Serval
Leptailurus serval

2025 Red list status
Near threatened
Regional Population Trend
Declining
Change compared
to 2016
No Change
Overview
Leptailurus serval – (Schreber, 1776)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – FELIDAE – Leptailurus – serval
Common Names: Serval (English), Tierboskat (Afrikaans), Letlotse, Lelotswe, Tetekgwe (Sepedi), Phaha, Tlohi, Tholi, Tlodi, Qwako (Sesotho), Indloti, Lindloti (Swati), Ndloti (Tsonga), Tadi, Letlôtse (Tswana), Didingwe, Didinngwe, Dagaladzhie (Venda), Ihlosi, Inhlosi, Ingwenkala, Indlosi (Xhosa), Indlozi (Zulu)
Synonyms: Caracal serval (Schreber, 1776), Felis serval (Smithers 1978)
Taxonomic Note:
A molecular phylogenetic study revealed that the Serval is closely related to both the African Golden Cat (Caracal aurata) and the Caracal (Caracal caracal) (Johnson et al. 2006), diverging from a common ancestor approximately 5.4 million years ago, though distinct enough to be monospecific (O’Brien and Johnson 2007). Only one subspecies is recognised from within the assessment region, L. s. serval (Meester et al. 1986).
Red List Status: NT – Near Threatened A2c + C2a(i) (IUCN version 3.1)
Assessment Information
Assessors: Power, R.J.1 & da Silva, J.M.2
Reviewers: Nicholson, S.K.3 & Do Linh San, E.4
Contributor: Patel, T.3 & Roxburgh, L.3
Institutions:1North West Provincial Government, 2South African National Biodiversity Institute, 3Endangered Wildlife Trust, 4Sol Plaatjie University
Previous Assessors: Ramesh, T., Downs, C., Power, J., Laurence, S., Matthews, W. & Child, M.F.
Previous Contributors: Jones, A., Do Linh San, E., Avenant, N., Hoffmann, M., Hayward, M., Eksteen, J. & Pence, G.
Assessment Rationale
Servals are wetland specialists that historically became regionally extinct in the Eastern Cape Province and most of the Western Cape Province by the 1980s. However, they were successfully reintroduced in several Eastern Cape protected areas in the early 2000s and, although rare, are now regularly seen throughout the province. Natural range expansion is also evident in the Western Cape. Additionally, several range expansions have been documented in the Free State and the eastern Northern Cape, as well as potentially in North West and Lesotho. This may indicate natural recolonisation, as there is no evidence of reintroductions for the North West Province, along man-made corridors (for example, dams and weirs that promote the growth of reeds and other dense vegetation that support their main prey species). Corroborating this, Servals appear to be adaptable to rural agricultural and/or industrial areas where appropriate wetland habitat is conserved or created, and thus areas where prey species thrive. Densities have been found to be similar between farms under various management intensities in the Drakensberg Midlands of KwaZulu-Natal, where density estimates range from 6.2 ± 1.9 to 7.7 ± 1.6 individuals / 100 km2 using both maximum likelihood and Bayesian methods. However, it remains to be seen whether Serval subpopulations in transformed or degraded landscapes are viable in the long-term, as recent research suggests that the spatial configuration of natural to non-natural wetlands may be important in determining resilience.
Continuing loss and degradation of natural wetlands and associated reed-banks is of major concern. The 1990–2013/14 (24-year period) South African National Land-Cover change report found a 32.8% decline in natural wetlands on a national scale, a combination of both anthropogenic degradation and drier conditions. This is almost exactly three generations for Serval (c. 25 years). This decline in wetlands is corroborated by general natural habitat loss on a provincial scale. We infer continuing decline from wetland loss. Further, if we infer that natural wetland loss is proportional to population decline for Serval, a Vulnerable A2c listing is appropriate. However, the relationship between natural wetland loss and population trends is unknown, the extent to which natural wetland loss is mitigated by artificial wetland establishment is uncertain, and there is no direct evidence for population decline. Thus, we list as Near Threatened A2c. Long-term monitoring sites should be established to detect any local or regional declines in various subpopulations related to land-use change.
Area of occupancy (AOO) was calculated by buffering wetlands occurring within the extent of occurrence (EOO) by 500 m and was estimated to range between 18,789 km2 (large, natural wetland clusters) and 209,738 km2 (including small waterbodies and artificial wetlands). This yielded an estimated mature population size that most likely ranges between 4,509 and 13,654 individuals (using a 50–70% mature population structure). An ongoing decline in mature individuals is suspected from either uses related directly to traditional medicine or cultural regalia (which may represent an emerging threat within the assessment region), or indirectly as collateral in snares laid out for other species or general predator control by farmers and landowners. Given that there is an inferred continuing decline from wetland loss, and a suspected continuing decline in mature individuals from incidental snaring, persecution or illegal trade, Servals would qualify for Vulnerable C2a(i). However, although it is likely that no subpopulation is larger than 1,000 mature individuals, it is uncertain how to define subpopulations in this species given their significant dispersal capacity. Thus, we list as Near Threatened C2a(i). We note that further research is needed to more accurately calculate population size and AOO, as well as the net effects of ongoing natural wetland loss versus use of modified habitats. Additionally, the emerging threat of being hunted for cultural regalia, should be monitored and quantified. This species should be reassessed as relevant data become available.
Key interventions include the conservation and restoration of wetlands and riparian/grass vegetation buffer strips around wetlands. We encourage managers and landowners to protect natural or artificial wetlands and waterside vegetation and maintain sufficient ground cover/vegetation refugia in both farmlands and ranchlands to sustain key resource areas for Servals within an otherwise unsuitable matrix. Although Servals respond well to ex situ breeding, reintroduction efforts show mixed success and should be monitored and evaluated more thoroughly to improve best practice. Awareness and education campaigns to end intentional or indirect persecution of Serval should also be continued.
Regional population effects: Dispersal across regions via drainage systems is suspected (sensu Hermann et al. 2008). We are unsure whether this dispersal represents significant immigration into the region. It is suspected that South Africa might be a source population as there is possible movement of individuals between South Africa and neighbouring countries. We can expect movements across the Mozambique–South Africa border at several points. Generally other countries tend to be marginal areas of distribution and no significant rescue effects are suspected.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Power RJ & da Silva JM. 2025. A conservation assessment of Leptailurus serval. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
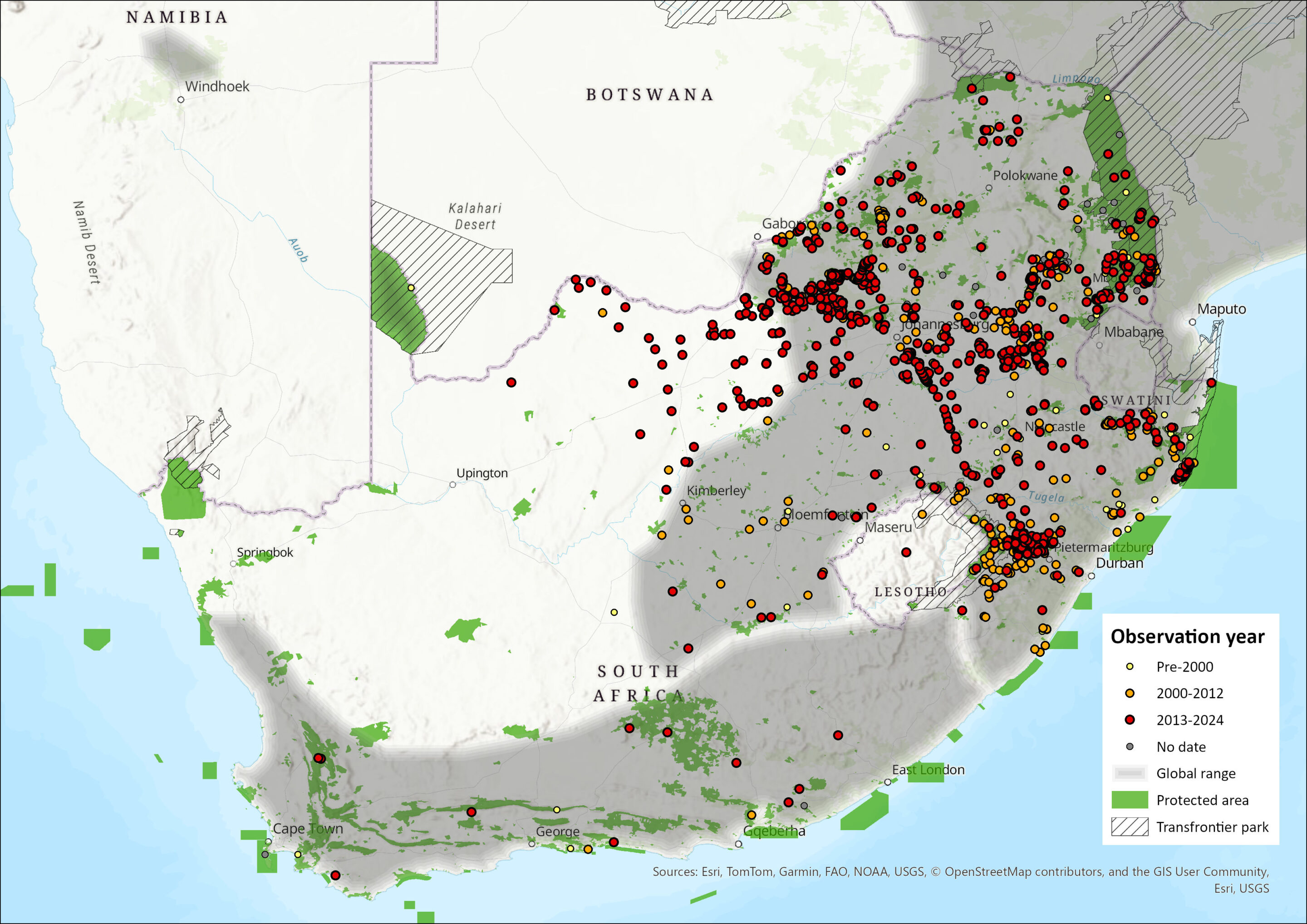
The Serval occurs widely through sub-Saharan Africa, with the exception of tropical rainforests and deserts (Nowell & Jackson 1996). They occur across most of the northern and eastern parts of South Africa (Skinner & Chimimba 2005).
