Sable Antelope
Hippotragus niger

2025 Red list status
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Hippotragus niger – (Harris, 1838)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Hippotragus – niger
Common Names: Sable antelope (English), Swartwitpens (Afrikaans), Ngwaladi, Ingwalathi, Umtjwayeli (Ndebele), Kgama (Sesotho), Kwalatê, Kukurugu, Pôtôkwane (Setswana), Impalampala, Ngwarati (Swati), Mhalamhala (Tsonga), Phalaphala (Venda), Iliza (Xhosa), Impalampala (Zulu) Antílope Sable Negro (Spanish; Castilian), Hippotrague noir (French), Mbarapi (Swahili), Rappenantilope (German)
Synonyms: No synonyms
Taxonomic Note:
Four subspecies are usually recognised: H. n. subsp. niger, H. n. subsp. kirkii, H. n. subsp. roosevelti and the isolated Giant Sable (H. n. subsp. variani) from Angola. As for many other antelope species, the validity and precise distribution of most of the described subspecies are uncertain. An extensive study of the geographical genetic structure of Hippotragus niger identified three genetic subdivisions representing a Kenya and east Tanzania clade (H. n. subsp. roosevelti), a west Tanzania clade (H. n. subsp. kirkii), and a southern African clade (H. n. subsp. niger; Pitra et al. 2002; Pitra et al. 2006), which corroborated the findings of Matthee and Robinson (1999) that delineated a genetic barrier between the east and southern African (Angola, Zambia and Malawi southwards) clades, thus cautioning against translocations between the two areas.
Red List Status: VU – Vulnerable, D1 (IUCN version 3.1)
Assessment Information
Assessors: Muller, K.L.1, Russo, I.M.2, da Silva, J.M.1, Kruger, J.3 & Selier, S.A.J.1
Reviewers: Nel, L.4 & Taylor, A.5
Institutions: 1South African National Biodiversity Institute (SANBI); 2Cardiff University; 3Limpopo Department of Economic Development, Environment and Tourism (LEDET), 4SA Hunters and Game Conservation Association, 5Panthera
Previous Contributors: Parrini, F., Koen, J., Dalton, D. & Eksteen, J., Mallon, D.P., Child, M.F., Selier, J. & Birss, C.
Assessment Rationale
The Sable Antelope is a charismatic species on the edge of its range within the assessment region. The current wild population within the natural distribution range is estimated at 1,107 individuals, of which 664 – 775 are considered as mature individuals. The population estimate includes state-owned protected and privately managed subpopulations within the natural distribution range but excludes all subpopulations outside the natural distribution range. Although South Africa’s state-owned protected areas are mandated with conserving biodiversity, many are currently falling short due to financial limitations, management challenges, and competing land-use pressures (Patel et al. 2023). This shortfall highlights the importance of exploring the role of the private sector in contributing to conservation goals. While private land can support important biodiversity outcomes, it is not appropriate to shift the responsibility for national conservation targets onto the private sector, whose primary focus is typically on economic productivity. Nevertheless, such commercial activities frequently yield important secondary conservation benefits, highlighting the potential for economic utilisation and ecological stewardship to support and enhance one another (Shumba et al. 2020; Taylor et al. 2021).
There is a continuing decline in most state-owned protected subpopulations, however, the largest subpopulation in Kruger National Park (KNP) as well as the Loskop Dam Nature Reserve subpopulation are showing increasing population trends. The number of mature individuals in KNP is currently estimated at 256 – 299. Over the past three generations (2000 – 2024), the total population has increased considerably, increasing from 79 to 427 individuals. In fact, KNP and Loskop Dam Nature Reserve indicated a 441% and 26% subpopulation increase over three generations, respectively. For the five remaining state-owned protected subpopulations within the natural distribution range; Atherstone Nature Reserve and Messina Nature Reserve in Limpopo Province, Borakalalo Nature Reserve and Kgaswane Mountain Nature Reserve in North West Province and Ohrigstad Nature Reserve in Mpumalanga there has been an overall average decline of approximately 75% over three generations. Overall, there is a population increase of 51% in the past three generations in the assessed state-owned protected areas, mainly due to the subpopulation increase in KNP between 2000 and 2024. The historical decline of Sable Antelope in KNP was initially attributed to habitat deterioration and the establishment of artificial water-points in the early 2000s that resulted in higher densities of prey species around previously waterless areas with associated higher predation on such species including Sable Antelope. However, habitat decline was later ruled out as the primary cause. As a low-density species, Sable Antelope are negatively affected by high densities of other herbivores due to their selective feeding habits and competition for resources. Reasons for recovery in this subpopulation remains understudied.
Sable Antelope present on private wildlife reserves and wildlife ranches within its natural distribution range were assessed for wildness, revealing that 40% may be considered wild, thereby adding a total of 443 individuals to the national total eligible for the Red List assessment. The general consensus is that privately managed subpopulations are increasing. However, the number and extent of wild subpopulations in the private sector that may be eligible for inclusion of the Red List remain uncertain. It should also be noted that the genetic integrity of such subpopulations has not been assessed. This uncertainty underscores the need for future improved assessments to evaluate the wildness of these subpopulations.
Overall, while some subpopulations within state-owned protected areas have shown increases, others have declined. It is important to note that several state-owned protected subpopulations included in this assessment were not part of the previous assessment, which limits the ability to interpret these trends as a true regional population increase. In the private sector, Sable Antelope numbers may have increased; however, this could also reflect improved reporting or inclusion of more animals in the current assessment compared to the previous assessment. Therefore, we list Sable Antelope as Vulnerable D1 due to small population size with mature individuals <1000. Habitat deterioration through ecological mismanagement in state-owned protected areas as well as the effects of climate change is a concern and likely to continue if key interventions are not implemented. Although suitable habitat exists for sable antelope on private land, habitat fragmentation caused by game fences remains a concern.
It is recommended that centralised databases be established to accurately monitor and manage Sable Antelope subpopulations across both state-owned protected areas and in the private sector. Surveys should identify further private subpopulations that qualify for inclusion in the Red List and that may contribute to improving the conservation status of the species in future. Key interventions in state-owned protected areas should focus on correct habitat and wildlife management for the species (for example, managing for low-density species, de-stocking competing herbivores, reducing artificially high predator numbers, and fire management), increasing overall habitat available within the natural distribution range and developing a national metapopulation plan for sustaining the genetic diversity and resilience of the species. The private sector should facilitate defragmentation of habitat through dropping fences and forming conservancies or prioritise extensive management approaches of private subpopulations with minimal interventions over intensive management of animals. Policymakers should consider implementing incentive-driven instruments to encourage the private management of sable antelope subpopulations in extensive systems over intensive environments.
Regional population effects: Although the range of this species is on the edge of the assessment region, its range is not continuous. Private and state subpopulations are isolated since they are surrounded by fences. The only dispersal routes that might exist are between KNP, Zimbabwe and Mozambique (the Great Limpopo Transfrontier Park). However, there is no confirmation that immigration takes place. Translocations of Sable Antelope within the private sector are primarily carried out to enhance genetic diversity and maintain population fitness, however it is uncertain whether state-owned protected areas follow similar management practises. Thus, although the regional population is not expected to be a sink for the global population, there is unlikely to be a rescue effect from immigration, so, the Vulnerable listing remains.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Muller KL, Russo IM, da Silva JM, Kruger J & Selier SAJ. 2025. A conservation assessment of Hippotragus niger. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The Sable Antelope occurs in the savannah woodlands of south-eastern Africa, with an isolated population (Giant Sable, H. n. subsp. variani) in Angola. Estes (2013) notes that the species is mainly associated with the well-watered Miombo woodland zone. Some botanists argue that South Africa historically contained miombo along the northern Limpopo, of which patches still exist (Saidi & Tshipala-Ramatshimbila 2006). It is thus speculated that natural subpopulations in those areas might be remnants of historical miombo conditions.
In southern Africa, Sable Antelope occur in Zimbabwe, north-eastern Botswana, scattered subpopulations in Mozambique, the north-eastern part of the Caprivi Strip in Namibia, and South Africa (Skinner & Chimimba 2005). Within the assessment region, Sable Antelope naturally occur in the Lowveld of eastern Mpumalanga, northern Limpopo, and west into the North-West Province. Interestingly, the type specimen was collected and described by Harris (1838) in the Magaliesberg, North West Province (and thus probably why it adorns the province’s coat of arms; Power 2014), which is considered the south-western limit of the historical distribution, while the south-eastern limit is the Crocodile and Komati Rivers (Skinner & Chimimba 2005; Estes 2013). Sable antelope have been eliminated from large parts of their former African range by bushmeat hunting, habitat loss to agricultural expansion, habitat degradation (for example, bush encroachment) and competition with other grazers, including livestock (Skinner & Chimimba 2005).
The species has been reintroduced patchily into many areas of their former range. However, they have also been widely introduced to both state-owned protected and private areas outside its historical range, such that there are extra-limital subpopulations in the Northern Cape, Western Cape, Eastern Cape, Free State, Gauteng and KwaZulu-Natal provinces. For example, 10 individuals were introduced to Karkloof Nature Reserve, KwaZulu-Natal in 1986 (Skinner & Chimimba 2005); as well as being introduced to Sandveld, Willem Pretorius and Koppies Dam Nature Reserves in the Free State Province, Rooipoort and Tswalu Kalahari Reserves and Mokala National Park in the Northern Cape Province. A subpopulation was reported in 2021 to occur in Leeuwfontein Nature Reserve in the Gauteng Province. Additionally, they have been introduced into the Mkhaya Game Reserve in Eswatini (Skinner & Chimimba 2005).
