Samango Monkey
Cercopithecus albogularis

2025 Red list status
C. a. erythrarchus
Near Threatened

2025 Red list status
C. a labiatus
Vulnerable

2025 Red list status
C. a schwarzi
Endangered
Regional Population
Trend
Decreasing
Change compared
to 2016
No Change
Overview
Cercopithecus albogularis – Sykes, 1831
Cercopithecus albogularis labiatus Geoffroy Saint-Hilaire 1842
Cercopithecus albogularis erythrarchus Peters 1852
Cercopithecus albogularis schwarzi Roberts 1931
ANIMALIA – CHORDATA – MAMMALIA – PRIMATES – CERCOPITHECIDAE – Cercopithecus – albogularis
Common Names: Samango Monkey, Sykes’ Monkey, Stair’s White collared Monkey, Schwarz’s White-collared Monkey (English), Samango-aap (Afrikaans), Insimango (siSwati, isiZulu), Ndlandlama (Xitsonga), Dulu (Tshivenda), Intsimango (isiXhosa)
Synonyms: Cercopithecus mitis, Geoffroy Saint-Hilaire 1842 (Blue Monkey, Diademed Monkey)
Taxonomic Note:
There are taxonomic controversies on both the species and subspecies level. While Groves (2001, 2005) classifies the Samango Monkey as C. albogularis (recognising albogularis as a separate species within the highly polytypic Cercopithecus nictitans group), Grubb et al. (2003) does not recognise C. albogularis as a separate taxon and classifies it as C. mitis. No extensive genetic research has been undertaken to date to help clarify this issue, but recent analysis of mtDNA data from a limited number of samples across the taxon’s range provides first evidence in support of Grove’s suggested classification (Linden et al. 2020a). Further sampling is required to resolve the taxonomy. The number of subspecies recognised in South Africa is also inconsistent. Meester et al. (1986) followed by Grubb et al. (2003) recognise two subspecies, namely C. a. labiatus and C. a. erythrarchus, whereas Roberts (1951), Dandelot (1974) and Groves (2001) recognise an additional third Samango Monkey subspecies in South Africa, namely C. a. schwarzi, Roberts 1931. Recently, Dalton et al. (2015) found clear pelage colour polymorphism in South African Samango Monkeys and were able to identify three distinct geographical colour morphs as well as three genetically distinct entities: Hogsback (Eastern Cape Province) corresponding to C. a. labiatus, Inland (Soutpansberg, Magoebaskloof) corresponding to C. a. schwarzi, and Coast (Cape Vidal, Sodwana Bay) corresponding to C. a. erythrarchus. These findings support the presence of three subspecies within the assessment region, as first proposed by Roberts (1951) and later supported by Groves (2001).
| Red List Status |
|
C. a. erythrarchus – NT – Near Threatened B2ab(ii,iii,v) C. a labiatus – VU – Vulnerable B2ab(ii,iii,v) C. a schwarzi – EN – Endangered B2ab(ii,iii,v) (IUCN version 3.1) |
Assessment Information
Assessors: Linden, B.1 & da Silva, J.2
Reviewer: Le Roux, A.3
Institutions: 1Aberystwyth University, UK, University of Venda, South Africa; 2South African National Biodiversity Institute, 3University of the Free State, South Africa.
Previous assessors & Reviewers: Wimberger, K., Ehlers-Smith, Y. & Child, M.F.
Previous contributors: Roxburgh, L. & Howlett, C.
Assessment Rationale
Samango Monkeys are restricted to a variety of forest habitats and comprise three subspecies within the assessment region: Samango Monkey (C. a. labiatus), Stair’s White-collared Monkey (C. a. erythrarchus), and Schwarz’s White-Collared Monkey (C. a. schwarzi). While C. a. labiatus is endemic to the assessment region, C. a. erythrarchus also occurs outside South Africa (from the Mlanje Plateau in southern Malawi, to Zimbabwe, and most of Mozambique) and it is uncertain whether C. a. schwarzi is endemic due to lack of sampling in suitable extra-regional habitats. However, according to current data, South African populations of the latter subspecies are most likely completely isolated with no rescue effect from neighbouring countries possible. Furthermore, given historical and ongoing forest habitat loss and fragmentation, all three subspecies exist in isolated or semi-isolated forest fragments with a suspected low rate of dispersal. Although the estimated extent of occurrence for all subspecies is > 20,000 km2, area of occupancy was calculated as the amount of remaining natural habitat within forest patches greater than 1.5 km2 in extent (below which, forest patches are generally unoccupied by samangos), which yielded 870 km2, 692 km2 and 340 km2 for C. a. labiatus, C. a. erythrarchus and C. a. schwarzi respectively.
For all three subspecies, there is an inferred continuing decline in area of occupancy due to ongoing forest habitat loss across the country, a suspected continuing decline in habitat quality from commercial forestry reducing food resources, and a suspected continuing decline in mature individuals from frequent reports of mortality from road collisions, electrocutions, snaring and hunting for traditional medicine. This results in the listings of Vulnerable B2ab(ii,iii,v) for both C. a. labiatus and C. a. erythrarchus, and Endangered B2ab(ii,iii,v) for C. a. schwarzi. Although the extra-regional habitat of C. a. erythrarchus is similarly fragmented, and we do not yet understand the dispersal capacity of Samango Monkeys, their core range is protected by the Lubombo Transfrontier Conservation Area (established in 2000), which has secured corridors between Eswatini, South Africa and Mozambique. Thus, because these northern forests of the South African range of C. a. erythrarchus are formally protected and there remains at least the potential for dispersal across boundaries, we confirm the listing of C. a. erythrarchus as Near Threatened B2ab(ii,iii,v). Although the minimum estimated population sizes fall below 10,000 mature individuals, and C. a. labiatus and C. a. schwarzi have lower estimates for the largest subpopulation size below 1,000 mature individuals, the huge range in possible population sizes was deemed too great to realistically apply the C criterion without more recent density and occupancy estimates from field surveys.
Although preliminary studies suggest that Samango Monkeys adapt to human-modified habitat by being able to eat exotic plant species planted by people, core forest patches are needed by the species for successful reproduction, recruitment and viability. Extinction risk is thus entirely dependent upon effective management of the Forest Biome. Key interventions correspondingly include enforcement of penalties for forest-related transgression, protected area expansion and the establishment and/or maintenance of corridors between forest patches. Critically, distribution data from the literature need to be collated and surveys of all suitable habitats need to be performed to more accurately delineate range boundaries and occupancy in remaining forest patches. This current assessment should therefore be revised once a more complete dataset is available.