Within the assessment region, it still occurs throughout the Limpopo, Mpumalanga, Gauteng, KwaZulu-Natal and North West increasingly so in the Eastern Cape, and even the Western Cape (Figure 1). In KwaZulu-Natal it is common within wetland habitats of the Drakensberg Midlands, (Ramesh & Downs 2013) though rare in the lowland wetlands of that province (Ramesh et al. 2016). It seems to have recolonised in the Free State, North West and Northern Cape (Hermann et al. 2008; Thorn et al. 2011; Power 2014).
In the latter half of the 20th century, the Serval was considered extinct, or near extinct, in many areas of its historical range (Von Richter 1972; Stuart 1985; Smithers 1978; 1986; Skinner & Chimimba 2005; Hermann et al. 2008; Thiel 2015), primarily due to accidental persecution as a damage-causing animal and competitive exclusion by other carnivore species (Stuart 1985). For example, it is thought the Serval occurred historically in the eastern Free State (Hunter & Bowland 2013) and as far west as the Cape Peninsula (Skead 2011). Skead (2007) reported that Servals historically occurred along the entire coastal and sub-coastal belt of the Eastern Cape, and that they were nearly extinct in that province in the 1980s (Stuart 1981; 1985; Skead 2007). In the early 2000s, Servals were reintroduced into Shamwari and Kwandwe Private Game Reserves in the Eastern Cape (Hayward et al. 2007). Although they are rare, they were present in the following properties in the Eastern Cape (D. Peinke pers. comm. 2015): Amakhala, Lalibella, Hopewell, Kariega Park, Samara, and Mount Camdeboo. They have also recently been recorded from the Western Cape (Figure 1). With no known reintroductions having taken place, these observations either represent greater sampling effort that revealed an already existing low-density subpopulation or a range expansion from the Eastern Cape or undetected sources in the southern Northern Cape. Such hypotheses need to be researched. It is not impossible too that undisclosed introductions took place from captive facilities.
In an older assessment (Friedmann & Daly 2004), it was thought that Servals were restricted to the wetter parts of the country, including the provinces of KwaZulu-Natal, Mpumalanga, Limpopo and the northeastern parts of the North West. However, a number of older records from central South Africa were not included in the species’ distribution range (Friedmann & Daly 2004; Skinner & Chimimba 2005), as they were considered to be very rare in the drier parts of the country. These records include four pre-1980 specimens from the eastern Free State (Lynch 1983), four sight records from the northern parts of the Eastern Cape (Lynch 1989), as well as a report that this species occurred within what is now the western part of the North West (Stuart 1981), However, more recent records from the Free State provincial conservation authorities, suggest that Servals have recolonised and bred successfully in the western Free State and the eastern boundary of the Northern Cape (Hermann et al. 2008; Figure 1), and their distribution spans both arid and mesic parts of the North West Province (Thorn et al. 2011; Power et al. 2019). Serval sightings have been taken in vleis or riverine vegetation and suggest that Servals use the drainage lines as corridors for movement through the drylands of the Free State for instance (Hermann et al. 2008). As such, this was likely to be a genuine range expansion as there was no evidence of any ad hoc reintroductions. This expansion was most likely facilitated by the increase in man-made habitats, such as impoundments, weirs and dams, that have enhanced vlei-type habitats favourable to Servals (such as reeds and other dense vegetation that support their main prey species) (Hermann et al. 2008). Reduced threat levels from larger predators or domestic dogs and the adoption of holistic control methods for damage-causing animals or increased landowner tolerance for this species (Hermann et al. 2008) may also have facilitated range expansion. If true, then similar to Greater Cane Rats (Thryonomys swinderianus) (van der Merwe & Avenant 2004), Servals may continue to expand their range into other areas of South Africa. Encouragingly, most specimens were collected in the vicinity of perennial and non-perennial rivers or dams, which are the preferred habitats of their main prey species, in landscapes that would otherwise be considered unsuitable for Servals (Hermann et al. 2008). Servals also occur in Gauteng, even 20 km from Johannesburg (Kuhn 2011).
Similarly, within the North West Province, they have expanded their range by 37% since 2000 (Thorn et al. 2011), and by 2014, westwards by at least double since 1983 (Power 2014). The AOO of Servals in 2010, increased by 110 % since 2000 (Thorn et al. 2011). In the protected areas, they were most common at Vaalkop Dam Nature Reserve, and, interestingly, the species did not even appear on any earlier checklists at Pilanesberg or Borakalalo (for example, Newbery 1995), although they were present in these areas (Power 2014). Reintroductions to these parks, as well as Madikwe Game Reserve and Kgaswane Mountain Reserve, took place in the 1980s–1990s (van Aarde & Skinner 1986), and may have been to augment these protected areas owing to regional extirpation. They are currently still present on Madikwe (Smith et al. 2023), and Pilanesberg (Figure 1).
It was hypothesised that the above average rainfall years that have been experienced during the 2000s had led to favourable habitat throughout (for example, dense grass, rejuvenated marshes) (Power 2014). This subsequent increase in both EOO and AOO, probably due to the creation of artificial waterbodies and a subsequent increase in prey, may have indicated continuing recovery from historical persecution and habitat loss (Thorn et al. 2011). At present the distribution range has not changed since the previous assessment, though it seems to have expanded deeper into the Kalahari with camera trap records obtained in the !Khamab Kalahari Reserve, which may still be either a relict or low-density population, perhaps of a sporadic nature. Past evidence in this area has been borne from questionnaires (Lloyd & Millar 1984; Power 2014), and sign presence at best (Thorn et al. 2011), but no proper confirmation until recently (Photo below; camera trap at !Khamab). The occurrence here may be in tandem with range extensions to the north in Botswana’s Kalahari in the Ghanzi area (Van der Weyde et al. 2021), and Khutse Game Reserve (Finerty et al. 2019), and these were all ascribed to range expansion verses overlooked occurrences (Finerty et al. 2019; Van der Weyde et al. 2021).