The natural distribution range is used as a guideline for including subpopulations in this assessment and excludes the ‘mixed bushveld’ areas where some subpopulations are performing well. The underlying geology of Loskop Dam Nature Reserve is the same as for Limpopo’s Waterberg (Waterberg Sandstone) and is thus included within the assessment. Further analysis of habitats is required to justify extra-limital subpopulations for inclusion in the assessment.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 600 – 700
Elevation Upper Limit (in metres above sea level): 1500 – 1700
Depth Lower Limit (in metres below sea level): NA
Depth Upper Limit (in metres below sea level): NA
Depth Zone: NA
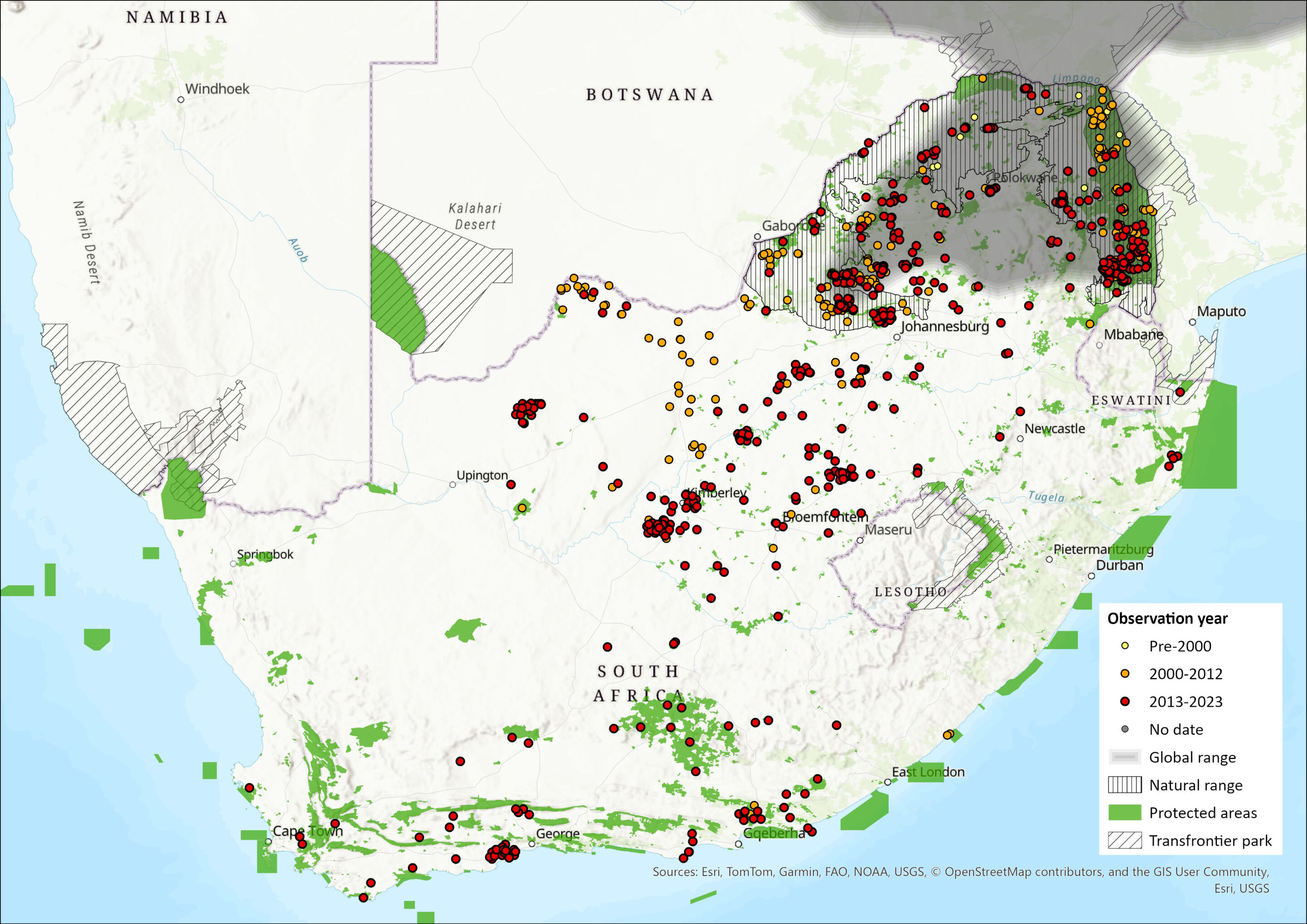
Map
Figure 1. Distribution records for Sable Antelope (Hippotragus niger) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Eswatini | Extant | Introduced | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia -> Caprivi Strip | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: No
FAO Area Occurrence
FAO Marine Areas: No
Climate change
Southern Africa is a predominantly semi-arid region characterised by extremely variable rainfall patterns coupled with frequent drought episodes and floods (Washington & Preston 2006). It is also widely recognised as one of the most vulnerable regions to climate change because of low levels of adaptive capacity (particularly among rural communities), combined with a high dependence on rain-fed agriculture (IPCC 2014). Conditions in Africa are predicted to get progressively hotter and drier with the proportion of arid and semi-arid lands likely to increase by 5–8% by 2080 (Boko et al. 2007). Mean seasonal temperatures are predicted to increase, and El Niño Southern Oscillation effects, fires and severe weather anomalies are more likely to be more common in southern Africa (Davis 2011; IPCC 2014). Extreme droughts, floods and climate warming, which are the major climate change factors, are thus likely to affect wildlife resources in southern Africa (Chidumayo et al. 2011). Climate can affect Sable Antelope populations indirectly by excessive temperatures, rainfall or through bottom-up effects on plant productivity. Bottom-up control mechanisms are based on the view that herbivore populations are limited by forage quality and quantity (Gandiwa 2014). Consequences are subpopulation declines as a result of the decline in suitable habitat within fenced areas, as suitable habitat shifts, and dispersal of the animals is constrained by fences.
Temperature and rainfall pattern changes: The physiology of the Sable Antelope is adapted to the temperate climates of savannas and woodlands in southern Africa (Cain et al. 2012). Rising temperatures and changing rainfall patterns can lead to habitat degradation, therefore affecting the grasslands that are crucial for their survival (Estes 2013). Prolonged periods of drought associated with climate change could have significant effects on water availability for Sable Antelope, who require frequent access to water sources (Cain et al. 2012). Changes in rainfall patterns and the drying of natural water sources may force antelopes to travel longer distances to find water, potentially increasing their exposure to predators and competition for resources. The subpopulation in KNP declined following droughts (Owen-Smith & Mills 2006).
Forage quantity and quality: Sable Antelope are selective grazers, typically preferring grasses with high nutritional value (Cain et al. 2012). Climate-induced changes in plant phenology, including earlier or delayed grass growth due to altered rainfall patterns, may affect food availability. This could cause shifts in grazing ranges, potentially forcing Sable Antelope to travel further for food resources. Parrini and Owen-Smith (2009) found that early autumn fires can help alleviate nutritional stress in Sable Antelope during the dry period, since there is sufficient soil moisture. This suggests that climate change, which can alter rainfall patterns, may impact the availability of soil moisture and, in turn, affect the nutritional stress of Sable Antelope.
Fire management: Variations in temperature and rainfall patterns may also influence fire management in savanna systems (Karp et al. 2023). Pacifici et al. (2015) investigated how fire management affects Roan (Hippotragus equinus), Sable and Tsessebe antelope (Damaliscus lunatus) occurrence and density in KNP. Sable Antelope showed preference for large burnt areas after fires in spring where they make use of the green flushes. Studies indicate that prolonged droughts and shifts in vegetation types may reduce the quality and quantity of forage, impacting their health and reproductive success (du Toit et al. 2003; Owen-Smith 2008). Additionally, climate change can exacerbate competition with other herbivores and increase the risk of disease transmission (Kupika et al. 2017), further threatening Sable Antelope populations.
Invasive species: Climate change can exacerbate the spread of invasive species, particularly in areas where temperature and moisture regimes become more favourable to non-native plants or animals. For example, invasive grass species could outcompete native grasses in savanna ecosystems (Milton 2004; O’Connor & van Wilgen 2020), changing the habitat structure and forage availability for Sable Antelope.
Population
The overall national wild population estimate included in this assessment is 1,107 individuals constituting 664 – 775 mature individuals (60–70% mature population structure). This estimate includes both state-owned protected areas and privately managed subpopulations within the species’ natural distribution range. The previous assessment (2016) estimated the total population at 818 – 1,346 mature individuals, including subpopulations from state-owned protected areas and subpopulations on private land that are considered wild. This may indicate an overall population decline; however, it is not clear how previous estimates were calculated, therefore, accurate conclusions regarding overall population decline cannot be made. Current estimates of subpopulations in the private sector suggest that 40% may be considered wild. Therefore, the observed decline in total population from 2016 to 2024 is likely due to decreases in subpopulations within state-owned protected areas, as well as uncertainty in data used between the two assessments. In addition, population estimates and the number of subpopulations on private land that could be considered remains uncertain. This highlights the need for improved participation of the private sector in future assessments.
State-owned: Within the assessment region, a total of 664 individuals (398 – 465 mature individuals) currently (2023 – 2024) occur on seven state-owned protected areas within the natural distribution range, including an additional state-owned property, although not formally proclaimed as a protected area (Table 1). The largest sable antelope subpopulation, in KNP, crashed from an estimated 2,240 individuals in 1986 to approximately 507 in 1999 (Grant & van der Walt 2000; Grant et al. 2002). However, the KNP subpopulation has appeared to stabilise between 2004 and 2012: estimated at 400 in 2004 (Friedmann & Daly 2004), and 385 individuals in 2012 (Ferreira et al. 2013). The initial decline was attributed to deteriorating habitat quality and increased predation pressure following artificial water-point installation (Harrington et al. 1999). Owen-Smith et al. (2012) proposed that the reason behind the lack of population recovery is a combination of reduced herd size (and thus increased vulnerability) and Allee effect (lowered probability of finding mates). Asner et al. (2015) found no differences in habitat features between the areas where herds persist and areas from which herds have disappeared, suggesting that deteriorating habitat conditions were not the primary reason for the decline. A key consideration is that sable antelope are naturally low-density species, resulting in competition for space preceding competition for food – an ecological dynamic observed on reserves in Limpopo Province (L. Nel, pers. comm. 2025). A census performed in 2023 estimated the total KNP subpopulation at 427 individuals (256 – 299 mature individuals; Greaver et al. 2024), thus indicating an increasing trend of 11% since 2012.