Regional population effects: While C. a. labiatus is confirmed to be endemic to South Africa, and C. a. schwarzi is assumed to be endemic until further research shows otherwise, C. a. erythrarchus is presumably connected to extra-regional subpopulations through the Lubombo Transfrontier Conservation Area and thus we assume rescue effects are possible. Future research should, however, confirm dispersal between countries.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Linden B & da Silva JM. 2025. A conservation assessment of Cercopithecus albogularis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
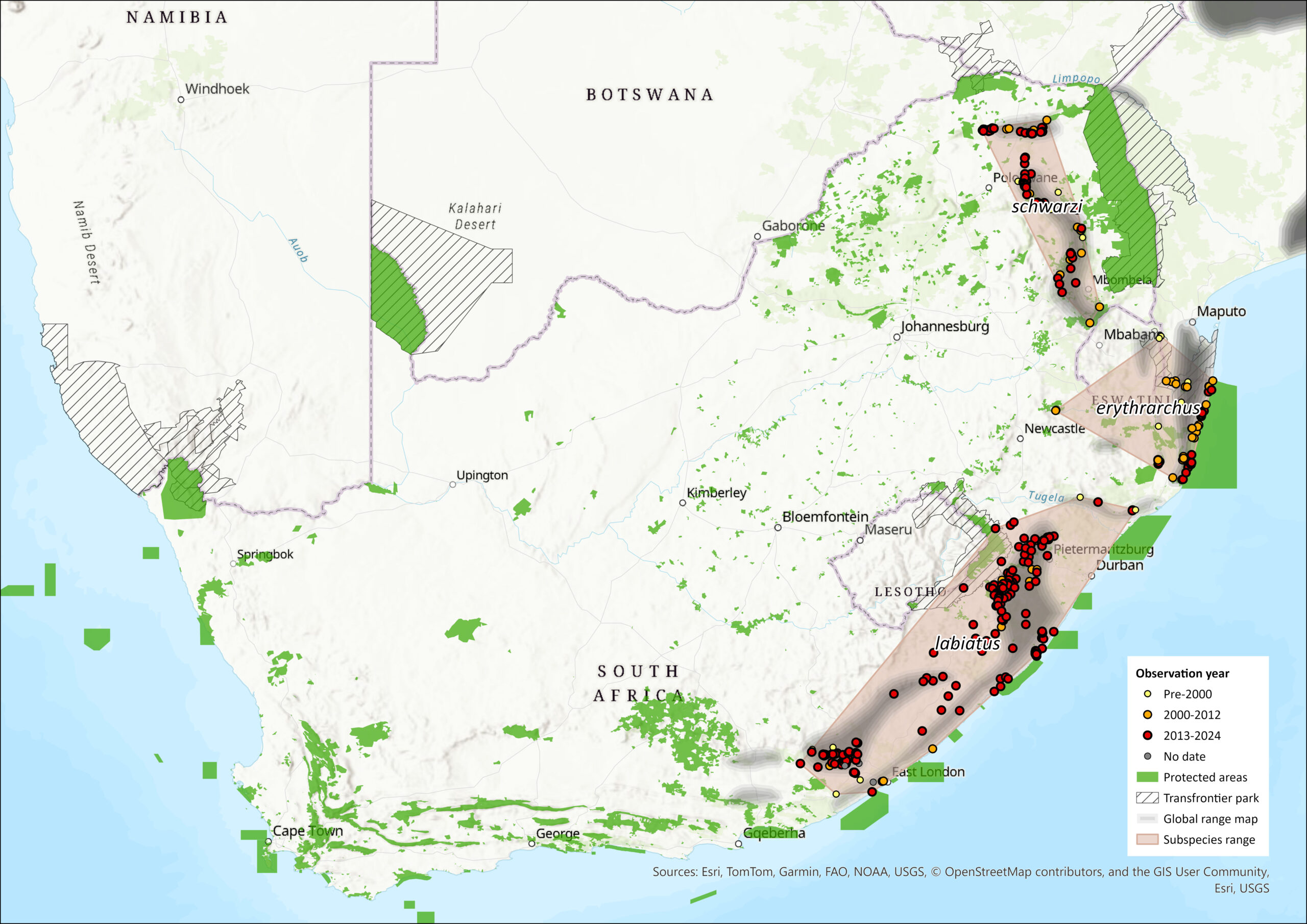
Regional Distribution and occurrence
Geographic Range
Cercopithecus albogularis is distributed from Ethiopia to South Africa and also occurs in southern and eastern Democratic Republic of the Congo and northwestern Angola (Groves 2001, 2005). The current South African Samango Monkey populations are relicts of the repeated historical (Pleistocene and Holocene) expansion and contraction of forests. They are naturally fragmented and, as a result, often isolated. This natural fragmentation is exacerbated by human population expansion and development. Where there might have been connections and dispersal corridors between subpopulations in the past (for example, through riverine corridors), these have been increasingly lost to development and agriculture.
Cercopithecus a. labiatus is endemic to South Africa and has been separated from populations further north in the assessment region for 0.5 1.7 million years (Dalton et al. 2015, Linden et al. 2020a). The southern limit of C. a. labiatus is the Pirie Forest in the Eastern Cape; it is not found in the evergreen Knysna and Tsitsikamma forests further south (Lawes 1990), and it extends north-eastwards to the midlands of the KwaZulu-Natal Province. Its present distribution is closely correlated with the distribution of Afromontane forests within the assessment region (Lawes 1990). It occurs in Scarp and Indian Ocean Coastal belt forests, as well as Pondoland forests (Hayward et al. 2005). However, identification of the subspecies there is unknown, and needs to be done via genetic analyses. The boundary between C. a. labiatus and C. a. erythrarchus is currently suggested to be the St. Lucia and Umfolozi River systems (Lawes 1990; Dalton et al. 2015), where apparently neither subspecies is found in the dune forest south of the St. Lucia estuary (Lawes 1992).
Accordingly, C. a. erythrarchus occurs from northern KwaZulu-Natal Province, possibly along southern Mpumalanga Province, through Zimbabwe and Mozambique (although the coastal limits are uncertain) up to Malawi. While this subspecies has been described to extend as far as Malawi, their forest habitat is fragmented (both naturally and through human expansion and development), and thus it is unlikely that there is significant immigration from outside the region, especially as not enough is known yet about individuals’ (especially male) dispersal abilities. In Eswatini, they have been recorded from forest patches in Mlawula and Muti Muti Nature Reserves and in some of the larger forested gorges in the eastern Lubombos (for example, Mnyame Gorge) (Monadjem et al. 2003).
The area bordering Eswatini to the north and the area just north of the Umfolozi River and west to the confirmed C. a. erythrarchus range (in the past assigned to C. a. labiatus) are both confirmed C. albogularis distribution records. However, at this stage it is not known to which subspecies these distribution records belong (Dalton et al. 2015). For the purposes of this assessment, we have assigned the records to C. a. erythrarchus.
The distribution of C. a. schwarzi comprises Mariepskop in the Pilgrims Rest District (Mpumalanga Province), north to the Magoebaskloof area including Woodbush (Roberts 1951; Groves 2001), as well as the Soutpansberg mountains (Limpopo Province, Dalton et al. 2015). It is unknown whether the subspecies is endemic, as possibly suitable locations in the Afromontane forests further north (Eastern Highlands in Zimbabwe and Gorongoza in Mozambique) have not yet been sampled. Consequently, genetic data on subpopulations further north are unavailable for comparison. Recent genetic data shows that the Soutpansberg C. a. schwarzi subpopulations are currently most likely isolated from the escarpment C. a. schwarzi subpopulations further south due to a lack of suitable connecting habitat and extensive human-induced landscape change (Linden et al. 2022a). For this assessment, the area south of Mariepskop along the escarpment to Eswatini has been assigned to C. a. schwarzi. However, future sampling of those populations and genetic analysis will need to be undertaken to make a definite assignment to which subspecies they belong.
Samango Monkeys are mostly restricted to intact forest habitat although they will traverse other habitats while foraging or moving between forest patches (Skinner & Chimimba 2005; Linden 2020b, Linden et al. 2020c; Wimberger et al. 2017), such as Pinus and Eucalyptus spp. plantations and residential gardens, but patch occupancy seems unaffected by land-use type of the surrounding matrix (Lawes et al. 2000). As such, we construe intact forest patches as a measure of core area of occupancy. Estimating extent of occurrence (EOO) and area of occupancy (AOO) is challenging as the precise range delimitations and current forest patch occupancy has not been determined. However, based on available data and information from the literature (for example, Lawes 1990, 1992; Lawes et al. 2000; Dalton et al. 2015), the estimated EOO is between 21,605 and 90,412 km2 (Table 1). The AOO can be estimated according to all forest patches available (based on the Mucina & Rutherford 2006 vegetation types) within the EOO or from forest patches with current confirmed presence, which, for C. a. labiatus, ranges from 1,109–1,400 km2 (depending on more or less conservative forest patch inclusion) to 702 km2 respectively. The minimum critical forest patch size has been calculated as 0.44 km2 and forest patches < 1.5 km2 were never occupied by Samango Monkeys (Lawes et al 2000). By including only patches above the 1.5 km2 threshold, AOO is estimated to be between 382 and 989 km2 (all available patches). Finally, using land-cover satellite data (GeoTerraImage 2015), we estimated the effective AOO as the amount of remaining natural habitat within forest patches, which was 88–89% of the habitat (11–12% modified), which yielded revised AOO estimates of 340 km2, 692 km2 and 870 km2 for C. a. schwarzi, C. a. erythrarchus and C. a. labiatus respectively (Table 1).
The effective AOO is likely to be less when dispersal distances (3.7 km) between known occupied forest patches are taken into consideration (Lawes et al. 2000). Indeed, Lawes et al. (2000) found that only 7% of forest patches were occupied in the Balgowan and Karkloof districts of KwaZulu-Natal Province. Similarly, only 11–17% of the forest patches are above the 1.5 km2 threshold for the various subspecies and thus suspected to be occupied by viable subpopulations (Table 1).
