Single-striped Grass Mouse
Lemniscomys rosalia

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Lemniscomys rosalia – (Thomas, 1904)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Lemniscomys – rosalia
Common Names: Single-striped Grass Mouse, Single-striped Lemniscomys, Zebra Mouse (English), Eenstreepmuis (Afrikaans)
Synonyms: No Synonyms
Taxonomic Note: Meester et al. (1986) listed four subspecies of which only one, Lemniscomys rosalia spinalis (Thomas 1916) occurs in the assessment region: KwaZulu-Natal, Mpumalanga, Limpopo, Gauteng and North West Provinces, as well as Eswatini and western Zimbabwe. Van der Straeten et al. (2008) noted that L. rosalia may represent a complex of species. However, as only one subspecies is listed for the assessment region, and due to the lack of either morphometric or genetic data, we treat it is treated as a single species for this assessment.
Red List Status: LC – Least Concern
Assessment Information
Assessors: Russo, I.M.1 & da Silva, J.M.2
Reviewer: Oosthuizen, M.K.3
Institutions:1Cardiff University,2South African National Biodiversity Institute,3University of Pretoria
Previous Assessors and Reviewers: Monadjem, A., Shoeman, C. & Child, M.F.
Previous Contributors: Cohen, L., Power, J., Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
The species is listed as Least Concern in view of its wide distribution within the assessment region (occupying an array of grassland habitats), its occurrence in many protected areas (including the Kruger National Park), its potential range expansion in the North West Province, its tolerance of habitat modification, and because there are no major threats that could cause population decline. Research has shown that its diet consists of the most abundant grass species occurring in a particular habitat, not necessarily the most nutritious, and thus food resources may not be a limiting factor. However, land managers should employ ecological stocking levels to avoid overgrazing and thus sustain tall grasses. Prudent fire management is recommended too.
Regional population effects: As suitable habitats are suspected to be largely connected along the northern border of South Africa with Botswana, and through Mozambique and Zimbabwe through the Great Limpopo Transfrontier Conservation Area, as well as between Mozambique and the northern part of the KwaZulu-Natal Province rescue effects may be possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Russo IM & da Silva JM. 2025. A conservation assessment of Lemniscomys rosalia. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
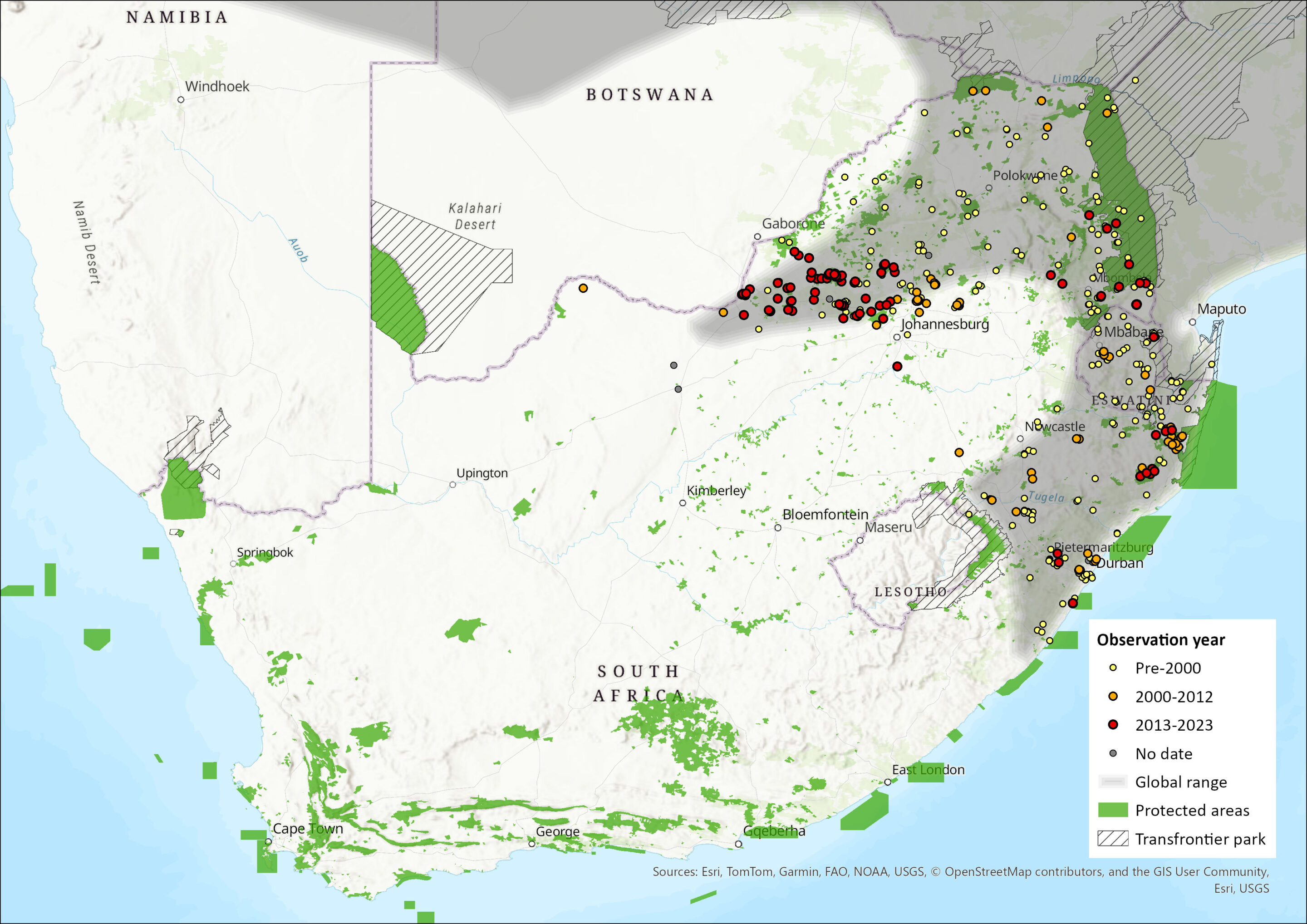
This species is widely distributed across the savannah regions of Namibia, South Africa, Eswatini, Zimbabwe, central and northern Botswana, Angola, Mozambique, Zambia, Malawi, Tanzania and southern Kenya (Monadjem et al. 2015). Within the assessment region, it occurs in the Savanna Biome in landscapes with tall, dense grass, including agricultural landscapes, distributed across the KwaZulu-Natal, Gauteng, Mpumalanga, North West and Limpopo Provinces.