Subpopulations in Atherstone Nature Reserve, Limpopo Province, declined by 40% (25 to 15 individuals) between 2016 and 2024. The subpopulation in Messina Nature Reserve, Limpopo, became functionally extinct in 2024, declining from nine individuals in 2016 to only one remaining individual. Similar trends were observed in the North West Province, with subpopulations in Borakalalo Nature Reserve decreasing from seven to one and Kgaswane Mountain Nature Reserve from 39 to 17 individuals, both between 2016 and 2023. A total of 12 individuals were introduced to Ohrigstad Dam Nature Reserve, Mpumalanga Province, in 2021, but census data from 2024 indicated a remaining total of nine individuals. In contrast, Loskop Dam Nature Reserve, Mpumalanga, saw a 35% increase (40 to 54 individuals) in its subpopulation from 2016 to 2023. Sable antelope are occasionally observed in Manyeleti Nature Reserve, Mpumalanga, but individuals mainly move into the reserve from KNP to access water sources and do not remain in Manyeleti Nature Reserve permanently. A subpopulation of Sable Antelope occurs on state-owned land within the South African Air Force (SAAF) training grounds in Limpopo Province. Although the area is not formally proclaimed as protected, it supports an estimated 140 individuals across approximately 43 km2. Due to limited capacity and funding, active management of this subpopulation is minimal (J. Kruger, LEDET, pers. comm. 2025).
Seven state-owned protected subpopulations of Sable Antelope have become locally extinct between 2015 and 2024. It is uncertain whether these subpopulations were included in the previous Red List assessment. For example, subpopulations within four nature reserves located in the Limpopo Province became locally extinct between 2019 and 2024 namely Blouberg Nature Reserve, Hans Merensky Nature Reserve, Letaba Ranch Nature Reserve and Wonderkop Nature Reserve. Pilanesberg National Park, North West Province, had four individuals in 2014 and went locally extinct in 2015. The subpopulation in Faan Meintjies Nature Reserve, North West, also went locally extinct in 2022. Four adult male sable antelope were introduced into Songimvelo Nature Reserve, Mpumalanga, in the winter of 2019, however, during a game count conducted in October 2019, only two individuals were counted. Since then, only one individual was occasionally sighted during patrols. During the 2023 game count, no sable antelope were located, suggesting that there are none left in Songimvelo Nature Reserve (J. Myeni pers. comm. 2024).
Table 1. Summary of population size estimates for Sable Antelope (Hippotragus niger) in state-owned protected areas within the natural distribution range. This is based on available data only and thus may underestimate total numbers.
| Province | Number of state-owned protected areas | Subpopulation total (2023-2024) |
|---|---|---|
| Limpopo | 3* | 156 |
| Limpopo/Mpumalanga | 1 | 427 |
| Mpumalanga | 2 | 63 |
| North West | 2 | 18 |
| Total | 8 | 664 |
*One state-owned property managed by SAAF not formally proclaimed as protected area.
Private: A substantially larger number of Sable Antelope are believed to occur on privately-owned wildlife reserves and game ranches throughout South Africa. However, precise population figures at the provincial level remain unknown, primarily due to insufficient or unavailable data from provincial conservation authorities. In 2013, rough estimates placed the national population number on private land at 25,000 (Bezuidenhout 2013). The North West Department of Economic Development, Environment, Conservation & Tourism (DEDECT) reported a total number of 14,611 Sable Antelope present on 586 private wildlife ranches in 2024 based on permit data (J. Power, DEDECT, pers. comm. 2025). The Northern Cape Department of Environment and Nature Conservation (DENC) indicated that a total number of 133 private wildlife ranches have valid permits for keeping sable antelope, but the estimated number of sable antelope on these private game farms remains uncertain (M. Smit, DENC, pers. comm. 2025). The Mpumalanga Parks and Tourism Agency (MPTA) reported a total of 580 sable antelope occurring on 31 private wildlife ranches between 2022 and 2024.
However, disputes remain over whether subpopulations on private game reserves and ranches should be considered wild due to their management regimes. Subpopulations dependent on direct intervention are not considered wild if they would go extinct within 10 years without intensive management (IUCN Standards and Petitions Subcommittee 2014). Based on data collected in 2016, at least 63% of privately managed assessed subpopulations were kept in breeding camps or enclosures (N = 76 private properties, Endangered Wildlife Trust, unpubl. data). A framework used to measure the wildness of these managed subpopulations indicated that only 2-10% could be considered wild (Child et al. 2019). A separate follow-up wildness assessment of subpopulations (N = 68) on a total of 46 private wildlife ranches was performed in 2024 using the framework by Child et al. (2019). Results indicated that 40% of assessed subpopulations may be considered wild, of which 56% occur within the natural distribution range (SANBI, 2024, unpubl. data). This adds a total of 443 individuals to the population estimate for state-owned protected areas that are eligible for the Red List. Consequently, 1,107 individuals (664 – 775 mature individuals) are assessed as the overall national population.
When excluding assessed properties (N = 11) managing Zambian Sable (thought to be H. n. subsp. kirkii) from the analysis, 52% of subpopulations (N = 48) may then be considered wild, of which more than 50% occur in the natural distribution range (SANBI, 2024, unpubl. data). This would add 410 individuals to the total population estimate for state-owned protected areas and bring the total estimate to 560 – 654 mature individuals eligible for the Red List. While there is no conclusive subspecies evidence for the Zambian Sable, it is considered a potentially evolutionary significant unit (ESU) of the southern Sable of South Africa (H. n. subsp. niger; Pitra et al. 2006; Jansen van Vuuren et al. 2010; Rocha et al. 2022).
Table 2. Summary of assessed subpopulations (N = 68) of Sable Antelope (Hippotragus niger) in the private wildlife industry. This is based on available data only and thus may underestimate total numbers
| Province | Inside natural range | No. assessed subpopulations | Sable Antelope total | No. subpopulations considered wild | Wild subpopulation total |
|---|---|---|---|---|---|
| Eastern Cape | No | 21 | 639 | 9 | 346 |
| Free State | No | 7 | 140 | 3 | 85 |
| Gauteng | No | 1 | 26 | 0 | – |
| Limpopo | Yes | 31 | 876 | 15 | 443 |
| North West | Yes | 7 | 231 | 0 | – |
| Northern Cape | No | 1 | 34 | 0 | – |
| Total | 68 | 1946 | 27 | 874 |
Sable Antelope have been introduced into a number of reserves outside of the species natural distribution range. Based on current data, at least 2,221 individuals occur collectively on state-owned and private reserves, that could be considered wild (Table 3). However, these subpopulations do not fall within the natural distribution range and cannot be included in the assessment.
State-owned: In the Northern Cape Province, an estimated total of 146 individuals currently (2023 – 2024) occur in four state-owned protected areas, namely Benfontein Nature Reserve (1), Rooipoort Nature Reserve (5), Dronfield Nature Reserve (95) and Mokala National Park (45). The subpopulation on Mokala National Park has increased from 10 to 31 individuals from 2008 to 2012, but based on aerial surveys, has since declined to 15 individuals and have remained stable between 10 – 15 individuals since 2017. In general, aerial count estimates may be an underestimate of actual subpopulation numbers due to the cryptic nature of Sable Antelope. Combining direct observations, camera traps and aerial surveys brings the estimated total subpopulation in Mokala National Park between 40 – 49 individuals (N. Mzileni, SANParks, pers. comm. 2025). Subpopulations in the Free State provincial nature reserves performed well between 2012 and 2018, with a combined total of 685 individuals and an average annual growth rate of 71% during this period. Speculative reasons for extra-limital subpopulation increases included climate change and fire suppression creating favourable savannah systems (E. Schulze pers. comm. 2015). However, from 2018 to 2025, subpopulations in Koppies Dam Nature Reserve and Sandveld Nature Reserve, Free State Province, declined by 42% and 62%, respectively. The subpopulation in Willem Pretorius Nature Reserve, Free State, has increased from 140 to 171 individuals from 2018 to 2025. A total of 13 individuals were reported to occur in Leeuwfontein Nature Reserve, Gauteng Province, in 2021. Subpopulations in Commando Drift Nature Reserve and Great Fish River Nature Reserve, Eastern Cape Province, are currently (2020 – 2021) functionally extinct, with only one remaining individual on both reserves.
Private: One privately-owned wildlife ranch covering >500 km2, Northern Cape Province, currently (2024) houses a total of 406 free-ranging Sable Antelope. Additionally, a free-ranging subpopulation exists within another privately protected area in the Northern Cape, spanning approximately 900 km², with an estimated total of 73 individuals (C. Motau, pers. comm. 2025). In the Western Cape Province, an estimated total of 740 Sable Antelope occur on 60 private wildlife ranches and wildlife reserves between 2020 and 2023, however, the extent of subpopulation wildness on these properties remains uncertain. From assessed wild subpopulations in the private sector (Table 2), another 431 individuals from the Free State and Eastern Cape Provinces can be added to the total eligible for the Red List.