Table 1: Area of Occupancy (AOO) estimates for Samango Monkeys (Cercopithecus albogularis) based on remaining forest patches within the extent of occurrence (EOO)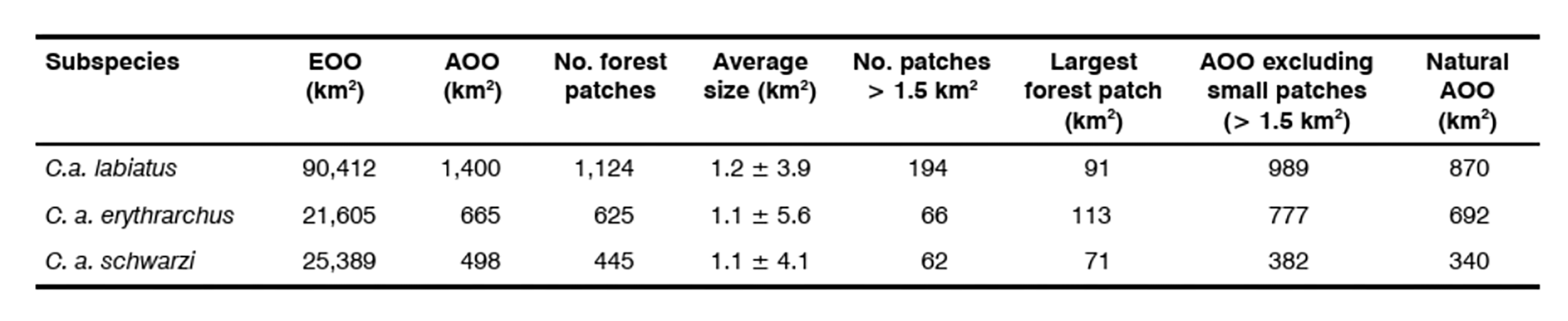
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): 3800
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realms
Biogeographic Realm: Afrotropical
Map
Figure 1. Distribution records for Samango Monkey (Cercopithecus albogularis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
| Country | Presence | Origin |
| Botswana | Absent | – |
| Lesotho | Absent | – |
| Mozambique | ||
| C. a. labiatus | Absent | |
| C. a. erythrarchus | Extant | Native |
| C. a. schwarzi | Presence uncertain | |
| Namibia | Absent | |
| South Africa | Extant | Native |
| Eswatini | ||
| C. a. labiatus | Presence uncertain | |
| C. a. erythrarchus | Extant | Native |
| C. a. schwarzi | Presence uncertain | |
| Zimbabwe | ||
| C. a. labiatus | Absent | |
| C. a. erythrarchus | Extant | Native |
| C. a. schwarzi | Presence uncertain |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
When assessing Samango Monkey persistence, possible future climate change impacts on forest habitat must be considered. While average global temperatures are predicted to rise by up to 2.5 °C by 2050 (IPCC 2013), trends in Africa suggest the continent may experience double this rate of change (Engelbrecht et al. 2016) with tropical southern Africa being one of the fastest warming areas (Davis-Reddy & Vincent 2017). Over the last century average temperatures across greater southern Africa have shown a clear and marked increase (Kruger & Nxumalo 2017) and future temperature projections show rises of 2–4 °C in the interior (Davis-Reddy & Vincent 2017). These mean temperature increases are further accompanied by increases in extreme events such as drought, heavy rainfall, strong winds and wildfires (Davis-Reddy & Vincent 2017). Rainfall predictions for southern Africa are ambiguous with disagreement on wetting vs. drying trends (Skowno et al. 2019) but in the north-eastern parts of South Africa, trends indicate that rainfall will reduce, and average annual temperatures and atmospheric CO2 levels will continue to rise (Kruger & Sekele 2013; DEA 2013; Engelbrecht et al. 2016).
The potential impact of these changes on forests is diverse and temperature changes alone are likely to drastically alter forest ecosystem dynamics (Norby et al. 2007). Higher temperatures lead to elevated water losses from evaporation and evapotranspiration and can reduce water use efficiency of plants (Mortsch 2006). Moisture stress and drought can lead to increased susceptibility to disturbances such as insect pests, pathogens and forest fires (Moore & Allard 2008). Temperature and precipitation changes can further make forests more sensitive to alien plant invasion (Dale et al. 2001). Increased CO2 levels generally promote plant growth but only as long as other factors such as water do not become limiting (Moore & Allard 2008). A consistent response of plant species to climate change entails changes in distribution and here forests plants are expected to shift their ranges northwards or to higher altitudes with warming temperatures (Parmesan 2006; Schreiter et al. 2017). Taking all these impacts together we suggest that the Samango Monkey forest habitat is very vulnerable to climate change. Many forest patches and riverine corridors are naturally elongated and narrow with high perimeter: area ratio, making them vulnerable to edge effects such as changes in temperature, humidity and amount of sunlight penetrating, increasing risks of fires and alien invasion. Afromontane forests in the assessment region for example are driven by shade effects and runoff from cliffs, rainfall, fog precipitation and groundwater. The predicted drying trend and increased mean temperatures for the region might result in forest tree die-offs, plant community changes, overall forest cover shrinkage (Rutherford et al. 1999) and increased susceptibility to disturbances such as invasion by alien invasive plants. More specifically, recent projections (Yahaya et al. 2024) confirm that all Shared Socioeconomic Pathways Scenarios in a warming climate lead to the significant loss of forested areas in southern Africa.
The impact of droughts on Samango Monkey populations has not been studied systematically, but it is suggested that a Samango Monkey group reintroduced to the Ficus sycomorus forest along the Pafuri River in Kruger National Park from 1982-86 failed to sustain itself due to the 1991-92 drought after which they were not seen again (Viljoen, 1982; http://www.krugerpark.co.za/krugerpark-times-2-21-samango-monkey-21398.html). Extremely low rainfall in 1991 resulted in trees failing to fruit for two years. This, together with possible other fitness reducing factors (e.g. demographic and genetic) described in small, isolated populations, may have led to them not surviving in the long-term.
Population information
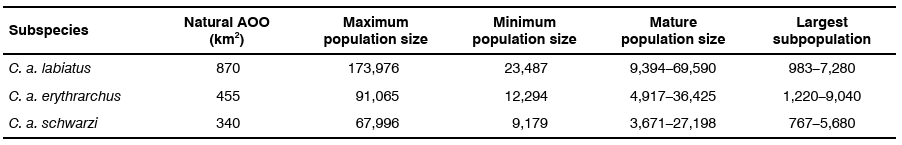
The population density of C. albogularis varies greatly with the habitat type occupied, but they can be common in suitable habitat. In South Africa, lowest densities are found in swamp, sand and riverine forests (< 30 individuals / km2), medium densities in Afromontane forests (44–83 individuals / km2) and highest densities in coastal forests (200 individuals / km2) (Lawes 1992). C. a. labiatus in general have lower population densities than C. a. erythrarchus (Lawes 1992). Home range size varied from 0.46 km2 and 0.55 km2 for C. a. schwarzi in the Soutpansberg (Limpopo Province) (Heikamp 2008, Coleman & Hill 2014) to 0.22 km2 and 0.27 km2 for C. a. labiatus in Hogsback (Wimberger et al. 2017) to 0.15 km2 for C. a. erythrarchus in Cape Vidal (KwaZulu-Natal Province), with densities of between 145 individuals / km2 and 202 individuals / km2 (Lawes & Henzi 1995). Table 2 shows the population and largest subpopulation estimates (using the largest forest patch per subspecies as a proxy) for the three subspecies based on maximum and minimum densities using the effective AOO estimates listed in Table 1. The number of mature individuals per troop varies from 40–50% (Friedmann & Daly 2004; Linden et al. 2015). Although the minimum estimates are below 10,000 mature individuals, and two subspecies contain lower estimates of < 1,000 mature individuals in the largest subpopulation, the range in mature individuals is too great to estimate a population size of <10,000 mature individuals for any subspecies. Further surveying will help to refine occupancy of forest patches and subsequently subpopulation sizes.
Table 2: Population size estimates for Samango Monkey subspecies based on maximum (200 individuals / km2) and minimum (27 individuals / km2) density estimates. Estimates for the largest subpopulation pertain to the largest forest patch for the subspecies. AOO is area of occupancy.
For the subspecies with the largest AOO, C. a. labiatus, Table 3 shows the probable subpopulation sizes in various forest types for currently occupied forest patches above the 1.5 km2 threshold. The analysis still yields over 10,000 mature individuals within the assessment region. However, this approach should be replicated when new density estimates become available for the various forest types within the subspecies’ ranges, especially for the largest forest patches, and when current occupancy of forest patches is more comprehensively mapped.