Camera trap image of a serval taken in 2022 on the !Khamab Kalahari Reserve (photograph courtesy of Panthera)
Although no previous records exist, an individual was seen at the Rooikop waterhole in the dry bed of the Nossob River in the Kgalagadi Transfrontier Park (T. Jackson pers. obs. 1990). They do not occur in the arid parts of the Nama or Succulent Karoo Biomes (Skinner & Chimimba 2005). Serval are present in Mpumalanga, including the Highveld of Verloren Vallei (Loock & Swanepoel 2019) and Telperion Nature Reserves (Webster et al. 2021), while also in the Lowveld (Wentzel et al. 2021). The species also occurs throughout Eswatini (Monadjem 1998; Skinner & Chimimba 2005), and, although listed as a species predicted to occur in Lesotho lowlands, was not recorded by Lynch (1994). However, they have subsequently been caught on camera traps in the Lesotho Highlands (A. Jones unpubl. data).
At larger spatial scales, mesocarnivores like Serval may be considered generalists (Ray et al. 2011), but at finer scales, they are habitat specialists in fragmented landscapes (Ramesh et al. 2015a). They may be locally restricted to smaller areas within their broad distribution range, and conservation planners should integrate these scale considerations into regional management plans (Ramesh et al. 2015b). To incorporate the fine-scale habitat selection and wetland spatial configuration into the estimate of AOO, the wetland cluster layer from Driver et al. (2012) was used to calculate a minimum AOO for Serval across its range. The wetland cluster layer represents clusters of at least three wetlands (within 1 km of each other) embedded within relatively natural areas (50% natural area or more – compared to artificial wetlands). Maximum Serval home range size was used (60 km2; Ramesh et al. 2015a) to buffer current (post-2000) distribution points using the equation:
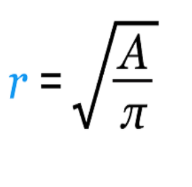 where r is the radius distance and A is the home range area, to generate a buffer radius of 4.4 km. This buffer layer was then overlaid onto the wetland clusters (buffered by 500 m) and any clusters containing Serval home ranges were summed. This yielded 10,426–11,144 km2 of wetland cluster areas overlapping with current Serval records. This is likely to be a significant underestimate in minimum AOO for viable Serval subpopulations as there are false negatives in the Serval distribution data and many occurrence records fall outside of the cluster areas, which indicates Servals can make use of smaller wetland areas. The total EOO is 1,034,067 km2, compared with 976,233 km2 in the 2016 assessment. This does not necessarily indicate a range expansion but may be the result of increased camera trapping and submission of records to iNaturalist or GBIF. The increase in EOO is largely related to a recent sighting, with photographic evidence, of Serval in the Cedarberg. We assumed the same AOO as in 2016, as it is not clear, based on this isolated record in the Cedarberg, whether the species established here, or whether it is a wandering outlier. Within the EOO, there was an estimated 18,789 km2 wetland cluster area in total (not clipped to current Serval sightings). Furthermore, summing all buffered wetlands (small waterbodies and artificial waterbodies, not just wetland clusters) within the EOO yielded a total AOO of 209,738 km2 within the Serval’s range. These AOO estimates represent a potentially useful baseline of core habitat occupancy for this species. Further research is required to refine these. For example, Ramesh et al. (2015a) use a 20 m buffer from the boundary line of each wetland to define core areas.
where r is the radius distance and A is the home range area, to generate a buffer radius of 4.4 km. This buffer layer was then overlaid onto the wetland clusters (buffered by 500 m) and any clusters containing Serval home ranges were summed. This yielded 10,426–11,144 km2 of wetland cluster areas overlapping with current Serval records. This is likely to be a significant underestimate in minimum AOO for viable Serval subpopulations as there are false negatives in the Serval distribution data and many occurrence records fall outside of the cluster areas, which indicates Servals can make use of smaller wetland areas. The total EOO is 1,034,067 km2, compared with 976,233 km2 in the 2016 assessment. This does not necessarily indicate a range expansion but may be the result of increased camera trapping and submission of records to iNaturalist or GBIF. The increase in EOO is largely related to a recent sighting, with photographic evidence, of Serval in the Cedarberg. We assumed the same AOO as in 2016, as it is not clear, based on this isolated record in the Cedarberg, whether the species established here, or whether it is a wandering outlier. Within the EOO, there was an estimated 18,789 km2 wetland cluster area in total (not clipped to current Serval sightings). Furthermore, summing all buffered wetlands (small waterbodies and artificial waterbodies, not just wetland clusters) within the EOO yielded a total AOO of 209,738 km2 within the Serval’s range. These AOO estimates represent a potentially useful baseline of core habitat occupancy for this species. Further research is required to refine these. For example, Ramesh et al. (2015a) use a 20 m buffer from the boundary line of each wetland to define core areas.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map .
Figure 1. Distribution records for Serval (Leptailurus serval) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified
Biogeographic Realms
Biogeographic Realm: Afrotropical, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Algeria | Possibly Extinct | Native | – | – |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Djibouti | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Liberia | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Morocco | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Tunisia | Extant | Reintroduced | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Given the documented advance of the species distribution range into arid parts of South Africa (Hermann et al. 2008; Thorn et al. 2011; Power et al. 2019), these gains could be reversed if one considers the impact of climate change, particularly the drying out of the landscape (Foden & Young 2016).
Engelbrecht et al. (2024) report that the southern African region will generally become drier, with reductions in soil moisture and increased frequency of multi-year droughts. Higher temperatures will also increase evaporation rates, reducing wetland water levels. Despite this, an increase in intense rainfall events is projected, leading to flash floods and erosion of wetland soils and an alteration in sediment dynamics. This is likely to lead to the collapse of hydrological systems supporting wetlands in some areas and reduce habitat suitability for wetland-dependent species. An increase in fire-danger days under all climate scenarios can also lead to increased burning of riparian wetlands and peatlands, further degrading and reducing wetland habitat in the region. Thus, we can infer an ongoing decline in habitat suitability and availability for Serval in the region.
Population information
The Serval is commonly recorded from most major national parks and reserves within the assessment region (Von Richter 1972; Skinner & Chimimba 2005; Ramesh et al. 2016). From elsewhere in Africa, the minimum density of Servals in optimal habitat in Ngorongoro Crater was 0.42 animal / km² and 0.1 animal / km² in Luambe National Park in Zambia (Thiel 2015). Their status outside protected areas is relatively uncertain, but they are inconspicuous and may be common in suitable habitat as they are tolerant of farming practices, provided there is cover and food available (Bowland 1990; Hunter & Bowland 2013; Ramesh & Downs 2013; Thiel 2015).
In 2010, outside protected areas in the North West, based on sign surveys, a serval density of 0.01 (±0.01)/100 km² was determined (Thorn et al. 2010), which may be reasonable to suspect, but the method has been superseded since by more superior methods to assess density.
Bowland (1990) estimated 8 individuals / 100 km² in the Drakensberg foothills, while more recently in the same area, a similar density of 6.2 ± 1.9 to 7.7 ± 1.6 individuals / 100 km² was recorded on farmland (Ramesh & Downs 2013), which perhaps indicates a stable subpopulation over the past 20 years. Elsewhere, to the north of the province, serval density was recorded as 2.49 ± 0.81 (1.24-4.63) Servals/100 km² (Taylor 2020).
Additionally, recent work at the Sasol refinery site in Secunda, a site with very disturbed artificial wetlands, but affording a good source of rodents, shelter, and protection (from the industry itself as well as persecution, and other predators), has shown the largest density recorded yet of 76.2 – 101.2 servals / 100 km² (Loock et al. 2018) This is an unusually high density for a medium-sized felid. There are also indications that the Sasol coal mining site, Syferfontien, (approx. 30 km away from the Secunda site) also has a healthy Serval subpopulation (W. Matthews unpubl. data). This very high density may not be the norm, but does indicate that, where Servals are protected, they can thrive.