Table 3. Summary of population size estimates for Sable Antelope (Hippotragus niger) outside the natural distribution range.This is based on available data only and thus may underestimate total numbers.
|
Province |
Type |
Number of protected areas/properties |
Subpopulation total (2020-2024) |
|
Eastern Cape |
Private |
7 |
346 |
|
Eastern Cape |
State-owned |
2 |
2 |
|
Free State |
Private |
3 |
85 |
|
Free State |
State-owned |
3 |
410 |
|
Gauteng |
State-owned |
1 |
13 |
|
KwaZulu-Natal |
Private |
Unknown |
Unknown |
|
Northern Cape |
State-owned |
4 |
146 |
|
Northern Cape |
Private |
2 |
479 |
|
Western Cape |
Private |
60 |
740 |
|
Total (Private) |
|
72 |
1,650 |
|
Total (State-owned) |
|
10 |
571 |
|
Overall total outside natural distribution range |
|
82 |
2,221 |
Sable Antelope occur at low densities compared to other ungulates of similar size in semi-arid savannahs (Owen-Smith 2008), ranging from an estimated density of four individuals/km² in the Matetsi area of Zimbabwe (Wilson & Hirst 1977), and three individuals/km² in Matopo National Park, Zimbabwe (Grobler 1973) to a density not exceeding 0.5 animals/km² within the Kruger National Park (Chirima et al. 2013).
Detailed subpopulation structure data are only available for Kgaswane Mountain Nature Reserve, North West Province, where the most recent (2016) composition is reported as (Nel 2016):
- Adult males: 9.5%
- Adult females: 31%
- Sub-adult males: 21%
- Sub-adult females: 24%
- Juvenile males: 5%
- Juvenile females: 9.5%
Thus, the mature proportion of the population is estimated to be between 62–86% (depending on whether one considers the sub-adult female as mature, since being more than two years they could be reproducing in theory). This value is relatively higher than what was reported in 2014, but the subpopulation in the Kgaswane Mountain Nature Reserve has still declined considerably by 56% from 2016 to 2023. Further subpopulation structures should be analysed. Thus, we use a higher estimate of 70% as the upper bound of the estimate. Generation length is estimated to be between 8.4 years (Pacifici et al. 2013) and 9 years (C. Birss unpubl. data), which yields a three-generation window of 25–27 years.
Overall, sable antelope numbers continue to decline in state-owned protected areas, except for recovering subpopulations in KNP and Loskop Dam Nature Reserve, Mpumalanga Province. In contrast, numbers appear to be increasing on private wildlife ranches, though likely underestimated due to a lack of accurate data. Since the initial wildness assessment was performed in 2016, the proportion of privately managed subpopulations that can be considered wild may have increased from between 2 – 10% to 40%. However, it is important to note that different private properties were assessed in each case, so the observed increase may not accurately reflect a true change in wildness. Further data analysis is required to determine the cause for the increase in the proportion of wild subpopulations in the private sector. Uncertainty remains about the wild status of managed subpopulations nationwide and their inclusion in future Red List assessments, underscoring the need for private sector participation in future wildness evaluations.
Population Information
Continuing decline in mature individuals? Unknown
Qualifier: Estimated
Justification: Based on uncertainty of subpopulations included in the previous assessment and some subpopulations added in the current assessment that were not included in the previous assessment. Lack of resources, capacity and expertise for efficient management in state-owned protected areas are projected and suspected to cause continuing decline of mature individuals. In contrast, subpopulations within the private sector are suspected to have increased since the previous assessment.
Extreme fluctuations in the number of subpopulations: Yes
Continuing decline in number of subpopulations: Unknown
Qualifier: Estimated
Justification: At least seven state-owned subpopulations have gone functionally extinct between 2015 and 2024. However, it is uncertain whether such subpopulations were included in the previous assessment.
All individuals in one subpopulation: No
Number of mature individuals in largest subpopulation: 278
Number of subpopulations: 35
Justification: Subpopulations are defined as any fenced area, as there is little exchange between these areas and no natural dispersal. Many more subpopulations exist on private protected areas and wildlife ranches.
Subpopulation Details: A total of eight state-owned protected areas house subpopulations, while a total of 27 subpopulations assessed in the private sector are considered wild.
Number of mature individuals: 664 – 775
Subpopulation trend: Increasing
Subpopulation trend: Increasing
Qualifier: Estimated
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: 10%
Population genetics
While outside the assessment region, a recent range-wide analysis based on complete mitogenomes supported two sympatric and deeply divergent evolutionary lineages (divergence at 1.5 million years ago) in western Tanzania (Rocha et al. 2022). This is possibly due to introgressive hybridisation of a mitochondrial ghost lineage from a relic population. Further phylogeographic subdivisions into three (eastern, central and southern) lineages (divergence at 375,000 years ago) are evident due to climatic events and geomorphological features such as East Africa Rift System and the Eastern Arc Mountains.
Within the assessment region, a single evolutionary significant unit (ESU) is believed to exist corresponding to areas south of the Zambezi River, including Namibia, Botswana, Zimbabwe, south-central Mozambique and South Africa (Rocha et al. 2022). Two haplogroups (S1 + S2 with respective sub-haplogroup b) have been identified south of the Zambezi River based on mitochondrial DNA (Rocha et al. 2022). However, to date, no study has included samples from the assessment region and the species remains understudied from a population genetics perspective and the distribution of these haplogroups/sub-haplogroups are unknown for the assessment region.
The number of wild mature individuals has been estimated at 664 – 775 (average of 720) for the assessment region (see “Assessment Rationale” section). A conversion ratio of 0.1 or 0.3 between Nc (mature individuals) and Ne (effective population size, mature individuals contributing to breeding) will result in Ne of 72 – 216, which are both below the recommended number of 500 individuals per population to ensure long-term survival of the species (Franklin 1980).
Habitats and Ecology
The Sable Antelope is an “edge” species that frequents the woodland/grassland ecotone. They are selective feeders with a preference for fresh growth grasses (40–140 mm) and are dependent on drinking water, travelling to water at two to four days intervals (Cain et al. 2012). Burns that provide green regrowth and/or vleis are key resource areas in the dry season (Parrini & Owen-Smith 2010). Panicum maximum is a key resource grass species in certain areas; Themeda triandra is a highly sought after species too (Parrini 2006). However, they show a broad dietary acceptance for other grass species such as Brachiaria nigropedata, Heteropogon contortus, Digitaria spp. and Eragrostis spp. in the Matobos National Park in Zimbabwe (Grobler 1981). Chrysopogon serrulatus was the main dietary item in Pilanesberg National Park (Magome et al. 2008), however, Sable Antelope became locally extinct from the park since 2015. Tall stemmy species like Hyperthelia dissoluta was commonly eaten in the Percy Fyfe Reserve (Wilson & Hirst 1977), the Kgaswane Mountain Nature Reserve (Parrini 2006) and in the Okavango Delta region in Botswana (Hensman et al. 2012).
The use of burning practices early in the dry season can provide green grass regrowth used by Sable Antelope (Sekulic 1981; Magome et al. 2008; Parrini & Owen-Smith 2010) when the grass regrowth is at least 50 mm (Grobler 1981). Additionally, potential competition with other grazers is a concern in some areas like the KNP where the occurrence of Sable Antelope seems to be restricted by the presence of more abundant grazers, either directly or indirectly through the presence of common predators (Chirima et al. 2013). This then forces Sable Antelope into areas with restricted availability of food (Owen-Smith et al. 2013).
Sable Antelope occur in herds of 10–30 with temporary aggregations of up to 200 (Skinner & Chimimba 2005). There is little dimorphism in body size (Owen-Smith 1988) and females and juveniles form herds, while sub-adult males are often associated with these herds and rarely form bachelor groups (Estes 1991; Parrini 2006). Mature males are believed to be territorial (Estes 1991), however observations in the Kgaswane Mountain Nature Reserve seem to suggest that at times an adult male attaches itself to a herd of females even outside its territory (Parrini 2006). Home range size estimates range from as little as 7.5 km2 in the Percy Fyfe Nature Reserve, Limpopo (Wilson & Hirst 1977), to 118 km2 in KNP (Owen-Smith & Cain 2007).
Current habitat trend: Declining in state-owned protected areas. Anecdotal evidence suggests change in the Lowveld since the early 1900s through bush encroachment but there is little evidence that recent change in habitat quality and structure in KNP could explain Sable Antelope decline (Owen-Smith et al. 2012; Asner et al. 2015). Assessed wild subpopulations in the private sector appear to have increased, but the habitat quality of wild subpopulations remains understudied.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
|---|---|---|---|
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Marginal | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Marginal | – |
Life History
| Generation Length | Justification | Data Quality |
|---|---|---|
| 7 | Considering age at first reproduction as 3 years (for females), z=0.5 and length of the reproductive period 7 years (Grobler 1980; Capon 2011) | medium |
Age at Maturity: Female or unspecified: 2 years (female)
Age at Maturity: Male: 16 – 19 months
Size at Maturity (in cms): Female: 130
Size at Maturity (in cms): Male: 135
Longevity: 16 years (wild); 19 – 22 years (captivity)
Average Reproductive Age: 3 years (female); 2.5 years (male)
Maximum Size (in cms): 140
Size at Birth (in cms): 65 – 75
Gestation Time: 266 days
Reproductive Periodicity: 8 years (female); >10 years (male)
Average Annual Fecundity or Litter Size: 1
Natural Mortality: 16 years
Does the species give birth to live young: Yes
Movement Patterns
Movement Patterns: Not a Migrant
Aggregatory: Yes
Systems
System: Terrestrial
General Use and Trade Information
Sable Antelope are a highly utilised species within the assessment region. It is a high-value, sought-after game species in the private wildlife ranching industry, mainly managed commercially for international and local hunting and live sales purposes (Taylor et al. 2016). Therefore, some subpopulations within the private sector continue to be managed in intensive to semi-extensive systems nationwide (Taylor et al. 2016). Private sector systems typically manage subpopulations separately as breeding and wild stock, usually on the same property. Breeding stocks are typically kept in camp systems (ranging from <1 to >5 km2; based on 68 assessed subpopulations, SANBI, 2024, unpubl. data) with disease control and genetic selection for specific traits and is often sold at auction. In addition, predators are mostly excluded from these camps. Genetic selection involves placing bulls with specific traits with a herd to produce offspring with similar traits, and does not refer to genetic manipulation or engineering, a process of altering on organism’s DNA using laboratory technologies (IFOAM 2014). Wild stock refers to free-ranging Sable Antelope (mainly on the entire wildlife ranching property) without disease management or genetic selection and are primarily managed for international and local hunting purposes. From survey data collected in 2015 from 50 private properties, an average camp size for Sable Antelope (taking the maximum when a range is given) was 2.2 ± 3.6 km2 (A. Taylor, unpubl. data). In 2024, a total of 59% of assessed subpopulations (N = 68; SANBI, 2024, unpubl. data) that are considered breeding stock are kept in camp systems larger than overall reported home range sizes (>2 km2). Therefore, this underscores the value of including privately managed subpopulations in subsequent Red List assessments.