Power (2014) presented evidence for a possible range expansion in the North West Province. the species has been recorded throughout the mesic savannah regions and more recently it was captured from a particular site (Khamab Kalahari Reserve) in the Molopo plains sandy bushveld vegetation type, which expands the range by 250 km and represents South Africa’s westernmost record (Figure 1). This may be a genuine range expansion, and this population might be linked to Botswana’s Kalahari population (Smithers 1971; Newbery 1996).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): 1200
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Single-striped Grass Mouse (Lemniscomys rosalia) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
No models have been done, and there has been no observed decline due to climate change.
Population Information
The Single-striped Grass Mouse is relatively common to very common across its range although the population appears to fluctuate. It was the most frequently encountered species in Hluhluwe-iMfolozi Park, the KwaZulu-Natal Province in the late 200o’s, representing 75% of all small mammal samples (Hagenah et al. 2009). In contrast, other sampling efforts in the same reserve in 2011 rendered the Single-striped Grass Mouse one of the least common species trapped (I. Russo pers. comm.). It was one of the least abundant murids captured at uMkuze Game Reserve, Kube Yini Game Reserve and Phinda Private Game Reserve, the KwaZulu-Natal Province (Delcros et al. 2015; Rautenbach et al. 2014). It occurs at high densities in suitable habitat. For example, in an ungrazed subtropical grassland near Matsapha, Eswatini, densities ranged from 4.4 ± 0.8 to 8.2 ± 1.0 individuals/ha from 1995 to 1998 (Monadjem & Perrin 2003). Although its abundance fluctuates widely, it is more numerous in dry winter months (Monadjem & Perrin 2003).
Population Information
Current population trend: Stable, inferred from stable savanna habitat.
Continuing decline in mature individuals: No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No. Can utilise transformed and agricultural landscapes.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While no population genetics studies have been conducted for this species, the species has been investigated in a phylogenetic context (e.g., Hánová et al. 2021). Unfortunately, these have not been able to inform whether there is substructure present within the assessment region. It is highly recommended that a more focused study be undertaken incorporating more comprehensive samples and fine scale molecular tools to look deeper into the population structure and diversity within the species. Despite this, it is possible that the species exists as a single metapopulation within the assessment region as suitable habitats are suspected to connect their landscape.
