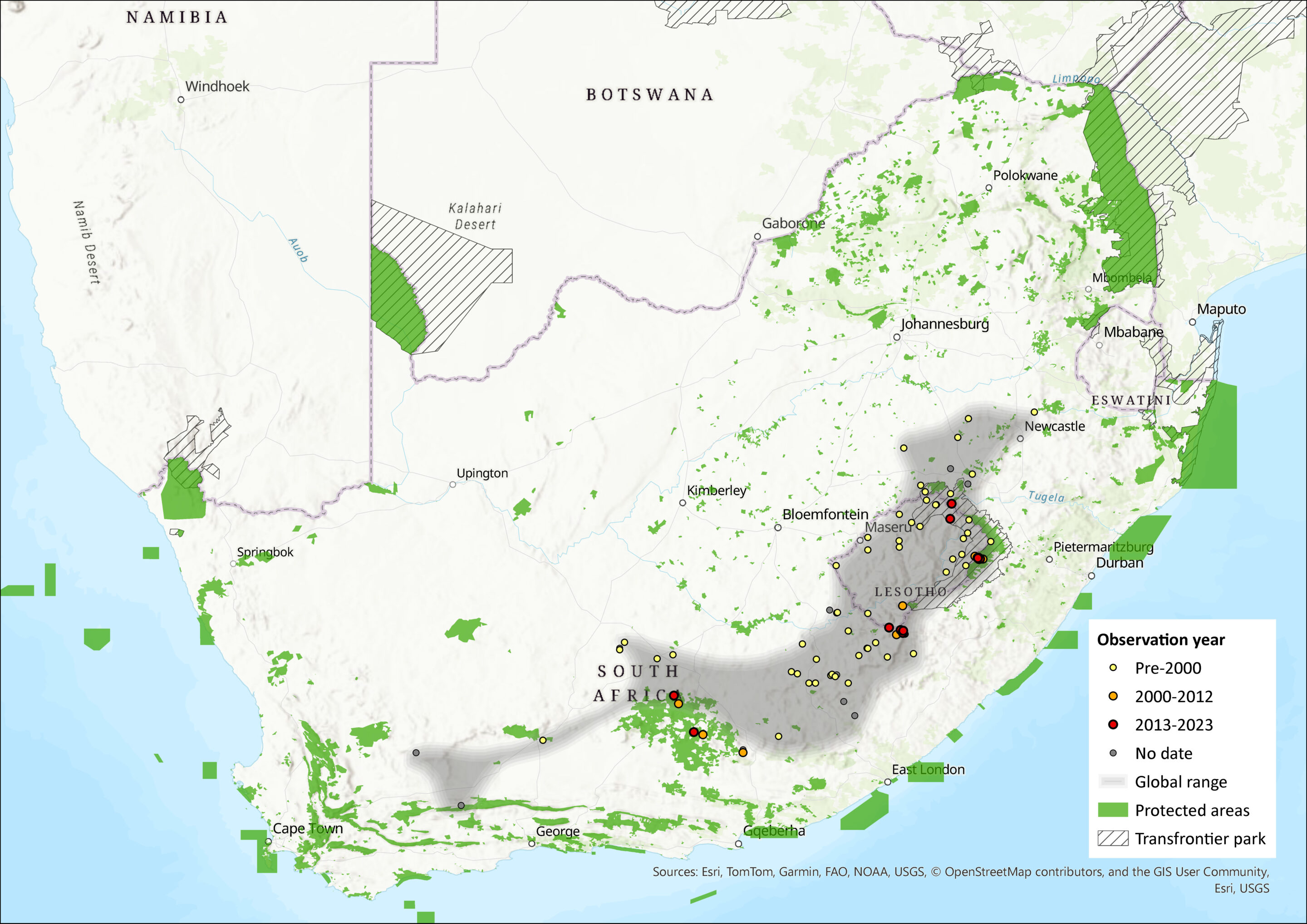
Sloggett’s Vlei Rat is found predominantly at high elevations (>2,000 m) in the central Drakensberg Mountains of the Eastern Cape and western KwaZulu-Natal provinces of South Africa as well as Lesotho (Lynch & Watson, 1992, Lynch 1994, Monadjem et al. 2015). Isolated subpopulations have been recorded from three localities > 1700 m asl in the Sneeuberg Mountain Complex of the southern Drakensberg (Kok et al. 2012, Taylor et al. 2020), and from inselbergs and mountain ranges in the Karoo at >1,500 m asl. However, only three out of six historical records from the Karoo can be verified from identified museum specimens. These include the type specimen collected in 1902 from Deelfontein (Natural History Museum, London, catalogue number 2.9.1.61, personally checked by PJ Taylor), and two specimens in the Ditsong National Museum of Natural History collected from Hanover (locus 3124 Ab) in 1903 and Orange River (locus 2924 Ca) in 1934 (Lynch & Watson (1992). Given the antiquity of these records (>90 years ago), and the fact that recent surveys in the Karoo have not detected them (Lynch & Watson 1992), it is possible that the Karoo subpopulation is locally extinct. Nevertheless, as the western-most limit of the current range of the species, the Sneeuberg range represents a transition area between the Nama-Karoo and Grassland biomes (Kok et al. 2013). In the Drakensberg Range, O. auratus typically occurs on mid-elevation grassland slopes while O. sloggetti is found at higher elevations in both grassland and alpine heath habitats. However in the Sneeuberg, O. sloggetti has been collected together with O. auratus and O. willani (Kok et al. 2012; Taylor et al. 2020), and in the alpine zone at 3100 m asl at Witsieshoek Alpine Research Station, O. auratus and O. sloggetti were collected together in 2022 and 2023, but with O. auratus occurring in more lush alpine vegetation and both O. sloggetti and O. auratus in piles of rocks created in the construction of the alpine research station (PJ Taylor, personal observation). Fossils attributable to O. sloggetti have been described from deposits from abut 15 kya to 150 kya on the south coast, suggesting that the species had a much wider distribution range during the late Pleistocene up until at least the Last Glacial Maximum (Matthews et al. 2021).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Sloggett’s Vlei Rat (Otomys sloggetti) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Lesotho | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