However, this high density is not the norm for many game reserves, farms and agricultural areas. The occurrence of Servals in farmlands is probably enhanced by the relatively low density of large or competing carnivores such as Caracal and African Wildcat (Felis lybica) (Ramesh & Downs 2013; Loock et al. 2018). Interestingly, a protected area complex exhibited a lower density than human modified landscapes, whether industrially so, or agriculturally. Finally, the newer records in the previous two decades from the western Free State, southern North West and eastern Northern Cape represent the most western records of Servals in recent years and may represent recolonisation of grassland habitats over the past 20 years in central South Africa (Hermann et al. 2008). Thus, the Serval population may have remained stable over the past decade (regionally) and seems to have increased along the edge of its range in western North West. However, it is difficult to infer a population increase as Servals could simply have been undetected in these areas or have always existed at low density in sub-optimal habitat. Additionally, the recent surge in the use of camera traps in ecological studies may partially explain the increasing number of Serval observations.
Using the minimum and maximum density estimates from the KwaZulu-Natal Midlands (4.3–9.3 individuals / 100 km²) and the AOO estimates, potential population size ranges from 448–1,747 individuals (using wetland clusters) and 9,018–19,506 individuals (using all buffered wetlands, to account for false negatives in the dataset). This corresponds to a range of 240–1,223 mature individuals using a 50–70% mature population structure for the wetland cluster AOO and 4,509–13,654 for all wetlands within the range.
This species can subsist away from wetlands, as long as adequate grass cover and/or ecotonal tree cover is available, and artificial wetlands in dam outflows are utilised. These habitats are being increasingly created on low density eco-estates, wildlife ranches, mine buffer zones and agricultural systems. However, where subdivision of farms is taking place, due to inheritance and deeds transfers, habitat quality is often reduced through alien plant invasion and increases in traffic. Nevertheless, the Serval remains a wetland (and moist grassland) specialist and these habitats are the most threatened ecosystem in South Africa with an ongoing loss in pristine and functional wetland areas. The extent to which this is compensated for by increasing artificial wetlands remains to be seen (see Hermann et al. 2008). Although Servals make use of such areas, these may not represent resilient and viable long-term habitats, especially as they are vulnerable to changes in management or land-use. Thus, ongoing wetland habitat loss and degradation, combined with a continuing loss of mature individuals to persecution, trade, road collisions or failed reintroductions, indicates that a decline in numbers or loss of core subpopulations cannot be ruled out. It must be borne in mind that some of the largest populations are in areas where there are minimal wetlands, i.e. Kruger National Park (Pienaar et al; 1996), and there are large tracts of waterless areas where the species also occurs (see Thorn et al. 2011).
Manqele (2017) has estimated regional population size of the serval in KwaZulu-Natal and made forecasts of the population trajectory based on known densities (Ramesh & Downs 2013), and various population parameters.
Within KwaZulu-Natal, the population appears stable, especially as densities are comparable across a range of farming intensities (Ramesh & Downs 2013), as long as there are sufficient wetlands with natural vegetation evenly spaced within the landscape (Ramesh et al. 2015a). However, in the Ithala Game Reserve, serval density decreased from 9.66 (± 2.1) servals/100 km2 recorded in 2014 to as low as 1.42 (± 0.6) in 2018, which tracked the simultaneous decline in the local Leopard population. This suggested that both these carnivore species were facing some form of influence that threatens their population numbers (Taylor 2020). This suggests there are areas where local declines have taken place, and concerningly in protected areas.
Generation length has been estimated as 8.3 years (Pacifici et al. 2013), which makes the three-generation window 24.8 years.
Population Information
Current population trend: Possibly declining due to net loss of wetland habitat, and deaths from persecution (snaring and roadkill) in some areas of their range.
Continuing decline in mature individuals: Yes, due to direct or indirect persecution.
Number of mature individuals in population: 4,509-13,654
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No. They have a broad habitat tolerance and can exist in agricultural landscapes. Although these habitats are fragmented, Servals are able to connect with other subpopulations through corridors such as drainage lines to ameliorate fragmentation effects (Hermann et al. 2008).
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
While L. serval has been studied in a phylogenetic context (Johnson et al. 2006), no population genetic investigation has been conducted on the species to provide insight into its genetic structure and health.
Based on the density estimates quantified for certain areas, L. serval is thought to be fairly abundant and capable of dispersing to across the assessment region. Consequently, there are no anticipated barriers to gene flow; therefore, the species is likely to exist as a single metapopulation. No subpopulations are thought to have gone extinct in the past and hence, the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF’s) complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (all populations remaining).
While no census (Nc) or effective population size (Ne) data exists for the species, density estimates in some areas suggest that across the assessment region there is well over 10,000 individuals. Even though the 10,000 value is not an actual census count, the quantity can still be used as a proxy for quantifying the GBF’s headline genetic indicator – proportion of populations with an Ne > 500. Based on the assumption that the species is likely to occur as a single metapopulation, the Ne of the species is ~1000-3000 individuals, based on a Ne/Nc conversion ratio of 0.1-0.3. Having the 1 population above Ne 500, translates to an indicator score of 1.0 (1/1 population with Ne > 500).
While the species can subsist away from wetlands, it is considered a wetland specialist. Given the current state of wetlands across the assessment region and the perceived declines in mature individuals associated with this, there is a chance animals have become isolated. Consequently, it is highly recommended a population genetic study be undertaken to get a clear picture of the current levels of genetic structure and diversity within the species.
Habitats and ecology
In sub-Saharan Africa, Servals are mostly found in and around marshland, well-watered savannah and long-grass environments, and are particularly associated with reed-beds and other riparian vegetation types (Smithers 1978; Skinner & Chimimba 2005; Thiel 2015). Servals can penetrate dense forest along waterways and through grassy patches and are able to tolerate agricultural areas to some extent provided cover is available (Hunter & Bowland 2013; Ramesh & Downs 2013; Ramesh et al. 2015b). However, they have quite specific habitat requirements, so may be locally restricted to smaller areas within their broad distribution range (Sunquist & Sunquist 2002; Ramesh et al. 2015b). The species can of late be said to occupy the Eastern Kalahari Bioregion (Power et al. 2019), and suggests it may not be overly reliant on mesic affinities.
Key vegetation types are thus wetlands, grasslands (with a preference for long, rank grass), and indigenous vegetation that can provide cover and allow dispersal. These can be natural or man-made, although more research needs to be done on the ideal configuration of the latter wetlands (Ramesh et al. 2015a). So far, the status and response of Servals in mosaic agricultural landscapes is uncertain, particularly their movement patterns and land use in response to habitat fragmentation. For example, they exhibit lower occupancy rates in cropland but increased occupancy with higher human abundance (Ramesh & Downs 2015b). Indeed, the species could be described as semi-synanthropic due to the strong positive association with human activities. However, they are likely to be sensitive to fragmentation due to habitat specialisation, mainly preference for wetlands (Ramesh et al. 2015b), and its associated rodents (Ramesh & Downs 2015a). In an ongoing collaring study (Ramesh & Downs 2013), it was found that native wetland with a higher percentage of less disturbed, large-sized patches (core area) positively explained landscape use by Servals where nearly 80% of the GPS fixes concentrated only on remnant wetland habitats. Servals also avoided or used less cropland (Ramesh et al. 2015a). This is mainly due to the conversion of wetlands and their associated habitat for farming, residential development and commercial purposes that have led to many small, disconnected patches of human-altered landscapes (Ramesh & Downs 2013). Interestingly, males utilised forest patches more readily than females, while they both utilised wetland habitats (Ramesh et al. 2015b).
In the North West Province, serval occurs across all bioregions of the North West Province, including the Grassland Bioregions, Central Bushveld Bioregion, and interestingly even the Eastern Kalahari Bioregion (Power et al. 2019). Thorn et al. (2011) has reiterated that the species occurs widely in semi-arid and mesic areas in the North West Province, and they are adept at utilising open woodlands and scrub provided, there is a dense grass cover, even distant from permanent water.
Servals specialise on small mammals, in particular rodents, with birds being of secondary importance (Hunter & Bowland 2013). Small mammals (especially Vlei Rats Otomys species and Striped Mice Rhabdomys pumilio), birds, reptiles, fish, and rarely invertebrates, are also preyed upon (Smithers 1978; Bowland 1990; Skinner & Chimimba 2005; Ramesh & Downs 2015a). They are predominantly nocturnal (Skinner & Chimimba 2005; Ramesh & Downs 2013), but increasing evidence points to crepuscular behaviour (S. Laurence unpubl. Data; Ramesh & Downs 2013). Interestingly, in an intensive farming area of KwaZulu-Natal, they were strictly nocturnal (Ramesh & Downs 2013). On the South African Highveld of Mpumalanga, servals were mostly crepuscular, and it was found that they have a narrow temperature range in which they were found to be active, between 5°C and 15°C (Webster et al. 2021).