Assuming a single metapopulation and knowing the species is considered common (at least in some parts), density estimates would suggest there is an excess of at least 10,000 individuals within the assessment region. This would translate to an effective population size (Ne) greater than 500, assuming a conservative Ne/Nc conversion ratio of 0.1.
Habitats and ecology
This species tolerates a wide range of savannah habitats, including savannah woodland, dry grassland at the ecotone between vleis and woodland (Fuller & Perrin 2001), and scrub savannah of the Kalahari, but all these habitats are characterised by tall grassland areas (Kern 1981; Bowland & Perrin 1988; Monadjem 1997a; Skinner & Chimimba 2005; van der Straeten et al. 2008; Rautenbach et al. 2014). The species has been sampled in tall, closed grassland, low open rocky woodland, cultivated areas and open woodland in the Maguga Dam area, eSwatini (Avenant & Kuyler 2002). It is known to inhabit the tall grasses surrounding agricultural lands, especially fallow fields, and occupies suburban gardens in the KwaZulu-Natal Province (Taylor 1998; Skinner & Chimimba 2005). It prefers areas where there is a good grass, herbs or reeds cover and avoids areas where vegetation cover is short or absent (Skinner & Chimimba 2005). It vacates recently burnt areas but returns as soon as the grass recovers (Monadjem & Perrin 1997). It has not been recorded from montane grasslands, forests and riverine woodlands (Monadjem 1999; Delcros et al. 2015).
It is predominantly crepuscular but can be diurnal (Oosthuizen et al. 2024; Perrin 1981) and terrestrial, burrows are excavated where there is overhead cover of matted grass (Skinner & Chimimba 2005). In a study in the Kruger National Park, stomach content analysis revealed a diet comprised of 50% seeds, 48% herbaceous material and 2% insects (Watson 1987). Similarly, in Hluhluwe-iMfolozi Park, the KwaZulu-Natal Province, grass leaves and stems took up 65% of the diet while seeds (25%) and arthropods (3%) comprised the bulk of the remaining food items (Hagenah et al. 2009). In eSwatini, herbaceous material comprised 84.6 ± 9.2% of the diet, and seeds comprised 15.4 ± 9.2% (Monadjem 1997b). Hagenah et al. (2009) demonstrated that it prefers the most abundant grass species occurring in its habitat and not necessarily the highest quality grass species, and it has been suggested that the arthropod component of the diet may contribute significantly to meet the nutritional requirements of the species. In Hluhluwe,the species mostly fed on two low-quality bunch grass species, Sporobolus africanus (69%), and Eragrostis curvula (21%), whereas, in iMfolozi, it predominantly fed on the high-quality lawn grass species Urochloa mosambicensis (77%) and Themeda triandra, a lower-quality bunch grass species, comprised the remainder of its diet (13%). Grass species composition is therefore unlikely to be a limiting factor in the diet of this species. Its most important habitat requirement is probably dense ground cover of long grass as it breeds in surface grass nests (Monadjem 1997a, 2013; Taylor 1998).
Ecosystem and cultural services: Preyed on by Black-shouldered Kites (Elanus caeruleus) and Barn Owls (Tyto alba) (Monadjem 2013).
IUCN Habitats Classification Scheme
|
Habitat |
Season |
Suitability |
Major Importance? |
|
2.2. Savanna -> Savanna – Moist |
– |
Suitable |
– |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no major threats to this species that could cause widespread population decline. This species is present even when large herbivores are dominating as it feeds on the most abundant grass species (Hagenah et al. 2009). Livestock and wildlife ranching are not suspected to be a threat if tall grass structure is sustained. Similarly, although crop plantations and resultant pesticides were previously listed as a threat (Friedmann & Daly 2004), Single-striped Grass Mice will occur in agricultural landscapes if buffers of rank grass are conserved around field edges. Minor threats include overgrazing that reduces tall grass swards (Bowland & Perrin 1989), and overly frequent fire regimes (although it may return after grass has recovered, Monadjem & Perrin 1997). Such management practices will decrease habitat quality and may reduce the area of occupancy.
Current habitat trend: Savanna ecosystems are not threatened (Driver et al. 2012), and this species is tolerant of modified landscapes and agricultural areas.
Conservation
This species is present in many protected areas within the assessment region, including the Kruger National Park, uMkuze Game Reserve and Phinda Private Game Reserve. No interventions are necessary at present. The expansion of wildlife ranching may similarly be creating additional habitat for this species, if the grass is not overgrazed. No specific conservation interventions are necessary at present.