There is evidence that the species’ range has contracted to higher elevations both in prehistoric and historical time scales (Matthews and Nel 2021). Even though the species is apparently adaptable to montane and alpine rangeland degradation, it may still suffer further range contractions due to accelerated global warming in mountains and the “Escalator to Extinction” phenomenon.
Population Information
Population densities of over 100 individuals/ha have been recorded in suitable rocky habitats (Willan 1990). In Lesotho, they are common in the higher areas and may be increasing. A field survey study in 2006 at three localities separated by 70, 80 and 130 km in the north-eastern Lesotho Drakensberg (Sani Valley, Oxbow Motete Valley and Katse Dam) revealed an increase in maximum densities from 110 to 342 animals / hectare between 1992 and 2006 in Oxbow and from 100 to 319 animals / hectare between 2004 and 2006 in Sani Valley (Mokotjomela et al. 2010). Katse Dam had low numbers of Sloggett’s Vlei Rat, possibly due to competition with O. auratus and/or habitat loss from human settlement expansion (Mokotjomela et al. 2010). Overall, the population increase is possibly due to warming temperatures in the region that reduce winter die-off and increase habitat productivity.
Population Information
Current population trend: Increasing
Continuing decline in mature individuals: No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No. Tends to occur in high-altitude areas that are connected with one another and unlikely to be significantly transformed.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Based on mitochondrial CytB divergence, three distinct groups of O. sloggetti can be distinguished occurring from west to east geographically along the Drakensberg (Phukuntsi et al. 2016; Taylor et al. 2020). A central clade (“GS1” of Phukuntsi et al. 2016) includes individuals from Tiffendell Ski Resort in the Eastern Cape close to the Free State border. A western clade (corresponds to the Sneeuberg mountain range but includes two individuals from Tiffendell. An eastern clade (“GS3”) corresponds to the northern Drakensberg from Sani Pass in Lesotho (Taylor et al. 2020).
Given that the available molecular evidence is based on a single mitochondrial marker and may reflect distant (older) relationships, a finer scale population genetic study is recommended to gain a more comprehensive and recent look at the genetic structure and diversity within the species. However, given their dispersal capabilities, it is assumed that they likely exist as metapopulations within each lineage, with at least 1 migrant per generation moving between subpopulations allowing for gene flow.