Table 3. Summary of subpopulation size estimates for C. a. labiatus based on observed densities in different forest types for those forest patches above 1.5 km2. Average density per forest type is from Lawes (1992). AOO is area of occupancy.

Small forests are generally unable to support Samango Monkey groups; they seem generally absent from forests smaller than 1.5 km² (Swart et al. 1993; Lawes 2002) (Table 1). Although it may be simplistic to define a subpopulation as a forest patch, Samango Monkeys are poor dispersers, in comparison to other forest-dwelling mammals, such as Blue Duiker, Philantomba monticola and Southern Tree Hyrax, Dendrohyrax arboreus, and are reluctant to disperse over open ground (Lawes et al. 2000). As such, most forest patches where they occur can be considered isolated or semi-isolated subpopulations. For example, nearly half (42%, N = 22) of the known (currently occupied) subpopulations of Samango Monkeys (for both erythrarchus and labiatus subspecies) in KwaZulu-Natal Province are found in forests patches smaller than 4 km2 (Lawes 1992; Lawes et al. 2000). Based on estimates of mean density (59 individuals / km2, Lawes 1992) this would mean that a large number of subpopulations contain fewer than 250 individuals and thus probably contain fewer than 100 mature individuals (based on 40–50% mature subpopulation structure). Such small subpopulations are vulnerable to demographic and environmental stochasticity facing a high risk of losing genetic diversity and thus adaptability through breeding of closely related individuals and are seldom recolonised (Swart & Lawes 1996). They exist in transient, non-equilibrium or declining metapopulations vulnerable to local extinctions as a result of diminishing forest areas and decline in habitat quality (Lawes 2002). Thus, the long-term viability of these small subpopulations is threatened if further habitat loss or a reduction in their population density occurs. Swart et al. (1993) found that if the density of this species falls below 30–40 individuals / km2 they are at risk of local extinctions within 50 years as they are unable to withstand a further 30–35% reduction in size.
Population Information
Current population trend: Decreasing. We infer that the population is declining within the assessment region, due to ongoing loss and degradation of forests.
Number of mature individuals in population: See Table 2.
Number of mature individuals in largest subpopulation: See Table 1 “Number of patches > 1.5 km2”.
Number of subpopulations: Unknown
Severely fragmented: Yes. Coastal forests are historically contiguous but have become fragmented by human activities in the last 100–150 years. Coastal forests are linear in shape and thus their ecological processes are dominated by those that characterise forest edges. Similarly, natural fragmentation of Afromontane forests (Eeley et al. 1999) has been exacerbated in the last 100 years by logging and harvesting activities, by afforestation (plantations), agriculture (e.g. avocado orchards) and human settlements.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Based on the available information, it is likely that there were three historical subpopulations of Samango as indicated by Dalton et al. (2015); however further subdivision has occurred due to anthropogenic influences.
Since the last assessment in 2016 two population genetic studies on local Samango Monkey populations in South Africa were published, both investigating how anthropogenic landscape change and habitat fragmentation may impact gene flow and genetic diversity. The first, by Madisha et al. (2017), sampled two neighbouring C. a. labiatus groups separated by 1 km of anthropogenically transformed areas in Hogsback on the southern slopes of the Amathole Mountains in the Eastern Cape Province. Results of this study did not detect negative genetic factors associated with isolation (e.g., inbreeding) in either group, suggesting that male migration between the groups was not impaired by the 1 km gap of seemingly unsuitable habitat. The second study by Linden et al. (2022a) investigated five local C. a. schwarzi populations on a larger scale along the length of the Soutpansberg mountain range in Limpopo Province, including an outgroup from the escarpment south of the mountain. Here, distances between sampled populations ranged from 27km – 109km. Results of this study concluded that extensive anthropogenic landscape changes in the eastern parts of the Soutpansberg and distance between high-canopy forest patches in the western parts appear to have reduced the suitability of environmental conditions for male samangos to disperse between sampling areas to facilitate gene flow. In particular three genetic clusters were found to best describe the dataset, although each site showed signs of genetic distinctiveness with some admixture detected within each. The study raises concerns about the long-term viability of populations across the Soutpansberg mountain based on the degree of population subdivision and the apparent lack of contemporary migration between populations. The study highlights that the eastern populations of the Soutpansberg are of particular concern due to immense anthropogenic pressure and that the western populations are likely entirely isolated without any rescue effect possibilities in the future. Considering the population genetic results and the fact that the geographic extent of C. a. schwarzi is still unknown, the Soutpansberg schwarzi populations should be of high conservation concern. Taken together, both studies show that distance between subpopulations / groups and the nature of the matrix surrounding forest patches can affect gene flow and with this genetic diversity. Patterns similar to those found for the Soutpansberg population likely apply in other parts of the assessment area due to the fragmented and isolated distribution of suitable forest habitat.
In total, it is expected that at least five genetically distinct subpopulations exist for this species (1 in the Amathole mountains, 3-5 in the Soutpansberg, 1 along the coast); however, this might be an underestimate. A comprehensive examination of the genetic structure across the species range is required to get a more accurate estimate of the number of distinct subpopulations. It is important to note that an increase in genetic distinctiveness is not desired in this instance as it is the result of human-mediated habitat transformation. Efforts should be made to re-establish connectivity.
Habitats and ecology
Samango Monkeys are primarily arboreal, utilising the canopy of evergreen forests, and their present distribution is indicative of very broad forest habitat tolerances (Lawes 1990). Within the assessment region, Samango Monkeys are associated with high-canopy, evergreen forests and are South Africa’s only forest-dwelling guenon. They inhabit a variety of indigenous forest types namely Afromontane Forests (including Mistbelt Forests), Coastal Forests (including Dune Forests), Scarp Forests as well as Riverine Forests (forest types follow von Maltitz et al. 2003). Cercopithecus a. schwarzi and C. a. erythrarchus have also been observed to utilise acacia woodland and coastal thicket adjacent to high canopy forests (Lawes & Piper 1992; Linden et al. 2015). Furthermore, experimental food patches placed in these secondary growth forests with lower canopies were utilised equally to food patches in higher-canopy forests (Emerson & Brown 2013). All three subspecies have been observed in human-modified habitat, including pine plantations, residential gardens and campsites (Lawes 1991, Chapman et al. 1998, , Wimberger et al. 2017, Linden 2020b), but more research needs to be conducted todetermine to which extent the species can use modified landscapes to disperse between forest patches. They can utilise human infrastructure to traverse their habitat, such as travelling along telephone and power lines, and across roads (Linden 2020b, Linden et al. 2020c, Linden et al. 2022b, K.Wimberger pers. obs.). However, Samango Monkeys seem to view human inhabited areas (residential gardens) as “riskier” habitats than their natural habitat, preferring to forage in indigenous forest if given experimental patches in both forest and gardens (Nowak et al. 2017). Meanwhile at a site with relatively high density of natural predators, C. a. schwarzi seemed to view humans as “shields” against terrestrial predators (for example, leopards Panthera pardus), reflected in their more intensive exploitation of experimental food patches at typically high-risk strata (ground level) in the presence of researchers (Nowak et al. 2014). Being arboreal monkeys, the density of food remaining in an experimental patch when a forager leaves was greatest at ground level relative to higher tree canopy levels, highlighting a strong vertical axis of fear (Emerson et al. 2011; Nowak et al. 2014).
