Slender Mongoose
Herpestes sanguineus

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Herpestes sanguineus – (Rüppell, 1835)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – HERPESTIDAE – Herpestes – sanguineus
Common Names: Common Slender Mongoose, Black-tipped Mongoose, Slender Mongoose (English), Rooimuishond, Swartkwasmuishond (Afrikaans), Iwobo (Ndebele), Kgano (Sesotho, Setswana), Kganwe, Khano, Ramotsibodi (Setswana), Chakidze (Swati), Mangovo (Tsonga), Khohe, Khoke (Venda), Uchakidze (Zulu)
Synonyms: Galerella sanguinea (Rüppell, 1836)
Taxonomic Note:
This species is sometimes included in the genus Galerella (e.g. Meester et al. 1986; Wozencraft 1993, 2005) as the genus Herpestes is paraphyletic (Veron et al. 2022)
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Do Linh San, E.1, Filonzi, J.2, Adams, E.C.3 & da Silva, J.M.4
Reviewer: Smith, C.3
Contributor: Moyo, D.2
Institutions: 1Sol Plaatje University, 2University of Fort Hare, 3Endangered Wildlife Trust, 4South African National Biodiversity Institute
Previous Assessors & Reviewers: Do Linh San, E., Zemouche, J., Madikiza, Z., Maddock, A.H., Perrin, M. & Mills,M.
Previous Contributor: Relton, C.
Assessment Rationale
The Slender Mongoose is listed as Least Concern as it is common and widespread in a variety of habitats (including human-modified landscapes), there are no major threats that could cause rapid population decline, and it is present in several protected areas (notably Kruger National Park and the Kgalagadi Transfrontier Park) across its distribution range within the assessment region.
Regional population effects: Dispersal is likely between regions, as this species’ range is continuous across southern Africa from northern South Africa to Botswana, Namibia, Zimbabwe and Mozambique south of the Zambezi River. The Slender Mongoose is not limited by fences and is also present within several transfrontier reserves, including the Greater Mapungubwe Transfrontier Conservation Area.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Do Linh San E, Filonzi J, Adams EC & da Silva JM. 2025. A conservation assessment of Herpestes sanguineus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
One of the most widely distributed African mongooses, ranging from Senegal in the west to the Red Sea coast of Sudan in the east and south to the Northern Cape in South Africa (Hoffmann & Taylor 2013). Stuart (1981) mentions a museum record of this species from Mountain Zebra National Park, but this specimen is not mentioned in the studies of Watson and Dippenaar (1987) and Watson (1990), and the most southerly distribution limit is probably the far eastern part of the Eastern Cape in South Africa (Hoffmann & Taylor 2013). This species also occurs in Zanzibar (Stuart & Stuart 1998; Goldman & Winther-Hansen 2003). It ranges from sea level to 2,700 m asl in the Ethiopian Highlands (Yalden et al. 1996).