Home-ranges of rehabilitated orphaned and hand-raised Servals had small home-ranges (2-2.9 km² (Van Aarde 1986; Perrin 2002). Radio-tracked individuals on the Magaliesberg plateau of the North West’s Kgaswane Mountain Reserve had home range sizes between 2–2.7 km² (van Aarde & Skinner 1986).
In KwaZulu-Natal, home-ranges have been determined for a number of areas (Bowland 1990; Perrin 2002; Ramesh et al. 2015a). In Mount Currie Nature Reserve, a reintroduced orphan Serval had a home range of 2.66 km² (MCP) (Perrin 2002). In the Drakensberg foothills, near Kamberg, home-ranges varied from 2.2 –31.5 (MCP) km² (Bowland 1990). While in 2013, male home-ranges averaged 38.07 km2 (n = 11, 95% Kernels), and females averaged 6.22 km² (n = 5, 95% Kernels) (Ramesh et al. 2015a). As expected with felids, male ranges were much bigger than females, and there was high intrasexual overlap, and minimal intersexual overlap (Ramesh et al. 2015a), typical of felids. In Secunda, Mpumalanga, where a relatively high population density is documented (Loock et al. 2018), home ranges are estimated at 1–2 km² (W. Matthews, unpubl. data).
Ecosystem and cultural services: As a predator of rodents and birds (Smithers 1978; Bowland 1990), Servals may play a functional role in agricultural landscapes in controlling the numbers of pest species (Thiel 2015; Ramesh & Downs 2015b). It is alleged that the Forestry Department, Mpumalanga, reintroduced Serval to control rodents that feed on the newly sprouting Pinus sp. seedlings but no documentary proof has been published for its effectiveness. However, camera trap data do provide evidence of Servals foraging in plantations (S. Laurence unpubl. data), these habitats were found to not be selected (Ramesh et al. 2015b). Servals are culturally important to the Zulu culture, in as far as their skins and body parts are concerned (Manqele et al. 2018).
The Serval can be used as an umbrella species for savannah biotopes, and as an indicator for the highly endangered humid savannah biotope (Thiel 2015).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 4.6. Grassland -> Grassland – Subtropical/Tropical Seasonally Wet/Flooded | – | Suitable | – |
| 4.7. Grassland -> Grassland – Subtropical/Tropical High Altitude | – | Suitable | – |
| 5.1. Wetlands (inland) -> Wetlands (inland) – Permanent Rivers/Streams/Creeks (includes waterfalls) | – | Suitable | – |
| 5.3. Wetlands (inland) -> Wetlands (inland) – Shrub Dominated Wetlands | – | Suitable | – |
Life History
Generation Length: 8.3 years (Pacifici et al. 2013)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Servals are utilised in the traditional medicine trade and skins used for traditional regalia (Whiting et al 2011; Manqele et al. 2018). Trade in West Africa and South Africa appears to be primarily for ceremonial or medicinal purposes, but also for bushmeat. The impacts of this are unknown, but probably low regionally, such as in North West only one skin has been confiscated in 14 years (R.J. Power unpubl. data). There are also reports of local communities capturing Serval for traditional medicine or ritual use in the Midlands, KwaZulu-Natal (T. Ramesh unpubl. Data, Manqele et al 2017), probably as a substitute for Leopard skin. They are sometimes mistakenly persecuted as a damage-causing animal (Hermann et al. 2008; Power 2014), although the overall level of persecution may be decreasing owing to increased knowledge and awareness of the species. An increased demand for this species as a hunting trophy by local and overseas hunters is suspected, although the CITES trade database documents a stable trend, at an average of 46 ± 9 Serval trophies exported between 2002 and 2012.
There has been a notable increase in the demand to hunt Servals from at least 2015, which was round about the time of the last assessment (see Ramesh et al. 2016). From 2010 to 2019, there have been 589 offtakes of servals as hunted and reported by PHs, and the most important provinces in this are Mpumalanga, Limpopo then KwaZulu-Natal. In the North West, 16 have been removed over this time period, which range from 0-5 annually.
Since 2020, there have been 10 applications to hunt serval, of which 40% were rejected owing to various policy related procedures, i.e. regional quotas, seasonal restrictions (North West Directorate of Biodiversity Management). Prior to 2020, there was a tendency to want to hunt the species where they marginally occur in the Kalahari, and these hunts were always suspected to be laundered captive bred animals and were summarily rejected. However, given range occurrences of late (Figure 1), the species may indeed become more established, but until then, trophy hunting is regionally excluded from that area for that species.
The species is increasingly becoming popular to see on ecotourism reserves, hence the need to reintroduce them onto Eastern Cape (Hayward et al. 2007) and North West reserves (Power 2014), and they are frequently seen, and photographed in the Pilanesberg.
Although trophy hunting may cause local population declines, it is not expected to impact the overall population negatively given the species propensity for recolonisation and reintroduction. Illegal hunting is, however, suspected to be high. Additionally, there is an international market for hybridised Servals in the pet trade where they are marketed as the “Savannah Cat” in the United States, which is a likely market for captive-bred Servals, if they are not sold locally for reintroduction efforts. The demand for the Savannah Cat does not seem to be prevalent in South Africa.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Used for cultural regalia or traditional medicine. | Yes | Pet trade and ex situ conservation breeding. |
National Commercial Value: Yes
International Commercial Value: (Not specified)
| End Use | Subsistence | National | International | Other (please specify) |
| 3. Medicine – human & veterinary | – | – | – | – |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Unknown
Threats
Habitat loss/degradation
The major threat to Serval is loss and degradation of wetland and associated grassland. Wetlands generally harbour high rodent densities compared with other habitat types, and form the core areas of Serval home ranges (Bowland 1990; Ramesh et al. 2015a; Thiel 2015). Of secondary importance is the degradation and loss of grasslands through several factors including annual burning followed by over-grazing by livestock, intensive wildlife/livestock farming leading to reduced abundance of small mammals and cover, and transformation with changing land-use, increased anthropogenic structures (roads, buildings) and invasive alien plants. Thus, while Ramesh and Downs (2013) found Serval density similar across a range of farmland management intensities, the continued loss of core wetland area may ultimately threaten a viable Serval population, especially if there is resistance to movement through hostile habitat such as open cropland (Ramesh et al. 2015a). Within agricultural landscapes, Servals selected areas with minimal disturbance and a high proportion of natural habitat (Ramesh et al. 2015a), thus highlighting that only landscapes with a mosaic of modified to natural habitats will be suitable and emphasises the importance of undisturbed habitats. Similarly, Serval occupancy decreased with increased pesticide use on farmlands (Ramesh & Downs 2015b).
Road mortalities
Other threats within the assessment region include road mortalities (Power 2014), where in the North West Province, 22 road mortalities were reported between 2014 and 2023, with an increase in the last 4 years, which was related to dedicated monitoring on this (North West Directorate of Biodiversity Management records, and for the previous year, NWPG 2024).
In KwaZulu-Natal, Serval roadkill has been reported (Ramesh et al. 2015a; Manqele et al. 2018), and it is estimated that 5.2 Serval are killed/100 km/year on the N4 which traverses the Free State, from Gauteng to KwaZulu-Natal (Williams et al. 2019). Roadkills are so severe that it even biases their occurrence records, as can be seen in the linear occurrences along the N3 in the Free State (see Figure 1).
Serval killed on the R509 east of Boons, North West Province, just west of the Gauteng border, May 2022 (Photo John Power)
Incidental killing
In the North West, incidental killing of Serval has been unofficially reported when targeting other damage causing predators (Power 2014). Although Servals are non-target animals (and indeed may even be beneficial to crop farmers due to their predilection for rodents), many die in traps set out for carnivores such as Black-backed Jackal (Lupulella mesomelas), which are considered a problem animal or pest on these farmlands (see also Smithers 1978; Hermann et al. 2008). They can be a problem where poultry is raised and can be persecuted (Smithers 1978), and in particular hand-raised individuals tend to target poultry more so (Perrin 2002). In the North West Province, since 2014, until 2023, there have been 12 reports of serval incidences (North West Directorate of Biodiversity Management records), which is low, and these have often been reported sightings of alleged larger felids (i.e. Cheetah Acinonyx jubatus, Leopard), verses actual damage caused.