Samango Monkeys are predominantly frugivores, with 50–70% of their diet consisting of fruit and leaves, and insects constituting the main source of protein (Lawes 1991, Skinner & Chimimba 2005, Linden et al. 2015). During periods of low fruit availability, other plant parts such as flowers and leaves/buds can be eaten (Linden et al. 2015; Wimberger et al. 2017). Using artificial food patch experiments, a goup of C. a. schwarzi were shown to prefer high-energy foods (peanuts) and were the least likely to choose animal protein (cat food, Emerson & Brown 2012). Low dietary diversity was found in a group of C. a. erythrarchus in Cape Vidal, KwaZulu-Natal Province where consumption of plant parts from four of the top 20 plant species (Isogloassa woodii, Mimusops caffra, Senegalia (Acacia) karroo, and Grewia occidentalis) accounted for 49% of the diet (Lawes 1991). Similarly, in a group of C. a. schwarzi one food item, namely figs, accounted for 26% of feeding time, indicating that Ficus spp. is perhaps a key resource for this subspecies (Linden et al. 2015). Furthermore, the exotic seeds of invasive Black Wattle (Acacia mearnsii) were the most frequently consumed item (26%, 32% of total diet) for two troops of C. a. labiatus (Wimberger et al. 2017). Due to their frugivorous diet, Samango Monkeys are suggested to play an important role in forest plant seed dispersal. In the Soutpansberg they were found to disperse the seeds of 22 plant species via defecation, cheek-pouch carrying and spitting or dropping, indicating that they play a substantial role in forest plant dynamics (Linden et al. 2015, Stringer et al. 2020). Furthermore, through their foraging behaviour Samango Monkeys were found to make fruit available to ground-dwelling mammals such as Bush Pig (Potamochoerus porcus), Red Duiker (Cephalophus natalensis) and Porcupine (Hystrix africaeaustralis), which may act as subsequent seed dispersers (Seufert et al. 2009, Hikel et al. 2023). All subspecies have been recorded to eat exotic plant species, invasive or planted by people, consume human refuse (for example kitchen scraps), and may become a pest in some areas as a result (Lawes et al. 1990; Lawes 1991; Chapman et al. 1998; Linden 2020b, K. Wimberger et al. 2017). Consumption of exotic plant species, particularly acorns from Quercus sp., may have a detrimental impact on teeth as well as other health parameters (A. Tordiffe pers. obs.).
Samango Monkeys typically live in large (up to 45 individuals) multi-female, single-male groups (Skinner & Chimimba 2005). The largest group size of C. a. schwarzi observed in the Soutpansberg comprises over 60 individuals (Coleman & Hill 2014; Nowak et al. 2014). Females are philopatric, while males leave their groups just before sexual maturity, between 6–8 years old (Henzi & Lawes 1987, 1988). These males remain alone or in the company of other lone males before taking over as group male (Henzi & Lawes 1987, 1988). In some populations, there are seasonal influxes of lone males, which has been documented to occur mainly during May to July in both C. a. erythrarchus (Cape Vidal) and C. a. schwarzi (Soutpansberg) (Henzi & Lawes 1987, 1988; B. Linden unpubl. data), but from November to March in C. a. labiatus (K. Wimberger pers. obs. 2011). These lone males may mate with the females in a group. The gestation period is around 176 days after which a single infant is usually born (Pazol et al. 2002). Samango Monkeys are seasonal breeders, but with birth seasons varying according to locality and subspecies, such as C. a. erythrarchus in St Lucia and C. a. schwarzi in the Soutpansberg birthing in September/October (Lawes 1990, B. Linden pers. obs.), and C. a. labiatus in Hogsback birth in July to August (K. Wimberger pers. obs.). Females have been observed carrying their dead infants for several days before eventually leaving them on the ground (K. Wimberger, pers.obs.).
The different subspecies can be visually distinguished according to Dalton et al. (2015) as follows: C. a. erythrarchus has an overall lighter appearance when compared to C. a. labiatus and C. a. schwarzi individuals. A marked difference is the black arms of C. a. labiatus and C. a. schwarzi individuals compared to the grey arms of C. a. erythrarchus. The yellow wash or shine on the back is most visible and most extensive in C. a. erythrarchus and near absent in C. a. labiatus. The ischial regions also show clear colouration differences, being most prominent and orange in C. a. erythrarchus, yellow in C. a. schwarzi and white in C. a. labiatus. Further colour differences worth mentioning are the very conspicuous white ear tufts and white underside of the tail (about the first quarter) in C. a. labiatus compared to the much less obvious white ear tufts and dark tail undersides in C. a. schwarzi and C. a. erythrarchus. Dalton et al.’s (2015) results are very similar to those of Roberts (1951) and Groves (2001).
Ecosystem and cultural services: Recent research reveals the importance of Samango Monkeys in dispersing the seeds of fruit trees (for example, 52% of fruiting species eaten by C. a. schwarzi), especially those occurring in high-canopy forests (Linden et al. 2015, Stringer et al. 2020). They may be particularly important dispersers for fig trees where these occur in their habitat (Linden et al. 2015). Samango Monkeys can be considered a flagship species for South African forests, and they are often a tourist attraction with popular articles on forests, which describe hiking trails and other tourism activities, seldom failing to mention Samangos.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.8. Forest -> Forest – Subtropical/Tropical Swamp | – | Suitable | – |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Suitable | – |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Suitable | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Suitable | – |
| 14.6. Artificial/Terrestrial -> Artificial/Terrestrial – Subtropical/Tropical Heavily Degraded Former Forest | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: 33.5 years recorded for s wild female (Cords 2012)
Average Reproductive Age: Females: first conception ~ 7 years (Cords 2012). Males: 6-8 years (Henzi & Lawes 1987, 1988)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: ~176 days (Pazol et al. 2002)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: One offspring every 2-3 years (Pazol et al. 2002, Cords 2012)
Natural Mortality: Average adult annual mortality is 5% (Cords 2012)
Does the species lay eggs? No.
Does the species give birth to live young: Yes.
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? No.
Does the species require water for breeding? No.
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Samango Monkeys are mainly used in the local trade for traditional medicine. Samango Monkey skins and carcasses have been found to be traded illegally at South Africa’s largest traditional medicine market, the Faraday market in Johannesburg (Whiting et al. 2011). Anecdotal evidence suggests they are used in traditional medicine if caught, as drying skins and hands were found near human settlements (Soutpansberg: J. Linden pers. obs. 1998; Hogsback: K. Wimberger pers. obs. 2011) and requests to researchers for samango skulls and hands by traditional healers were also experienced in the Soutpansberg area (B. Linden unpubl. data). They are possibly used on a subsistence level for bushmeat according to anecdotal reports, snares found intact on samango monkeys and an infant samango monkey being sold (for example, Hogsback K. Wimberger pers. obs. 2011). The effects of this trade are suspected to be minimal because they are infrequently captured and not a targeted species in cultural hunts.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Used locally and opportunistically as bushmeat. | Yes | Local commercial use in traditional medicine trade. |
National Commercial Value: No
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | – | – | – |
| 3. Medicine – human & veterinary | true | – | – | – |
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
The major threat to the species is deforestation and resulting habitat fragmentation causing population isolation, as Samango Monkeys are poor dispersers and re-colonisers and are susceptible to local extinction within small forest fragments (Lawes et al. 2000, Lawes 2002, Linden et al. 2022a). Although forests are currently well-protected in some areas, there is a continuing loss of forest habitat and habitat quality across the assessment region (see current habitat trend below). For instance, loss of habitat and habitat disturbance caused by selective logging of forests for timber and firewood, mainly in the Eastern Cape; and charcoal production and the medicinal plant trade in Maputaland. Coastal development through residential and industrial expansion is a major threat to the coastal forest habitats of both C. a. labiatus and C. a. erythrarchus, while expanding human settlements on the Pongola Floodplain may have led to the extirpation of the riverine subpopulations of C. a. erythrarchus in the area. Further south in the region, strip mining (between St Lucia and Sodwana) is a threat to C. a. erythrarchus and possibly C. a. labiatus. Extensive commercial afforestation in mistbelt regions of South Africa has resulted in the loss of small forests and reduction in area of some of the larger forests (Armstrong et al. 1998), which may specifically threaten C. a. schwarzi (Armstrong & van Hensbergen 1996). Indeed, in Limpopo’s Soutpansberg Mountains, ongoing habitat loss, fragmentation and degradation of riverine vegetation, which are possible corridors between forest patches, are the greatest threats to C. a. schwarzi (Munyati & Kabanda 2009, B. Linden pers. obs.).
The effects of forest loss and fragmentation include an increase in edge effects on forest patches and abundance of exotic plant species and availability of “human food” (for example, bread) and bring Samango Monkeys into more frequent contact with human settlements and infrastructure, which may lead to increased rates of morbidity and mortality. These deaths and injuries can be caused by bushmeat hunting, collection for the muthi trade, intentional or incidental snaring and hunting by dogs (which are possibly directed at other forest species; for example, small antelope), attacks by residential dogs (Cuozzo et al. 2022), direct persecution as pests, road collisions and electrocution when using power lines to cross roads and/or properties (Linden et al. 2020c, Linden et al. 2022b). It seems that adult males (lone or bachelor males not associated with groups) are particularly vulnerable to being road-killed in certain areas in the Soutpansberg where they have been found to move out of forests through highly transformed landscapes (Linden et al. 2020c). In addition to being persecuted as pests when foraging for food in people’s homes (K. Wimberger pers. Obs., B. Linden pers. obs.), and in campsites (Chapman et al. 1998), they may become pests in large-scale and subsistence fruit orchards or pine plantations surrounding natural forest remnants as they sometimes strip the bark to eat the sap (Droomer 1985, von dem Bussche & van der Zee 1985, B. Linden unpubl. data).