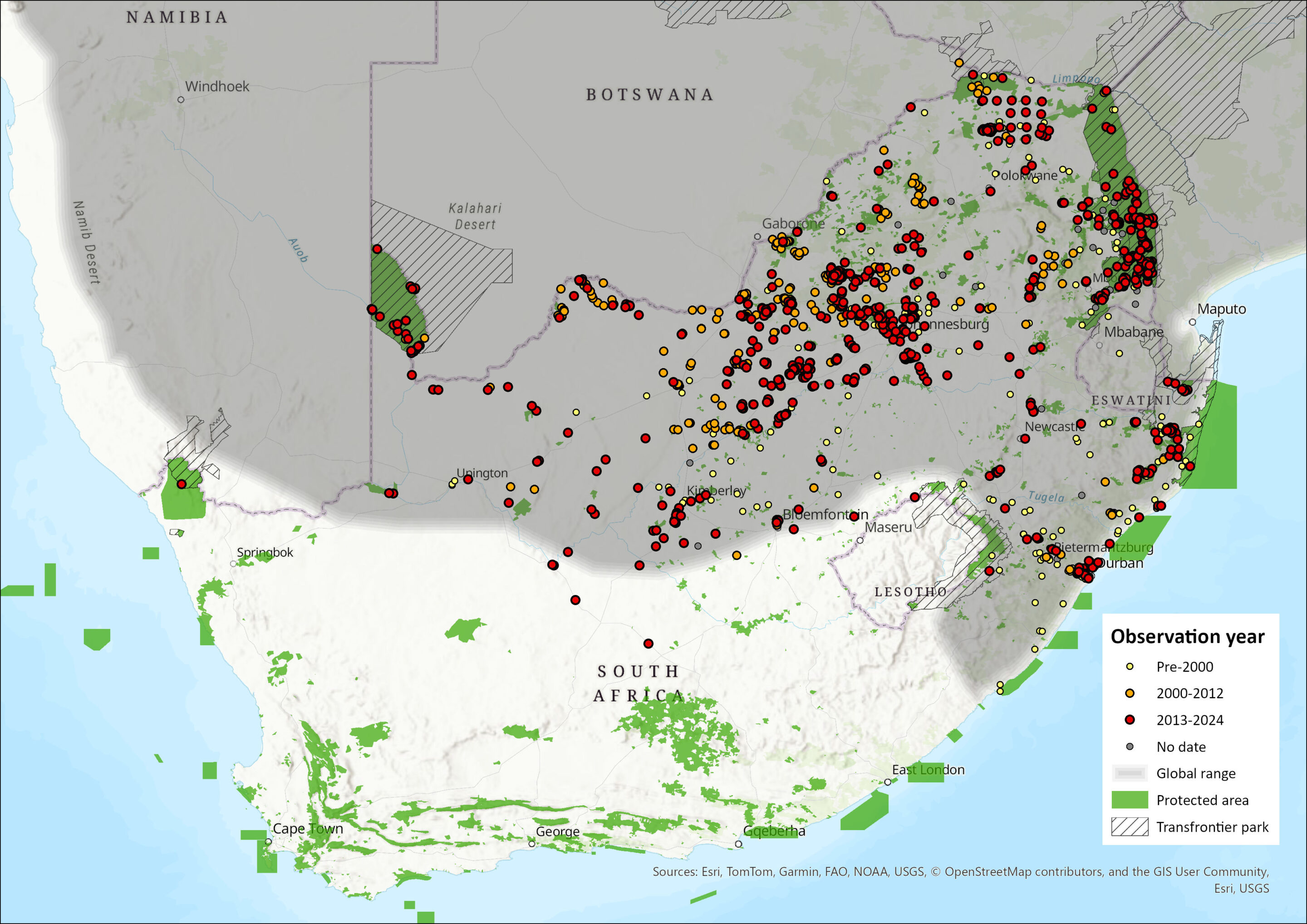
Within the assessment region, the Slender Mongoose occurs across all savannah habitats north of the Orange River, but is absent from montane grassland. This includes Limpopo, North West, Gauteng and Mpumalanga provinces, much of eastern KwaZulu-Natal Province, the central and northwestern areas of the Free State Province, along part of the east coast of the Eastern Cape Province, as well as Eswatini (Skinner & Chimimba 2005). In the Kalahari, it occurs especially amongst calcrete outcrops and not in the dunes (Mills et al. 1984).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0 m
Elevation Upper Limit (in metres above sea level): 2,700 m (in Ethiopia)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Slender Mongoose (Herpestes sanguineus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Cabo Verde | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Djibouti | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Liberia | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
No research has focused on how the Slender Mongoose will be affected by climate change in the future. However, research has been conducted on similar sized species such as Suricates (Suricata suricatta) and Cape Ground Squirrels (Xerus inauris) in arid environments (Fuller et al. 2021). It has been noted that smaller animals such as the Slender Mongoose may be at risk as temperatures continue to increase as a result of the greater surface area to volume ratio, resulting in them increasing their body temperature faster than larger animals (Fuller et al. 2021). This could put particularly the arid populations more at risk of extinction if they fail to regulate their activity patterns accordingly, such as through practicing crepuscular rather than diurnal activity.
However, Kamau et al. (1979) observed that, in controlled laboratory conditions, despite the larger surface to volume ratio, the slender mongoose’s metabolic rate did not differ to that of same size mammals, suggesting a physiological adaptation to warmer climates. Additionally, it was recorded that the slender mongoose could maintain normal metabolic functions between 10°C and 38°C (Kamau et al. 1979).