Based on the available molecular evidence, it is possible to quantify the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF) complementary genetic indicator – proportion of populations maintained (PM) within a species. Given that the 3 lineages/metapopulations still exist and no other lineages are thought to have gone extinct, this indicator would receive a value of 1.0 (3/3 distinct groups remain).
While actual population sizes are unknown, based on the high densities estimates in suitable habitat and the assumption that each lineage represents a metapopulation, it is likely that each population is stable and thriving, likely exceeding 5,000-10,000 individuals each (and this supports by an effective population size (Ne) in excess of 500). While this is a gross assumption that should be validated using molecular techniques, it allows for the quantification of the GBF’s headline indicator – the proportion of populations with Ne > 500. This indicator would therefore receive a value of 1.0 (all populations likely exceed Ne 500).
Habitats and ecology
Occurs in montane grasslands on xeric or mesic soils, either dry or wet typically amidst piles of loose stones or boulders, both natural and man-made (for example, stone walls). It does not occur in modified habitats but will sometimes nest in crevices in rock foundations of roads (Willan 1990). They are diurnal (Hinze & Pillay 2006; Oosthuizen 2020) and feed on stems, leaves and floral parts of green plants. In the Sani Valley, O. sloggetti feeds on wetland grasses, sedges and herbaceous vegetation but avoid Helichrysum spp. (Schwaibold and Pillay 2010).
It lives in colonies consisting of at least 4-16 individuals and the breeding season occurs between October and March (Hinze 2005). For example, in the Sani Valley, O. sloggetti lives in mixed-sex colonies of up to 17 individuals (Hinze et al. 2013), which construct an intricate underground burrow system in organic and mineral soils (Hinze et al. 2006). Plants taken below ground are used for nesting and there is no evidence of food hoarding (Hinze et al. 2006). Suitable wetland sites in the Sani Valley are home to several colonies and competition for preferred food plants leads to solitary feeding and avoidance between individuals of the same and different colonies (Hinze et al. 2013). Rocky surfaces and boggy soil limits dispersal (Mokotjomela et al. 2010).
Ecosystem and cultural services: It may be an important prey species for predators occurring at high altitudes. Their extensive burrow systems s contributes to soil turnover and aeration. However, when burrows collapse, the resulting gullies alter water flow, contributing to erosion (Grab and Deschamps 2004).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 4.7. Grassland -> Grassland – Subtropical/Tropical High Altitude | – | Suitable | – |
| 5.4. Wetlands (inland) -> Wetlands (inland) – Bogs, Marshes, Swamps, Fens, Peatlands | – | Suitable | – |
| 6. Rocky areas (e.g. inland cliffs, mountain peaks) | – | Suitable | – |
Life History
| Generation Length | Justification | Data Quality |
| 1-2 years | Inferred (Pacifici et al. 2013) | good |
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: 2-3 years in captivity, max 5 years
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 38-40 days
Reproductive Periodicity: breeding during summer
Average Annual Fecundity or Litter Size:1-4 young, max 5
Natural Mortality: (Not specified)
Does the species lay eggs? no
Does the species give birth to live young: yes
Does the species exhibit parthenogenesis: no
Does the species have a free-living larval stage? no
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
There is anecdotal information of herdsman in Lesotho hunting O. sloggetti. However, this threat remains to be quantified.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no major identified threats to the species. Unlike other Otomys species threatened by climate change (Taylor et al. 2016), population increases in Lesotho are the result of better overwintering of O. sloggetti as a consequence of warmer minimum temperatures over the past two decades (Mokotjomela et al. 2010). Overgrazing the vegetation by domestic livestock and O. sloggetti themselves around wetlands reduces habitat suitability.
Habitat trend: Stable. This species inhabits high-altitude areas that are unlikely to be transformed significantly. However, overgrazing may reduce habitat quality, and this should be monitored. Another important consideration is that the increasing population sizes contribute greatly to habitat change through vegetation loss and consequent soil erosion (Grab and Deschamps 2004). Experimental plots erected in the Sani Valley to investigate the single or combined effects of O. sloggetti and domestic livestock on soil erosion and loss showed that the damage caused by O. sloggetti exceeds that of livestock (Mokotjomela et al. 2009). Therefore, while low temperature and prolonged snowfall are density independent regulators of O. sloggetti populations in Lesotho, diminishing resources (food and suitable nest sites) are possible density-dependent regulators. Correspondingly, an increase in population densities may accelerate soil erosion rates through their feeding and burrowing habits (Mokotjomela et al. 2009).