Current habitat trend: Most forest types in South Africa have been fragmented throughout much of their history by repeated and severe climate changes, especially in the Quaternary (Eeley et al. 1999). However, selective logging of these forests from 1870 to 1944, the spread of agriculture, and lately the encroachment of commercial plantation forestry have all exacerbated the fragmentation of the natural forested landscape (Lawes et al. 2000). In the Karkloof Forest, KwaZulu-Natal Province, the rate of habitat loss has slowed from an estimated 10–15% between 1880 and 1940, to 5.7% between 1944 and 1996 (Rycroft 1944; Lawes et al. 2004). In KwaZulu-Natal overall, there was a 20.4% loss of natural habitat from 1994 to 2011, with an average loss of 1.2% per annum (Jewitt et al. 2015). In the eastern part of the Soutpansberg range, expansion of forest plantations between 1990 and 2006, wood collection and settlement expansion are significant reasons for an observed reduction of 20% in indigenous forest and woodland cover (Munyati & Kabanda 2009). Data confirm an ongoing loss of forest habitat within the assessment region between 2000 and 2014: Afrotemperate, Coastal, Scarp and Lowveld Riverine Forest has declined by between 0.3–3.5% (F. Daniels unpubl. data) between 2000 and 2013; and 1.6 ± 1.9% for forest cover overall between 1990 and 2013 (A. Skowno unpubl. data). Future analyses should incorporate spatially explicit patterns of forest loss into this assessment.
Conservation
We argue for separate conservation management of the three distinct genetic entities defined by Dalton et al. (2015), as subspecies need to be conserved to prevent the loss of genetic diversity in the species, which is an essential part of biodiversity conservation. This needs to apply in the wild as well as in captivity (zoological gardens and primate rehabilitation facilities). In particular, rehabilitation facilities which aim at establishing groups and releasing them in the wild must ensure that different subspecies are not mixed and able to interbreed in captivity and that individuals from any of the three subspecies are not reintroduced into another subspecies range resulting in interbreeding in the wild. Similarly, if wild groups are relocated, such as when provincial conservation authorities conducted relocation programmes for monkeys causing damage to plantations (Droomer 1985; von dem Bussche & van der Zee 1985), they should be translocated to the correct geographic area for the subspecies. Strict guidelines are therefore needed for zoological gardens, rehabilitation centres and provincial conservation authorities to conserve the genetic distinctness of the three subspecies.
Protected area expansion and proper management of indigenous forests are the primary interventions for this species. Some populations of the three subspecies occur in protected areas across their range. For example, C. a. labiatus in Hogsback Forest Reserve; C. a. schwarzi in Happy Rest Provincial Nature Reserve, Entabeni and Ratombo parts declared Forest Reserves, and Luvhondo Private Nature Reserve in the Soutpansberg; and C. a. erythrarchus in the Lubombo Transfrontier Conservation Area. Protected area expansion could focus on reclaiming and restoring non-viable or cleared commercial forest plantation areas, particularly those that had been planted in areas conducive to indigenous forest growth including riparian forests along streams and rivers. Samango Monkey life-history traits necessitate large forest areas, which means identifying large habitat patches that are close to one another and minimising disturbance in and on the edge of these patches as a key management strategy (Lawes et al. 2000). Conservation planning should thus continue to connect forest fragments to create viable subpopulations and ensure the persistence of metapopulations (Swart & Lawes 1996), especially in Afromontane and Scarp forests (Eeley et al. 1999). For example, in Maputaland, a new transfrontier landscape is being designed that will, amongst other species, presumably benefit C. a. erythrarchus immensely by extending the existing Lubombo Transfrontier Conservation Area to the south (Smith et al. 2008). For C. a. schwarzi, larger forest patches in the eastern Soutpansberg (e.g. Luonde, Entabeni and Thathe Vondo) are key areas to ensure the Samango’s long term persistence in that part of the mountain range. Forests in the eastern part of the Soutpansberg are embedded in an anthropogenically highly transformed landscape. Efforts to create corridors connecting these main forests patches should be a conservation focus to facilitate gene flow between these largely isolated populations and to offer safe travel routes supporting (re)colonisation of forests patches (Linden 2020b). This could be achieved, for example, through canopy crossing structures over roads, green corridors along riverine forests or engagement with timber plantation management as Samangos are known to utilise both Blue Gum and pine trees for travel (Linden et al. 2020c). On the escarpment, Woodbush and Magoebaskloof forests are important sites as they are both large, contiguous indigenous forest patches.
To improve effective forest management, active engagement with communities and private landowners should be initiated as much as possible to create awareness and give advice on appropriate management and protection of indigenous forests under their stewardship. Forest guards should be trained to reduce poaching and disturbances within forests. Human-wildlife conflict could be reduced by educating surrounding communities on proper waste and refuse control in residential areas, as well as securing potential attractants such as vegetable gardens, subsistence farming and chicken coops with monkey-proof enclosures or by physically chasing the monkeys away. Intentional feeding of monkeys should be prevented. Communities should also be encouraged to plant indigenous garden trees rather than planting exotic ones to make gardens only as attractive to Samango Monkeys as the surrounding indigenous forest habitat. Indigenous species often have less attractive fruit and flowers than those offered by exotic plants (for example, protein and fat rich acorns, such as in Hogsback (Wimberger et al. 2017).
In general, conservationists should identify road collision hotspots for this species across the assessment area and erect canopy bridges to facilitate safe road crossing. Public awareness campaigns about Samango Monkey road collisions are conducted in the Soutpansberg area through bumper stickers (“I brake for Samango Monkeys”) in conjunction with local newspaper articles informing people about the problem (B. Linden unpubl. data). Similarly, power line infrastructure should be secured/insulated to reduce electrocution incidents.
Recommendations for land managers and practitioners:
- Captive breeding programmes are not needed for the species if the long-term persistence of Samangos throughout their current range is ensured through proper management. In situ conservation is the only logical approach for ensuring long-term Samango Monkey persistence in the country. An important part of ensuring the latter is the identification and creation of corridors linking local subpopulations as well as proper management and protection of their forest habitat.
- Managers should develop a Biodiversity Management Plan for this species to sustain a genetically diverse, resilient metapopulation amongst remaining forest patches. Engagement with the private sector will be key in this regard.
- Managers should continue to monitor the cutting down of trees and hunting within or on the edge of forest patches and enforce penalties.
- Due to the differing landownership types in the range of the Samango Monkey (private, communal, state) different management approaches must be formulated and areas should ideally be managed by one entity only to ensure appropriate management strategies.
Research priorities:
Samango Monkey research in the Soutpansberg is ongoing, but more research in other locations of the assessment area is needed. Current research priorities include:
- Collection and collation of all Samango Monkey distribution data (including from the literature) and analysis of specimens to delineate boundaries and/or hybrid zones between the subspecies.
- Surveys to determine current distribution and density across forest fragments, including demographic research.
- Further taxonomic resolution of the Cercopithecus complex is needed to establish if southern and eastern African Cercopithecus monkeys are a separate species (C. albogularis) from Central African Cercopithecus monkeys (C. mitis).
- For C. a. erythrarchus, further research is needed to identify the extent of occurrence further south along the South African coast (and especially around the Umfolozi/Imfolozi area) and further north in Zimbabwe and Mozambique.
- For C. a. schwarzi, further research is needed to identify the extent of occurrence including southern limits in South Africa, and specifically northern limits outside of the country (Mozambique and Zimbabwe) to clarify endemism.
- Data on ranging behaviour of lone males is important for generating parameters relevant to metapopulation persistence (such as gene flow rates and effects of fragmentation on population stability).
- Reproduction and breeding success of each subspecies in different habitat types.
- Implementing road-kill mitigation measures in the form of canopy bridges at road-kill hotspots for samango groups to cross roads.
Encouraged citizen actions:
- For property owners and tourists, ensure no feeding is done either directly or indirectly (for example, leaving fruit and seeds out for birds to eat or badly managed bins and rubbish dumps).
- Plant indigenous garden species so that your garden is only as enticing as the surrounding indigenous forest.
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP).