Population
Slender Mongooses are among the most common mongooses in Africa. In the Serengeti National Park, Tanzania, population densities between 1975 and 1990 ranged from 3–6 individuals / km² (Waser et al. 1995). In the Kalahari, South Africa, based on data collected between 2007 and 2011, population density was around 1.6–2.0 adults / km² (B. Graw et al. unpubl. data). In Vernon Crookes Nature Reserve, KwaZulu-Natal, Maddock (1988) estimated a density of 7.3 individuals / km². Considering the wide distribution of this species and a minimum average density of at least 0.1 individual / km², we estimate that there are well over 10,000 individuals in the assessment region.
Population Information
Current population trend: Stable, based on wide habitat tolerance and extent of occurrence.
Continuing decline in mature individuals? No
Extreme fluctuations in the number of subpopulations: Unlikely due to the relatively generalist habits of this species
Continuing decline in number of subpopulations: Unlikely
All individuals in one subpopulation: Unknown, but possible based on inference from genetic data
Number of mature individuals in population: Probably > 10,000
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
No population genetic study has been conducted on this species to better understand its genetic structure and health within the assessment region; however, a molecular investigation was undertaken to look at the social organisation and structure of a particular group of Slender Mongooses in the southern Kalahari (Graw et al. 2019). It was found that this specie formed spatial groups consisting of one to three adult males that overlapped the ranges of several adult females. Female ranges were more exclusive, overlapping on average 12.3% annually, while male ranges showed a much larger overlap of 66.2%. There was some but much less overlap between individuals of neighbouring spatial groups, with males sharing about 11.2% of their range with males considered outside of their spatial group, and females only marginally overlapped (2%) with females in neighbouring spatial groups.
While this does provide some insight, a more comprehensive population genetic study would help understand if there are any barriers to gene flow. Based on the results from Graw et al. (2019), there would appear to be sufficient gene flow within the species to have it considered as a single metapopulation, especially as it is considered one of the most common and abundant of mongooses in the region. Consequently, the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF’s) complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (all populations remaining).
While no census (Nc) or effective population size (Ne) data exists, it is estimated that well over 10,000 individuals exist within the assessment region. Even though the 10 000 value is not an actual census count, the quantity can still be used as a proxy for quantifying the GBF’s headline genetic indicator – proportion of populations with an Ne > 500. Based on the assumption that the species is likely to occur as a single metapopulation, the Ne of the species is ~1000–3000 individuals, based on a Ne/Nc conversion ratio of 0.1– 0.3. Having the 1 population above Ne 500, translates to an indicator score of 1.0 (1/1 population with Ne > 500).
Further population genetic analyses are needed to understand the genetic structure and health of this species.
Habitats and ecology
Slender Mongooses are present in a wide variety of habitats, but absent from true deserts, such as the Namib Desert (Pallett & Thomson 2022), and in sub-desertic parts of the Sahara. They occur on forest fringes and may penetrate into forests along roads and are sometimes found around villages (Hoffmann & Taylor 2013). They are also absent from the greater parts of the karroid regions of the Northern, Western and Eastern Cape provinces. This is speculated to be as a result of competitive exclusion by the Cape Grey Mongoose (H. pulverulentus; Skinner & Chimimba 2005). There is, however, some range overlap between these species at the perimeter of the Slender Mongoose’s range (Skinner & Chimimba 2005). In urban regions they occur in open habitats, as long as there is some cover (Ramesh & Downs 2014; Cronk & Pillay 2020), such as hollow logs, rocks, fallen trees or disused Aardvark (Orycteropus afer) holes. They have also been recorded among rocky outcrops, particularly in grassland regions where Yellow Mongooses (Cynictis penicillata) are also present (Rautenbach 1982; Rood 1989; Webster et al. 2021; Moyo 2022). In farmland landscapes within the Drakensberg Midlands, KwaZulu-Natal, the detection likelihood of this species with camera-traps was positively correlated with bushland cover and human abundance; possibly due to a reduction in natural predator density and increased food and water resources (Ramesh & Downs 2014).