Between 2016 and 2024, a total of 12,906 Sable Antelope were sold live on formal auctions across South Africa. At the end of 2024, a total of 1,577 Sable Antelope were sold on auction, valued at a total exceeding R74 million, making up 12% of the total rare game sold on auction that year. Nearly 900 hunting bulls were sold in 2024 at an average of between R45,000 and R55,000 (AWA 2024). Between 2016 and 2023, a total of 8,506 Sable Antelope were hunted by international clients. The number of international hunts declined in 2020 due to the COVID-19 pandemic but subsequently increased, reaching a total 1,267 by the end of 2023. The international hunting market prices have declined between 2018 and 2019, followed by somewhat stabilising market trends until the end of 2023 (DFFE Professional Hunting Registers, unpubl. data; Fig. 2).
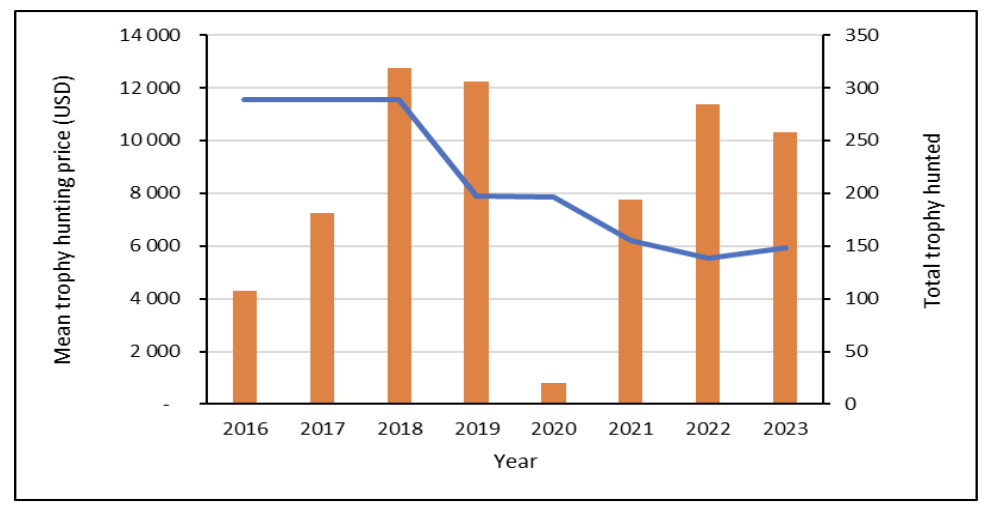 Figure 2. Mean international hunting price (blue line) and total international hunts of (orange bars) roan antelope between 2016 and 2023.
Figure 2. Mean international hunting price (blue line) and total international hunts of (orange bars) roan antelope between 2016 and 2023.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| No | Poaching for bushmeat may be occurring on a small scale but is not a major threat. | Yes | Live sales and local and international hunting. |
National Commercial Value: Yes
International Commercial Value: Yes
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | – | true | – | – |
| 15. Hunting/specimen collecting | – | true | true | – |
| 17. Other (free text) | true | Live animal sales at auctions |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Sable Antelope subpopulations managed in the private wildlife ranching sector are frequently harvested mainly for international hunting purposes due to its significant economic value. Offtake/harvest in the form of hunting from subpopulations in state-owned protected areas does not occur.
Threats
Sable Antelope face several broad-scale threats within the assessment region, most notably habitat mismanagement and the impacts of climate change. At both regional and local levels, a critical concern is the lack of robust, evidence-based management practices within state-owned protected areas. These areas often lack the necessary resources, capacity, and ecological expertise to effectively manage rare, low-density species, despite previous successes on many state reserves. The translocation of individuals into ecologically unsuitable habitats further compounds this issue. In the private sector, the primary threat is the uncertainty surrounding the genetic integrity of sable subpopulations. Although hybridisation with non-native subspecies—likely resulting from intensive management practices—may have occurred in the past, the current genetic status of these populations remains unclear. Additionally, poaching and incidental snaring continue to pose localised threats (Nel 2015). While sufficient habitat exists to support sable subpopulations, key challenges remain: (i) limited resources and capacity, including ecological expertise, within state-owned protected areas to manage rare, low-density species effectively, despite historical success on many state-owned reserves; and (ii) the lack of incentives for the private sector to manage populations extensively, as intensive systems are easier and more cost-effective to maintain.
Habitat mismanagement and ecological pressures: Habitat fragmentation remains a significant issue, particularly within the private sector, while poor habitat management is more prevalent in state-owned protected areas due to a lack of resources for effective management. Anthropogenic impacts such as incorrect fire management and hyper-dispersed water-point distribution continue to pose challenges. However, livestock farming is not a major threat, as many landowners in South Africa have transitioned to wildlife ranching as an alternative land use (Taylor et al. 2016). Studies show that hyper-dispersal of artificial water-points leads to both inflated interspecific grazing competition and predation (Owen-Smith 1996; Harrington et al. 1999; Grant & van der Walt 2000). The Sable Antelope is a low-density species that may be outcompeted by more abundant species depressing grass height through their own grazing (Macandza et al. 2012). Without careful management of competing species numbers, this could be an ongoing problem. The closing of artificial water-points in certain areas in KNP has not led to a recovery of the closely related roan antelope (Ferreira et al. 2010), possibly due to an allee effect where reduced herd vigilance causes increased juvenile mortality from predation and declining population numbers (Owen-Smith et al. 2012). Similar negative effects may be observed in Sable Antelope subpopulations in KNP without management interventions. Predation is an indirect threat, mediated by an increase in the abundance of high-density species in the same areas as Sable Antelope, which attracts more predators to those areas (Owen-Smith & Mills 2006). Sable Antelope have a high dietary tolerance but might be restricted to areas with low availability of nutritious food types by the presence of more abundant species and hence increased predation risk in more suitable areas. Sable Antelope went locally extinct in Madikwe Game Reserve in 2009 due to a combination of grazing competition, high predation rates (specifically Spotted Hyaenas, Crocuta crocuta, preying on calves), and a deterioration in habitat quality due to bush encroachment (P. Nel, pers. comm. 2015). These individuals were reintroduced from managed stock and perhaps were also ecologically naïve. Parasitism may also pose a significant threat to Sable Antelope survival, particularly under conditions of poor ecological management. Grobler (1981) identified parasitism as the primary cause of mortality in Sable Antelope populations in Matopos, Zimbabwe. The main external parasites included Rhipicephalus appendiculatus, Rhipicephalus evertsi, and larvae of Chrysomya bezziana, while notable internal parasites were species of Haemonchus and Taenia multiceps.
Genetic integrity and translocation concerns: Poorly planned translocations can expose Sable Antelope to unsuitable habitats (Basson 1991; Owen-Smith 2003) and risk genetic mixing of H. n. subsp. niger with other subspecies, potentially causing outbreeding depression (Arnold 1992). Anthropogenic hybridisation may result from human-driven changes in Sable Antelope abundance and distribution on private properties (Rhymer & Simberloff 1996; Allendorf et al. 2001). It is suggested that some individuals from Zambian and/or the Eastern evolutionary significant units are widespread in the private sector and are frequently advertised on formal auctions (Shepstone 2022; Kriek n.d.). However, the genetic makeup of such private subpopulations is unknown (Pitra et al. 2002; B. Reilly pers. comm. 2024) and distinguishing between subspecies or hybrids without genetic testing is challenging, as some individuals exhibit traits from multiple ESUs (Shepstone 2022). Further survey work and ground-truthing is needed to establish genetic purity of subpopulations managed in the private sector and identify private subpopulations that can enhance the resilience of the overall wild population. Thus far, hybridisation between subspecies of sable antelope has not been reported, however it is suspected because extensive translocations of wildlife throughout South Africa is threatening the genetic integrity of a number of ungulate taxa such as the Blue and Black Wildebeest (Connochaetes taurinus and C. gnou; Grobler et al. 2011), Black-faced and Common Impala (Aepyceros melampus petersi and A. m. melampus; Green & Rothstein 1998), Grevy’s and Plains Zebra (Equus grevyi and E. quagga; Cordingley et al. 2009) and Bontebok and Blesbok (Damaliscus pygargus pygargus and D. p. phillipsi; van Wyk et al. 2013). The consequences of anthropogenic hybridisation include reduced fertility in the rare taxon, and genetic swamping or assimilation (Levin et al. 1996). To have viable and resilient populations for the future, conservation management strategies must preserve the genetic integrity and diversity in local subpopulations. Management plans on private wildlife ranches must be in place and should include measures to prevent the risk of hybridisation between closely related Sable Antelope subspecies.