Deliberate targeted killing
Deliberate and incidental mortality by humans has caused deaths of Servals in KwaZulu-Natal. For example, Ramesh et al. (2015a) found that nearly 40% of the collared Servals (N = 17) in the KwaZulu-Natal Midlands died due to snaring, roadkill and possibly rodent poisoning.
Serval may be persecuted owing to depredations upon livestock, in particular that of poultry (Smithers 1978, Manqele et al. 2018), as well as small stock too even (Thorn et al. 2013). Interestingly, in Kwazulu-Natal, one of the drivers of illegal hunting was sport for unemployed youth, and amongst bushmeat hunters, serval were hunted as by-catch, and opportunistically then utilised for the trade in their skins and body parts (Manqele et al. 2018).
In the Waterberg, Limpopo, in 2011, 1.2% of small stock taken was attributed to Serval (Thorn et al. 2013), though one cannot be certain of the provenance or certainty of this.
The off-take (based on damage causing animal control) of Serval from Waterberg ranches in the Limpopo between 2009 and 2011 was estimated at 0,04 Servals/ 100 km² (Thorn et al. 2013). This off-take is fewer than the off-takes cited for some of the larger carnivores, and is likely a very low off-take and this low off-take is probably still a realistic regional level off-take for most ranchland areas, and would likely not be enough to impact the population a great deal.
Trophy hunting
There has been an increase in the demand to hunt Servals from at least 2015, which was round about the time of the last assessment (see Ramesh et al. 2016). From 2010 to 2019, there have been 589 offtakes of Servals as hunted and reported by PHs, and the most important provinces in this are Mpumalanga, Limpopo then KwaZulu-Natal (SANBI, PH Register, 2010-2019). In the North West, 16 have been removed over this time period, which range from 0-5 annually. In 2019, there were 59 removed from Mpumalanga through hunting, and Limpopo removed 29, and it is unclear whether these are excessive, but it is possible. It would be hoped that any off-takes of the species on the rangelands are matched with the desire of the landowner to continue to nurture the species. This possible threat needs to be monitored, though it is not urgent enough as an emerging threat.
Snaring
Servals are frequently entrapped in snares targeted to kill ungulates for meat (Manqele et al. 2018). For example, Ramesh et al. (2015a) found that nearly 40% of the collared Servals (N = 17) in the KwaZulu-Natal Midlands died due to snaring, inter alia. An average collared individual’s age was 3–4 years, which indicates high mortality and low survival rate (Ramesh et al. 2015a).
Skin trade
Similarly to Leopards, the trade in Serval skins for ceremonial traditions is an important threat (G. Balme unpubl. Data; Manqele et al. 2018), and also may contributes to a suspected ongoing decline in mature individuals, however Manqele et al. (2018) has performed population modelling, with progressive increases of anticipated mortality rates, and the Serval population was shown not to decline, with extinction probabilities evident at very high levels of mortality only (Manqele et al. 2018). Trade in Serval pelts for ceremonial or medicinal purposes is widespread throughout Africa (Thiel 2015), and pelts are often worn as a substitute for Leopard pelts, however, in Zulu culture they are still a valued commodity (Manqele et al. 2018).
Body parts trade
In the Faraday market, Gauteng, two traders had serval body parts (Whiting et al. 2011), while the trade in this is more prominent in KwaZulu-Natal (Manqele et al. 2018). Muthi practitioners and traders who were interviewed, reported using serval body parts to treat epilepsy, headaches, urinary problems and arthritis (Manqele et al. 2018).
Drowning in dams
Servals have been reported to have been found drowned in farm dams (Manqele et al. 2018).
Hybridisation
Hybridisation with feral cats may be a minor threat within the assessment region, although this is not as severe as other species, such as African Wild Cat. Hybridisation with the African Wild Cat has been documented in captivity (Skinner & Chimimba 2005). Deliberate hybridisation with the feral cat has resulted in a newly registered breed, the “Savannah Cat” (Eckermann-Ross 2014), and facilitated by the fact that many small felids are susceptible to domestication (Cameron-Beaumont et al. 2002). However, the males tend to become sterile after a few generations (Davis et al. 2015). The “Savannah Cat” is even registered with the International Cat Association as a breed, and is subject to various regulations depending on the state or province in the United States and Canada respectively. There has, however, been no indication of any threat as yet.
Conservation
Effective conservation of Serval populations demands sufficient viable native habitat, particularly wetlands in fragmented landscapes of southern Africa (Ramesh & Downs 2013, 2015a; Ramesh et al. 2015a, 2015b). Wetlands form an island habitat in a mosaic of farmland for several wetland-dependent species; and they are reservoirs of small mammal populations that are major dietary components of Servals (Bowland 1990). Consequently, if wetlands are protected in a mosaic of farmland, the landscape may support the persistence of Serval subpopulations. Improving the quality of both remaining natural habitats and artificial wetlands is thus a priority intervention. For example, retaining ground cover and rank vegetation by reducing grazing pressure or keeping a buffer of natural vegetation intact around the wetland can reduce the impacts of damaging land-use practices (Bowland & Perrin 1989; Driver et al. 2012), and thus conserve small remnant habitat patches such as shrubby areas and scattered semi-natural grasslands to sustain murid diversity. Restoration of these rich habitat patches is the only means of improving functional connectivity in modified landscapes to facilitate movement between isolated patches (Ramesh et al. 2015a). Conservation managers should thus enhance heterogeneity by protecting diverse habitats including wetlands and other indigenous habitats. The increases in habitat quality could provide source subpopulations for long-distance dispersers to re-colonise fragmented habitats (sensu Hermann et al. 2008). The Serval as an ecological indicator (Ramesh et al. 2015b), could be a flagship so to speak in the bigger picture in terms of water resource security. An Agricultural Research Council (ARC) driven project aimed at peatland restoration via a GEF 8-UN funded project should bode well for the species, specifically in the Maputaland area of KwaZulu-Natal, Molemane/Molopo eye area of North West, and perhaps the Gouritz area of the Western Cape (though if not already present in the latter, reintroduction may be worthwhile there).
Additionally, artificial wetlands, such as industrial sites, that provide protection, prey base and shelter can be integrated into landscape-scale conservation plans (Loock et al. 2018). Biodiversity stewardship schemes should also be promoted if landowners possess wetlands close to core protected areas or remaining habitat patches (see Ramesh et al. 2015; 2015b). Protecting such habitats may create further dispersal corridors between patches.
Although Servals can thrive outside protected areas (Ramesh & Downs 2013; Loock et al. 2018), in KwaZulu-Natal at least, they are more inclined to be found deeper in reserves away from the edges (Pretorius 2019), and in Mpumalanga, they are more prevalent, based on photo-capture rates in larger Lowveld reserves adjacent Kruger National Park, than ones further (Wentzel et al. 2021), which may point to an edge effect of sorts, hence protected areas are still important for the species.
Reintroduction of Servals shows mixed success. Captive-bred Servals are amenable to reintroductions (Van Aarde & Skinner 1986; A. Jones pers. comm. 2015), but it is unclear whether there have been any introductions beyond the known range, though no permit applications have been received in at least the North West (see Power 2014), so it does appear as if natural range expansion has occurred. In North West Province, reintroductions took place in the 1990s (Power 2014) and subpopulations are still present in the sites, and seem to be thriving, except Kgaswane. In KwaZulu-Natal, releases in 2013 in Isimangaliso Wetland Park have been successful thus far, as eight individuals have been recorded in 2014 during camera trap surveys (T. Ramesh unpubl. data).