Slender Mongooses are almost exclusively diurnal (Graw & Manser 2017; Moyo 2020). Intersexual differences in activity patterns were observed: in the Kalahari females displayed an earlier onset of activity (Graw & Manser 2017), while in Telperion Nature Reserve (Mpumalanga) females were recorded to cease activity earlier than males (Moyo 2020). As generalist carnivores, Slender Mongoose’s diet mainly comprises small vertebrates (rodents, insectivores, reptiles, amphibians, birds), invertebrates (insects, spiders, millipedes), plant material and fruit (Maddock 1988; Graw & Manser 2017; Zemouche 2018). In urban environments, hard invertebrates such as Coleopterans, small to medium-sized mammals and anthropogenic food items are commonly consumed (Cronk & Pillay 2019). While this species is largely terrestrial, it is more arboreal than most other mongoose species (Hoffmann & Taylor 2013). Although individuals are predominantly solitary, occasionally up to three – related and unrelated – males form loose coalitions within the same home range, which overlaps with the home ranges of up to four females (Rood 1989; Waser et al. 1994; Graw et al. 2016, 2019). Communal denning has also been observed in the Kalahari; this type of activity peaked when temperatures were at their minimum, indicating thermoregulation (Graw et al. 2019). Females were observed to only share burrows with their offspring and males, while males shared with both males and females (Graw et al. 2019). In the Serengeti, male home ranges vary from 70–80 ha, while females occupy smaller ranges of 30–50 ha (Rood & Waser 1978). In Telperion Nature Reserve, mean home range size is slightly smaller with 54 ha for males and 42 ha for females (Moyo 2020). However, home range sizes are larger in the Kalahari (South Africa), with 362 ha on average for males and 118 ha for females (Graw et al. 2019).
Young are born during the wet, summer months (October–March), which coincides with a peak in the abundance of insectivorous prey. Females regularly begin reproducing at 1 year of age (Waser et al. 1995) and after a gestation period of 60–70 days, 1–4 pups are born (Taylor 1969, 1975; Graw & Manser 2017). One to two litters are produced per reproductive season, with on average 130 days between the birth of each litter (Graw & Manser 2017). Pups are born in hollow trees or rock crevices, emerge for the first time between 4 and 6 weeks, and start foraging with their mother 6–9 weeks after their birth. Weaning likely takes place at the age of 7–12 weeks, and juveniles become independent when 3–5 months old (Graw & Manser 2017). Dispersing sex and dispersal age and distances vary greatly. In the Serengeti, juveniles typically disperse within their first 6 months with males dispersing earlier and further than females (Rood & Waser 1978; Waser et al. 1994). In contrast, in a study conducted in the Kalahari, none of the females remained in their natal range past the age of 10 months, while genetic analyses demonstrated that 93% of males were philopatric, and anecdotal field evidence suggested that males disperse less often and possibly later but further than females (Graw et al. 2016). In the Tanzanian study juveniles had a survival rate of 0.63, whereas adults of both sexes had higher survival rates, namely 0.82 for males and 0.79 for females (Waser et al. 1995). Similar results were obtained in the Kalahari (Graw & Manser 2017). In the wild, both male and female Slender Mongooses have a maximum lifespan of 8–10 years (Waser et al. 1995; Graw & Manser 2017).
Ecosystem and cultural services: This species may be a valuable predator of agricultural pest species, such as grasshoppers, termites, beetles and possibly rodents. Further research is required to quantify this effect.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.7. Shrubland -> Shrubland – Subtropical/Tropical High Altitude | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: 8–10 years
Average Reproductive Age: 1 year
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 60–70 days
Reproductive Periodicity: October to March
Average Annual Fecundity or Litter Size: 1–4 pups per litter
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Slender Mongooses have been recorded in bushmeat markets in West Africa (e.g. Colyn et al. 2004) and Cunningham and Zondi (1991) listed this species among those used in traditional medicine in KwaZulu-Natal, South Africa.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Used as bushmeat and for traditional medicine. | No | – |
National Commercial Value: No
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | Yes | No | Yes | – |
| 3. Medicine – human & veterinary | – | Yes | – | – |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: Unknown, but probably stable.