Conservation capacity and management constraints: Traditionally, state-owned protected areas have been regarded as the most secure environments for sustaining Sable Antelope populations. However, recent population trends within the assessment region indicate a decline in their conservation efficacy, raising concerns about the long-term viability of these subpopulations. In light of this, there is growing consideration of augmenting or reintroducing Sable Antelope using individuals from privately managed populations. These concerns are not without merit. Privately managed stock are frequently maintained under intensive conditions, including predator-free enclosures, supplemental feeding, veterinary care, and controlled breeding. Such artificial environments reduce exposure to natural selective pressures, particularly predation, which can result in the relaxation of predator-driven selection. Consequently, individuals may fail to develop key anti-predator behaviours such as vigilance, flight responses, or cohesive group defence strategies (Selier et al. 2018). When released into predator-rich environments, these behavioural deficiencies may increase their vulnerability to predation, as has been observed in reintroduced Oribi (Ourebia ourebi) populations (Grey-Ross et al. 2009). However, further investigation into the ecological preparedness of private sector animals is required to substantiate these concerns. Despite the potential role of the private sector in conservation efforts, the likelihood of state conservation agencies purchasing Sable Antelope from private owners remains extremely low. Moreover, the private wildlife industry is generally unlikely to donate animals for state reintroduction efforts. As a result, the persistence of Sable Antelope within state-owned protected areas is increasingly reliant on substantial improvements in management practices. To complement these efforts, the development of incentive-based frameworks is essential to encourage extensive, conservation-oriented management of Sable Antelope by the private sector. Such strategies could enhance the species’ resilience and contribute to broader national conservation goals.
Proper habitat viability assessments should be performed before considering translocation to a new area. Additionally, while a typical herd is 10–30 individuals (Skinner & Chimimba 2005), extensive translocation has led to the fragmentation of subpopulations into small herds and consequent loss of genetic diversity (Skinner & Chimimba 2005). In Zimbabwe, 35% of 136 ranches had fewer than 50 individuals and 20% had fewer than 10 (du Toit 1992 cited in Skinner & Chimimba 2005). Similarly, in South Africa, 78% of sampled subpopulations had fewer than 20 individuals and 43% fewer than 10 (Basson 1991). Previous survey data indicate that 29% of all properties contain herds of fewer than 10 individuals and 55% of properties have herds of fewer than 30 individuals (N = 76 properties, A. Taylor, 2015, unpubl. data). More recent survey data showed similar results, indicating that 20% of private properties assessed contain herds of 10 or fewer individuals and 57% have herds fewer than 30 individuals (N = 46 properties, SANBI, 2024, unpubl. data). Grobler and van der Bank (1994) warned that calf mortality in Mpumalanga and Limpopo Provinces might be the result of inbreeding. Owen-Smith et al. (2012) also reported a potential Allee effect (a positive density dependence below a certain threshold that inhibits population recovery) in KNP, caused by currently low herd sizes and thus difficulty in finding mates, risk of inbreeding, and reduced security in numbers when facing predators.
Conservation
Sable Antelope are not widely distributed within the assessment region, and they currently only remain in seven important state-owned protected areas (see “Population” section). Kruger National Park (largest subpopulation) and Loskop Dam Nature Reserve, Mpumalanga Province, show increasing subpopulation trends. Other important state-owned protected areas have shown stark declining subpopulation trends since the previous Red List assessment in 2016. Borakalalo Nature Reserve, North West Province, reported one remaining individual in 2023, declining from 48 individuals in 2014. Kgaswane Mountain Nature Reserve, North West, experienced a 66% decline in their subpopulation between 2014 and 2023. Protected area expansion has been posed as a key intervention to mitigate the effects of climate change on biodiversity persistence. The Mpumalanga Biodiversity Sector Plan (MBSP) incorporated climate change considerations by identifying habitat corridors to enhance connectivity between natural areas. However, concerns were raised that the feasibility and resources, such as funding and human capacity, to enable proposed expansion strategies on state-owned protected areas in Africa, are lacking (Clements et al. 2022). Therefore, there is increasing recognition that privately-owned land has the potential to contribute to international and regional conservation targets (Kremen & Merenlender 2018; Taylor et al. 2021). As a large private protected area, Tswalu Kalahari Reserve, while falling outside the natural distribution range, may contain important source pools for reintroduction, as this subpopulation is showing stable trends. Subpopulations considered wild within the private wildlife ranching sector appear to have increased. However, further analysis is needed to identify the factors contributing to this increase and to accurately assess the extent of wildness in the private sector. This underscores the value of using the wildness framework by Child et al. (2019) to include wild subpopulations managed by the private sector in the Red List. In addition, the private sector should prioritise both the conservation of genetic integrity in Sable Antelope herds and the implementation of extensive management systems.
In state-owned protected areas, correct habitat and wildlife management is the most important current intervention for Sable Antelope. In some areas (for example, Kgaswane Mountain Nature Reserve) active management of potential competing species was implemented to minimise grazing competition consequences (Nel 2000). However, given current subpopulation declines in Kgaswane Mountain Nature Reserve, including the remaining state-owned protected areas, further research should prioritise effective habitat management for Sable Antelope. At Loskop Dam Nature Reserve, the limits on impala and blue wildebeest numbers, combined with correct fire management, have worked well for Sable Antelope and other selective tall-medium grass grazers (Tsessebe, Oribi, and Common Reedbuck; Redunca arundinum). As a comparison, the Songimvelo Nature Reserve subpopulation (which was founded in 1997/98 with 21 individuals from Loskop Dam Nature Reserve) became locally extinct in 2011 due to grazing competition from livestock. Livestock were added to the system due to a land claim (J. Eksteen pers. comm. 2014) and numbered around 1,500 in 2008, which pushed the Sable Antelope from savannah into higher lying sourveld areas. Sable Antelope numbers started dropping and the Mpumalanga Tourism and Parks Agency (MPTA) removed the remaining group. In the same period, Loskop Dam Nature Reserve was managed with conservative stocking of Impala and Blue Wildebeest, and no artificial water-points were established in a large area that was added to the reserve for the benefit of selective tall grass grazers. Sable Antelope and Tsessebe make use of this area. Fire can also be used in some areas to alleviate nutritional stress during the otherwise food-limiting dry season (Parrini & Owen-Smith 2010). Loskop Dam Nature Reserve managers have been able to implement planned fires in most years (J. Eksteen, pers. comm. 2014). Additionally, as most areas are fenced, it is also important that fences are regularly monitored to avoid unkept fences that facilitate the disappearance of individuals.
Carefully planned and coordinated translocations are crucial. Translocations to areas believed to be suitable (for example, in Pilanesberg National Park and Madikwe Game Reserve; Magome et al. 2008), were not always successful. It is important, when planning translocations, not only to release in areas that occur within the natural distribution range, but to precede the introduction event with a habitat suitability study that focuses on dry season resources, which is the limiting period. Sable Antelope that have been maintained in controlled, intensive management systems over extended periods often exhibit low survival rates when reintroduced into natural environments. In addition, the social structure of introduced groups plays a critical role in the success of reintroduction efforts. Empirical observations from Limpopo Province indicate that successful establishment was only achieved when larger groups with intact and appropriate social hierarchies were translocated (L. Nel, pers. comm. 2025). However, given climate change predictions, suitable habitat areas outside of the current natural distribution range as conservation introductions should be considered. It is also important to prioritise translocations to areas free of predators. Reintroducing wild herds or social units, rather than individuals or intensively managed stock, is also recommended to avoid introduction of different ecotypes or ecological naïve individuals. Translocations and reintroductions of suspected hybrid subpopulations into state-owned protected areas should be avoided and monitored.
Recommendations for land managers and practitioners:
- Sable Antelope translocations and reintroduction need to be managed more effectively at a national scale. Herds must be introduced into protected areas that have suitable habitat, habitat management and fire regimes; that do not overstock predators or other large herbivores; and that are large enough for seasonal movements and key resources regeneration. A biodiversity management plan that focuses on effective ecological management, together with a scientifically informed translocation policy (currently under development by the African node of the IUCN Conservation Genetics Specialist Group (CGSG) – ConGen Africa), will mitigate potential inbreeding and hybridisation concerns in free-ranging Sable Antelope herds. A formal risk assessment of the non-native subspecies of Sable Antelope was completed in March 2025, awaiting approval from the Department of Forestry, Fisheries and the Environment (DFFE). This risk assessment considers prohibitions of import, trade and translocations of non-native subspecies of Sable Antelope on a national scale (SANBI unpublished).
- Given the continued decline in subpopulations in state-owned protected areas and the challenges faced by the state to allocate resources for effective habitat and wildlife management, reintroductions and augmentations of Sable Antelope could be focused on private protected areas where habitat is suitable, and resource allocation is not dependent on the state.
- Recommended strategies for improved management in state-owned protected areas include enhances human capacity development, operational investment in infrastructure, governance and policy reform and improved partnerships and collaborations (Patel et al. 2023).
- Monitoring subpopulations in state-owned protected areas needs to become systematic. One of the North West Province subpopulations (Kgaswane Mountain Nature Reserve) is monitored regularly by the field rangers who keep track of total numbers and age/sex classes. This same subpopulation is also monitored in terms of areas occupied and resources utilised as individuals in the herds have GPS collars that enable researchers to remotely monitor the herd locations. Similar monitoring systems should be established in other state-owned protected areas. Group monitoring is a method in which rangers systematically patrol the habitat and record the location, composition, and dynamics of animal groups encountered in the veld daily, without relying on GPS collars. Sable Antelope typically form stable, recognisable groups, making this a cost-effective monitoring approach suitable for both state and private reserves. On state-owned land, this technique proved particularly effective. It not only facilitated regular population tracking but also helped identify problem areas. For example, if a group was observed with a certain number of calves that later failed to mature, further investigation often revealed predation as the underlying cause (L. Nel. pers. comm. 2025). Increased monitoring systems can allow for a future metapopulation plan to be implemented for state-owned protected areas (J. Power pers. comm. 2024).