- Contact the Samango Monkey Working Group (Bibi Linden at bibilinden82@gmail.com) to report any Samango Monkey sightings. Please note any injuries/deaths of individuals and possible causes (e.g. through road collisions, electrocutions, domestic dogs), and estimate number of individuals and the types of habitat in which they were seen.
Bibliography
Armstrong AJ, Benn G, Bowland AE, Goodman PS, Johnson DN, Maddock AH, Scott-Shaw CR. 1998. Plantation forestry in South Africa and its impact on biodiversity. The Southern African Forestry Journal 182: 59-65.
Armstrong, A.J. and van Hensbergen, H.J. 1996. Impacts of afforestation with pines on assemblages of native biota in South Africa. South African Forestry Journal 175: 35-42.
Chapman KL, Lawes MJ, Macleod MM. 1998. Evaluation of nonlethal control methods on problematic samango monkeys in the Cape Vidal Recreation Reserve, greater St. Lucia Wetland Park. South African Journal of Wildlife Research 28: 89–99.
Coleman B.T., Hill R.A. 2014. Living in a landscape of fear: the impact of predation, resource availability and habitat structure on primate range use. Animal Behaviour, 88: 165-173.
Cuozzo F.P., Linden J., Sauther M.L., Halajian A., Linden B. 2024. Domestic dogs present a human-induced threat to thick-tailed bushbabies (Otolemur crassicaudatus) in northern South Africa. African Primates, 16: 59-64.
Cords M. 2012. The 30-year blues: what we know and don’t know about life history, group size and group fission of blue monkeys in the Kakamega Forest, Kenya, in: Kappeler P.M., Watts D.P. (Eds.), Long-term field studies of primates. Springer, Berlin, pp. 289–312.
Dale V.H., Joyce L.A., Mcnulty S., Neilson R.P., Ayres M.P., Flannigan M.D., Hanson P.J., Irland L.C., Lugo A.E., Peterson C.J., Simberloff D., Swanson F.J., Stocks B.J., Wotton B.M. 2001. Climate change and forest disturbances. BioScience, 51(9), 723-734.
Dalton DL, Linden B, Wimberger K, Nupen LJ, Tordiffe AS, Taylor PJ, Madisha MT, Kotze A. 2015. New insights into samango monkey speciation in South Africa. PloS one 10: e0117003.
Dandelot P. 1974. Order Primates. Pages 1-43 in Meester J, Setzer H, editors. The Mammals of Africa. An Identification Manual. Part 3. Smithsonian Institution Press, Washington, DC, USA.
Davis-Reddy C.L., Vincent K. 2017. Climate risk and vulnerability: A handbook for southern Africa. Council for Scientific and Industrial Research (CSIR), Pretoria, South Africa.
DEA (Department of Environmental Affairs) 2013. Long-Term Adaptation Scenarios Flagship Research Programme (LTAS) for South Africa. Climate Trends and Scenarios for South Africa. Pretoria, South Africa.
Droomer EAP. 1985. Volume and value loss owing to samango monkey damage in pine stands in the Northern Transvaal. South African Forestry Journal 134: 47–51.
Eeley HA, Lawes MJ, Piper SE. 1999. The influence of climate change on the distribution of indigenous forest in KwaZulu-Natal, South Africa. Journal of Biogeography 26: 595–617.
Emerson SE, Brown JS, Linden JD. 2011. Identifying Sykes’ monkeys’, Cercopithecus albogularis erythrarchus, axes of fear through patch use. Animal Behaviour 81: 455–462.
Emerson SE, Brown JS. 2012. Using giving-up densities to test for dietary preferences in primates: an example with Samango monkeys (Cercopithecus (nictitans) mitis erythrarchus). International Journal of Primatology 33: 1420–1438.
Emerson SE, Brown JS. 2013. Identifying preferred habitats of samango monkeys (Cercopithecus (nictitans) mitis erythrarchus) through patch use. Behavioural Processes 100: 214–221.
Fairgrieve C, Muhumuza G. 2003. Feeding ecology and dietary differences between blue monkey (Cercopithecus mitis stuhlmanni Matschie) groups in logged and unlogged forest, Budongo Forest Reserve, Uganda. African Journal of Ecology 41: 141-149.
Engelbrecht F. A. et al. 2016. Chapter 3: Climate Change over South Africa: From trends and projected changes to vulnerability assessments and the status quo of national adaptation strategies, in: South Africa’s 3rd National Communication to UNFCCC, Department of Environmental Affairs, Pretoria.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. CBSG Southern Africa, IUCN SSC Conservation Breeding Specialist Group, Endangered Wildlife Trust, South Africa.
GeoTerraImage. 2015. 1990-2013/14 South African National Land-Cover Change. DEA/CARDNO SCPF002: Implementation of Land-Use Maps for South Africa. Project Specific Data Report.
Groves, C.P. 2001. Primate Taxonomy. Smithsonian Institution Press, Washington, DC, USA.
Groves, C.P. 2005. Order Primates. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World, pp. 111-184. The Johns Hopkins University Press, Baltimore, Maryland, USA.
Grubb, P., Butynski, T.M., Oates, J.F., Bearder, S.K., Disotell, T.R., Groves, C.P. and Struhsaker, T.T. 2003. Assessment of the diversity of African primates. International Journal of Primatology 24: 1301–1357.
Hayward MW, White RM, Mabandla KM, Bukeye P. 2005. Mammalian fauna of indigenous forest in the Transkei region of South Africa: an overdue survey. South African Journal of Wildlife Research 35: 117-124.
Heikamp B. 2008. The role of cheek pouches in seed dispersal: An analysis of dispersal methods within a free ranging group of Sykes’ monkeys (Cercopithecus albogularis) in the Western Soutpansberg, South Africa. M.Sc.Thesis. University of Würzburg.
Henzi SP, Lawes M. 1987. Breeding season influxes and the behaviour of adult male samango monkeys (Cercopithecus mitis albogularis). Folia Primatologica 48: 125–136.
Henzi SP, Lawes M. 1988. Strategic responses of male samango monkeys (Cercopithecus mitis) to a decline in the number of receptive females. International Journal of Primatology 9: 479–495.
Hikel, K. B., Peters, M. K., Linden, J., & Linden, B. 2023. The role of bushpigs (Potamochoerus larvatus) as seed dispersers in indigenous forests in the Soutpansberg Mountain range, South Africa. Animal Biology, 74(1), 37-58.
IPCC 2013. Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge and New York.
Jewitt D, Goodman PS, Erasmus BFN, O’Connor TG, Witkowski ETF. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: implications for biodiversity. South African Journal of Science 111: 1–9.
Kingdon, J. 1997. The Kingdon Field Guide to African Mammals (first edition}. Academic Press, San Diego, California, USA.
Kruger, A. C., & Sekele, S. S. 2013. Trends in extreme temperature indices in South Africa: 1962-2009. International Journal of Climatology, 33(3).
Kruger A.C., Nxumalo M. 2017. Surface temperature trends from homogenized time series in South Africa: 1931–2015. International Journal of Climatology, 37(5): 2364–2377.
Lawes MJ, Henzi SP. 1995. Inter-group encounters in blue monkeys: how territorial must a territorial species be? . Animal behaviour 49: 240–243.
Lawes MJ, Macfarlane DM, Eeley HA. 2004. Forest landscape pattern in the KwaZulu-Natal midlands, South Africa: 50 years of change or stasis? Austral Ecology 29: 613-623.
Lawes MJ, Mealin PE, Piper SE. 2000. Patch occupancy and potential metapopulation dynamics of three forest mammals in fragmented afromontane forest in South Africa. Conservation Biology 14: 1088–1098.
Lawes MJ, Piper SE. 1992. Activity patterns in free-ranging samango monkeys (Cercopithecus mitis erythrarchus Peters, 1852) at the southern range limit. Folia Primatologica 59: 186–201.
Lawes MJ. 1991. Diet of samango monkeys (Cercopithecus mitis erythrarchus) in the Cape Vidal dune forest, South Africa. Journal of Zoology 224: 149–173.
Lawes MJ. 2002. Conservation of fragmented populations of Cercopithecus mitis in South Africa: the role of reintroduction, corridors and metapopulation ecology. Pages 375–392 in Glenn ME, Cords M, editors. The Guenons: Diversity and adaptation in African monkeys. Academic/Plenum Publishers, New York, USA.
Lawes, M. J. 1990. The distribution of the samango monkey (Cercopithecus mitis erythrarchus Peters, 1852 and Cercopithecus mitis labiatus I. Geoffroy, 1843) and forest history in southern Africa. Journal of Biogeography 17: 669-680.