Threats
There are no major threats to this species. As stated above, Slender Mongooses are locally used as bushmeat and in traditional medicine. Although wildlife ranching and the private sector have possibly had a positive effect on this species due to the conservation and connection of suitable habitats (e.g. in the Waterberg), this small carnivore may be accidentally caught as bycatch in predator and rodent control programmes using poisons. It is however unlikely that these consumptive uses, and accidental mortalities have a substantial effect on the population. This species could be threatened by climate change in the future, particularly the populations in arid environments.
Conservation
This species is present in numerous protected areas across its range, notably in Kruger National Park and the Kgalagadi Transfrontier Park. No conservation interventions are currently deemed necessary within the assessment region; however, this species is likely to benefit from the expansion of protected areas to connect suitable habitat patches.
Recommendations for land managers and practitioners:
- Create conservancies to protect and connect habitat.
Research priorities:
- Monitoring “subpopulations” to detect trends across various land uses.
- General studies on the biology and ecology of this species in different habitat types.
- The effects of climate change on this species throughout its range.
- Population health and genetics across its range.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
Bibliography
Colyn, M., Dufour, S., Condé, P.C. and Van Rompaey, H. 2004. The importance of small carnivores in forest bushmeat hunting in the Classified Forest of Diecké, Guinea. Small Carnivore Conservation 31: 15-18.
Cronk, N. and Pillay, N. 2019. Dietary overlap of two sympatric African mongoose species in an urban environment. Mammalia, 83(5), 428-438.
Cronk, N.E., and Pillay, N. 2020. Spatiotemporal co-occurrence and overlap of two sympatric mongoose species in an urban environment. Journal of Urban Ecology, 6(1), juaa013.
Cunningham, A.B. and Zondi, A.S. 1991. Use of animal parts for the commercial trade in traditional medicines. Institute of Natural Resources, University of Natal, Pietermaritzburg, South Africa.
Fuller, A., Mitchell, D, Maloney, S.K., Hetem, R.S., Fonsêca, V.F., Meyer, L.C., Van de Ven T.M. and Snelling, E.P. 2021. How dryland mammals will respond to climate change: the effects of body size, heat load and a lack of food and water. Journal of Experimental Biology, 224(Suppl_1), jeb238113.
Goldman, H.V. and Winther-Hansen, J. 2003. First photographs of the Zanzibar servaline Genet Genetta servalina archeri and other endemic subspecies on the island of Unguja, Tanzania. Small Carnivore Conservation 29: 1-4.
Graw B, Lindholm AK, Manser M. 2016. Female-biased dispersal in the solitarily foraging slender mongoose, Galerella sanguinea, in the Kalahari. Animal Behaviour 111: 69–78.
Graw B, Manser M. 2017. Life history patterns and biology of the slender mongoose (Galerella sanguinea) in the Kalahari Desert. Journal of Mammalogy 98: 332–338.
Graw, B., Kranstauber, B., and Manser, M.B. 2019. Social organization of a solitary carnivore: spatial behaviour, interactions and relatedness in the slender mongoose. Royal Society Open Science, 6(5), 182160.
Hoffmann, M. and Taylor, M.E. 2013. Herpestes sanguineus Slender Mongoose. In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. V. Carnivores, Pangolins, Equids and Rhinoceroses, pp. 314-319. Bloomsbury, London, UK.
Kamau, J.M., Johansen, K., and Maloy, G.M.O. 1979. Thermoregulation and standard metabolism of the slender mongoose (Herpestes sanguineus). Physiological Zoology 52(4): 594-602.
Maddock, A.H. 1988. Resource partitioning in a viverrid assemblage. Ph.D. Thesis, University of Natal.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum , Pretoria, South Africa.
Mills, M.G.L., Nel, J.A.J. and Bothma, J. du P. 1984. Notes on some smaller carnivores from the Kalahari Gemsbok National Park. Koedoe Supplement 221-227.
Moyo, D. 2020. Spatio-temporal ecology of the slender mongoose (Galerella sanguinea) in Telperion Nature Reserve, Mpumalanga, South Africa. MSc dissertation, University of Fort Hare, Alice, South Africa.