- For the private sector to effectively contribute to Sable Antelope conservation, they should prioritise extensive management strategies by establishing free-ranging herds on conservancies or sufficiently large properties that require minimal intervention. Additionally, the private sector should ensure that intensively managed stock are from the correct ecotype for the region and are not mixed with subspecies from outside the region. Private land managers can be incentivised to manage Sable Antelope under extensive conditions through regulatory and ecological benefits. Certification programs recognising sustainable management practices could enhance ecotourism opportunities and increase the value of wildlife-based revenue streams, including international hunting. Access to genetic management support, such as rewilding programs or facilitated translocations, could ensure subpopulation viability and conservation value. Additionally, private land managers participating in conservation initiatives could receive grants or technical support for habitat restoration and wildlife monitoring, reinforcing the ecological and economic benefits of maintaining Sable Antelope subpopulations as part of the broader national wild population.
- Policymakers should consider implementing incentive-driven instruments to encourage the private management of Sable Antelope subpopulations in extensive systems over intensive environments.
- The Wildlife Group of the SA Veterinary Council published a booklet on Sable Antelope as a game ranch animal in 1993. Updated information can be sourced from a recent Masters dissertation (Shepstone 2022) completed in 2022 regarding ranching with Sable Antelope in South Africa.
Research priorities:
A large number of publications have been produced targeting different aspects of Sable Antelope ecology from home range selection within landscapes to foraging at plant species level and causal factors leading to decline in state-owned protected areas. The findings seem to suggest that one cause alone cannot be held responsible for the observed decline in numbers within South African state-owned protected areas and that different areas might be influenced by different constraining factors. To ensure that these projects can become part of a long-term research plan to further investigate poorly understood threats, continual logistical support is needed both from private and state-owned reserves.
Other research priorities include:
- Obtaining an accurate estimate of wild subpopulation numbers on privately owned land is essential. Additionally, further research is needed to evaluate the advantages and disadvantages of reintroducing and supplementing subpopulations with intensively managed individuals and whether such reintroductions from private stock into state-owned protected areas seems a feasible intervention.
- A genetic test should be developed that can detect possible hybrid animals (hybrids between closely related subspecies but also hybrids between different species, for example, Sable Antelope and roan; Vaz Pinto et al. 2016). Thus far, genetic studies on the geographical structure of Sable Antelope in Africa have focused solely on mitochondrial DNA (mtDNA). Therefore, management decisions which affect the survival of the species are currently being made based on these published results. However, more extensive sampling and the inclusion of additional markers in the form of nuclear DNA is necessary to identify geographically distinct populations. In addition, identification of hybridisation would only be possible with the identification of a suitable panel of nuclear markers. Research with the inclusion of both types of markers would thus provide a better understanding of genetic diversity and population structure from which conservation decisions can be evaluated.
- Long-term dataset collation for all state-owned protected areas to accurately calculate past and potential future declines. A revised population viability analysis for the subpopulation in KNP should be performed to determine whether this species qualifies as Endangered under C1 or C2a(i).
Encouraged citizen actions:
The private sector can contribute to conservation by removing fences, establishing conservancies, or transitioning from intensive to extensive management. This approach allows Sable Antelope to form wild, free-ranging herds within their natural distribution range, promoting their conservation.
Bibliography
African Wildlife Auctions (AWA). 2024. Wildlife industry trends & analysis for 2024. Available at www.wildlifeauctionstestlatest.co.za
Allendorf FW, Lear, RF, Spruel P, Wenburg JK. 2001. The problems with hybrids: Setting conservation guidelines. Trends in Ecology & Evolution 16: 613-622.
Arnold ML. 1992. Natural hybridization as an evolutionary process. Annual review of Ecology and Systematics 23: 237-261.
Asner GP, Owen-Smith N, Loarie SR, Davies AB, le Roux E, Levick SR. 2015. Habitat differences do not explain population declines of sable antelope in an African savanna. Journal of Zoology 297: 225-234.
Basson SR. 1991. n’ Voorlopige Studie van die Ekologie van die Swartwitpens in die Letaba Distrik. Report for the Letaba sable study group. [In Afrikaans].
Bezuidenhout R. 2013. High value game farming: How to get started. Farmers Weekly Magazine, Available at https://www.farmersweekly.co.za/agri-business/agribusinesses/farming-high-value-game-species/.
Boko M, Niang I, Nyong A, Vogel C, Githeko A, Medany M, Osman-Elasha B, Tabo R, Yanda P. 2007. Africa. In: E Chidumayo, D Okali, G Kowero, M Larwanou (eds.), Climate change and African forest and wildlife resources. African Forest Forum, Nairobi, Kenya.
Cain JW, Owen-Smith N, Macandza VA. 2012. The costs of drinking: comparative water dependency of sable antelope and zebra. Journal of Zoology 286: 58-67.
Capon SD. 2011. The ecology and potential factors limiting the success of Sable antelope in south eastern Zimbabwe: Implications for conservation. M.Sc. Thesis. Stellenbosch University, Stellenbosch, South Africa.
Chidumayo E, Okali D, Kowero G, Larwanou M (eds.). 2011. Climate change and African forest and wildlife resources. African Forest Forum, Nairobi, Kenya.
Child MF, Selier SJ, Radloff FG, Taylor WA, Hoffmann M, Nel L, Power RJ, Birss C, Okes NC, Peel MJ, Mallon D, Davies‐Mostert H. 2019. A framework to measure the wildness of managed large vertebrate populations. Conservation Biology 33(5): 1106-1119.
Chirima GJ, Owen-Smith N, Erasmus BNF, Parrini F. 2013. Distributional niche of a relatively rare large herbivore: habitat template versus biotic interactions. Ecography 36: 68-79.
Clements HS, Child MF, Lindeque L, Lunderstedt K, de Vos A. 2022. Lessons from COVID-19 for wildlife ranching in a changing world. Nature Sustainability 5: 1040–1048.
Cordingley JE, Sundaresan SR, Fischhoff IR, Shapiro B, Ruskey J, Rubenstein DI. 2009. Is the endangered Grevy’s zebra threatened by hybridization? Animal Conservation 12: 505-513.
Davis CL. 2011. Climate risk and vulnerability: A handbook for southern Africa. Council for Scientific and Industrial Research, Pretoria, South Africa, pp 92.
Du Toit JT, Rogers, KH, Biggs HC. 2003. The Kruger Experience: Ecology and Management of Savanna Heterogeneity. 2nd edition, Island Press, Washington, US.
Estes R. 1991. The Behaviour Guide to African Mammals. University of California Press, Berkeley, USA.
Estes RD. 2013. Hippotragus niger. In: JS Kingdon, M Hoffmann (eds.), The Mammals of Africa. VI. Pigs, Hippopotamuses, Chevrotain, Giraffes, Deer, and Bovids. Bloomsbury Publishing, London, UK.
Ferreira S, Gaylard, A, Greaver, C, Hayes, J, Cowell C, Ellis G. 2013. Animal abundances in Parks 2012/2013. Scientific Services, SANParks, Skukuza, South Africa.
Ferreira S, Gaylard, A, Greaver, C, Herbst M. 2010. Animal abundances in Parks 2009/2010. Scientific Services, SANParks, Skukuza, South Africa.
Franklin I. 1980. Evolutionary change in small populations. In: EE Soulé, BA Wilcox (eds.), Conservation Biology: An evolutionary-ecological perspective. Sinauer Associates, pp. 135-150.
Friedmann Y, Daly B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. Conservation Breeding Specialist Group (SSC/IUCN) and Endangered Wildlife Trust, Parkview, South Africa.
Gandiwa E. 2014. Vegetation factors influencing density and distribution of wild large herbivores in a southern African savannah. African Journal of Ecology 52: 274-283.
Grant CC, Davidson T, Funston PJ, Pienaar DJ. 2002. Challenges faced in the conservation of rare antelope: a case study on the northern basalt plains of the Kruger National Park. Koedoe 45: 45–66.
Grant CC, van der Walt JL. 2000. Towards an adaptive management approach for the conservation of rare antelope in the Kruger National Park-outcome of a workshop held in May 2000. Koedoe 43: 103-112.
Greaver C, Ferreira S, Simms C, Botha J, Crowhurst E. 2024. Estimates of animal species in the Kruger National Park – 2023. Aerial Survey Report – March 2024, South African National Parks (SANParks).
Green WCH, Rothstein A. 1998. Translocation, hybridization, and the endangered Black-Faced Impala. Conservation Biology 12(2): 475-480.
Grey-Ross R, Downs CT, Kirkman K. 2009. Reintroduction failure of captive-bred oribi (Ourebia ourebi). South African Journal of Wildlife Research 39(1): 34–38.
Grobler JH. 1973. Aspects of the biology, population ecology and behaviour of the sable antelope, Hippotragus niger niger (Harris, 1838) in the Rhodes Matopos National Park, Rhodesia. M.Sc. Thesis. University of Pretoria, Pretoria, South Africa.
Grobler JH. 1981. Feeding behaviour of sable Hippotragus niger niger (Harris, 1838) in the Rhodes Matopos National Park, Zimbabwe. South African Journal of Zoology 16: 50-58.
Grobler JP, Rushworth I, Brink JS, Bloomer P, Kotze A, Reilly B, Vrahimis S. 2011. Management of hybridization in an endemic species: Decision making in the face of imperfect information in the case of the black wildebeest—Connochaetes gnou. European Journal of Wildlife Research 57: 997-1006.
Grobler JP, van der Bank FH. 1994. Genetic heterogeneity in sable antelope (Hippotragus niger Harris 1838) from four southern African regions. Biochemical Systematics and Ecology 22: 781-789.
Harrington R, Owen-Smith N, Viljoen PC, Biggs HC, Mason DR, Funston PJ. 1999. Establishing the causes of the roan antelope decline in the Kruger National Park, South Africa. Biological Conservation 90(1): 69-78.