Lawes, M. J. 1992. Estimates of population density and correlates of the status of the samango monkey (Cercopithecus mitis) in Natal, South Africa. Biological Conservation 60(3): 197-210.
Lawes, M. J., Henzi, S. P. and Perrin, M. R. 1990. Diet and feeding behaviour of samango monkeys (Cercopithecus mitis labiatus) in Nygoye forest, South Africa. Folia Primatologica 54: 57-69.
Linden B., Linden J., Fischer F., Linsenmair K.E. 2015. Seed dispersal by South Africa’s only forest-dwelling guenon, the samango monkey (Cercopithecus mitis). African Journal of Wildlife Research 45: 88–99.
Linden B., Dalton D.L., Ralph T.M.C., Silva I., Kotze A., Taylor P.J., 2020a. Adding another piece to the southern African Cercopithecus monkey phylogeography puzzle. African Zoology, 55(4): 351-362.
Linden B. 2020b. Distribution, population status and conservation of the samango monkey (Cercopithecus albogularis schwarzi) in the Limpopo Province, South Africa. PhD thesis, University of Venda, Thohoyandou, South Africa.
Linden B., Foord S., Horta-Lacueva Q.J.B., Taylor P.J. 2020c. Bridging the gap: how to design canopy bridges for arboreal guenons to mitigate road collisions. Biological Conservation, 246, 108560.
Linden B., Dalton D.L., Van Wyk A., de Jager D., Moodley Y., Taylor P.J. 2022a. Potential drivers of samango monkey (Cercopithecus albogularis schwarzi) population subdivision in a highly fragmented mountain landscape in northern South Africa. Primates, 63: 245-260, https://doi.org/10.1007/s10329-022-00981-7
Linden B, Cuozzo FP, Sauther ML, Collinson Jonker W, 2022. Impact of linear infrastructure on South Africa’s primate fauna: the need for mitigation. Folia Primatologica, 93(3-6): 235-253, https://doi.org/10.1163/14219980-20211112
Madisha M.T., Dalton D.L., Jansen R., Kotze A. 2017. Genetic assessment of an isolated endemic Samango monkey (Cercopithecus albogularis labiatus) population in the Amathole Mountains, Eastern Cape Province, South Africa. Primates, 59:197-207, DOI 10.1007/s10329-017-0636-5
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of southern African mammals. Transvaal Museum Monographs 5: 1–359.
Monadjem A, Boycott RC, Parker V, Culverwell J. 2003. Threatened Vertebrates of Swaziland Red Data Book: Fishes, Amphibians, Reptiles, Birds and Mammals. Swaziland Environment Authority, Ministry of Tourism, Environment and Communications, Swaziland.
Moore B., Allard G. 2008. Climate change impact on forest health. Forest Health and Biosecurity Working Papers, Food and Agriculture Organization (FAO) of the United Nations.
Mortsch L.D. 2006. Impact of climate change on agriculture, forestry and wetlands, in: Bhatti J., Lal R., Apps M., Price M. (Eds.), Climate change and managed ecosystems, Taylor and Francis, CRC Press, Boca Raton, pp. 45–67.
Mucina, L. and Rutherford, M.C. 2006. The Vegetation of South Africa, Lesotho and Swaziland. Strelitzia 19. South African National Biodiversity Institute, Pretoria, South Africa.
Munyati C, Kabanda TA. 2009. Using multitemporal Landsat TM imagery to establish land use pressure induced trends in forest and woodland cover in sections of the Soutpansberg Mountains of Venda region, Limpopo Province, South Africa. Regional Environmental Change 9: 41–56.
Norby R.J., Rustad L.E., Dukes J.S., Ojima D.S., Parton W.J., Del Grosso S.J., McMurtrie R.E., Pepper D.A. 2007. Ecosystem responses to warming and interacting global change factors, in: Canadell J.G., Pataki D., Pitelka, L. (Eds.), Terrestrial Ecosystems in a Changing World, The IGBP Series, Springer-Verlag, Berlin Heidelberg, pp. 23–36.
Nowak K, le Roux A, Richards SA, Scheijen CP, Hill RA. 2014. Human observers impact habituated samango monkeys’ perceived landscape of fear. Behavioral Ecology 25: 1199–1204.
Nowak K., Wimberger K., Hill R.A., le Roux A. 2017. Samango Monkeys (Cercopithecus albogularis labiatus) Manage Risk in a Highly Seasonal, Human-Modified Landscape in Amathole Mountains, South Africa. International Journal of Primatology, 38: 194–206, DOI 10.1007/s10764-016-9913-1
Parmesan C. 2006. Ecological and evolutionary responses to recent climate change. Annual Review of Ecology, Evolution, and Systematics, 37: 637–69.
Pazol K, Carlson AA, Ziegler TE. 2002. Female reproductive endocrinology in wild blue monkeys: A preliminary assessment and discussion of potential adaptive functions. Pages 217–232 in Glenn ME, Cords M, editors. The Guenons: Diversity and Adaptation in African Monkeys. Kluwer Academic Publishers, New York, USA.
Roberts A. 1951.. The Mammals of South Africa. The Trustees of the Mammals of South Africa. Central News Agency, Johannesburg, South Africa.
Rutherford M.C., Midgley G.F., Bond W.J., Powrie, L.W., Roberts R., Allsopp, J. 1999. South African country study on climate change. Plant biodiversity: vulnerability and adaptation assessment. Technical Report.
Rycroft HB. 1944. The Karkloof Forest, Natal. Journal of the South African Forestry Association 11: 14–25.
Scheiter S., Gaillard C., Martens C., Erasmus B. F. N., Pfeiffer M. 2018. How vulnerable are ecosystems in the Limpopo province to climate change?. South African journal of botany, 116, 86-95.
Skinner J.D. and Chimimba C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Skowno, A.L., Raimondo, D.C., Poole, C.J., Fizzotti, B. & Slingsby, J.A. (2019). South African National Biodiversity Assessment 2018 Technical Report Volume 1: Terrestrial Realm. South African National Biodiversity Institute, Pretoria.
Smith RJ et al. 2008. Designing a transfrontier conservation landscape for the Maputaland centre of endemism using biodiversity, economic and threat data. Biological Conservation 141: 2127–2138.
Swart J, Lawes MJ, Perrin MR. 1993. A mathematical model to investigate the demographic viability of low-density samango monkey (Cercopithecus mitis) populations in Natal, South Africa. Ecological Modelling 70: 289–303.
Stringer, S.D., Hill, R.A., Swanepoel, L., Dalrymple, S.E., Linden, B. and Koyama, N.F. 2020. Assessing the role of a mammalian frugivorous species on seed germination potential depends on study design: A case study using wild samango monkeys. Acta Oecologica, 106, p.103584.
Swart J., Lawes M.J. 1996. The effect of habitat patch connectivity on samango monkey (Cercopithecus mitis) metapopulation persistence. Ecological Modelling 93: 57–74.
Whiting, M.J., Williams, V.L. and Hibbitts, T.J. 2011. Animals traded for traditional medicine at the Faraday market in South Africa: species diversity and conservation implications. Journal of Zoology 284: 84-96.
Wimberger K., Nowak K., Hill R.A. 2017. Reliance on exotic plants by two groups of threatened samango monkeys, Cercopithecus albogularis labiatus, at their southern range limit. International Journal of Primatology, 38: 151-171, DOI 10.1007/s10764-016-9949-2
Viljoen P.J. 1982. Red-list monkeys re-established at Pafuri. Custos Wildlife Conservation Magazine, Kruger National Park, South Africa.
von Maltitz G, Geldenhuys CJ, Lawes MJ, Eeley H, Aidie H, Vink D, Fleming G, Bailey C. 2003. Classification system for South African indigenous forests. An objective classification for the Department of Water Affairs and Forestry. ENV-P-C 2003-017. Environmentek, CSIR, Pretoria, South Africa.
von dem Bussche GH, van der Zee D. 1985. Damage by Samango monkeys, Cercopithecus (mitis) albogularis, to pine trees in the Northern Transvaal. South African Forestry Journal 133: 43–48.
Yahaya, I., Xu, R., Zhou, J., Jiang, S., Su, B., Huang, J., Cheng, J., Dong, Z., & Jiang, T. (2024). Projected patterns of land uses in Africa under a warming climate. Scientific Reports, 14(1), 12315. https://doi.org/10.1038/s41598-024-61035-0.