Pallett J and Thomson G 2022. A conservation assessment of Slender Mongoose Herpestes sanguineus. In: NCE,
LCMAN, MEFT (eds) 2022. Conservation Status and Red List of the Terrestrial Carnivores of Namibia. Pp 138-140. MEFT, LCMAN & NCE,Windhoek, Namibia
Power, J.P. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Ramesh T, Downs CT. 2014. Modelling large spotted genet (Genetta tigrina) and slender mongoose (Galerella sanguinea) occupancy in a heterogeneous landscape of South Africa. Mammalian Biology 79: 331–337.
Rautenbach, I. L. 1982. Mammals of the Transvaal. Ecoplan monograph, N.S. Supplemento 1: 111-211.
Rood JP, Waser PM. 1978. The slender mongoose, Herpestes sanguineus, in the Serengeti. Carnivore 1: 54–58.
Rood JP. 1989. Male associations in a solitary mongoose. Animal Behaviour 38: 725–727.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Stuart, C. and Stuart, T. 1998. A note on the herpestids and viverrids of south-eastern Unguja (Zanzibar) Island. Small Carnivore Conservation 18: 16-17.
Stuart, C.T. 1981. Notes on the mammalian carnivores of the Cape Province, South Africa. Bontebok 1: 1-58.
Taylor M.E. 1969. Notes on the breeding behaviour of two genera of viverrids Genetta spp. and Herpestes sanguineus in Kenya. East African Wildlife Journal 7: 168–169.
Taylor ME. 1975. Herpestes sanguineus. Mammalian Species 65: 1–5.
Taylor, M.E. 2013. Herpestes ochraceus Somali Slender Mongoose. In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. V. Carnivores, Pangolins, Equids and Rhinoceroses, pp. 310-311. Bloomsbury, London, UK.
Veron, G., Patou, M.-L. and Jennings, A.P. 2022. Systematics and evolution of the mongooses (Herpestidae, Carnivora). In: E. Do Linh San, J.J. Sato, J.L. Belant and M.J. Somers (eds), Small Carnivores: Evolution, Ecology, Behaviour and Conservation, pp. 61–78. Wiley-Blackwell, Oxford, UK.
Waser PM, Keane B, Creel SR, Elliot LF, Minchella DJ. 1994. Possible male coalitions in a solitary mongoose. Animal Behaviour 47: 289–294.
Waser, P.M., Elliott, L.F., Creel, N.M. and Creel, S.R. 1995. Habitat variation and mongoose demography. In: A.R.E. Sinclair and P. Arcese (eds), Serengeti II: dynamics, management, and conservation of an ecosystem, pp. 421-447. University of Chicago Press, Chicago, IL, USA.
Watson, J.P. 1990. The taxonomic status of the slender mongoose, Galerella sanguinea (Ruppell, 1836) in southern Africa. Navorsinge van die Nasionale Museum, Bloemfontein 6: 351-492.
Watson, J.P. and Dippenaar, N.J. 1987. The species limits of Galerella sanguinea (Ruppell, 1836), G. pulverulenta (Wagner, 1839) and G. nigrata (Thomas, 1928) in southern Africa (Carnivora: Viverridae). Navorsinge van die Nasionale Museum, Bloemfontein 5: 351-492.
Webster, A.B., Pretorius, M.E., and Somers, M.J. 2021. The determinants of mesocarnivore activity patterns in highveld grassland and riparian habitats. African Journal of Wildlife Research, 51(1), 178-192.
Wozencraft, W.C. 1993. Order Carnivora. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World: A Taxonomic and Geographic Reference. Second Edition, pp. 279-344. Smithsonian Institution Press, Washington, DC, USA.
Wozencraft, W.C. 2005. Order Carnivora. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World: A Taxonomic and Geographic Reference. Third Edition, pp. 532-628. Johns Hopkins University Press, Baltimore.
Yalden, D.W., Largen, M.J., Kock, D. and Hillman, J.C. 1996. Catalogue of the mammals of Ethiopia and Eritrea. 7. Revised checklist, zoogeography and conservation. Tropical Zoology 9: 73-164.
Zemouche, J. 2018. Trophic ecology of rusty-spotted genet Genetta maculata and slender mongoose Herpestes sanguineus in Telperion Nature Reserve, with a focus on dietary segregation as a possible mechanism of coexistence. MSc dissertation. University of Witwatersrand, Johannesburg, South Africa.