Harris CW. 1838. Narrative of an expedition into Southern Africa during the years 1836 and 1837, from the Cape of Good Hope, through the territories of the Chief Moselakatse, to the Tropic of Capricorn, with a sketch of the recent emigration of the border colonists, and a zoological appendix. American Mission Press, Bombay.
Hensman MC, Owen-Smith N, Parrini F, Bonyongo CM. 2014. Home range occupation and habitat use of sable antelope in the Okavango Delta region of northern Botswana. African Journal of Ecology 52: 237-245.
Hensman MC, Owen-Smith N, Parrini F, Erasmus BF. 2012. Dry season browsing by sable antelope in northern Botswana. African Journal of Ecology 50: 513-516.
Intergovernmental Panel on Climate Change (IPCC). 2014. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change [Core Writing Team, R.K. Pachauri and L.A. Meyer (eds.)]. IPCC, Geneva, Switzerland, 151 pp.
International Federation of Organic Agriculture Movements (IFOAM). 2014. The IFOAM norms for organic production and processing. Version 2014. IFOAM Organics International, Germany.
International Union for Conservation of Nature (IUCN) Standards and Petitions Subcommittee. 2014. Guidelines for Using the IUCN Red List Categories and Criteria. Version 11.
Jansen van Vuuren B, Robinson TJ, Vaz Pinto P, Estes R, Matthee CA. 2010. Western Zambian sable: Are they a geographic extension of the giant sable antelope? African Journal of Wildlife Research, 40(1): 35–42.
Karp AT, Uno KT, Berke MA, Russell JM, Scholz CA, Marlon JR, Staver AC. 2023. Nonlinear rainfall effects on savanna fire activity across the African Humid Period. Quaternary Science Reviews 304: 107994.
Kremen C, Merenlender AM. 2018. Landscapes that work for biodiversity and people. Science 362(6412): eaau6020.
Kriek J. n.d. The history of Manattu Private Game Reserve. Available at: https://mattanu.com/history/.
Kupika OL, Gandiwa E, Kativu S, Nhamo G. 2017. Impacts of climate change and climate variability on wildlife resources in southern Africa: Experience from selected protected areas in Zimbabwe In: B Sen, O Grillo (eds.) Selected Studies in Biodiversity, pp. 1-23.
Levin DA, Francisco-Ortega J, Jansen RK. 1996. Hybridization and the extinction of rare plant species. Conservation Biology 10: 10-16.
Macandza VA, Owen-Smith N, Cain JW. 2012. Habitat and resource partitioning between abundant and relatively rare grazing ungulates. Journal of Zoology 287:175–185.
Magome H, Cain III JW, Owen-Smith N, Henley SR. 2008. Forage selection of sable antelope in Pilanesberg Game Reserve, South Africa. South African Journal of Wildlife Research 38(1): 35-41.
Matthee CA, Robinson TJ. 1999. Mitochondrial DNA population structure of roan and sable antelope: implications for the translocation and conservation of the species. Molecular Ecology 8: 227-238.
Milton SJ. 2004. Grasses as invasive alien plants in South Africa: Working for water. South African Journal of Science 100(1): 69-75.
Nel HP. 2000. Ecological management objectives and monitoring procedures for Rustenburg Nature Reserve, North West province. M.Sc. Thesis. University of Pretoria, Pretoria, South Africa.
Nel P. 2015. Population estimates for large herbivores and predators in protected areas in the North West Parks Board November 2015. North West Parks Board.
Nel P. 2016. Population estimates for animals in parks in the North West Parks Board 2016. North West Parks Board.
O’Connor TG, van Wilgen BW. 2020. The impact of invasive alien plants on rangelands in South Africa In: BW van Wilgen, J Measey, DM Richardson, JR Wilson, TA Zengeya (eds.) Biological Invasions in South Africa, Springer, pp. 459-487.
Owen-Smith N. 1988. Megaherbivores: The Influence of Very Large Body Size on Ecology. Cambridge University Press, Cambridge, UK.
Owen-Smith N. 1996. Ecological guidelines for waterpoints in extensive protected areas. South African Journal of Wildlife Research 26: 107-112.
Owen-Smith N. 2003. Foraging behavior, habitat suitability, and translocation success, with special reference to large mammalian herbivores. In: M Festa-Bianchet, M Apollonio (eds.) Animal Behaviour and Wildlife Conservation, pp. 93–109. Island Press, Washington, USA.
Owen-Smith N. 2008. The comparative population dynamics of browsing and grazing ungulates. In: IJ Gordon, HHT Prins (eds.) The Ecology of Browsing and Grazing. Springer-Verlag, Berlin, Germany, pp. 149-178.
Owen-Smith N, Cain JW. 2007. Indicators of adaptive responses in home range utilization and movement patterns by a large mammalian herbivore. Israel Journal of Ecology & Evolution 53: 423-438.
Owen-Smith N, Chirima GJ, Macandza V, le Roux E. 2012. Shrinking sable antelope numbers in Kruger National Park: What is suppressing population recovery? Animal Conservation 15: 195–204.
Owen-Smith N, Le Roux E. & Macandza V. 2013. Are relatively rare antelope narrowly selective feeders? A sable antelope and zebra comparison. Journal of Zoology 291: 163-170.
Owen-Smith N, Mills MGL. 2006. Manifold interactive influences on the population dynamics of a multispecies ungulate assemblage. Ecological Monographs 76: 73-92.
Pacifici M, Santini L, Di Marco M, Baisero D, Francucci L, Grottolo Marasini G, Visconti P, Rondinini C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Pacifici M, Viscont, P, Scepi E, Hausmann A, Attorre F, Grant R, Rondinini C. 2015. Fire policy optimization to maximize suitable habitat for locally rare species under different climatic conditions: a case study of antelopes in the Kruger National Park. Biological Conservation 191: 313-321.
Parrini F. 2006. Nutritional and social ecology of the sable antelope in a Magaliesberg Nature Reserve. Ph.D. Thesis. University of the Witwatersrand, Johannesburg, South Africa.
Parrini F, Owen-Smith N. 2010. The importance of post-fire regrowth for sable antelope in a Southern African savanna. African Journal of Ecology 48: 526-534.
Patel T, Cowan O, Little I, Friedmann Y, Blackmore A. 2023. The state of provincial reserves in South Africa: Challenges and recommendations. The Endangered Wildlife Trust, Johannesburg, South Africa.
Pitra C, Hansen AJ, Lieckfeldt D, Arctander P. 2002. An exceptional case of historical outbreeding in African sable antelope populations. Molecular Ecology 11: 1197-1208.
Pitra C, Vaz Pinto P, O’Keeffe BWJ, Willows-Munro S, Jansen Van Vuuren B, Robinson TJ. 2006. DNA-led rediscovery of the giant sable antelope in Angola. European Journal of Wildlife Research 52(3): 145-152.
Power RJ. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Rhymer JM, Simberloff D. 1996. Extinction by hybridization and introgression. Annual Review of Ecology and Systematics 27: 83-109.
Rocha LJ, Vaz Pinto P, Siegismund HR, Meyer M, Jansen van Vuuren B, Veríssimo L, Ferrand N, Godindo R. 2022. African climate and geomorphology drive evolution and ghost introgression in sable antelope. Molecular Ecology 31: 2968-2984.
Saidi TA, Tshipala-Ramatshimbila TV. 2006. Ecology and management of a remnant Brachystegia Speciformis (Miombo) Woodland in North Eastern Soutpansberg, Limpopo Province. South African Geographical Journal 88: 205-212.
Sekulic R. 1981. Conservation of the sable Hippotragus niger roosevelti in the Shimba Hills, Kenya. African Journal of Ecology 19: 153-165.
Selier J, Nel L, Rushworth I, Kruger J, Coverdale B, Mulqueeny C, Blackmore A. 2018. An assessment of the potential risks of the practice of intensive and selective breeding of game to biodiversity and the biodiversity economy in South Africa. Gland, Switzerland: IUCN.
Shepstone CA. 2022. Ranching with Sable Antelope (Hippotragus niger) in South Africa. M.Sc. Thesis. University of Stellenbosch, Stellenbosch, South Africa.
Shumba T, de Vos A, Biggs R, Esler KJ, Ament JM, Clements HS. 2020. Effectiveness of private land conservation areas in maintaining natural land cover and biodiversity intactness. Global Ecology and Conservation 22: e00935.
Skinner JD, Chimimba CT. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
South African National Biodiversity Institute (SANBI). unpublished. Risk analysis of Hippotragus niger (Harris, 1838) for South Africa as per the risk analysis for alien taxa framework v2.0, approved by the South African Alien Species Risk Analysis Review Panel on 28 March 2025, pp. 28, http://dx.doi.org/10.5281/zenodo.15102393.
Taylor WA, Child MF, Lindsey PA, Nicholson SK, Relton C, Davies-Mostert HT. 2021. South Africa’s private wildlife ranches protect globally significant populations of wild ungulates. Biodiversity Conservation 30(30): 4111–4135.
Taylor WA, Lindsey PA, Davies-Mostert H. 2016. An assessment of the economic, social and conservation value of the wildlife ranching industry and its potential to support the green economy in South Africa. The Endangered Wildlife Trust, Johannesburg.
Van Wyk AM, Kotzé A, Randi E, Dalton DL. 2013. A hybrid dilemma: A molecular investigation of South African bontebok (Damaliscus pygargus pygargus) and blesbok (Damaliscus pygargus phillipsi). Conservation Genetics 14: 589-599.
Vaz Pinto P, Beja B, Ferrand N, Godinho R. 2016. Hybridisation following population collapse in a critically endangered antelope. Scientific Reports 6: 18788.
Washington R, Preston A. 2006. Extreme wet years over southern Africa: Role of Indian Ocean sea surface temperatures. Journal of Geophysical Research 111: D15104.
Wilson DE, Hirst S. 1977. Ecology and factors limiting roan and sable antelope populations in South Africa. Wildlife Monographs 54: 1-109.