Thus, captive-bred animals may repopulate nature reserves well and convert readily to free ranging subpopulations. Similarly, Eastern Cape subpopulations have been restored at Kariega and Great Fish River Game Reserves with limited success (Hayward et al. 2007). However, they have thrived on Shamwari where six were reintroduced in 2001, and 15, while were counted in 2005, and in the same year, five were reintroduced into Kwandwe, and there were four left at the end of that year (Hayward et al. 2007), which suggests some breeding may have taken place in at least one of the reserves. However, there are also cases of unsuccessful reintroductions. For example, Serval recently released into Phinda Game Reserve, KwaZulu-Natal Province did not persist, which may be related to the high number of large predators in the reserve (V. Hugo pers. comm. 2016) or insufficient rodent prey as the coastal grasslands are generally poorer. Similarly, Tembe Elephant Park is another example of low success of reintroduction, and it seems that management priorities of protected areas affect the success of reintroduction (W. Matthews pers. comm. 2015). Servals used to occur in Mkuze Game Reserve historically but have not been seen for years (T. Bodasing pers. comm. 2016), but two rehabilitated individuals were introduced in February 2014 (T. Bodasing pers. comm. 2015). Of two hand-reared Serval released into Mount Currie Nature Reserve, KwaZulu-Natal in 1998, one remained in the reserve while the other established its core home range in neighbouring farmland (Perrin 2002). Reintroductions have also had low success in Mpumalanga, especially in areas with other more prolific or dominant predators present (J. Eksteen pers. comm. 2015). Radio-tracking data also revealed a high turnover of individuals, which may suggest a high mortality rate post-release (Ramesh et al. 2015a). Thus, while Servals can be reintroduced easily as they respond well to ex situ breeding, the factors determining successful reintroductions require analysis before reintroduction can be effectively used as a tool to increase habitat occupancy and supplement existing subpopulations. For example, Perrin (2002) recommends hand-reared Servals are reintroduced to sites > 10 km away from human settlements to avoid habituation and potential mortality.
Strict controls on the importation of hybrid “Savannah Cats” from the United States of America should be put in place to prevent genetic contamination with the national population. While the males tend to become sterile, the females do not (Davis et al. 2015). Likewise, our CITES management authority should monitor exports of this species in the light of potential over-harvest for this purpose. To this end, educational awareness campaigns, are suspected to have decreased persecution of Servals and should be continued to promote Serval conservation and good land management practice. Similarly, reducing the illegal trade in skins by providing faux furs for use at cultural ceremonies may be an effective intervention.
Recommendations for land managers and practitioners:
- Habitat management to conserve prime habitat. Managers and landowners must avoid draining wetlands and ensure good veld condition as Servals forage away from wetlands too in woodlands with good grass cover. Waterside vegetation and ground cover should be maintained in farmlands and ranchlands to conserve key resource areas for Servals within an otherwise unsuitable matrix.
- Serval subpopulations should be systematically monitored to determine abundance and trends. Due to their specialised habitat requirements at small spatial scales, they may serve as a useful ecosystem indicator of the effect of habitat fragmentation in agricultural landscapes (Ramesh et al. 2015b).
- Monitoring Serval should be introduced as a compliance measure in Environmental Impact Assessment mitigation reports.
- Industrialised sites should incorporate the long-term persistence of Serval and associated habitats into onsite biodiversity management practices (see Loock et al. 2018). Buffer habitats on mining sites could be modelled based on minimum wetland size and available cover.
Research priorities:
- General survey for population estimates, by estimating densities in both prime and marginal habitat. Some regions, such as Limpopo, have no current information on density and should be targeted for surveys. Long-term monitoring of subpopulations in protected areas and farmlands will also reveal the resilience and population trends of Serval subpopulations.
- Efficacy of local community-based eco-awareness to mitigate hunting and persecution of Serval (for example, snaring, use of hunting dogs and roadkill).
- Long-term monitoring of translocated/reintroduced individuals, as well as released animals from rescue centres, at least until they have reproduced (one to two years). There are currently very few data on whether captive releases or reintroductions have been successful or not. These should be done using radio-, GPS-Cell or satellite telemetry collars.
- Ecological role as controller of agricultural pests (i.e. agroecosystem services).
- Basic systematic distribution data of Serval across southern Africa should be collected through structured questionnaire surveys. This will highlight key areas that will need to be studied further.
- Ascertain their status in the Kalahari and if range expansion has indeed been the case to explain the species’ contemporary occurrences there.
- Research on whether the species is dependent on wetlands given occurrences in dryland habitats away from wetlands.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Create conservancies to enhance Serval dispersal and persistence.
- Do not purchase or import hybrid “Savannah Cats” from the USA.
- Report snaring or roadkill incidents to provincial conservation agencies or conservation NGOs, or data repository apps.
Bibliography
Bowland, A.E. and Perrin, M.R. 1989. The effect of overgrazing on the small mammals in Umfolozi Game Reserve. Zeitschrift für Säugetierkunde 54: 251–260.
Bowland, J.M. 1990. Diet, home range and movement patterns of serval on farmland in Natal. Department of Zoology and Entomology, University of Natal.
Cameron-Beaumont CH, Lowe SE, Bradshaw JW. 2002. Evidence suggesting preadaptation to domestication throughout the small Felidae. Biological Journal of the Linnean Society 75: 361–366.
Davis BW, Seabury CM, Brashear WA, Li G, Roelke-Parker M, Murphy WJ. 2015. Mechanisms underlying mammalian hybrid sterility in two feline interspecies model. Molecular Biology and Evolution 32: 2534–2546.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Eckermann-Ross C. 2014. Small nondomestic felids in veterinary practice. Journal of Exotic Pet Medicine 23: 327–336.
Engelbrecht, F.A., Steinkopf, J., Padavatan, J., and Midgley, G.F. 2024. Projections of Future Climate Change in Southern Africa and the Potential for Regional Tipping Points. In: von Maltitz, G.P., et al. Sustainability of Southern African Ecosystems under Global Change. Ecological Studies, vol 248. Springer, Cham. https://doi.org/10.1007/978-3-031-10948-5_7.
Finerty G, Bahaa-El-Din L, Henley S, Kesch MK, Seymour-Smith J, Van Der Weyde LK, Macdonald DW,
Loveridge AJ. 2019. Range expansion: Servals spotted in the Kalahari. Cat News 69:1-5 (tentative page numbers)
Foden, W.B. and Young, B.E. (eds.) 2016. IUCN SSC Guidelines for Assessing Species’ Vulnerability to Climate Change. Version 1.0. Occasional Paper of the IUCN Species Survival Commission No. 59. Cambridge, UK and Gland, Switzerland: IUCN Species Survival Commission. x+114pp.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. Conservation Breeding Specialist Group (SSC/IUCN) and Endangered Wildlife Trust, Parkview, South Africa.
GeoTerraImage. 2015. 1990-2013/14 South African National Land-Cover Change. DEA/CARDNO SCPF002: Implementation of Land-Use Maps for South Africa. Project Specific Data Report.
Hayward MW et al. 2007a. Practical considerations for the reintroduction of large, terrestrial, mammalian predators based on reintroductions to South Africa’s Eastern Cape Province. The Open Conservation Biology Journal 1: 1–11.
Hermann E, Kamler JF, Avenant NL. 2008. New records of servals Leptailurus serval in central South Africa. South African Journal of Wildlife Research 38: 185–188.
Hunter, L. T. B. and Bowland, J. 2013. Leptailurus serval. In: J. S. Kingdon and M. Hoffmann (eds), The Mammals of Africa, pp. 180–185. Academic Press, Amsterdam, The Netherlands.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Johnson, W.E., Eizirik, E., Pecon-Slattery, J., Murphy, W.J., Antunes, A., Teeling, E. and O’Brien, S.J. 2006. The late Miocene radiation of modern Felidae: A genetic assessment. Science 311: 73-77.
Kuhn, B.F. 2014. A preliminary assessment of the carnivore community outside Johannesburg, South Africa. South African Journal of Wildlife Research 44(1): 95-98.
Lloyd PH, Millar JCG. 1984.A questionnaire survey (1969-1974) of some of the larger mammals of the Cape Province. Bontebok 3: 1-49.
Loock D, SwanepoeL L. 2019. Assessing local patterns in carnivore occupancy and richness by camera trapping: a monitoring of diversity at Verloren Vallei Nature Reserve, Dullstroom, Mpumalanga. Wildlife & Resource Association (WRA), Bloemfontein.