Conservation
It occurs in many protected areas across its range such as the Maloti-Drakensberg Transfrontier Conservation Area (Monadjem et al. 2015) and Mountain Zebra National Park (Kok et al. 2012). Although no specific interventions are necessary at present, wetland conservation and restoration are likely to benefit this species.
Recommendations for land managers and practitioners:
- Land managers should maintain a vegetation buffer around wetlands to reduce impacts of land-use practices.
- Land managers should practice holistic management of ranchlands, including de-stocking and rotational grazing.
- Further long-term, systematic monitoring is needed to establish subpopulation trends and threat levels.
Research priorities:
- Fine scale studies on habitat loss and inferred impact on the species
- Effects of overgrazing on the density and viability of this species.
- Population fluctuations in response to climate change.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
Bibliography
Grab, S.W. and Deschamps, C.L. 2004. Geomorphological and geoecological controls and processes following gully development in alpine mires, Lesotho. Arctic, Antarctic, and Alpine Research 36: 49–58.
Hinze A. 2005. Social behaviour and activity patterns of the African ice rat Otomys sloggetti robertsi. Faculty of Science, School of Biology, University of the Witwatersrand.
Hinze, A. & Pillay, N. 2006. Life in an African Alpine habitat: Diurnal activity patterns of the ice rat Otomys sloggetti robertsi. Arctic, Antarctic and Alpine Research 38:540-546.
Hinze, A., Pillay, N. and Grab, S. 2006. The burrow system of the African ice rat Otomys sloggetti robertsi. Mammalian Biology 71: 356–365.
Hinze, A., Rymer, T. and Pillay, N. 2013. Spatial dichotomy of sociality in the African ice rat. Journal of Zoology 290: 208–214.
IUCN. 2017. The IUCN Red List of Threatened Species. Version 2017-2. Available at: www.iucnredlist.org. (Accessed: 14 September 2017).
Kok, A.D., Parker, D.M. and Barker, N.P. 2012. Life on high: the diversity of small mammals at high altitude in South Africa. Biodiversity and Conservation 21: 2823–2843.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
Lynch CD, Watson JP. 1992. The distribution and ecology of Otomys sloggetti (Mammalia: Rodentia) with notes
on its taxonomy. Navorsinge van die Nasionale Museum Bloemfontein 8: 141–158.
Matthews T, Nel TH. 2021. The cryptic case of Otomys sloggetti (Sloggett’s Vlei Rat): Interpreting murid molar morphology in the fossil record. South African Journal of Science 117(1/2), https://doi.org/10.17159
sajs.2021/7137
Mokotjomela, T., Schwaibold, U. and Pillay, N. 2009. Does the ice rat Otomys sloggetti robertsi contribute to habitat change in Lesotho? Acta Oecologica 35: 437–443.
Mokotjomela, T., Schwaibold, U. and Pillay, N. 2010. Population surveys of the ice rat Otomys sloggetti robertsi in the Lesotho Drakensberg. African Zoology 45: 225–232.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa – a biogeographic and taxonomic synthesis. De Gruyter, Berlin/Munich/Boston.
Oosthuizen, M.K. 2020. Temporal flexibility in activity rhythms of a diurnal rodent, the ice rat (Otomys sloggetti). Chronobiology International 37:824-835.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Schwaibold, U. and Pillay, N. 2010. Habitat use in The Ice Rat Otomys sloggetti robertsi. South African Journal of Wildlife Research 40: 64–72.
Taylor, P.J., Denys, C. and Mukerjee, M. 2004. Phylogeny of the African murid tribe Otomyini (Rodentia), based on morphological and allozyme evidence. Zoologica Scripta 33: 389–402.
Taylor, P.J., Nengovhela, A., Linden, J. and Baxter, R.M. 2016. Past, present, and future distribution of Afromontane rodents (Muridae: Otomys) reflect climate-change predicted biome changes. Mammalia 80: 359–375.
Willan, K. 1990. Reproductive biology of the southern African ice rat. Acta Theriologica 35: 39–51.