Recommendations for land managers and practitioners:
- Landowners and communities should be incentivised to stock livestock or wildlife at ecological carrying capacity to avoid overgrazing as this species needs a tall grass structure for breeding.
- Managers are encouraged to maintain an appropriate fire regime for the relevant habitat.
Research priorities:
- This species may comprise at least two species based on chromosomal data (Castiglia et al. 2002). Resolving the taxonomy within the southern African subregion may alter this assessment in the future.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside of protected areas on private or agricultural lands.
- Landowners and city planners can conserve corridors of tall grassland on their properties and/or in public spaces.
Bibliography
Avenant NL, Kuyler P. 2002. Small mammal diversity in the Maguga Dam inundation area, Swaziland. South African Journal of Wildlife Research 32: 101-108.
Bowland AE, Perrin MR. 1988. The effect of fire on the small mammal community in Hluhluwe Game Reserve. Mammalian Biology 53: 235–244.
Bowland, A.E. and Perrin, M.R. 1989. The effect of overgrazing on the small mammals in Umfolozi Game Reserve. Zeitschrift für Säugetierkunde 54: 251–260.
Castiglia, R., Fadda, C., Corti, M., Scanzani, A., Verheyen, W. and Capanna, E. 2002. Chromosomal evolution in the African Arvicanthine rats (Murinae, Rodentia): comparative cytogenetics of Lemniscomys (L. zebra, L. rosalia, L. striatus) and Arvicanthis dembeensis. Journal of Zoological Systematics and Evolutionary Research 40: 223-231.
Delcros, G., Taylor, P.J., and Schoeman, M.C. 2015. Ecological correlates of small mammal assemblage structure at different spatial scales in the savannah biome of South Africa. Mammalia 79(1): 1-14.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. Conservation Breeding Specialist Group (SSC/IUCN) and Endangered Wildlife Trust, Parkview, South Africa.
Fuller, J.A. and Perrin, M.R. 2001. Habitat assessment of small mammals in the Umvoti Vlei Conservancy, KwaZulu-Natal, South Africa. South African Journal of Wildlife Research 31: 1-12.
Hagenah N, Prins HH, Olff H. 2009. Effects of large herbivores on murid rodents in a South African savanna. Journal of Tropical Ecology 25: 483–492.
Kern NG. 1981. The influence of fire on populations of small mammals of the Kruger National Park. Koedoe 24: 125-157.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum, Pretoria, South Africa.
Monadjem A, Perrin M. 2003. Population fluctuations and community structure of small mammals in a Swaziland grassland over a three-year period. African Zoology 38: 127-137.
Monadjem A, Perrin MR. 1997. Population dynamics of Lemniscomys rosalia (Muridae: Rodentia) in a Swaziland grassland: effects of food and fire. South African Journal of Zoology 32: 129–135.
Monadjem A. 1997. Habitat preferences and biomasses of small mammals in Swaziland. African Journal of Ecology 35: 64-72.
Monadjem A. 1997. Stomach contents of 19 species of small mammals from Swaziland. South African Journal of Zoology 32: 23–26.
Monadjem A. 1999. Geographic distribution patterns of small mammals in Swaziland in relation to abiotic factors and human land-use activity. Biodiversity & Conservation 8: 223-237.
Monadjem A. 2013. Lemniscomys rosalia. In: D. Happold (ed.), Mammals of Africa, pp. 449–451. Bloomsbury Publishing, London, UK.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. De Gruyter, Berlin, Germany.
Newbery CH. 1996. Inventory – report: Small Mammals: Molopo Nature Reserve. Ecological Support Services, North West Parks Board, Mafikeng, South Africa.
Perrin MR. 1991. Notes on the activity patterns of 12 species of southern African rodents and a new design of activity monitor. South African Journal of Zoology 16: 248–258.
Oosthuizen, T., Pillay, N., Oosthuizen, M.K. 2024. Wild mice in an urbanized world: Effects of light at night under natural and laboratory conditions in the single-striped grass mouse (Lemniscomys rosalia), Chronobiology International, 41:3, 347-355,
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Rautenbach A, Dickerson T, Schoeman MC. 2014. Diversity of rodent and shrew assemblages in different vegetation types of the savannah biome in South Africa: no evidence for nested subsets or competition. African Journal of Ecology 52: 30-40.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor, P.J. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.