Loock, D.J., Williams, S.T., Emslie, K.W., Matthews, W.S. and Swanepoel, L.H. 2018. High carnivore population density highlights the conservation value of industrialised sites.Scientific reports 8(1): p.16575.
Lynch C.D. 1989. The mammals of the north-eastern Cape Province. Navorsinge van die Nasionale Museum Bloemfontein 25: 1-116.
Lynch, C.D. 1983. The mammals of the Orange Free State, South Africa. Navorsinge van die Nasionale Museum Bloemfontein 18: 1-218.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
Manqele, N. S. 2017. Assessing the drivers and impact of illegal hunting for bushmeat and trade on serval (Leptailurus serval, Schreber 1776) and oribi (Ourebia ourebi, Zimmermann 1783) in South Africa. MSc. thesis, University of KwaZulu-Natal, Pietermaritzburg.
Manqele, N.S., Selier, J.A., Hill, T.R. and Downs, C.T. 2018. Drivers of the illegal hunting of serval (Leptailurus serval) and oribi (Ourebia ourebi) in the KwaZulu-Natal Midlands, South Africa. African Journal of Wildlife Research, 48(2): 1-18.
Matthews W, Loock D, Emslie K, Somers M, Toft M. In prep. The Status and Health of a Serval Population on a Highly Industrialised Site and Implications for the Conservation of this Species. Sasol, Secunda, South Africa.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum , Pretoria, South Africa.
Monadjem, A. 1998. Distributional patterns and conservation status of mammals of Swaziland, southern Africa. Koedoe 41: 45-59.
Mucina, L. and Rutherford, M.C. 2006. The Vegetation of South Africa, Lesotho and Swaziland. Strelitzia 19. South African National Biodiversity Institute, Pretoria, South Africa.
Nowell, K. and Jackson, P. 1996. Wild Cats. Status Survey and Conservation Action Plan. IUCN/SSC Cat Specialist Group, Gland, Switzerland and Cambridge, UK.
NWPG (North West Provincial Government) 2024. The impact of roadkill on mammal populations in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
O’Brien, S.J. and Johnson, W.E. 2007. The evolution of cats. Scientific American July: 68-75.\
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Perrin MR. 2002. Space use by a reintroduced serval in Mount Currie Nature Reserve. South African Journal of Wildlife Research 32: 79–86.
Pienaar, U. De V., Joubert, S.C.J., Hall-Martin, A., De Graaf, G. and Rautenbach, I. L. 1996. Field guide to the mammals of the Kruger National Park. Struik, Cape Town.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Power, R.J., Van Straaten, A., Schaller, R., Mooke, M., Boshoff, T. and H. P. Nel. 2019. An inventory of mammals of the North West Province, South Africa. Annals of the Ditsong Museum of Natural History 8: 6-29.
Ramesh T, Downs CT. 2013. Impact of farmland use on population density and activity patterns of serval in South Africa. Journal of Mammalogy 94: 1460–1470.
Ramesh T, Downs CT. 2015a. Diet of serval (Leptailurus serval) on farmlands in the Drakensberg Midlands, South Africa. Mammalia 79: 399–407.
Ramesh T, Downs CT. 2015b. Impact of land use on occupancy and abundance of terrestrial mammals in the Drakensberg Midlands, South Africa. Journal for Nature Conservation 23: 9-18.
Ramesh T, Kalle R, Downs CT. 2015a. Spatiotemporal variation in resource selection of servals: insights from a landscape under heavy land-use transformation. Journal of Mammalogy 97: 1–14.
Ramesh T, Kalle R, Downs CT. 2015b. Sex-specific indicators of landscape use by servals: consequences of living in fragmented landscapes. Ecological Indicators 52: 8–15.
Ramesh T, Kalle R, Rosenlund H, Downs CT. 2016. Native habitat and protected area size matters: Preserving mammalian assemblages in the Maputaland Conservation Unit of South Africa. Forest Ecology and Management 360: 20-29.
Ramesh, T, Downs C. T., Power J., Avenant, N., Laurence, S., Matthews, W. and M. F. Child.2016. A conservation assessment of Leptailurus serval. In: Child F.M., Roxburgh L., Do, Linh. San, E., Raimondo, D. and Davies-Mostert H.T, (Eds). The Red List of Mammals of South Africa, Swaziland and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Rowe-Rowe, D.T. 1992. The Carnivores of Natal. Natal Parks Board, Pietermaritzburg, South Africa.
Skead, C.J. 2007. Historical Incidence of the Larger Land Mammals in the broader Eastern Cape.In: Boshoff AF, Kerley GIH, Lloyd PH (ed.), Centre for African Conservation Ecology, Nelson Mandela Metropolitan University., Port Elizabeth, South Africa.
Skead, C.J. 2011. Historical incidence of the larger land mammals in the broader Western and Northern Cape provinces.In: Boshoff A.F., Kerley G.I.H, Lloyd P. (ed.), Centre for African Conservation Ecology, Nelson Mandela Metropolitan University, Port Elizabeth.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smith K, Venter JA, Peel M, Keith M, Somers MJ. 2023. Temporal partitioning and the potential for avoidance behaviour within South African carnivore communities. Ecology and Evolution. 13(8):e10380.
Smithers RHN. 1978. The serval Leptailurus serval Schreber, 1776. South African Journal of Wildlife Research 8: 29–37.
Smithers, R.H.N. 1986. South African Red Data Book – Terrestrial Mammals. South African National Scientific Programmes Report 125: 1–216.
Stuart, C.T. 1981. Notes on the mammalian carnivores of the Cape Province, South Africa. Bontebok 1: 1-58.
Stuart, C.T. 1985. The status of two endangered carnivores occurring in the Cape Province, South Africa, Felis serval and Lutra maculicollis. Biological Conservation 32: 375-382.
Sunquist, M. and Sunquist, F. 2002. Wild Cats of the World. University of Chicago Press.
Taylor, J. 2020. From big spots to little spots: Influence of camera trap deployment on spatial capture-recapture estimates of servals (Leptailurus serval) in Ithala Game Reserve. Msc thesis, University of Cape Town, Cape Town.
Thiel C. 2015. Leptailurus serval. The IUCN Red List of Threatened Species 2015.
Thorn M, Green M, Bateman PW, Cameron EZ, Yarnell RW, Scott DM. 2010. Comparative efficacy of sign surveys, spotlighting and audio playbacks in a landscape-scale carnivore survey. South African Journal of Wildlife Research 40(1): 77-86.
Thorn, M., Green, M., Keith, M., Marnewick, K., Bateman, P.W., Cameron, E.Z. and Scott, D.M. 2011. Large-scale distribution patterns of carnivores in northern South Africa: implications for conservation and monitoring. Oryx 45(4): 579-586.
Thorn M, Green M, Scott D, Marnewick K. 2013. Characteristics and determinants of human-carnivore conflict in South African farmland. Biodiversity and conservation 22: 1715-1730.
Van Aarde, R.J. & J. D. Skinner., 1986. Pattern of space use by relocated servals, Felis serval. African Journal of Ecology 24(2): 97-102
Van der Weyde LK, Theisinger O, Mbisana C, Gielen MC, Klein R. 2021. The value of pastoral ranches for wildlife conservation in the Kalahari. Wildlife Research 49(3): 215-226.
Von Richter, W., 1972. Remarks on present distribution and abundance of some South African carnivores. Journal of the Southern African Wildlife Management Association 2(1): 9-16.
Webster, A.B., Pretorius, M.E. and Somers, M.J. 2021. The determinants of mesocarnivore activity patterns in highveld grassland and riparian habitats. African Journal of Wildlife Research 51(1): 178-192.Wentzel J, Gall C, Bourn M, De Beer J, Du Plessis F, Fosgate GT. 2021. Carnivore detection at the domestic/wildlife interface within Mpumalanga Province, South Africa. Animals 11(9): p.2535.
Whiting, M.J., Williams, V.A., and Hibbitts, T.J. 2011. Animals traded for traditional medicine at the Faraday market in South Africa: species diversity and conservation implications. Journal of Zoology 284, 84-96.
Williams, S.T., Collinson, W., Patterson-Abrolat, C., Marneweck, D.G. & L. H. Swanepoel., 2019. Using road patrol data to identify factors associated with carnivore roadkill counts. PeerJ, 7, p.e6650.